Abstract
Friesian horses have a perceived high rate of congenital or hereditary diseases, including megaesophagus, that may lead to choke and death. A retrospective study was performed to determine the prevalence and pathologic characteristics of esophageal disease in 852 horses, including 17 Friesians, that had been necropsied over a 6-year period at the Diagnostic Center for Population and Animal Health. Forty-two horses had grossly described esophageal lesions (25 muscular hypertrophy, 7 hemorrhage, 6 megaesophagus, 4 erosion/ulceration, 3 obstruction, 2 tears, 2 secondary neoplasms, 2 lymphoid patches, 1 thin wall, 1 esophagitis). Some of these lesions occurred concurrently in the same horse. Ten of these horses died or were euthanatized because of severe esophageal disease (6 megaesophagus causing tears in 2 horses, 3 esophageal obstruction with food bolus, and 1 esophagitis). All 6 horses with megaesophagus were Friesians. No cause for megaesophagus was noted in the necropsy reports; however, 5 of these 6 Friesians had marked caudal esophageal muscular hypertrophy (wall thickness: 1.9 ± 0.3 cm). Microscopic review of the esophagus of these Friesians confirmed smooth muscle hypertrophy, with no obvious fibrosis, degeneration, or loss of myenteric plexi. Unlike the Friesians, the 4 non-Friesian horses with severe esophageal disease had esophageal obstruction with an intraluminal food bolus or severe esophagitis. None had caudal esophageal muscular hypertrophy. It is concluded that in comparison to other horse breeds, Friesians have a higher prevalence of severe esophageal disease, specifically megaesophagus, that is commonly associated with marked caudal muscular hypertrophy.
Keywords
Friesian horses reportedly have a higher rate of suspected hereditary diseases such as dwarfism, aortic rupture, and aortopulmonary fistulation.1,11,12 This may be the result of reduction in numbers of breeding stallions after World War II. 9 Low levels of variation in protein and microsatellite markers in the current population indicate low levels of heterozygosity. 9
Megaesophagus is a possibly inherited disease that has been recognized for many years within Friesian horses. 2,3 However, detailed information about the prevalence, clinical presentation, and pathology of megaesophagus in Friesian horses has not been reported. A cause has yet to be identified for this condition. While clinical esophageal disease is uncommon in horses, esophageal idiopathic muscular hypertrophy is commonly encountered during necropsy of aged horses and generally is regarded as an incidental finding rather than the cause of clinical signs. 6 In one study of esophageal disease in horses, only 2 of 31 horses with muscular hypertrophy of the caudal esophagus presented with clinical signs such as dysphagia. 2 Interestingly, both of these horses were Friesians. 2 Therefore, the occurrence of megaesophagus as an important esophageal disease in Friesians, and the clinical significance of muscular hypertrophy of the caudal esophagus in Friesian horses, require further investigation.
The goal of this study was to characterize the prevalence, clinical signs, and pathology of esophageal disease in a necropsy population of Friesian horses and to compare and contrast these findings with those observed in other breeds. We conducted a retrospective study reviewing all horse necropsy cases submitted to the Michigan State University Diagnostic Center for Population and Animal Health (DCPAH) over a period of 6 years. Furthermore, to evaluate the significance of caudal muscular hypertrophy in affected Friesian horses, a comparative study was performed evaluating esophageal tissue samples from Friesian horses and non-Friesian horses with no clinical signs of esophageal disease.
Material and Methods
Retrospective Study
Necropsy records of the DCPAH were searched for all equine accessions from October 2005 to December 2011. The search was performed with the words horse and esophagus to include all cases that had esophageal lesions in the gross reports. For all those horses, age, breed, sex, and diagnosis were recorded.
We selected all horses with severe esophageal disease, which were defined for the purpose of this study as those animals with primary esophageal lesions that led to death or euthanasia. Ten horses with severe esophageal lesions (6 Friesian and 4 non-Friesian horses) were identified, and archived esophageal tissue sections collected at necropsy were reevaluated to confirm the diagnosis, to identify consistent defining histologic characteristics, and to determine a plausible cause.
