Abstract
In this prospective study, a canine-adapted histological grading method was compared with histopathological and clinical characteristics and was evaluated as a prognostic indicator in canine mammary carcinomas (CMCs). Recruited dogs with at least 1 malignant mammary tumor (n = 65) were clinically evaluated, surgically treated, and followed up (minimum follow-up 28 months, maximum 38 months). Histopathological diagnoses were performed according to Goldschmidt et al (2011). Tumors were graded as grade I (29/65), grade II (19/65), and grade III (17/65). The tumor size, clinical stage, histological diagnosis, presence/absence of myoepithelial proliferation, and regional lymph node metastases at diagnosis were significantly associated with histological grade. The histological grade, age, clinical stage, tumor subtype group, and lymph node metastases at time of diagnosis were significantly associated with the development of recurrences and/or metastases, cancer-associated death, and survival times (disease-free survival and overall survival) in univariate analyses. A subdivision of clinical stage I (T1N0M0) into stages IA and IB was proposed in terms of prognosis. The clinical stage, histological grade, and spay status were selected as independent prognostic variables (multivariate analyses) with disease-free survival as the dependent variable. When overall survival was evaluated as a dependent variable, clinical stage and histological grade were selected as the independent covariates. This grading system is a useful prognostic tool, facilitates histological interpretation, and offers uniform criteria for veterinary pathologists. Comparative studies on CMCs performed in different countries should take into account possible changes in the prognoses due to different proportions of spayed females among the selected dog population.
Keywords
Histological diversity of canine mammary tumors makes their diagnosis difficult and provides little prognostic information. The use of a histological grading system may be helpful for classification and prognosis. In canine mammary carcinomas (CMCs), the degree of infiltration
12
has been used and has been found to be of prognostic significance.34,35 Several histological malignancy grading systems have been proposed for human breast cancer to simplify the information provided by the pathologist to the clinician and to make this information more accurate.13,38 Among these grading systems, the most prevalent system used worldwide is the Elston and Ellis numeric method for grading human breast cancer; this method is also known as the Nottingham method.
7
This grading system is based on the assessment of the following 3 morphological features: tubule formation, nuclear pleomorphism, and mitotic counts. Each of these features is scored from 1 to 3, and this grading system is mostly directed at invasive (simple) adenocarcinomas. In recent years, this grading system has been used to study CMCs.6,8–11,16,18,20–22,27,31–33 Three of these studies18,25,27 evaluated the prognostic significance of this human method demonstrating its predictive value in univariate analyses.18,27 However, this method is not an independent prognostic indicator when using the required multivariate prognostic analyses.25,27 Furthermore, several specific characteristics of CMCs require a system adapted to the dog as follows:
A low proportion of CMCs metastasize.1,4,19 A grading system that discriminates between tumors that could potentially recur or metastasize and tumors that could not recur or metastasize is desirable. Complex and mixed malignant mammary tumors occur frequently in the dog.
13
The Elston and Ellis method does not include myoepithelial proliferation or mesenchymal areas. It is essential to use uniform criteria in assessing these areas when complex or mixed tumors are graded. There are specific criteria of malignancy in canine mammary tumors.
13
Nuclear pleomorphism often leads to overdiagnosis of malignancy even in benign tumors.
There are 2 slightly different canine-adapted grading numeric systems of CMC (modifications of the human method)
13
: the Misdorp
23
and Peña
3
systems. According to a recent publication on the classification and grading of canine mammary tumors, separating malignant neoplasms into categories with a meaningful clinical outcome requires additional retrospective and prospective studies.
13
A retrospective study comparing both the Misdorp and Peña grading systems on a large series of canine malignant mammary neoplasms was recently published.
30
This previous study showed that both histological grading systems are significant predictors (P < .001) of the risk of lymphatic vessel invasion and regional lymph node metastases at the time of diagnosis. However, the Peña system (partially published as Clemente et al
3
) has been shown to have a better predictive ability.
The present study is a prospective study in which the Peña histological grading method of canine mammary tumors is completely described and evaluated as a prognostic indicator following the recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology. 39
Materials and Methods
Recruitment of Animals, Criteria of Inclusion, and Clinical Procedures
In this prospective cohort study, female dogs with at least 1 malignant mammary tumor presented throughout 2008 at the Complutense Veterinary Teaching Hospital of Madrid (Complutense University, Madrid, Spain) were recruited. A complete clinical history and physical examination as well as a specific and detailed examination of the mammary gland region and drainage lymph nodes were performed. Routine 3-view (2 laterolateral projections and a dorsoventral projection) thoracic radiographs were taken in each case to determine the presence of metastatic disease. The following data were registered: age at diagnosis, spay status, breed, weight, regularity of estrus, and previous hormonal treatments. Information regarding the number of neoplasms per animal, location, size (in centimeters; largest diameter), adherence to skin, adherence to underlying tissues, and tumor ulceration was obtained. Clinical staging of animals into the following clinical categories according to a modified WHO clinical staging system 19 was performed: stage I (T1N0M0), stage II (T2N0M0), stage III (T3N0M0), stage IV (TanyN1M0), and stage V (TanyNanyM1). For determination of the stage, lymph node involvement was confirmed by cytology. All animals included in the cohort were in clinical stages I, II, III, and IV at the time of diagnosis and were surgically treated. Dogs with mammary sarcoma, carcinosarcoma, inflammatory mammary cancer, and distant metastases (clinical stage V) were excluded from the study. The surgical treatment performed included nodulectomy, simple mastectomy, regional mastectomy, and complete mastectomy depending on the clinical stage and number of tumors in the mammary chain. All intact dogs were ovariohysterectomized at the time of mastectomy. Dogs included in this study underwent surgery as the only treatment, and animals treated with adjuvant chemotherapy after surgery were excluded. This study was conducted in accordance with the Animal Ethics Institutional Committee, and consent forms were signed by owners.
Histopathology and Grading of CMCs
Samples of surgically resected mammary neoplasms and corresponding lymph nodes (when available) were fixed in 10% buffered formalin and routinely processed for histopathology. Neoplasms were histologically diagnosed throughout 2008 and reevaluated in 2010 (original manuscript before publication) following a new classification of canine mammary tumors recently published. 13 This classification 13 includes the following 5 new morphological subtypes of CMCs as compared to the 1999 WHO classification 24 : micropapillary invasive carcinoma; comedocarcinoma; ductal carcinoma; intraductal papillary carcinoma; and carcinoma and malignant myoepithelioma.
The tumor malignancy grade was determined as indicated in Table 1. This canine adaptation of the human Elston and Ellis grading system 7 considers the heterogeneity of CMC, states how to evaluate myoepithelial proliferation areas and mixed neoplasms, and modifies the evaluation of nuclear features. Thus, in heterogeneous canine mammary carcinomas, tubular scoring was assessed in the most representative malignant area. In complex and mixed tumors, the percentage of tubular formation was scored considering only epithelial areas, and nuclear pleomorphism was evaluated in all the malignant components. Tumors classified as grades I, II, and III are shown in Figs. 1 to 6. Due to the inability of this system to grade sarcomas, dogs with mammary sarcomas and carcinosarcomas were excluded. The presence or absence of evident myoepithelial proliferation (MP) was annotated in each case.
