Abstract
Aortic rupture in horses is a rare condition. Although it is relatively common in the Friesian breed, only limited histopathologic information is available. Twenty Friesian horses (1–10 years old) were diagnosed with aortic rupture by postmortem examination. Ruptured aortic walls were analyzed with histology and immunohistochemistry. Based on the histologic and immunohistochemical findings, these cases were divided into 3 groups: acute (n = 4, 20%), subacute (n = 8, 40%), and chronic (n = 8, 40%). Features common to samples from horses in all groups included accumulation of mucoid material; disorganization and fragmentation of the elastic laminae; aortic medial smooth muscle hypertrophy; and medial necrosis of varying degrees, ranging from mild and patchy in the acute cases to severe midzonal necrosis in the chronic cases. Inflammation, most likely secondary to medial necrosis, varied from predominantly neutrophilic infiltrates in the media and periadventitial tissue in the acute group to the presence of mainly hemosiderophages in the periadventitial tissue in the chronic group. Medial fibrosis with aberrant collagen morphology was seen in the subacute group and, more commonly, in the chronic group. Only minimal changes were seen in the aortic vasa vasorum. Smooth muscle hypertrophy and accumulation of mucoid material were not related to the age of the lesions. The findings of this study suggest that a connective tissue disorder affecting elastin or collagen in the aortic media is potentially the underlying cause of aortic rupture in Friesian horses.
Keywords
More than 40 cases of aortic rupture in Friesian horses have been reported to date. 38,51 These ruptures occurred spontaneously, in the absence of trauma, infectious disease, or iatrogenic injury. There was no sex predilection, and the median age of affected horses was 4 years, ranging from 1 to 20 years. Ruptures occurred consistently in the thoracic aorta near the ligamentum arteriosum. Frequent findings at postmortem examination included a concurrent circumferential cuff of perivascular hemorrhage (periaortic hematoma) and aortopulmonary fistulation, which is well described in the Friesian breed. 38
Aortic rupture in non-Friesian horse breeds is rare and mainly affects breeding stallions, in which it manifests as acute death. 42 In these cases, the rupture appears near the sinuses of Valsalva, and the macroscopic lesions and clinical signs differ from those seen in Friesian horses with aortic rupture.
An aneurysm is a localized abnormal dilation of any vessel. True aneurysms are composed of all or most layers of the intact vessel wall. False aneurysms, also called pseudoaneurysms, result from rupture of an artery or aneurysm, with disruption of all 3 layers of the arterial wall and communication with the arterial lumen. 27,52 Aortic aneurysms are rare in horses and occur mainly near the aortic arch. 40 In some cases, Strongylus vulgaris arteritis can cause aneurysm formation in the abdominal or thoracic aorta. 45
Spontaneous aortic rupture in humans is a multifactorial disease 1 and a relatively uncommon cause of death. 13,19,29 Histopathologic findings include abnormalities of the cellular and matrix constituents of the aortic media. Aortic rupture in humans occurs mostly in the abdominal portion of the vessel 10 and is frequently associated with atherosclerosis. 12 Thoracic aortic rupture is uncommon and predominantly linked to hereditary diseases. 16,23,30 In abdominal aortic aneurysms, the structure and amount of the matrix proteins are abnormal, there being decreased elastin and increased collagen. 5,31 Both the vasa vasorum and the increased synthesis of matrix metalloproteinases by smooth muscle cells have been implicated in the progression of abdominal aortic aneurysms. 18,37 Additionally, inflammatory processes have been shown to play an important role in the development of this disease. These can be divided into noninfectious causes, such as atherosclerosis, 1,49 and infectious causes, such as mycotic aneurysms of the aorta. 36 In thoracic aortic rupture, alterations in elastin (Marfan syndrome), fibrillin (Ehlers-Danlos syndrome type IV), and proteoglycans (biglycan gene deficiency) have been determined. 16,23,30 In rare cases, a functional connection occurs between the ascending aorta and the pulmonary artery. Such an aortopulmonary fistula is a diagnostically challenging condition. 20 It occurs most frequently as a result of erosion and/or rupture of a chronic process or pseudoaneurysm of the aorta. 26,46
Histologic examination of the ruptured aortic wall has proved to be a useful tool for investigation of the underlying causes of aortic rupture in humans. 3,44 To date, only 3 cases of aortic rupture with aortopulmonary fistulation in Friesians have been histologically examined and described in the literature. 51 In 2 horses, the lesion was described as scattered medial necrosis throughout the wall, surrounded by neutrophils and granulation tissue; in the third horse, no inflammatory reaction was observed. Many vasa vasora with intimal thickening and/or medial fibrosis were seen in the aortic adventitia, and this may predispose to aortic medial necrosis and rupture. 51 The purpose of the present study was to describe the histologic lesions in a larger series of Friesian horses with aortic rupture to gain better insight into the underlying mechanisms of this unique disease.