Comparative Study
To compare the wall thickness of the caudal esophagus and the histological findings between Friesian horses with clinical signs of esophageal disease identified in the retrospective study and horses without clinical signs of esophageal disease, esophageal tissue samples were collected from 6 Friesian horses and 20 non-Friesian horses with no clinical signs of esophageal disease. The diagnosis of idiopathic muscular hypertrophy in horses is generally based on subjective gross evaluation by a pathologist. To avoid such subjectivity, in our study the determination of muscular hypertrophy was based on previously published data: a mean wall thickness of 0.9 ± 0.15 cm in horses with idiopathic muscular hypertrophy of the esophagus and a mean wall thickness of 0.5 ± 0.1 cm in normal horses. 2 We only made a diagnosis of muscular hypertrophy if the esophagus had a wall thickness greater than 0.8 cm. Five of these 6 Friesian horses and 10 of the 20 non-Friesian horses had grossly visible hypertrophy of the caudal third of the esophagus. Sampling for histopathology as well as measurements of the wall thickness was conducted at 10 cm proximal to the gastroesophageal junction. Our data were expressed as mean ± SD. The Student’s t-test for 2 samples assuming unequal variances was used to compare the difference in the wall thickness of horses with muscular hypertrophy and possible associated clinical signs with that in horses with muscular hypertrophy and no clinical signs, and with that in normal horses. P < .05 was considered significant.
All tissue specimens had been fixed in 10% neutral-buffered formalin, processed by routine methods, and embedded in paraffin. Five-micrometer-thick serial sections were then cut and stained with hematoxylin and eosin as well as Gomori trichrome.
Results
Retrospective Study
Necropsy reports of 852 horses submitted to the DCPAH within the established time period were included in this study (Table 1). The 3 most commonly encountered breeds were Quarter horses (159), Thoroughbreds (107), and Standardbreds (79) (Supplemental Table 1, available online at http://vet.sagepub.com/supplemental). Horses included 329 mares, 143 stallions, and 334 geldings. The sex of 46 horses was not reported. The age of the animals ranged from aborted fetuses to 35 years of age. A total of 17 Friesian horses were submitted to the DCPAH during this time period. These 17 Friesian horses were 6 mares, 4 stallions, and 7 geldings, with an age range of 36 hours to 18 years (mean 7.1 ± 6.6 years).
Search Results of Necropsy Reports of Horses Submitted to the DCPAH Between October 2005 and December 2011: Comparison of Animal Numbers Between Friesian and Non-Friesian Horses.
aNumber of horses that had esophageal lesions in the gross necropsy report.
bNumber of horses that had esophageal lesions as the cause of death or euthanasia.
cNumber of horses that had muscular hypertrophy of the caudal esophagus.
Forty-two (42/852; 4.9%) horses had grossly described esophageal lesions (Table 1), of which several occurred in the same horse. The most common esophageal lesion was caudal muscular hypertrophy (n = 25), with a prevalence of 35.3% in Friesian horses, while the breed-specific prevalence in the 5 most commonly encountered breeds (Quarter horses, Thoroughbreds, Standardbreds, Arabian, and Paint horses) was less than 3% (Supplemental Table 1). Other esophageal lesions consisted of hemorrhages secondary to septicemia, endotoxemia, or trauma (n = 7); megaesophagus (n = 6); erosion/ulceration (n = 4); obstruction with food bolus (n = 3); tears (n = 2); secondary neoplastic invasion (n = 2); lymphoid patches (n = 2); thin wall (n = 1); and esophagitis (n = 1). Ten (10/852; 1.2%) horses—6 Friesians and 4 non-Friesians—died or were euthanatized because of severe esophageal disease. Thus, 6 of the 17 Friesian horses that had been submitted for necropsy (35.3%) had severe esophageal disease in contrast to only 4 of 835 horses of the other breeds (0.5%).
Signalment, clinical signs, and gross necropsy findings of the 10 horses with severe esophageal disease are summarized in Table 2. Clinical signs in the 6 affected Friesian horses included ptyalism/dysphagia (n = 6), nasal discharge (n = 3), respiratory distress suspected to be due to aspiration pneumonia (n = 3), anorexia/weight loss (n = 3), and evidence of pleural effusion (n = 2). Two of these Friesian horses had been clinically diagnosed with megaesophagus through endoscopy, and in the remaining 4 Friesian horses, esophageal obstruction (choke) was clinically suspected.
Signalment, Clinical History, and Necropsy Findings of 10 Horses With Severe Esophageal Lesions.
Abbreviations: F, female; m, month-old; M, male; MC, male castrated; y, year-old.
