Abstract
Porcine melanomas have proven interesting in a wider biological perspective due to a common phenomenon of spontaneous regression, which is characterized by infiltration of macrophages, among others. Separation of neoplastic melanocytes from pigment-laden macrophages may, however, be challenging as the morphology of melanocytes varies considerably and sometimes resembles macrophages. The aim of this study was correspondingly to characterize and differentiate the cells in 20 porcine melanocytomas and regional lymph nodes by histologic examination and immunohistochemistry for melan A, PNL2, S100, lysozyme, alpha-1-antitrypsin, and ionized calcium binding adaptor molecule 1 (Iba1). Grossly, the melanocytomas were divided into 2 distinct types: pigmented maculae (n = 7) and raised tumors (n = 13). In the maculae, the pigmented cells were mainly melanocytes reactive for melan A, PNL2 and S100. In contrast, the majority of the cells in the raised tumors were melanophages, which expressed Iba1, alpha-1-antitrypsin, and lysozyme. Yet, cells histomorphologically indistinguishable from the melanophages expressed melan A and PNL2. These cells were Iba1 and S100 negative, and ultrastructurally, they were devoid of lysosomal bodies and filled with stage III and IV melanosomes. In the regional lymph nodes, melanocytes were present in the trabecular sinuses. In focally or diffusely black lymph nodes, pigmentation was, however, mainly due to aggregates of melanophages, which were confined to the trabeculae, deep cortex, and peripheral lymphoreticular tissue. Normal and neoplastic porcine melanocytes express melan A and PNL2, and immunohistochemical staining for melan A, PNL2, and Iba1 was found useful to identify and distinguish melanocytes and melanophages in porcine melanotic lesions.
Melanocytic tumors in pigs are interesting, as they have several features in common with melanomas in humans. Most studies on porcine melanocytic tumors have been performed on the breeds Sinclair and melanoblastoma-bearing Libechov Minipig. These breeds have been proposed as useful human melanoma models because (1) they show a high incidence of spontaneous malignant melanoma, (2) spontaneous regression of benign and malignant melanomas is common, and (3) metastases with a similar distribution to what is seen in humans are frequent. 10,21,32,37,38,50 Furthermore, the histopathologic features of malignant melanoma in pigs are similar to the superficial spreading and nodular variants of cutaneous melanoma in humans. 10,37,50 The enzymes tyrosinase, α-mannosidase, and γ-glutamyltransferase, which are involved in differentiation and spread of melanoma cells in humans, have also been demonstrated in melanoblastoma-bearing Libechov Minipig, and the activity of tyrosinase was considerably reduced in regressing melanomas. 5
In slaughter pigs, melanocytic tumors mainly occur in the Duroc and Duroc crossbreeds. 12,24,25,30,40,46,49 The majority of melanocytic tumors in these breeds are benign, 8,12,46 and as in the miniature breeds, spontaneous regression of melanocytic tumors is common and characterized by massive infiltration of pigmented macrophages and lymphocytes. 8,40,46 Black pigmentation of regional lymph nodes is also common, and it has been associated with metastatic growth of pigment-producing malignant melanomas as well as by pigment-laden macrophages from melanomas undergoing regression. 8,24,25,49
The identification of metastatic melanocytes in lymph nodes is important, and several studies of human melanoma patients have used immunohistochemistry for detection of metastases to the lymph nodes. 4,6,23,31,47 The application of immunohistochemistry is also important because the morphology of the cells that make up melanomas is quite variable and can be confused with other cell types. 15,18,22 Among the most widely used antibodies for identification of melanocytes are S-100, HMB-45, and melan A. S-100 is a sensitive but not very specific marker for melanocytic lesions, as it labels other cell types, such as nerve sheath cells and Langerhans cells, in addition to melanocytes. 4,6,15,22,33,36,41,47 Antibodies for S-100 have been applied in pigs to differentiate melanocytes from other cell types. 10,20,29,40 The HMB-45 antibody is a marker of the cytoplasmic premelanosomal glycoprotein gp100. 4 HMB-45 is a specific marker for melanoma cells, but it is not reactive with normal adult melanocytes and intradermal nevus cells. 4,6,19,22,23,31,36,41,47 Positive labeling for HMB-45 has been demonstrated in porcine melanocytic tumors. 20,30 Melan A is a melanocyte-differentiating protein that is recognized by cytotoxic T lymphocytes. Melan A is expressed by normal melanocytes and most melanoma cells, and it is considered to be a specific and sensitive marker for human melanocytes. 6,31,36,47 More recently, PNL2, a monoclonal antibody that recognizes a fixative resistant melanocytic antigen, was shown to be a sensitive and specific marker of melanocytes and most melanoma cells in other animal species and humans. 1,9,16,27,35,43 –45,48
In present study, immunohistochemistry for melan A and PNL2, which have not been tested on porcine tissue before, and to the macrophage markers ionized calcium-binding adaptor molecule 1 (Iba1), alpha-1-antitrypsin (A1AT), and lysozyme was performed to differentiate and characterize the cells in porcine melanocytic cutaneous lesions and lymph nodes.