Methods
Animals
Twenty Friesian horses were included in this study (1-10 years old; 14 mares, 3 geldings, 2 stallions, and 1 animal of unknown sex); gross lesions in 7 of these animals have been described previously. 38 All horses were diagnosed with aortic rupture by means of postmortem examination over a period of 14 years (1997-2011) at Wolvega Equine Hospital (n = 6); the Faculty of Veterinary Medicine, Ghent University, Belgium (n = 5); or the Faculty of Veterinary Medicine of Utrecht University, the Netherlands (n = 9). Six horses were found dead or showed acute clinical signs in the few hours preceding death. However, the majority (n = 14) showed signs such as fever, tachycardia, and colic in the days, weeks, or even months before euthanasia. The animals, clinical signs, and gross lesions are presented in Table 1.
Histopathology
Tissue samples were taken at the level of the aortic rupture, fixed in phosphate-buffered formalin, routinely embedded in paraffin wax, and cut into 4-μm-thick sections. Periaortic hematomas, pseudoaneurysms, and arterial dissections were also analyzed. Sections were stained with hematoxylin and eosin, Van Gieson’s stain (Klinipath 64089, Duiven, Netherlands) to visualize possible collagen deposition of the media, Alcian Blue (Sigma A4045-25G, Zwijndrecht, the Netherlands) for detection of accumulation of mucoid material, and Von Kossa stain for demonstration of mineralization.
Immunohistochemistry
The immunohistochemical antibodies were used according to manufacturers’ instructions. Presence of smooth muscle was demonstrated with mouse anti-smooth muscle actin (Biogenex, Fremont, CA) as the primary antibody and horse anti-mouse/biotin (Vector Laboratories, Peterborough, UK) as the secondary antibody. Vasa vasora were identified by visualization of factor VIII–related antigen with rabbit anti-factor VIII (Dako, Glostrup, Denmark) as primary antibody and goat anti-rabbit IgG (Vector Laboratories) as secondary antibody. Additionally, factor VIII–related antigen was used to determine the nature of the lining of the pseudoaneurysms and dissections. Lymphocytes were analyzed with a polyclonal rabbit anti-CD3 antibody (Dako) that labels human T lymphocytes (both helper and cytotoxic T lymphocytes), a polyclonal rabbit anti-CD20 antibody that labels human B-lymphocytes (Thermo Fisher Scientific, Erembodegem, België), and a monoclonal mouse antibody that labels reactive and tissue macrophages (MAC387, Abcam, Cambridge, UK). Aortic elastin was visualized with a monoclonal anti-elastin antibody BA-4 (Leica Biosystems, Diegem, Belgium). A standard avidin–biotin complex method with diaminobenzidine as chromogen was used for visualization (Envision, Dako). Negative controls were prepared from serial sections in which the primary antibody was omitted and replaced by dilution buffer. The specificity of the primary antibodies was validated with recognized positive control tissues.
Scoring of Lesions
Histologic scoring of necrosis of the media, accumulation of mucoid material, altered smooth muscle orientation, elastin fragmentation, and fibrosis was based on protocols described previously. 43 This semiquantitative grading system uses a scale from 0 (no changes) to 3 (severe changes). The smooth muscle cells in the media were also assessed for the presence of hypertrophy.
The presence or absence of fibrin and mineralization was assessed. The degree of medial and adventitial inflammation (density of inflammatory cells) was scored with grade 0 representing no inflammation and grade 3, severe inflammation. 22 Finally, the numbers of vessels in the media and adventitia were assessed by counting the number of vasa vasora in 5 randomly chosen fields irrespective of the size of the vessel.
Results
Major findings are summarized in Supplementary Table 1 (available online at http://vet.sagepub.com/supplemental).