The gross diagnoses of the 10 horses with severe esophageal diseases consisted of megaesophagus (n = 6) leading to esophageal tears in 2 horses, esophageal obstruction with food bolus (n = 3), and severe esophagitis due to esophageal tube placement (n = 1). All 6 Friesian horses in this group, all of which were males or male castrated, had megaesophagus characterized by dilatation of the lumen in the middle to caudal portion of the esophagus (Fig. 1). In addition, 2 of them had esophageal tears with rupture of the mucosa and separation of the tunica muscularis, accompanied by severe necrosis and hemorrhage. In these 2 horses, dissecting tracts filled with blood and feed material extended throughout all the esophageal layers up to the adventitia (Figs. 2, 3). The ultimate cause of megaesophagus was not determined in the necropsy reports, although the esophagus of the 5 of these 6 Friesians that were 5 years or older had marked thickening of the caudal third of the esophageal wall (see results of comparative study) (Figs. 1, 3). In these 5 affected Friesian horses, the tunica muscularis of the caudal third of the esophagus bulged moderately on cut surface (Figs. 1, 3). In the sixth Friesian horse with megaesophagus, a 6-month-old foal, the esophagus was characterized by marked dilatation of the entire lumen, especially at the middle portion (up to 10 cm in diameter), with no apparent underlying cause; muscular hypertrophy of the caudal esophagus was not grossly evident at necropsy. In contrast to these 6 affected Friesian horses, the 4 non-Friesian horses with severe esophageal disease had either esophageal obstruction with an intraluminal food bolus (n = 3) or severe esophagitis associated with the placement of an esophageal tube (n = 1). None of these 4 horses had muscular hypertrophy of the caudal esophagus described in the necropsy report.
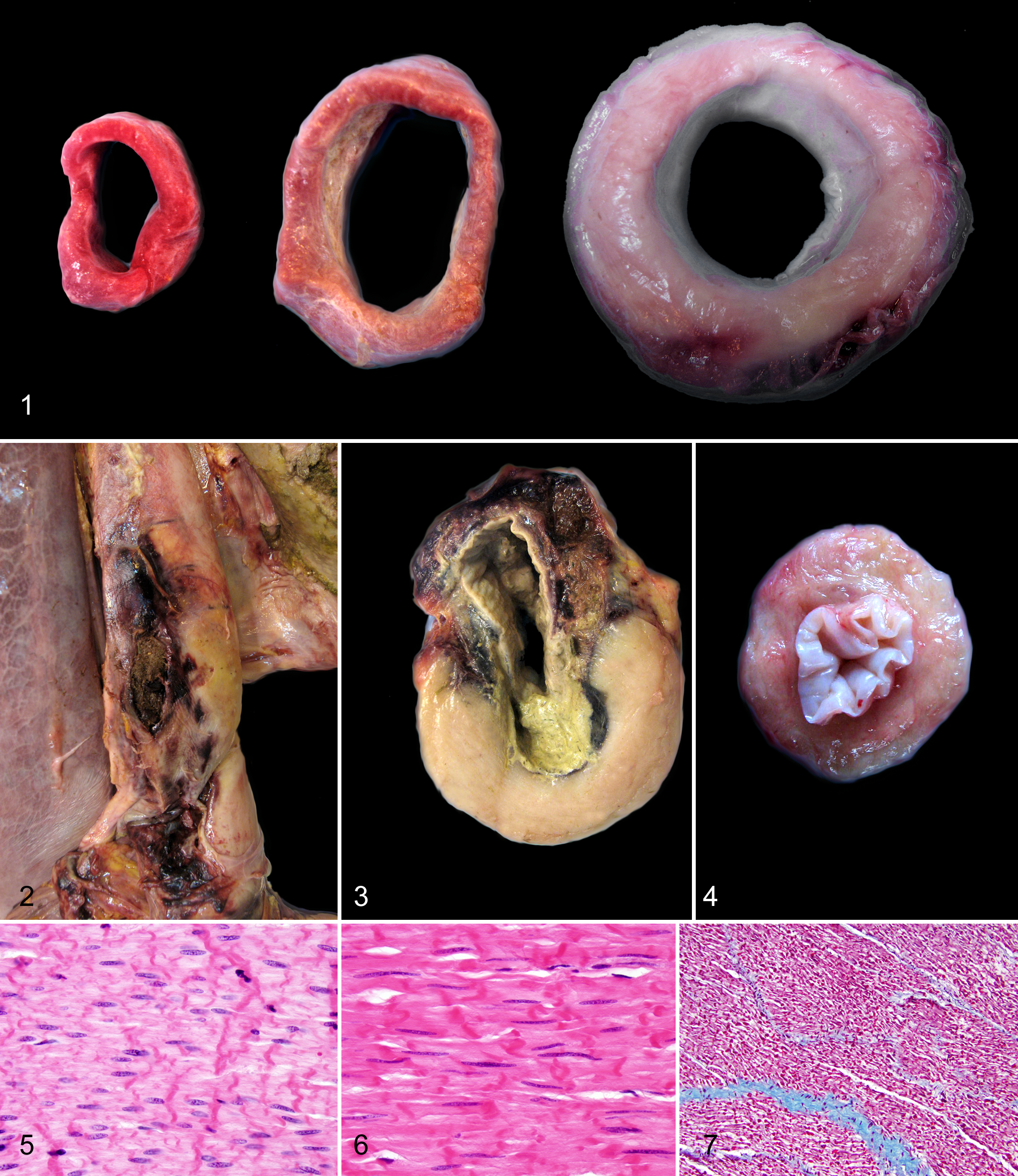
Two additional Friesian horses had grossly described esophageal lesions: One had a thin walled esophagus and the other caudal muscular hypertrophy (1.5 cm thick) of the esophagus. The Friesian horse with the thin esophageal wall was 4 days old and was euthanatized due to severe respiratory distress resulting from severe interstitial pneumonia. The other Friesian horse that had muscular hypertrophy of the caudal esophagus was an 18-year-old Friesian mare with intestinal adenocarcinoma and gastric rupture. The 9 remaining Friesian horses had no grossly observed esophageal lesions and died or were euthanatized due to lesions in other organs such as aortic rupture, pneumonia, or gastric rupture. No esophageal tissues of these 11 Friesian horses were available for reexamination.
Idiopathic esophageal muscular hypertrophy had been diagnosed grossly in 19 of 835 non-Friesian horses submitted for necropsy, none of which exhibited clinical signs of esophageal disease (Table 1).