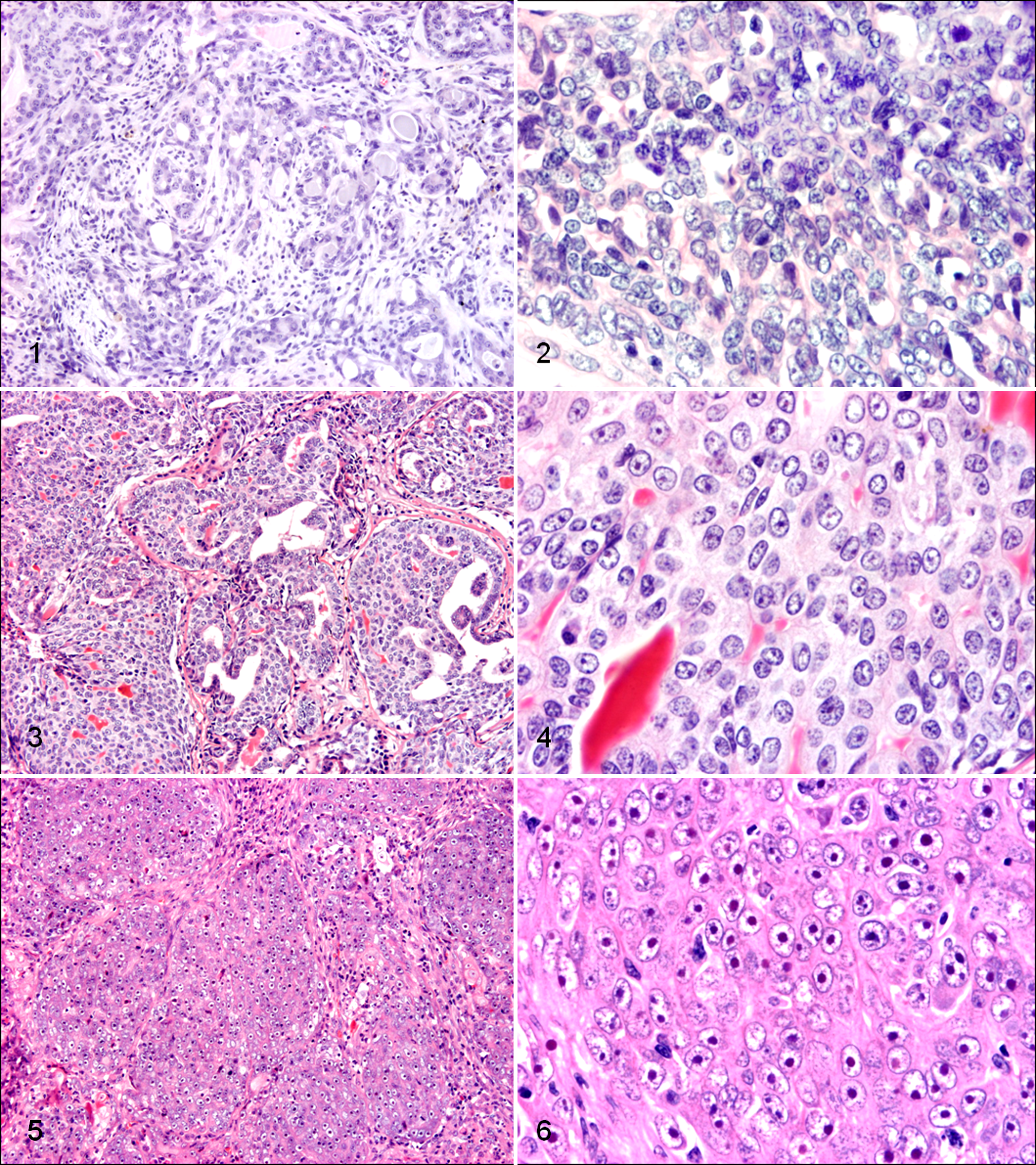
Histological Grading System for Canine Mammary Cancer.
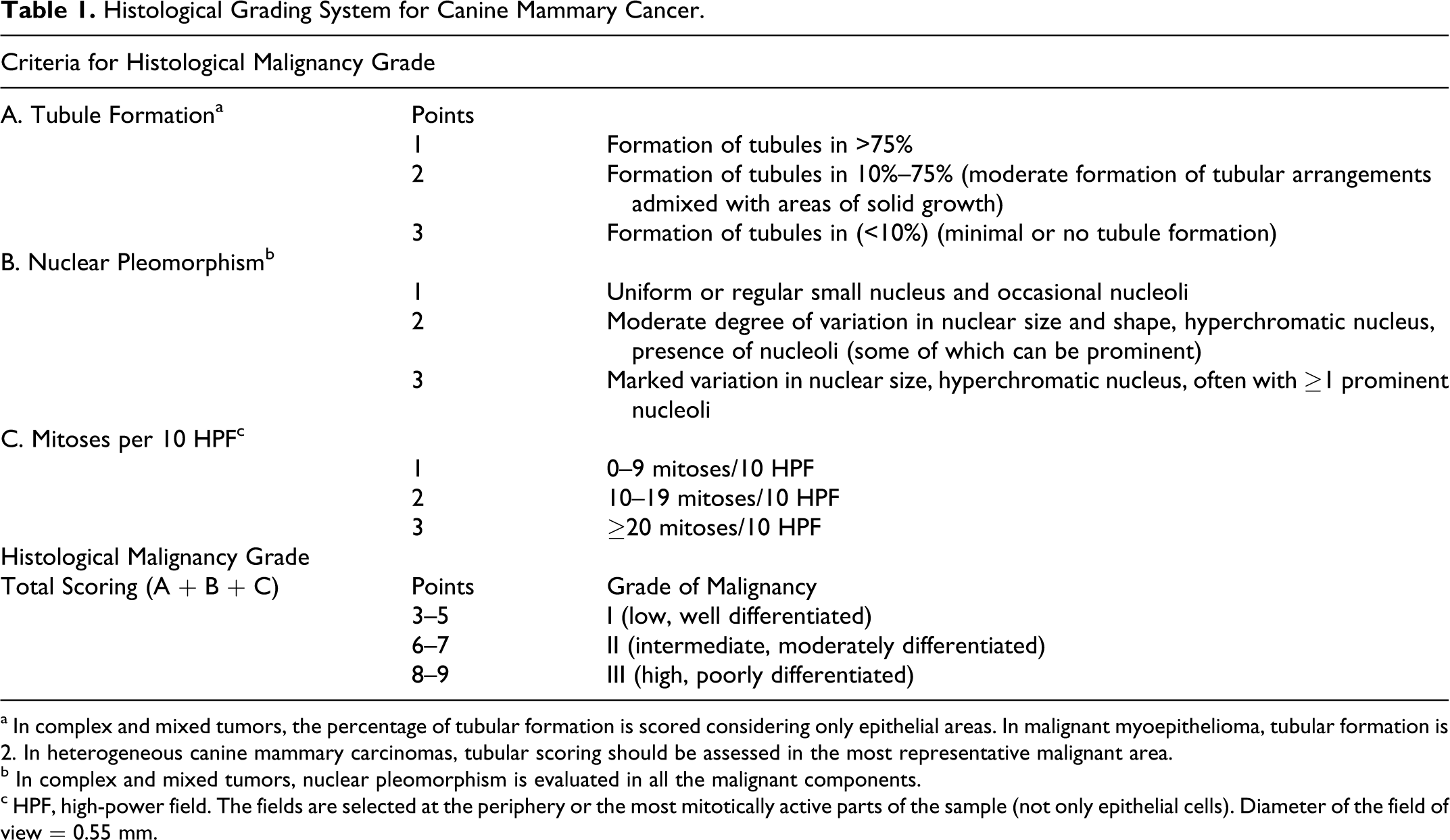
a In complex and mixed tumors, the percentage of tubular formation is scored considering only epithelial areas. In malignant myoepithelioma, tubular formation is 2. In heterogeneous canine mammary carcinomas, tubular scoring should be assessed in the most representative malignant area.