Macroscopic Findings
Macroscopic lesions in the present series (including those in the horses included in the previously mentioned study) were similar to those previously described. In summary, all Friesians had an aortic rupture just proximal to the ligamentum arteriosum, and 6 horses showed a hemothorax. A periaortic hematoma was present in 4 cases. Aortopulmonary fistulation was present in 13 animals and was in most cases associated with 1 or more pseudoaneurysms (extensive encapsulated perivascular hematoma, retaining communication with the aortic lumen) (Fig. 1). Pulmonary artery ruptures were transverse, located at the level of the ligamentum arteriosum, and varied from 2 to 7 cm in length. In 4 cases, a dissection of the aorta was present, and in 1 case, there was an additional dissection of the pulmonary artery wall. Dissection took the form of a longitudinal split in the midmedia, creating a “false” lumen running distally over a length of up to 15 cm.
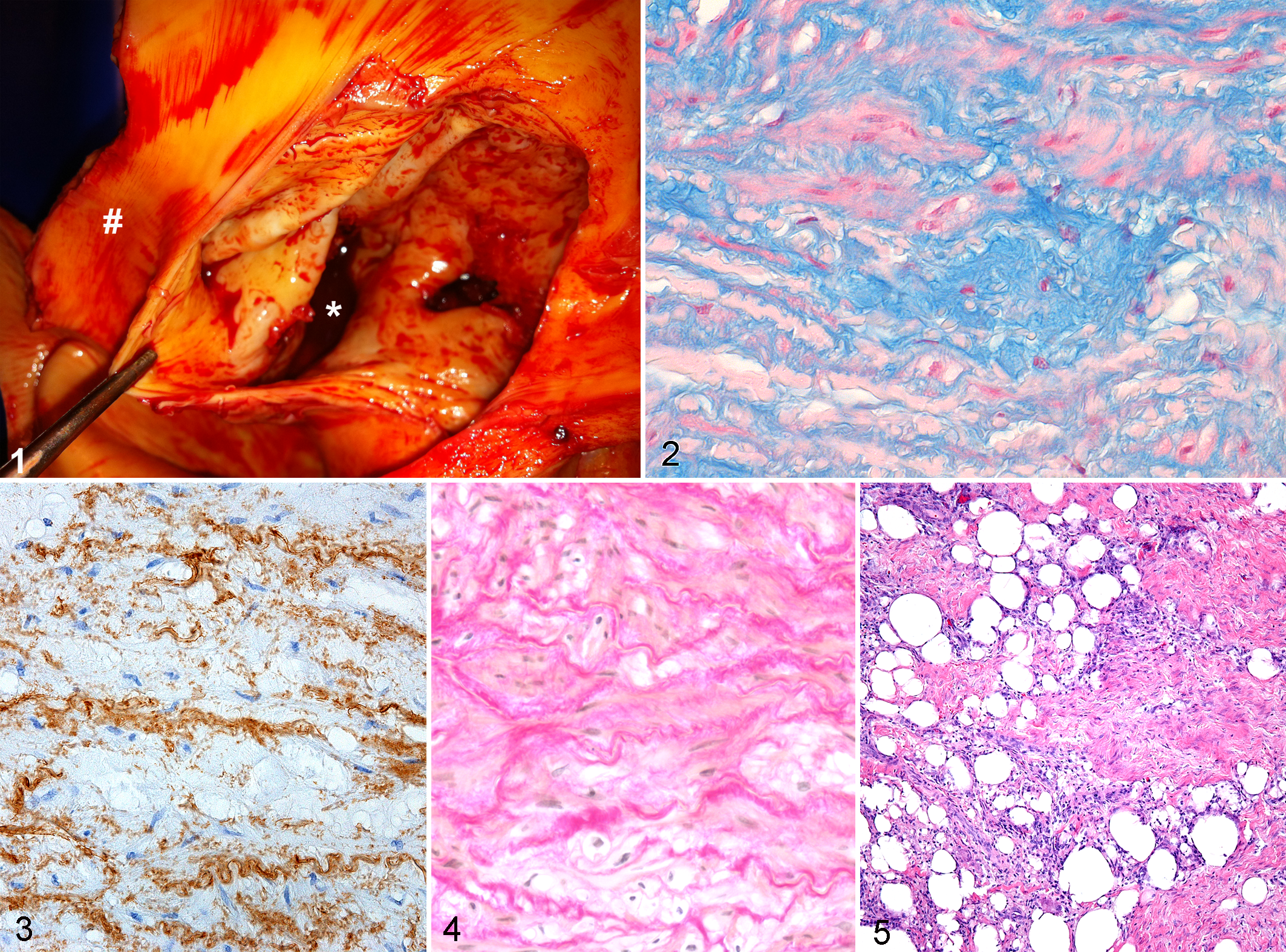
Histologic Findings of Ruptured Aortas
In 4 animals (acute group, Nos. 1–4), aortic lesions were histologically classified as acute based on the presence of fibrin clots in the hematoma and neutrophils as the dominant infiltrative cell type (Supplementary Table 1). Two of these horses had hemothorax; the other 2 had periaortic hematomas, and 1 of these had an aortopulmonary fistula. All horses in the acute group had mild (n = 3) to moderate (n = 1) (grades 1 and 2) necrosis of the media, which was mainly patchy. Small numbers (grade 1) of B and T lymphocytes, plasma cells, and macrophages were seen both in the media and in the periadventitial tissue, but neutrophils were the predominant inflammatory cell type. Accumulation of mucoid material (grades 1–3) (Fig. 2) and disorganization and fragmentation (grades 1 and 2) of the elastic laminae (Fig. 3) were seen in the aortic media of all horses in the acute group. Medial fibrosis was not present. Mild mineralization of the midzone of the media was present in a single case and occurred in association with mild medial necrosis. Smooth muscle hypertrophy was present in all affected Friesians, but no animals in this group showed altered smooth muscle orientation. The number of vasa vasora in the media ranged from 26 to 78 (mean, 45), and 2 of these showed moderate intimal thickening by subendothelial smooth muscle cells without medial fibrosis. The number of vasa vasora in the adventitia was determined in only a single case (17 vasa vasora) but could not be detected in others due to the extensive hemorrhage or the absence of adventitia.
Subacute stages of the lesions were identified in 8 horses (subacute group, Nos. 5–12) on the basis of the presence of immature fibroblasts in the adventitia and periadventitial tissue and infiltration of mononuclear cells and hemosiderin-laden macrophages (Supplementary Table 1). Five of these horses demonstrated a periaortic hematoma, which was associated with hemothorax in 4 cases and with an aortopulmonary fistula in 1 case. The other 3 horses showed an aortopulmonary fistula combined with an aortic dissection. Fibrin clots were observed in the hematoma in 6 