Materials and Methods
The material derived from Danish abattoirs, which were requested to submit benign melanocytic lesions and the regional lymph nodes to the Department of Veterinary Disease Biology.
The breed and sex of the 20 pigs included in the study were not recorded, but their age was estimated by the veterinary inspectors to be 5 to 6 months in 6 cases, 1 to 1.5 years in 3 cases, and 2.5 to 3.5 years in 9 cases. In 2 cases, the age was not recorded. In all the pigs, pigmented foci were observed only in the skin and lymph nodes. The material for this study also comprised mammary tissue and regional lymph nodes from 4 Duroc-crossbreed sows as well as the superficial inguinal lymph nodes from 16 Yorkshire-landrace crossbreeds and 10 Duroc crossbreeds without melanocytic cutaneous lesions.
Grossly, the size of the melanocytic lesion was measured at its largest diameter, and its extension into the dermal and subcutaneous tissue was assessed. Likewise, the size of the regional lymph nodes and the presence of pigmentation were evaluated.
One sample of tissue was taken from each cutaneous melanocytic lesion and from the respective lymph node. The tissue was fixed in 10% neutral buffered formalin for 36 to 48 hours, processed conventionally, and embedded in paraffin. From each sample, sections (2–3 μm) were cut, bleached with 10% (v/v) H2O2 in TBS, and/or stained with hematoxylin and eosin for histopathologic evaluation.
Transmission Electron Microscopy
For transmission electron microscopy, small samples (1 mm3) of cutaneous melanocytic lesions and regional lymph nodes from 5 pigs were fixed in 3% glutaraldehyde/0.1 M sodium phosphate buffer (pH 7.3) for 1 hour at room temperature. After rinsing in 0.1 M sodium phosphate buffer, the samples were postfixed in 1.0% osmium tetroxide/0.1 M sodium phosphate buffer and en bloc stained with 1% uranyl acetate in water for 1 hour. Dehydration in graded series of ethanol was followed by equilibration in propylene oxide and infiltration by Epon and polymerization at 60°C for 48 hours. The areas of interest were identified in semithin sections (1.0 μm) stained with 1% basic toluidine blue. Ultrathin sections (70 nm) were cut and collected on 200-mesh electron microscopy grids and contrasted with 1% uranyl acetate and 1% lead citrate before examination in a Philips CM 100 transmission electron microscope.
Immunohistochemistry
Immunohistochemical staining for melan A, PNL2, S100, lysozyme, A1AT, and Iba1 was performed to differentiate melanocytes from macrophages. Immunostaining of single antigens and double immunostaining were both performed (Table 1). Melanotic cutaneous lesions and lymph nodes from 6 animals were immunostained for S100 and A1AT, whereas antibodies for melan A, PNL2, lysozyme, and Iba1 were applied to all cases. In addition, mammary tissue and regional lymph nodes from 4 sows with melanosis uberis, as well as regional lymph nodes from pigs without melanotic cutaneous lesions, were immunostained for melan A and lysozyme.
Immunohistochemical Methods.