Histologically, the esophagus of 5 of the 6 Friesian horses with megaesophagus had a thickened tunica muscularis, mainly in the inner circular layer, due to smooth muscle hypertrophy. Muscular hypertrophy was restricted to the caudal third of the esophagus, whereas the tunica muscularis of the middle and cranial third of the esophagus was not affected. The 2 Friesian horses with esophageal tears had additionally a focal, fairly well-demarcated cavitary space within the tunica muscularis, filled with large amounts of fibrin and blood and lined by early granulation tissue. Food material had protruded from the esophageal lumen into this cavitary space, surrounded by mixed bacteria and locally extensive subacute suppurative inflammation characterized by infiltration of degenerated neutrophils and macrophages. In the sixth, 6-month-old, Friesian with megaesophagus, although the esophageal wall of the available sections was only approximately 0.5 cm thick and did therefore not appear to be thickened grossly, the caudal portion of the esophagus had less severe but otherwise similar histologic changes of muscular hypertrophy as seen in the esophagus of the other 5 affected Friesians. In addition, in the middle portion of the esophagus, there were scattered skeletal myofibers with degeneration/necrosis that were characterized by hypereosinophilic sarcoplasm with loss of cross-striation and pyknotic nuclei. Occasionally, mild focal inflammatory cell infiltration was noted in the interstitium in the middle segment of the esophagus. There was no evidence of fibrosis, inflammation, or loss of myenteric plexi in either the middle or caudal portion of the esophagus of this horse.
Comparative Study
The esophagus of 5 of the 6 Friesian horses with clinical signs of severe esophageal disease in our retrospective study had marked thickening of the caudal third of the esophageal wall with an average wall thickness of 1.9 ± 0.3 cm (range 1.3–2.2 cm), compared with an average wall thickness of 0.5 ± 0.1 cm (P = .001) of the normal esophagus regardless of horse breed (Table 3, Figs. 3, 4). Furthermore, the thickness of the esophageal wall of Friesian horses with muscular hypertrophy and concurrent severe clinical disease (average 1.9 ± 0.3 cm) was significantly greater than the thickness of the wall of the esophagus of non-Friesian horses with caudal muscular hypertrophy (average 1.1 ± 0.2 cm, P = .005). When the wall thickness of the esophagus of Friesian horses with muscular hypertrophy and concurrent severe clinical disease was compared with the esophageal wall thickness of Friesian horses with muscular hypertrophy and no associated clinical signs (1.3 ± 0.5 cm), the 1-tailed t-test was statistically significant (P = .04), but the 2-tailed t-test was not (P = .08).
Comparison of the Wall Thickness of the Caudal Esophagus of Friesian and Non-Friesian Horses.
Abbreviation: MH, muscular hypertrophy.