cases. Medial necrosis was seen in all cases with subacute lesions and varied from mild in 3 cases to severe (grades 1–3) in 5 cases. In all cases, there was infiltration of the adventitia and periadventitial tissue predominantly by hemosiderin-laden macrophages (grades 1–3) was present in all cases. In 4 cases, these macrophages were accompanied by neutrophils, and in 2 cases, they were admixed with several eosinophils. Accumulation of mucoid material (grades 1–3) and disorganization and fragmentation of the elastic laminae (grades 1–3) were seen in the aortic media of all horses in the subacute group. In 2 cases, mild to moderate (grades 1 and 2) medial fibrosis was observed. Increased fibrosis was typically characterized by disorganization, fragmentation, or clumping of fibers (Fig. 4). In 1 horse, there was multifocal mineralization of well-defined areas of medial smooth muscle cell necrosis and extensive elastin fragmentation. Smooth muscle hypertrophy was present in all affected Friesians, but no animals in the subacute group showed changes in smooth muscle orientation. The number of vasa vasora in the media ranged from 21 to 159 (mean, 70), and a single case showed medial fibrosis of vasa vasorum in the aortic media. In the adventitia, the number of vasa vasora ranged from 37 to 276 (mean, 149), and a single case also showed moderate intimal thickening with subendothelial smooth muscle cells and proteoglycans.
The remaining 8 animals (chronic group, Nos. 13–20) showed chronic lesions in the aorta. These were similar to the lesions seen in the subacute cases but additionally showed fibrosis in the adventitia and periadventitial tissue (Supplementary Table 1). All cases with chronic lesions showed aortopulmonary fistulation combined with a pseudoaneurysm, and all cases had moderate (n = 3) to severe (n = 5) necrosis (grades 2 and 3) of the midzone of the media. In all cases, a mild infiltration (grade 1) of B and T lymphocytes and plasma cells was found in the media and/or adventitia. In 5 cases, there was infiltration of the periadventitial tissue, mainly by hemosiderin laden macrophages, sometimes admixed with neutrophils. Accumulation of mucoid material (grades 2 and 3) and disorganization and fragmentation (grades 1–3) of the elastic laminae were seen in the aortic media of all horses in the chronic group. Mild to moderate (grades 1 and 2) medial fibrosis was observed in 7 cases and was similar to the morphologically abnormal fibrosis described in the subacute cases. In 5 cases, there was multifocal mineralization of well-defined areas of medial smooth muscle cell necrosis with extensive elastin fragmentation. Smooth muscle hypertrophy was present in all affected Friesians, but no animals in the chronic group showed altered smooth muscle orientation. Moderate to marked intimal thickening of the vasa vasorum of 2 to 5 vessels was seen in 3 of the chronic cases, and 2 of these were located in the adventitia. Mild medial fibrosis of the vasa vasorum was present in the media (n = 2) and/or adventitia (n = 3). The number of vasa vasora in the media ranged from 26 to 133 (mean, 59) and in the adventitia from 63 to 434 (mean, 163).