b In complex and mixed tumors, nuclear pleomorphism is evaluated in all the malignant components.
c HPF, high-power field. The fields are selected at the periphery or the most mitotically active parts of the sample (not only epithelial cells). Diameter of the field of view = 0.55 mm.
Follow-Up
After surgery, animals were followed up every 4 to 6 months for at least 28 months (maximum follow-up of 38 months). Each follow-up visit included a history, complete physical examination, mammary chain evaluation, regional lymphatic drainage evaluation, and radiological thoracic examination (2 projections). The clinical outcome was assessed by considering the development of local recurrences, distant metastases, or cancer-associated death as end points. Local recurrence was defined as the development of a subsequent CMC at the original tumor location. Disease-free survival (DFS; time from surgery to the development of recurrences and/or metastases) and overall survival (OS; time from surgery to death caused by cancer or end of the follow-up) were calculated. The follow-ups may have ended due to the following reasons: (a) cancer death, (b) death by other causes, (c) lost for follow-up (at least 28 months of follow-up available), and (d) end of the study (38 months). Data resulting from reasons b, c, and d were censored data for the survival study.
Statistical Study
In animals with more than 1 malignant tumor, the most malignant tumor (histopathological and clinical criteria, ie, the clinical stage) 23 was selected for statistical purposes. The epidemiological, clinical, histopathological, and follow-up variables were included in the statistical analyses. The epidemiological and clinical variables were as follows: age (years), breed (large breed ≥20 kg and small breed <20 kg), previous estrus preventive hormonal treatments (no/yes), regularity of heats (no/yes), number of malignant tumors/animal, tumor location (cranial included thoracic and cranial abdominal mammary glands, and caudal included caudal abdominal and inguinal mammary glands), and size. For statistical purposes, tumor size was analyzed using the following 3 variables: (a) numerical variable (in centimeters), (b) WHO size19,26 (categorized as T1 < 3 cm, T2 = 3–5 cm, and T3 > 5 cm), and (c) a new variable of tumor size with 3 categories (small size, tumors <1 cm; intermediate size, tumors between 1 and 2.9 cm; and large size, tumors ≥3 cm). The latter variable was used to determine whether there were prognostic significant differences in tumors categorized as being less than 3 cm. The following additional variables were included in the statistical analyses: skin ulceration (no/yes), adherence to skin (no/yes), adherence to underlying tissues (no/yes), clinical stage, and type of surgery (nodulectomy, simple mastectomy, regional mastectomy, and complete mastectomy). In addition to the histological diagnosis, some tumor subtypes were grouped into 3 histological categories for statistical evaluation on the basis of morphological features and biological behavior (performed according to previous studies23,24 and prior experience). This grouping generated a new histological variable called HD3, and the HD3 variable was classified by 3 different groups as follows: group 1, which included in situ carcinoma, simple carcinoma, carcinoma arising in a mixed tumor, complex carcinoma, mixed-type carcinoma, ductal carcinoma, and adenosquamous carcinoma; group 2, which included solid carcinoma, comedocarcinoma, carcinoma and malignant myoepithelioma, and anaplastic carcinoma; and group 3, which included other histological types. The histological grade of malignancy (I, II, or III) and presence of evident myoepithelial cell proliferation (MP) (no/yes) were included as histological variables. The following follow-up variables were considered: recurrences and/or metastases (no/yes), death by tumor (no/yes), DFS (months), and OS (months). Associations of variables were analyzed using the IBM SPSS Statistics 19 statistical package program. Kaplan-Meier survival curves were constructed for the disease-free period and overall survival. Regression analyses using the multivariate Cox proportional hazards model (Cox regression) were performed to evaluate the influence of variable sets (risk factors, named covariates) on dependent follow-up variables (DFS and OS). Whereas the Kaplan-Meier method with the log-rank test is useful for comparing survival curves in 2 or more groups, the Cox proportional-hazards regression allows analysis of the combined effect of several risk factors on survival. The probability of the end point (recurrences/metastases or death by cancer) is called the hazard. The hazard ratio (HR) is calculated with the following formula: HR = exp(β1 X1 + … + βnXn); where β1 … β n , estimated by Cox regression, are the coefficients affecting the sets of related independent variables (covariates, risk factors) included in the model (X1, X2, …, Xn). The dichotomous covariates are coded 1 if present and 0 if absent. In variables with 3 and 4 categories (histological grade and clinical stage, respectively), the system transforms them into dichotomous variables (clinical stages A, B, and C and histological grades A and B) (Table 2 includes additional explanations). Exp(β n ) represents the instantaneous relative risk of an event at any time for an individual with the risk factor “present” as compared with an individual with the risk factor “absent” when all other covariates are held constant. In the present Cox regression model, covariates were selected according to the “forward” method as follows: the independent variables entered the system sequentially in several steps according to their significance. Those variables that were significant at one step were then considered constants for evaluation of the combined effect of the other variables on the dependent variables. In all the statistical analyses, P values less than .05 were considered statistically significant. Only animals with complete data sets for the variables studied were included.
Multivariate Cox Proportional Hazard Analyses (Cox Regression) of Disease-Free Survival (DFS) and Overall Survival (OS) in Dogs (n = 65) With Malignant Mammary Tumors in a Prospective Study With 28–38 Months of Follow-Up.