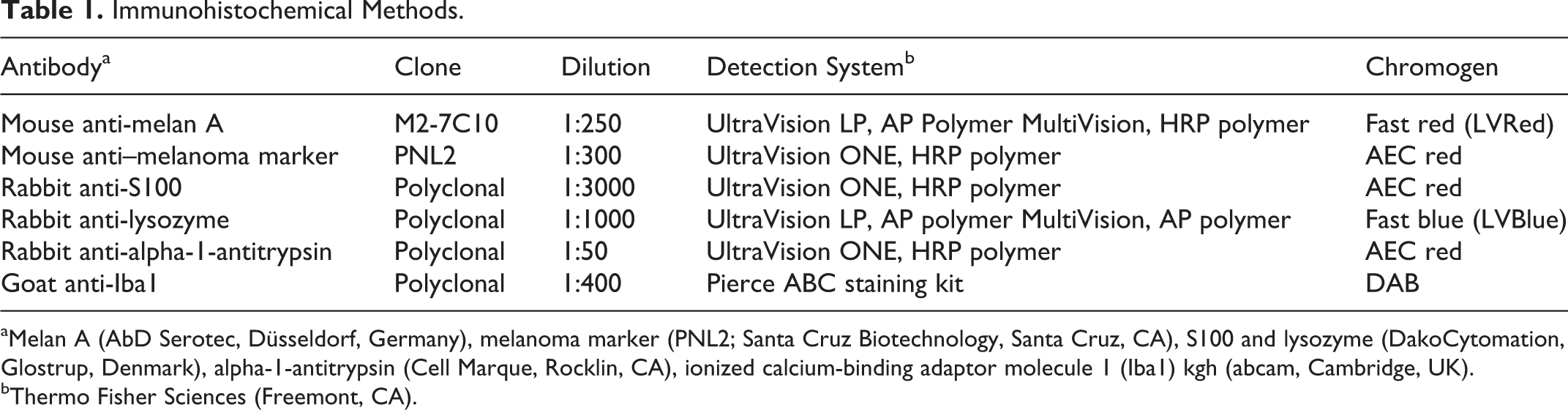
aMelan A (AbD Serotec, Düsseldorf, Germany), melanoma marker (PNL2; Santa Cruz Biotechnology, Santa Cruz, CA), S100 and lysozyme (DakoCytomation, Glostrup, Denmark), alpha-1-antitrypsin (Cell Marque, Rocklin, CA), ionized calcium-binding adaptor molecule 1 (Iba1) kgh (abcam, Cambridge, UK).
bThermo Fisher Sciences (Freemont, CA).
For immunohistochemical staining, thick sections (1–2 μm) from each tumor were mounted on adhesive-coated slides (Superfrost Plus; Menzel-Glaser, Braunschweig, Germany), processed through xylene, and rehydrated in ethanol. Antigen retrieval was performed with enzymatic- and heat-induced retrieval as well as different pH concentrations, which were tested on positive control tissues. Optimal staining was obtained by leaving the slides in protease 0.018% in TBS for 5 minutes (S100), in 0.01 M citrate buffer (pH 6) for 60 minutes at 85°C in a heating cupboard (melan A, lysozyme), or by boiling in a microwave oven (700 W) twice for 5 minutes in the citrate buffer (Iba1, PNL2, A1AT). The samples were bleached in 10% (v/v) H2O2 in TBS for 6 hours at room temperature before the sections were incubated with the primary antibody for 24 hours at 4°C (Table 1). The detection systems UltraVision LP, UltraVision ONE, Pierce ABC staining kit, and MultiVision HRP were applied in accordance with the manufacturer’s instructions (Lab Vision, Thermo Fisher Scientific, Fremont, CA) (Table 1). As chromogen, fast red, fast blue, AEC red (3-amino-9-ethylcarbazole), and DAB (3,3′-diaminobenzidine) (Lab Vision) were applied, and counterstaining was performed with Mayer’s hematoxylin (double-immunostained sections were not counterstained). Slides were given two 5-minute washes in TBS (pH 7.6) or four 4-minute washes in 0.1% Tween 20 in TBS (double immunostaining), before addition of each new reagent.