Pseudoaneurysms
Pseudoaneurysms were present in 1 of the acute cases, 4 of the subacute cases, and all of the chronic cases and in all Friesians with chronic histologic lesions. The adventitial side of the wall of the pseudoaneurysm was characterized by a thick layer of spindle-shaped cells (fibroblasts) that formed streams and bundles in various directions in a myxomatous matrix. Whorls were frequently formed, and multifocal small- to medium-sized blood vessels were present. The center of the wall was composed of a layer of smooth muscle cells that were longitudinally oriented. The smooth muscle cells were multifocally mixed with a moderate amount of collagen fibers that showed various orientations. Close to the intimal side of the wall of the pseudoaneurysm, these smooth muscle cells were replaced in large areas by moderate amounts of collagen, high numbers of small- to large-sized blood vessels with occasionally marked intimal proliferation, high numbers of B and T lymphocytes, multinucleated giant cells, a moderate amount of fat, low numbers of viable and degenerate neutrophils, plasma cells, and a few cholesterol clefts (Fig. 5). The luminal side was lined by well-differentiated endothelial cells, confirmed by staining with factor VIII–related antigen. Pseudoaneurysms contained large blood clots with high numbers of degenerated and viable neutrophils and low numbers of eosinophils and multinucleated giant cells.
Periaortic Hematomas
Periaortic hematomas were observed in 2, 5, and 1 animals demonstrating acute, subacute, and chronic histologic lesions, respectively. The periphery was bordered by a moderately thick layer of longitudinally arranged thick collagen fibers. In some cases, the adventitia showed large areas of fragmented (degenerate) collagen, hemorrhage, and an inflammatory infiltrate consisting of large numbers of lymphocytes and plasma cells. In a few cases, the adventitia showed multifocal proliferation of randomly aligned bundles of fibroblasts admixed with fat tissue. Multifocal areas with large numbers of neutrophils and a moderate amount of necrotic debris admixed with small deposits of hematoidin were also present.
Aortic Dissections
In 4 cases (3 subacute and 1 chronic), the aortic wall was dissected within the media close to the adventitia with the formation of a pseudolumen, which was walled off by large numbers of smooth muscle cells, a large amount of collagen, and moderate numbers of medium- to large-sized vessels. The smooth muscle cells were longitudinally aligned parallel to the lumen, but this alignment became less organized further from the lumen. The variable arrangement of collagen fibers became increasingly loose and disorganized toward the periphery. Both sides of the dissection were lined by endothelial cells (positive factor VIII–related antigen immunostaining).
Discussion
The histologic lesions described in the present study correlated well with the gross lesions and clinical signs. All horses in the acute group suffered from acute death or presented with acute signs, whereas all horses of the chronic group had shown clinical signs over a prolonged period due to aortopulmonary fistulation. To date, there is only a single report in the literature describing the histologic features of Friesian horses (n = 3) with aortic rupture and aortopulmonary fistulation. These animals had severe progressive symptoms of cardiac distress, died within a few hours to 8 days after referral, and are comparable to the chronic group in the present study. These authors reported the histologically detected alterations in the vasa vasorum as a possible cause of aortic medial necrosis. Furthermore, the scar of the former site of the ductus arteriosus was considered to be predisposed to rupture and fistulation. 51
In this study, aortic medial necrosis was observed in all horses. By weakening the aortic wall, this may predispose affected vessels to dissection and spontaneous rupture. 33 Medial necrosis has also been suggested as a predisposing lesion in non-Friesian horses dying from acute rupture of the aorta at the sinuses of Valsalva. 42 However, chronic histologic lesions as seen in the Friesian horses, such as fibrosis of the adventitia and periadventitial tissue with infiltration of hemosiderin-laden macrophages, have not been described.