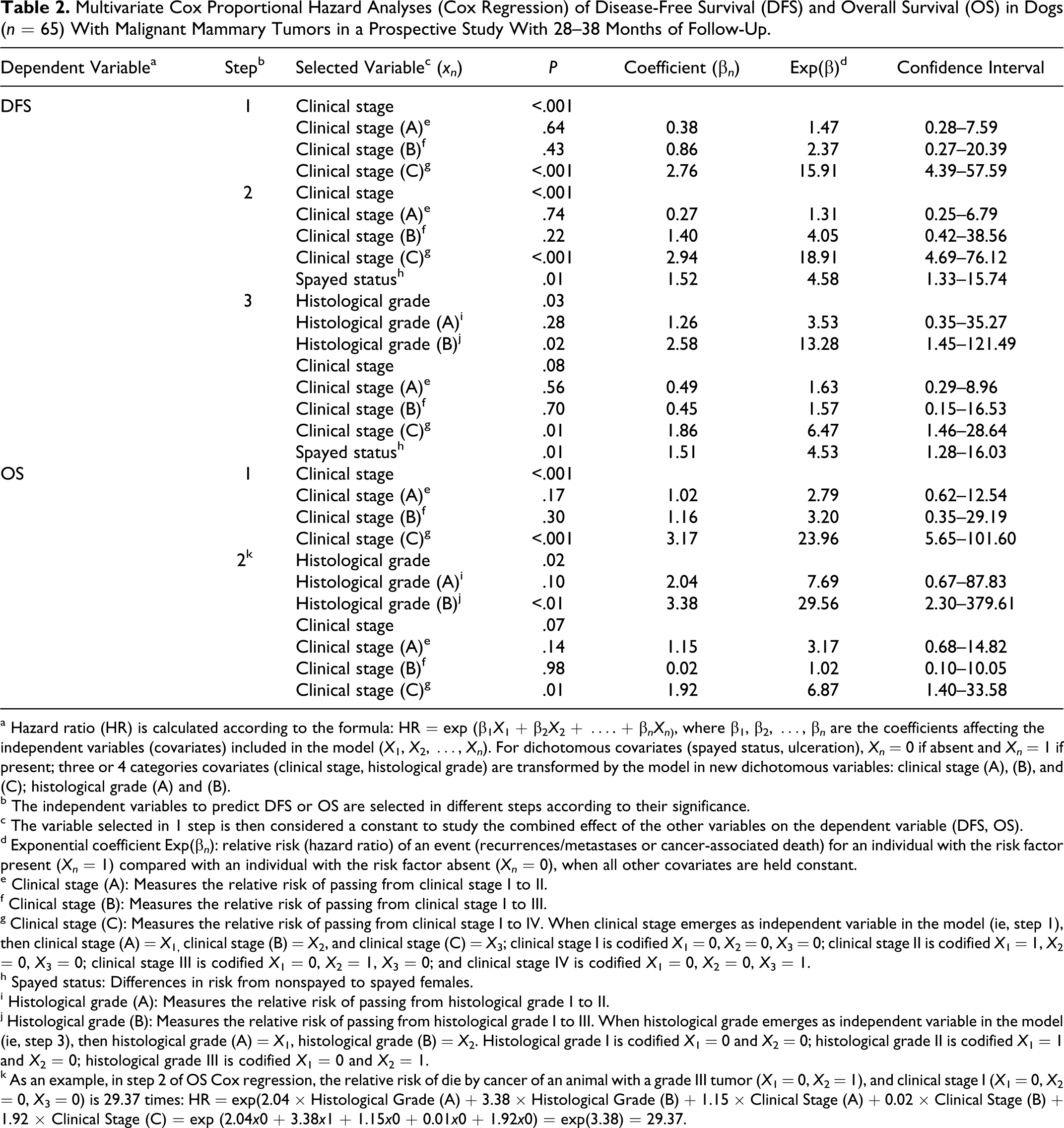
a Hazard ratio (HR) is calculated according to the formula: HR = exp (β1 X 1 + β2 X 2 + …. + β nXn ), where β1, β2, …, β n are the coefficients affecting the independent variables (covariates) included in the model (X 1, X 2, …, Xn ). For dichotomous covariates (spayed status, ulceration), Xn = 0 if absent and Xn = 1 if present; three or 4 categories covariates (clinical stage, histological grade) are transformed by the model in new dichotomous variables: clinical stage (A), (B), and (C); histological grade (A) and (B).
b The independent variables to predict DFS or OS are selected in different steps according to their significance.
c The variable selected in 1 step is then considered a constant to study the combined effect of the other variables on the dependent variable (DFS, OS).
d Exponential coefficient Exp(β n ): relative risk (hazard ratio) of an event (recurrences/metastases or cancer-associated death) for an individual with the risk factor present (Xn = 1) compared with an individual with the risk factor absent (Xn = 0), when all other covariates are held constant.
e Clinical stage (A): Measures the relative risk of passing from clinical stage I to II.
f Clinical stage (B): Measures the relative risk of passing from clinical stage I to III.
g Clinical stage (C): Measures the relative risk of passing from clinical stage I to IV. When clinical stage emerges as independent variable in the model (ie, step 1), then clinical stage (A) = X 1, clinical stage (B) = X 2, and clinical stage (C) = X 3; clinical stage I is codified X 1 = 0, X 2 = 0, X 3 = 0; clinical stage II is codified X 1 = 1, X 2 = 0, X 3 = 0; clinical stage III is codified X 1 = 0, X 2 = 1, X 3 = 0; and clinical stage IV is codified X 1 = 0, X 2 = 0, X 3 = 1.
h Spayed status: Differences in risk from nonspayed to spayed females.
i Histological grade (A): Measures the relative risk of passing from histological grade I to II.
j Histological grade (B): Measures the relative risk of passing from histological grade I to III. When histological grade emerges as independent variable in the model (ie, step 3), then histological grade (A) = X 1, histological grade (B) = X 2. Histological grade I is codified X 1 = 0 and X 2 = 0; histological grade II is codified X 1 = 1 and X 2 = 0; histological grade III is codified X 1 = 0 and X 2 = 1.
k As an example, in step 2 of OS Cox regression, the relative risk of die by cancer of an animal with a grade III tumor (X 1 = 0, X 2 = 1), and clinical stage I (X 1 = 0, X 2 = 0, X 3 = 0) is 29.37 times: HR = exp(2.04 × Histological Grade (A) + 3.38 × Histological Grade (B) + 1.15 × Clinical Stage (A) + 0.02 × Clinical Stage (B) + 1.92 × Clinical Stage (C) = exp (2.04x0 + 3.38x1 + 1.15x0 + 0.01x0 + 1.92x0) = exp(3.38) = 29.37.
Results
Descriptive Data
Sixty-five female dogs aged from 3 to 15 years (mean of 9.68 years) both intact (n = 52) and spayed (at least 4 years before tumor presentation; n = 13) as well as belonging to 19 different breeds and mixed breeds were included. Animals were further classified by size as follows: small (n = 38) and large (n = 27). Forty-six animals (70.76%) presented 1 malignant tumor, 13 animals (20.00%) had 2 malignant tumors, and 6 animals (9.23%) had 3 or more malignant tumors. Concurrent benign mammary neoplasms and/or dysplasias were observed in 50 of the 65 animals (76.92%). Of the 65 animals, 10 had benign neoplasms (15.38%), 24 (36.92%) had mammary dysplasias, and 16 (24.61%) had benign mammary tumors and dysplasias.