The mean ages of Friesian horses with caudal muscular hypertrophy with and without clinical signs were 10.8 ± 5.8 years and 11.4 ± 6.2 years, respectively. Friesian horses with caudal muscular hypertrophy were approximately 4 years younger than the non-Friesian horses (mean ages of 15.1 ± 9 years) with this idiopathic alteration.
Histologically, the smooth muscle cells of the 5 Friesian horses with megaesophagus and esophageal muscular hypertrophy had increased amounts of cytoplasm and sparsely distributed and elongated nuclei compared with the esophageal samples of grossly normal (1 Friesian and 10 non-Friesian) horses (Figs. 5, 6). Hypertrophied smooth muscle cells were arranged in parallel bundles, and neither mitoses nor cellular atypia was noted. Similar but less severe smooth muscle cell hypertrophy was also noted in the 5 Friesian and 10 non-Friesian horses with muscular hypertrophy with no associated clinical signs. There was no evidence of any degenerative or reparative disease process, such as fibrosis (Fig. 7), or any indication of inflammation or loss of myenteric plexi.
Discussion
Friesian horses have been previously recognized as prone to develop clinically significant esophageal disease. 2,3 Our data confirm a much higher prevalence (35.3%) of severe esophageal disease that may cause death or lead to euthanasia of Friesian horses compared with other horse breeds (0.5%). Based on these results, Friesian horses are predisposed to severe esophageal disease, specifically megaesophagus. Megaesophagus in Friesian horses is furthermore commonly associated with muscular hypertrophy of the caudal esophagus.
Common causes of esophageal diseases in horses include obstruction, trauma, inflammation, and disturbances in motility. Intraluminal obstruction is the most common abnormality and is usually due to impaction with feed material. 4 Regardless of the cause, commonly observed clinical signs of esophageal diseases include nasal discharge, excessive salivation, coughing, colic, sweating, gulping, extension of the head and neck, and respiratory distress due to aspiration pneumonia. 3,4 The horses in our study exhibited ptyalism/dysphagia, nasal discharge, respiratory distress suspected to be due to aspiration pneumonia, and anorexia/weight loss. In addition, 2 Friesian horses had pleural effusions associated with esophageal tears.
No cause of megaesophagus was evident based on gross and microscopic examination of the affected Friesian horses. This is in contrast to all affected non-Friesian horses, in which causes of their esophageal condition were apparent at necropsy: either esophageal obstruction with a food bolus in the lumen or severe esophagitis associated with an esophageal tube. In humans, the most common predisposition to megaesophagus is a condition known as achalasia. 7 Achalasia is a dysfunction of esophageal motility at the lower esophageal sphincter due to derangement of esophageal peristalsis. It is caused by a neurodegenerative disorder in which the neurons of the myenteric plexi are lost. 7 In animals, causes of megaesophagus include innervation or denervation disorders as well as partial physical obstruction and stenosis of the esophagus secondary to either inflammatory disease of the esophageal musculature or vascular anomaly such as persistence of the right aortic arch; however, many cases are idiopathic. 6 In our study, no persistence of the right aortic arch, luminal obstruction, or primary esophageal inflammation was reported in the necropsy reports of the affected Friesian horses. Also, there was no evidence of fibrosis, inflammation, degenerative disease, or loss of myenteric plexi histologically.
A previous study reported megaesophagus in 29 horses, including 14 Friesian horses (48%). 3 In that study, the mean age of the Friesian horses with megaesophagus was 7.3 months. 3 Interestingly, in our study, 5 of the 6 affected Friesian horses were older than 5 years of age. The reason for this discrepancy is unclear.
Five of the 6 Friesian horses with megaesophagus had severe muscular hypertrophy of the caudal esophagus. The thickening of the esophageal wall in these 5 Friesian horses was significantly greater than the thickness of the esophageal wall of the normal esophagus regardless of the horse breed and also significantly greater than that seen in non-Friesian horses with caudal muscular hypertrophy. Interestingly, while not statistically significant in the 2-tailed t-test (P = .08), in the 1-tailed t-test (P = .04) the thickness of the wall of the esophagus of Friesian horses with muscular hypertrophy and concurrent severe clinical disease was significantly greater than that seen in Friesian horses with caudal muscular hypertrophy without clinical signs. Since both groups included only 5 animals, the statistical evaluation was limited and future studies are necessary to substantiate our conclusions. Regardless, these results suggest that caudal muscular hypertrophy in Friesian horses may be a different condition than that in non-Friesian horses and may be associated with clinical signs. The sixth Friesian horse in our retrospective study that had megaesophagus with no reported esophageal muscular hypertrophy based on gross examination was only 6 months old whereas the horses with both of these esophageal alterations were 5 years or older. Although not evident at necropsy, this Friesian foal had histological features considered to represent early stages of smooth muscle hypertrophy and may have been too young to have developed hypertrophy severe enough to be identified on gross examination.