Nearly all affected Friesian horses (85%) showed laminar medial necrosis as a histologically evident laminar pink band with loss of nuclei in the midmedia. The middle part of the media is situated between the area supplied by the vasa vasorum and the area nourished by the intraluminal blood and is therefore most prone to ischemic damage. 53 For the same reason, aortic dissection usually occurs at the junction of the middle and outer third of the media, as seen in the Friesian horses in this study. 54
The laminar medial necrosis of the aorta in the Friesian horse could be attributed to ischemia. A high number of vasa vasora with intimal thickening and/or medial fibrosis in the media and adventitia of the aorta was described in 3 Friesian horses with aortic rupture. 51 The lumen of the vasa vasorum was completely obliterated, and this was suggested to cause hypoxia or anoxia of the aortic wall, resulting in local circulatory compromise, necrosis, and, finally, wall rupture. In the present study, only 5 of the 20 cases demonstrated mild to moderate intimal thickening of the vasa vasorum without complete occlusion. Medial fibrosis in 2 to 5 vessels of the vasa vasorum was infrequently present in subacute and chronic cases. There is a high discrepancy between our findings and those previously reported, therefore making ischemic damage as an underlying cause unlikely. The alterations observed in the vasa vasorum in the present study are believed to be a secondary phenomenon related to blood flow changes, such as severe circulatory disturbances caused by ruptured aortas or pseudoaneurysms.
Another cause of medial necrosis is connective tissue abnormalities that, in humans, are often related to gene dysfunctions. In such cases, cystic medial necrosis is often observed (degeneration of elastic fibers and collagen in the media of the aorta and subsequent accumulation of mucoid material). 55 Accumulation of mucoid material in the tunica media resembling cystic medial necrosis was present in all cases reported here, suggesting that a primary connective tissue disorder may contribute to aortic rupture in Friesian horses. There is controversy about the significance of cystic medial necrosis in human aortic rupture. Some authors have suggested that it is primarily an aging process, 43 but other studies have indicated that cystic medial necrosis is an expression of metabolic activity rather than the result of a degenerative process. 9,43 In this study, the role of aging is questionable, as the majority of animals were less than 7 years old. Furthermore, cystic medial necrosis has been observed in nondiseased horses. 41 Therefore, it is possible that cystic medial necrosis is not a primary feature of the disease.
Some degree of medial necrosis is related to aging and is typically observed as small focal defects in the human aorta. 7 In a minority of affected Friesian horses, a patchy distribution of necrotic medial foci was observed and could be attributed to age. However, 1 of these horses was only 4 years old, suggesting that age may not be a factor in the development of this lesion. In addition, this patchy medial necrosis was seen only in acute and subacute cases, indicating that this lesion may represent an early stage in the disease.
Finally, medial necrosis can be initiated by mediators from structural elements of the media. This mechanism is seen in abdominal aortic aneurysms in humans, where vascular smooth muscle cells promote matrix metalloproteinase release resulting in medial damage. 24 In such cases, inflammation is a prominent and consistent secondary finding characterized by extensive infiltrates of B and T lymphocytes, plasma cells, and dispersed macrophages in the adventitia. 17 Since smooth muscle hypertrophy, fibrosis, and the infiltration of lymphocytes and macrophages were observed in the affected Friesian horses, involvement of proteinases cannot be excluded.
Smooth muscle cell hypertrophy of the human aorta, often accompanied with protein deposition, is associated with hypertension and aberrant blood flow, creating cyclic stretch and shear stresses. 4,15,34 In bovine Marfan syndrome, smooth muscle hypertrophy is considered to be a secondary reaction, replacing damaged elastic fibers of the aortic wall. 39 The extracellular matrix can play a pivotal role in the regulation of arterial vascular smooth muscle cell differentiation and proliferation. 4,25,28 In the present study, all Friesians showed aortic medial smooth muscle cell hypertrophy, accumulation of mucoid material, and disorganization of the elastin. It is therefore possible that primary changes in the extracellular matrix with subsequent aberrant stretching may have caused smooth muscle hypertrophy in these cases.