The tumor size ranged from 0.5 to 14 cm (n = 65; mean = 2.2 cm). According to the WHO tumor size system, there were 45 T1 neoplasms (<3 cm), 15 T2 neoplasms (3–5 cm), and 5 T3 neoplasms (>5 cm). When considering the additional variable of tumor size used in this study, tumors were classified as follows: small size (<1 cm; n = 20), intermediate size (1–2.9 cm; n = 25), and large size (≥3 cm; n = 20). With regard to clinical staging, 42 animals (64.61%) were in stage I, 11 animals (16.92%) were in stage II, 5 animals (7.69%) were in stage III, and 7 animals (10.76%) were in stage IV. The histological types diagnosed are depicted in Table 3. Myoepithelial proliferation was observed in 25 of the 65 dogs (38.46%). The tumors were graded as follows: grade I (29/65; 44.61%), grade II (19/65; 29.23%), and grade III (17/65; 26.15%).
Histological Malignancy Grades and Histological Diagnosis.a
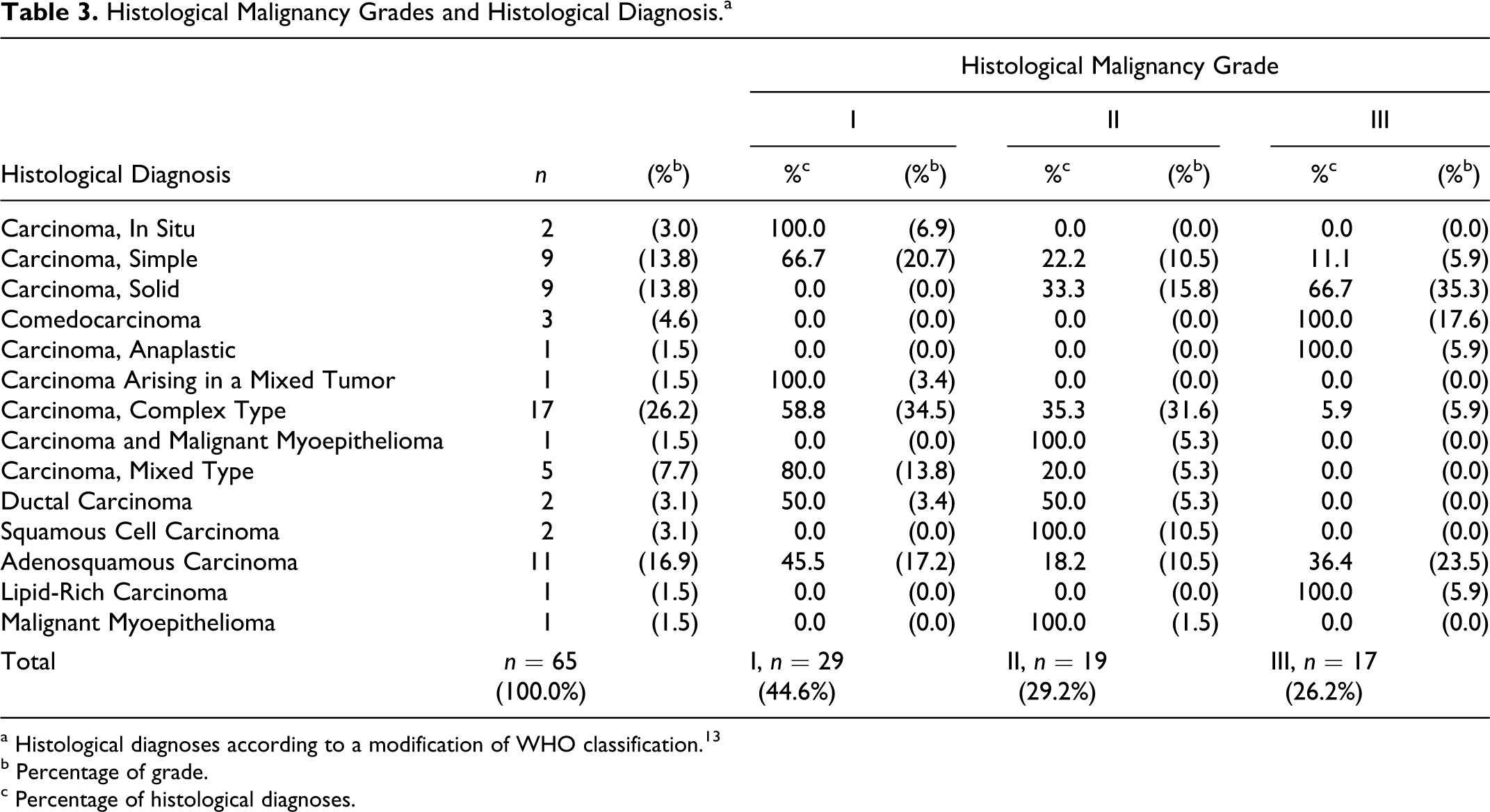
a Histological diagnoses according to a modification of WHO classification. 13
b Percentage of grade.
c Percentage of histological diagnoses.
During the follow-up after surgical excision (28–38 months), 14 animals (14/65; 21.53%) developed recurrences and/or metastases. Of these 14 animals, 3 animals developed only recurrences, 6 animals developed only metastases, and 5 animals developed recurrences and metastases (n = 14). Thirteen of 65 (20.00%) animals died due to mammary cancer.
Histological Grade and Epidemiological, Clinical, and Histological Variables
Univariate Analyses
None of the epidemiological or reproductive variables analyzed were related to histological grade. Tumor size (<1, 1–2.9, and ≥3 cm; P = 0.03) and clinical stage (P < .01) were associated with histological grade: tumor size was significantly higher in grade III tumors compared with grade I tumors. The WHO size of tumors was not significantly associated with grade (P = 0.06). All animals with grade I neoplasms, except for 1 animal, presented clinical stages I and II.
Histological diagnosis and grade were significantly associated (P < .01). All comedocarcinomas were grade III, and most of the simple carcinomas, complex carcinomas, and mixed-type carcinomas were grade I. Detailed information about histological diagnosis and grade is provided in Table 3. Due to the high number of histological subtypes and low number of dogs in some of these categories (prospective study), the HD3 variable was statistically analyzed and was also found to be significantly related to histological grade (P < .001). Histological grade was associated with the presence or absence of myoepithelial proliferation (P = 0.005) because most of the grade III tumors did not show MP (16/17; 94.1%).
During mastectomy, surgical excision of the regional lymph node was performed in 40 dogs. After histological examination of the paraffin sections, 5 dogs were found to be positive for regional lymph node metastases, and all of these dogs corresponded to grade III mammary neoplasms (2 comedocarcinomas, 1 solid carcinoma, 1 anaplastic carcinoma, and 1 adenosquamous carcinoma) (P < .001). Four of these 5 dogs with metastatic lymph nodes had been previously detected by cytology (P < .001).