Skin biopsies from pigs with dark skin and lymph nodes from healthy pigs were used as positive controls for melanocytes and macrophages, respectively. Brain tissue was used as positive control for S100, and Iba1 expression was examined in the spleen, lever, lung, and brain and in tissue with granulomatous inflammation, Negative controls were run in parallel with the primary antibody substituted by an irrelevant monoclonal (matching isotype) or polyclonal antibody of the same concentration as the primary antibody or with 5% normal swine serum in TBS.
Results
Gross Examination
At gross examination, 2 distinct types of cutaneous lesions were present: first, circular pigmented maculae ranging in size from 2 mm to 3 cm (7 cases) (Fig. 1); second, raised brown-black tumors, 1.5 to 6 cm in diameter, with discrete edges and an irregular, sometimes rough surface (13 cases) (Fig. 2). Pigmentation was mainly seen in the superficial dermis, but in 2 raised lesions, pigmentation was also noted in the subcutaneous fat. Depigmentation of the skin surrounding the lesions was not observed.
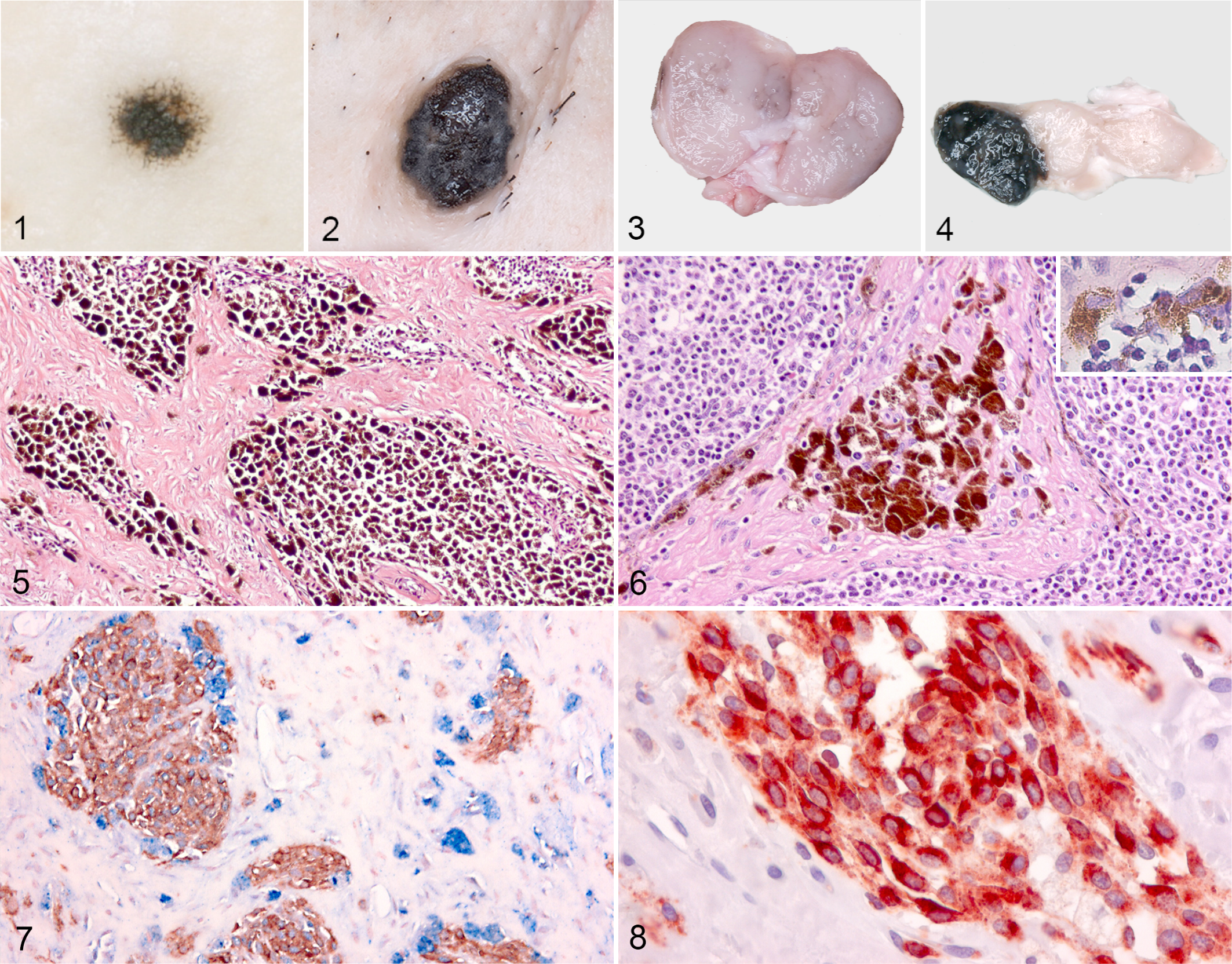
Three of the lymph nodes from pigs with melanocytomas were uniformly black; 4 contained traces of pigment (Fig. 3); and 13 showed distinct pigmented foci that sometimes bulged on the surface (Fig. 4) (Table 2). The lymph nodes appeared normal of size or slightly enlarged. Pigmented maculae and raised tumors were seen with the same frequency in all age groups.
Gross and Histologic Characteristics of Regional Lymph Nodes.

aMacroscopic appearance in parentheses.
Histopathology
In samples from the pigmented maculae, the superficial dermis contained a circumscribed mass of pigmented oval to spindle-shaped cells and only few large and round to polyhedral cells.
The raised lesions were typically composed of nests of heavily pigmented cells in the superficial dermis and perivascularly in the deeper dermis (Fig. 5). The cells were round or polyhedral with abundant cytoplasm and a relatively small eccentric nucleus, which was often obscured by variable-sized pigment granules. In 7 of the 13 raised lesions, moderate to heavy infiltration with lymphocytes was present. Mitotic figures were not observed in the samples; however, evaluation of mitosis in some of the cells was difficult due to the presence of pigment granules.