In about half the chronically affected Friesian horses, abnormal collagen deposition was seen predominantly in the midzone of the media. As seen in humans, this collagen deposition may reflect a reactive strengthening of the vessel wall as a reaction to the abnormal hemodynamic changes caused by the pseudoaneurysm and/or aortopulmonary fistula. 1,8 Additionally, as fibrosis was most obvious in the chronic group, the collagen deposition may be a secondary reaction to chronic injury. Possible triggers for repair in vascular tissues are necrosis of smooth muscle fibers, disruption of elastic laminae, or loss of endothelial continuity. 35 However, the collagen deposition observed in the affected Friesian horses was clumped and disorganized, which is not a common characteristic of fibrosis.
Disorganization and fragmentation of elastic laminae in the media were seen in all horses and could be due to proteolytic activity caused by the damaged extracellular matrix at the site of the rupture. In the past, elastin fragmentation was interpreted as being secondary to smooth muscle cell necrosis. 14 However, elastin degeneration was present in both chronic and acute cases, which may suggest an underlying primary connective tissue disorder in the Friesians. Well-known causes of elastin defects include Williams syndrome, 11 “cutix laxa,” 48 and human and bovine Marfan syndromes. 2,39 In these syndromes, vascular anomalies are accompanied by alterations in other organs. Since no other abnormalities were observed, a syndromal elastin defect seems unlikely in the Friesian horses.
In humans, aortopulmonary fistulation is rare and occurs most frequently as a complication of aortic pseudoaneurysm. The latter has been associated with a true aneurysm, aortitis, atherosclerosis, arteriosclerosis, aortic dissections, or traumatic aortic tear or as a postsurgical event. 6 Occasionally, a foreign body has been the inciting factor. 20 In the present study, there were no indications for infectious agents, atherosclerosis, or trauma as the primary cause. In one human case, a small aortic aneurysm extended into the pulmonary artery, and this was believed to be due to pressure and the close proximity of the pulmonary artery and the aorta. 21 In Friesian horses, the location of the aortopulmonary fistulation was assumed to be due to tension created by the ligamentum arteriosum on the previously damaged walls of aorta and pulmonary trunk. 51 However, in this study, an ongoing chronic process with weakening of the aortic wall and pseudoaneurysm formation toward the pulmonary artery is believed to be the primary cause in at least some of the cases. Nevertheless, as there are several cases with a fistula in the absence of a pseudoaneurysm, rupture of the pulmonary artery may be caused by an intrinsic defect.
Histologic lesions seen in affected Friesian horses differ from non-Friesian horses suffering from abdominal aneurysms. In 45% of the Strongylus vulgaris–mediated equine arteritis, one or more larvae can be found, and this finding is consistently associated with thrombosis. 32 The current study showed no true aneurysms, and there were no macroscopic or histologic indications for parasite migration in the affected Friesian horses. Rupture of the uterine artery is a common condition in predominantly aged multiparous mares, and copper deficiency is presumed to be a predisposing factor. 47 Peripartum hemorrhage in these mares is characterized by atrophy of smooth muscle cells with fibrosis of the tunica media and disruption and/or calcification of the internal elastic lamina. 50 In this study, the median age of the affected Friesians was only 4 years old, suggesting a different pathogenesis.
The finding of a shared set of clinical signs and gross and histologic findings in all cases of aortic rupture reported here supports the recognition of this syndrome as a distinct clinical entity unique to this breed. It is conceivable that an underlying genetic defect of the connective tissue in the aortic media predisposes these animals to aortic rupture, dissection, and aortopulmonary fistulation at an anatomically and hemodynamically predisposed site. The Friesian horse population appears to be the only animal species in which aortopulmonary fistulation is regularly encountered. In light of these findings, biochemical and ultrastructural examination of the extracellular matrix in the aorta of affected Friesians may be useful in furthering our understanding of the pathogenesis of this syndrome.
Footnotes
Acknowledgements
We thank Johan van Amerongen, Louis van den Boom, and the assistants of the histology laboratory of the Veterinary Pathology Diagnostic Centre for their technical assistance. Many thanks to Christian Puttevils, Delphine Ameye, Sarah Loomans, and Astra Dhanijns for technical assistance. We also thank Pascale Drijfhout, Siebren Boerma, Roel van Nieuwstad, Katrien Palmers, and Tresemiek Picavet and the referring equine practitioners for their collaboration and for providing us with most of the clinical material. In addition, we thank R. Thomas and R. Keesler (American College of Veterinary Pathologists diplomate) for checking the English language.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Koninklijke Vereniging “Het Friesch Paarden-Stamboek” (Royal Friesian Horse Studbook).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