Prognostic Study
Univariate Analyses of Follow-Up End Points and Survival
Recurrences/metastases
Histological grade was significantly associated (P < .001) with the development of recurrences and/or metastases as follows. Most of the neoplasms that recurred or metastasized were grade III (10/14; 71.42%) (Table 3). Age (animals between 9 and 11 years; P = 0.01) and spay status (P = 0.01) were epidemiological variables associated with the development of recurrences and/or metastases (14/65). Histological type (P < .001), absence of MP (P = 0.02), and HD3 (P < .001) were significantly associated. Solid carcinoma was the neoplasm that most frequently recurred or metastasized (6/14; 40.0%), followed by comedocarcinoma (3/14; 20.0%), simple carcinoma (2/14; 13.3%), adenosquamous carcinoma (2/14; 13.3%), and complex carcinoma (1/14; 6.7%). Lymph node involvement (histologically confirmed; P = 0.002), clinical stage (P < .001), and death by tumor (P < .001) were variables associated with the development of recurrences and/or metastases during the follow-up period.
Cancer-associated death
Death due to neoplastic disease (13/65) was related to histological grade (P < .001) (Table 4). The following variables were also associated with death due to mammary cancer: age (category 9–11 years; P = 0.01); small versus large breed (9/13; 69.23% of the dogs that died due to a mammary tumor were classified as a large breed; P = 0.02); clinical stage (6/7 animals in stage IV died during the follow-up period; P < .001); WHO tumor size (P = 0.03); histological diagnosis (P = 0.01); HD3 (P < .001); absence of MP (P = 0.04); and lymph node metastasis at time of surgery (P < .001).
Histological Grade and Follow-Up Categorical Variables.
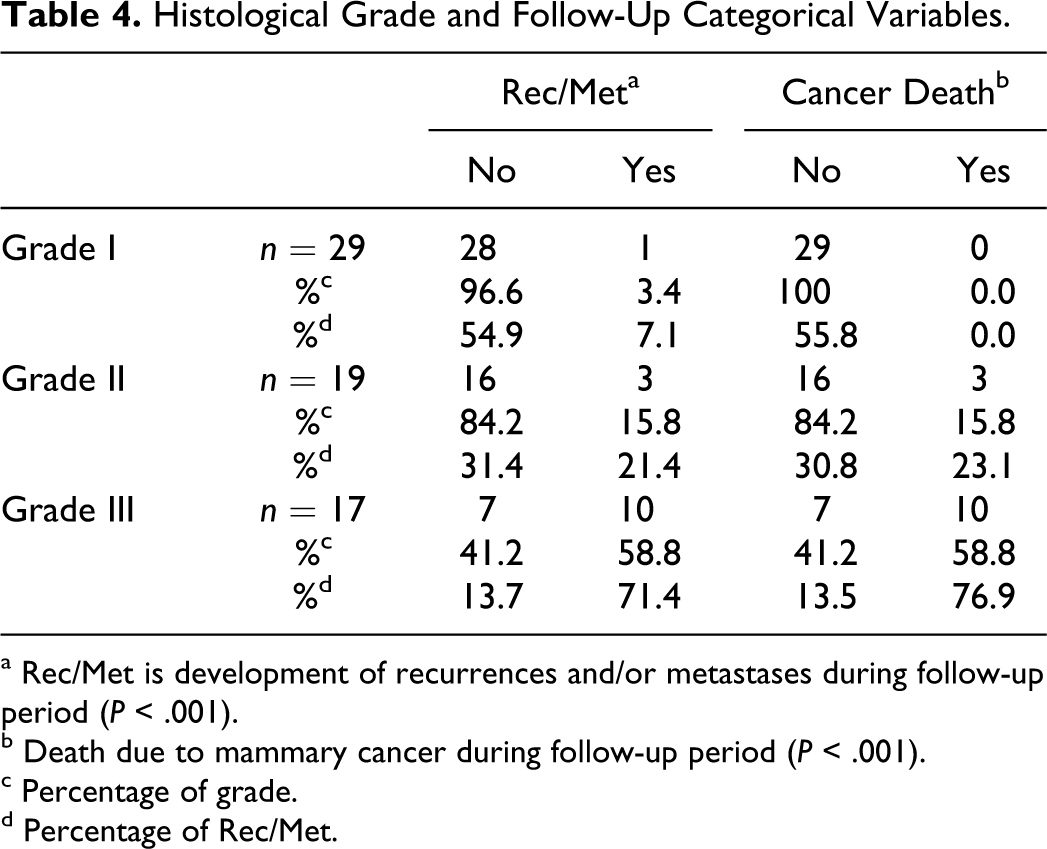
a Rec/Met is development of recurrences and/or metastases during follow-up period (P < .001).
b Death due to mammary cancer during follow-up period (P < .001).
c Percentage of grade.
d Percentage of Rec/Met.
Survival study: Kaplan-Meier survival curves
The Kaplan-Meier survival (log-rank) test of DFS (months) found statistical association of DFS with histological grade (P < .001) as follows: grade I with mean, minimum, and maximum DFS values of 37.29, 35.93, and 38.65 months, respectively; grade II with mean, minimum, and maximum DFS values of 32.68, 27.15, and 38.21 months, respectively; and grade III with mean, minimum, and maximum DFS values of 7.78, 11.39, and 24.17, respectively. Other variables associated with DFS were age (categorical variable; lowest DFS at 9–11 years; P = 0.009), spay status (P = 0.020) (Fig. 7a), clinical stage (stage I vs IV; P < .001), HD3 (group 1 vs 2; P < .001), absence of myoepithelial proliferation (P = 0.03), and lymph node involvement (P < .001). OS was associated with histological grade (P < .001) as follows (Fig. 7b): grade I, all animals were alive at the end of the follow-up period (greater than 38 months); grade II with mean, minimum, and maximum OS values of 32.84, 27.48, and 38.20 months, respectively; and grade III with mean, minimum, and maximum OS values of 20.36, 13.95, and 26.77 months, respectively. Age (lowest OS at 9–11 years; P = 0.006) (Fig. 7c), breed (small breeds had better OS; P = 0.018) (Fig. 7d), clinical stage (I vs II vs IV; P < .001) (Fig. 7e), tumor size (<1, 1–2.9, ≥3 cm; P = 0.008) (Fig. 7f), WHO size (P = 0.015), HD3 (group 1 vs 2; P < .001), and lymph node metastases (P < .001) were also significantly associated with OS.