As previously discussed, esophageal idiopathic muscular hypertrophy is generally regarded as an incidental finding in aged horses. 6 In our retrospective study, 19 of 835 (2.3%) non-Friesian horses submitted for necropsy had esophageal idiopathic muscular hypertrophy, whereas the breed-specific prevalence of the 5 major breeds in the study was less than 3%. None of these horses were reported to have shown clinical signs. This is in strong contrast to the Friesian horses in this study, where 6 of 17 (35.3%) animals had muscular hypertrophy of the caudal esophagus that was associated with severe esophageal disease in 5 horses. Interestingly, the only Friesian horse in this group that presented without clinical esophageal signs was a mare, while all other Friesian horses in this group were male or male castrated. Clinical disease has been previously reported in 2 Friesian horses (a gelding and a mare) with muscular hypertrophy of the caudal esophagus. 2 Similar to our findings, this study did not observe clinical disease in 29 non-Friesian horses that presented with muscular hypertrophy of the caudal esophagus. 2 These data together with our results strongly suggest that muscular hypertrophy of the caudal esophagus may be of clinical significance in Friesian horses.
In humans, muscular hypertrophy of the caudal esophagus can be primary hypertrophy or can occur secondary to achalasia. 7 There was no evidence of loss of myenteric plexi in our Friesian horses, as seen with achalasia. Thus, a primary muscular hypertrophy of smooth muscle cells morphologically similar to that seen in a human condition known as the esophageal leiomyomatosis seems more likely. 8,13 This condition is characterized by circumferential thickening of smooth muscle involving the lower third of esophagus and thereby causes narrowing at the gastroesophageal junction and mimics the clinical symptoms of achalasia. 8,13 Esophageal leiomyomatosis can occur sporadically or can have a hereditary basis such as in Alport’s syndrome. 8 The main clinical manifestation of Alport’s syndrome is a progressive nephropathy characterized by hematuric nephropathy and ultrastructural changes of the glomerular basement membrane, such as thickening, splitting, and thinning, associated with mutation in a gene encoding for type IV collagen. 8,10 This type of collagen forms the basement membrane of the glomeruli in the kidney and also of the smooth muscle in the esophagus. The mutation of its encoding gene is considered to result in clonal overgrowth of smooth muscle cells in the patient’s esophagus. 10 Histologically, the lower third of the esophagus in these patients is consistently characterized by interweaving fascicles and occasionally by small nodular proliferations of spindle cells without mitosis or atypia. 8 The overall cellularity is low. 8
In horses, the upper third of the esophagus is composed of striated muscle, while the lower third is composed of smooth muscle, and the middle contains both muscle types, with transition from striated to smooth muscle similar to what has been described in humans. 5,14 The affected Friesian horses in our study had marked circumferential thickening only in the caudal esophagus caused by smooth muscle hypertrophy that appeared histologically similar to the alterations described in human patients with esophageal leiomyomatosis associated with Alport’s syndrome: hypertrophy of smooth muscle layers with low cellularity, and no mitosis or cellular atypia. 8,13 However, based on the history and necropsy reports, there was no evidence of renal disease in affected Friesian horses as seen in human patients with Alport’s syndrome.
We speculate that muscular hypertrophy of the caudal esophagus in Friesian horses represents a hereditary condition similar to leiomyomatosis in humans that can cause severe esophageal disease, in particular megaesophagus and potentially esophageal tears. Considering the male predisposition observed in our study, a potential X-linked mechanism may be involved. Genetic analysis is ongoing to better understand the pathogenesis of this condition.
Footnotes
Acknowledgements
We thank all personnel of the anatomic pathology section of the DCPAH who were involved with the original necropsy cases, in particular Jennifer Lamoureux and Changseok Kim for their help during the early stages of the study. We also thank the staff of the Histology Laboratory and the Information Technology section of the DCPAH for their technical support. Furthermore, we are deeply grateful to the Fenway Foundation for Friesian Horses for their continued support and to all practitioners, clinicians and owners for case submission and clinical information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: The CVM Endowed Research Fund grant RH082218.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