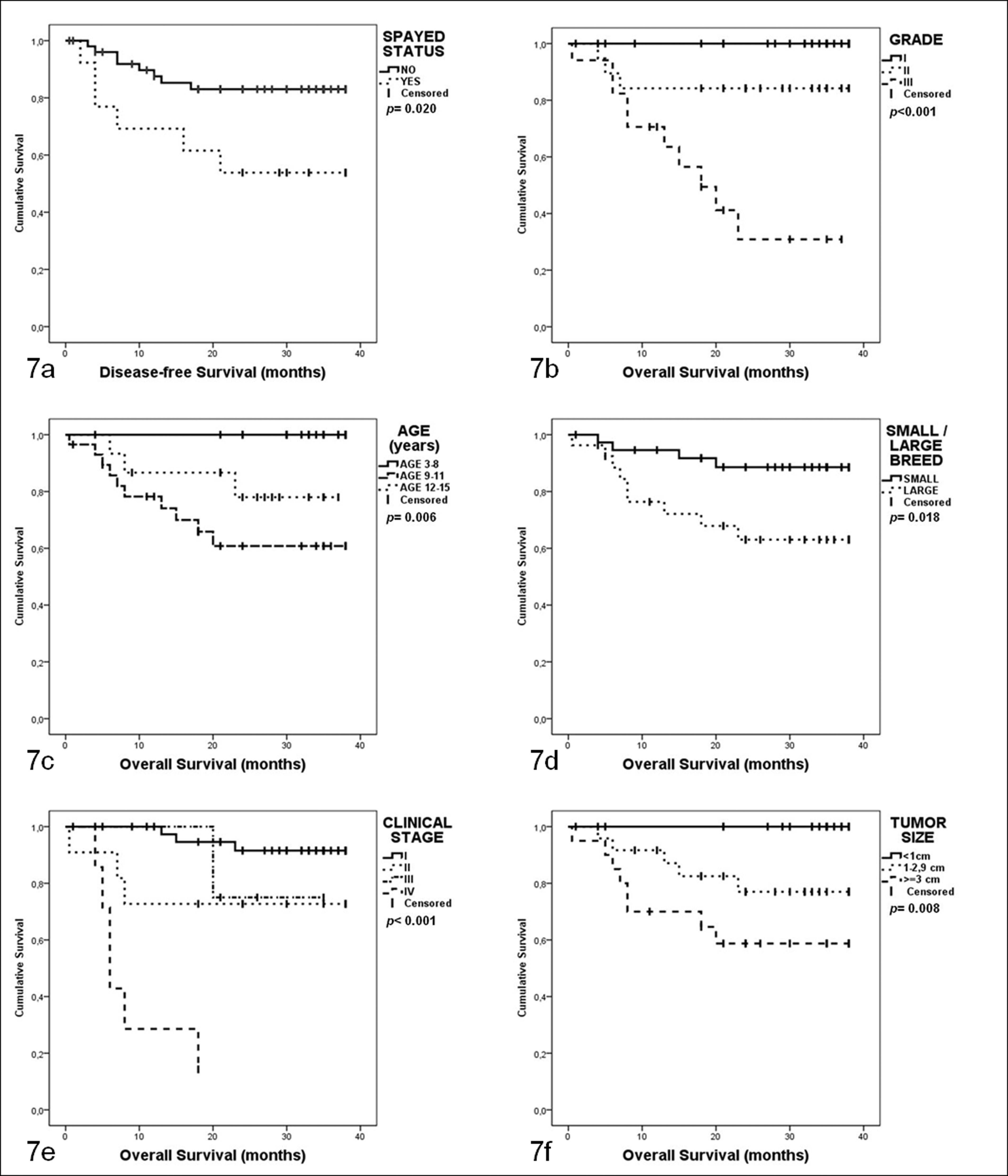
Kaplan-Meier survival curves of 65 female dogs with mammary carcinoma in a prospective study with 2-year follow-up. (a) Disease-free survival: time from surgery to the development of recurrences and/or metastases (event); censored, animals not developing recurrences/metastases or finished follow-up (minimum 28 months). (b–f) Overall survival: time from surgery to death by neoplasia (event) or end of the follow-up; censored: death by other causes, stop the follow-up (> 28 months) or be alive at the end of the study (38 months).
Survival Multivariate Study
To elucidate the independent prognostic value of the grading system in a combined model of possible predictor variables, Cox regressions (forward system) of DFS and OS were performed (Table 2). With DFS as the dependent variable, histological grade, clinical stage, and spay status were selected as independent variables (covariates) at the final step (step 3), thus indicating that they independently provided information about the outcome of the animals. By fitting the histological grade and clinical stage, the spay status of the animal provided the following additional information (P = 0.01): the hazard of developing recurrences and or metastases for spayed females was 4.5 times (Exp(β), Table 2) that of intact females. With OS as the dependent variable, the histological grade and clinical stage were selected as covariates at the final step (step 2). For each step of the Cox regression models, the following formula can be applied to obtain the proportional hazards with the selected independent covariates (Table 2):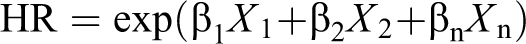
Discussion
The prognostic value of a canine-adapted histological grading system (named by Peña et al 13 ) was evaluated in this study. Although some information has been previously published,3,13,30 this is the first time that the Peña grading system has been published in its entirety including explanations on how to assess the grade of tumors with myoepithelial proliferation (complex and mixed tumors). This grading system is a modification of the widely used human histological grading numeric system known as the Elston and Ellis method or the Nottingham method, 7 which is a modification of Bloom and Richardson’s 2 grading method.
Several prognostic epidemiological, clinical, and histological factors have been proposed to be useful predictors of the outcome of animals with CMCs.19,37 Most of these factors have been evaluated by retrospective studies 37 without rigorous clinical follow-up of the animals. In common diseases, such as CMC, prognostic factors must demonstrate their ability to predict the outcome in well-controlled clinical prospective studies. 39 In contrast, because multiple variables can influence survival and confound associations between the prognostic marker of interest and survival, it is important to account for these variables by using multivariate analysis. 39 However, most of the prognostic factors of CMC evaluated in published studies have been tested in univariate studies. 37 Moreover, many factors lose their prognostic abilities as independent factors when tested in multivariate analyses.
The human breast cancer histological grading system of Elston and Ellis 7 has been applied to CMC by several authors in the last decade.6,8–11,16,18,20–22,27,31–33 Nevertheless, these studies have not evaluated this grading method in myoepithelial proliferation areas even though these studies included complex tumors and carcinosarcomas. In 1 of these studies, 18 this human grading method was found to be associated with survival in a prospective univariate analysis. However, in 2 previous prospective studies conducted by our group,25,28 the same human grading system was ruled out as an independent prognostic factor of CMC by means of multivariate statistical analyses. A system for evaluating the degree of infiltration of CMC 12 has been found to be of prognostic value in prospective multivariate studies.34,35
In the present cohort multivariate study, histological diagnoses of CMC were performed using a recent histological classification 13 with several new subtypes included. This new histological classification has been applied to a retrospective series of 245 canine mammary carcinomas 30 to compare the following 2 histological grading systems of canine mammary tumors: the Misdorp 23 and Peña 3 systems. The percentages of different histological types of tumors and grades were not fully comparable with the results of the present study, which may have been due to the retrospective selection of cases. Furthermore, it is also important to consider differences between the 2 analyzed dog populations as follows: 44.90% of spayed females 30 in the previous study versus 20.0% of spayed females in the present study.
In the present study, 21.53% of the animals had recurrences and/or metastases (4.61% recurrences, 9.23% metastases, and 7.69% recurrences and metastases), and 20.00% of the animals died due to mammary cancer. Other prospective studies have provided similar results showing that the cancer-associated death after 2 years of follow-up is 23.33 (Italy 2002), 34 26.66 (Italy 2010), 35 35.57 (Sweden 1993), 15 40.74 (Portugal 2010), 29 and 44.44% (Spain 1998). 27
Tumor size (<1, 1–2.9, ≥3 cm; P = 0.02) and clinical stage (P = 0.01) were associated with grade. WHO size 26 (T1 < 3 cm, T2 = 3–5 cm, and T3 > 5 cm) was not significantly (P = 0.06) related to grade, which agreed with a previous report using the Nottingham method to study CMC. 18 Histological diagnosis and grade were significantly associated in the present study (P < .01) and in a previous prospective study using the Nottingham method. 18 This association may be partially attributed to the evaluation of tubular formation, which is a fundamental part of both histological assessments (diagnosis and grade). However, tumors with the same pattern of tubular formation may be graded differently. Solid carcinomas and tumors without tubular formations were graded as grades II and III, respectively, which was an interesting result because a poor prognosis is generally attributed to solid carcinomas. 23 In addition to the histopathological diagnosis, a more accurate prognosis can be obtained by applying this grading system, which is able to separate solid carcinomas with high (III) and intermediate (II) grades. Grade II and grade III solid carcinomas were identified in a retrospective study using the same grading system. 30 The same occurred with comedocarcinomas: all comedocarcinomas (n = 3; a new type included in the histological classification 13 ) were classified as grade III. However, according to the results of a previous retrospective study, 30 comedocarcinomas can be classified as grade II or III. In agreement with the previous study using the same grading system, 30 regional lymph node metastasis at the time of diagnosis (surgical sample; hematoxylin and eosin) was significantly associated with grade III. In the previous study, 30 the Peña grading system was more accurate than the Misdorp system in detecting lymph node metastases at time of surgery. 23 However, the human Nottingham grading method applied to CMC is less effective in detecting lymph node metastases at the time of diagnosis. 18
In the present study, the univariate analyses revealed several variables being related to the prognostic variables (end points of follow-up and survival times) as follows: histological grade, age (9–11 years), clinical stage (IV), HD3 (group 2), and lymph node metastases at time of diagnosis. Some of these tumor or patient characteristics (age, clinical stage, and lymph node affectation) have been previously associated with prognosis in univariate or multivariate retrospective or prospective studies. 37 According to our results, myoepithelial proliferation has been proposed to have prognostic value in univariate studies. 19 Spayed dogs were associated in univariate analyses with the development of recurrences and/or metastases and shorter DFS periods. Moreover, the univariate analysis indicated that large breed animals died of mammary cancer more frequently (shorter OS) than did small breed dogs. The better prognosis for small breeds has been previously reported in a retrospective multivariate survival analysis, 17 and this prognosis is attributed to a lower malignancy of mammary tumors in small breed animals. However, the histological type, grade, and other characteristics of malignancy were not significantly associated with the size of the breed in the present study. The possible explanation for the longer survival of small breed dogs may be unrelated to the neoplastic disease itself and instead may be associated with the longer life span of small breed dogs compared with large breed dogs. 14
The size of the tumor according to the WHO size categories 26 has been proposed as a prognostic indicator for canine malignant mammary tumors in retrospective multivariate analyses5,28: tumors greater than 3 cm (univariate analysis) 28 and greater than 5 cm (multivariate analysis) 5 have been found to be associated with lower survival rates. In the present study, when the WHO size system for mammary tumors was applied, most of the neoplasms were classified as T1 (n = 45) and T2 (n = 15), and only 5 neoplasm were classified as T3. The WHO size categorization was related to cancer death and OS. The other studied tumor size categorical variable (<1, 1–2.9, and ≥3 cm) was related to OS. Combining the results of the univariate analyses of both size systems, a subdivision of the modified WHO clinical stage I (T1N0M0) 19 into stage IA (<1 cm) and stage IB (1–3 cm) could be proposed for improved prognosis. Although the present study was not comparable in terms of method used, this finding was reinforced by the results reported by Sorenmo et al, 36 who reported that only 1 of 97 mammary tumors less than 1 cm in diameter is diagnosed as malignant.
In the present study, the multivariate analyses of DFS and OS selected the clinical stage and histological grade as the 2 covariates for the dependent variables (DFS and OS) together with the spay status of the animal as a covariate for DFS. The transition from clinical stage I to IV was considered prognostic in the multivariate analyses of DFS and OS. The switch from histological grade I to III was considered prognostic in the multivariate analyses of DFS and OS. To our knowledge, this is the first time that the spay status and histological grading system used in the present study have been reported as prognostic factors in a prospective study. In a retrospective multivariate study on 79 dogs, 5 the spay status was shown to be significantly associated with low survival after surgery (follow-up of 2 years). The results regarding differences in prognosis between intact and spayed female dogs with mammary cancer may be attributed to a lower content of estrogen receptors in mammary tumors detected in spayed females. 25 Comparative studies on CMC performed in different countries should take into account the proportion of spayed females among the selected dog population and the possible differences in the prognoses. The relation between ovariohysterectomy at the time of mastectomy of CMC and prognosis was not analyzed in the present study.
As a final consideration, we would like to note that the grading system cannot substitute for the histological diagnosis but instead complements the histological diagnosis, which was demonstrated to have prognostic value in this prospective study in univariate analyses despite the low number of cases in some of the types studied. For validation of histological type as an independent prognostic indicator in multivariate analyses, it is necessary to use a large series of animals recruited prospectively over several years. In some of the most infrequent histological types with unknown prognoses, grouping different tumor entities by grade for statistical purposes would be acceptable to assess prognosis. Finally, it is important to remember that the histological variability of CMC can reflect different oncogenic mechanisms, thereby suggesting that therapeutic approaches may vary in the future for some specific histological types of carcinomas.
The goal of prognostic studies is to identify markers that are significantly and independently associated with a clinical outcome that could ideally be applied to a clinical or diagnostic setting. According to the present results, this modification of the Elston and Ellis numeric grading system adapted to canine mammary cancer is a useful tool for predicting prognosis, facilitates the clinical interpretation of complicated histological diagnoses, and offers pathologists uniform criteria for the assessment of grading myoepithelial proliferation areas and mixed tumors.
Footnotes
Acknowledgements
We are grateful to the staff of the Veterinary Pathology Service and Oncology Service at the Veterinary Teaching Hospital of the Complutense University of Madrid for their support and to Pedro Aranda for his histological technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by the Spanish Ministry of Science and Education (research project SAF2009-10572 and PhD fellowships).