In lymph nodes with traces of pigment, small separately arranged, oval to fusiform, and moderately pigmented cells were seen in trabecular sinuses and sometimes in the deep cortex and peripheral loose lymphoreticular tissue. This arrangement of pigmented cells was seen in the lymph nodes of pigs with cutaneous pigmented maculae but also in 4 of 5 and 2 of 10 inguinal lymph nodes from sows with melanosis uberis and Duroc crossbreeds with no cutaneous lesions, respectively (Table 2).
In all the diffusely black lymph nodes and in 7 of 13 lymph nodes with focal pigmentation, the trabeculae contained aggregates of large, heavily pigmented, polyhedral cells. These lymph nodes also contained clusters of pigmented small or moderately large cells in the trabecular sinuses and in the deep cortex and peripheral loose lymphoreticular tissue (Fig. 6).
In the rest of the lymph nodes, pigmented cells were arranged separately or in clusters in the trabecular sinuses, the cortex, and the peripheral lymphoreticular tissue (Table 2).
Immunohistochemistry and Electron Microscopy
In skin biopsies from pigs with dark skin, cells expressing melan A and PNL2 were interspersed in the stratum basale and in the epithelium of the hair follicles. PNL2 labeling was granular, and the labeling intensity for both melan A and PNL2 was strong and confined to the cytoplasm.
S100 immunolabeling was present in the nucleus and/or the cytoplasm of melanocytes but also in cutaneous sweat glands and nerves among others.
Strong Iba1 immunolabeling was seen in macrophages in the lymph node, spleen, liver (Kupffer cells), and dermis and in alveoli and connective tissue of the lung. Iba1 was also expressed by microglia in the brain and by lymphocytes in the extranodular lymphatic tissue in lymph nodes and in the white pulp of the spleen. In the granulomas, macrophages (but not giant cells) labeled positive. In the pigment-laden macrophages, Iba1 immunolabeling was mainly confined to the periphery of the cell.
The immunoreactivity of macrophages with antibodies to lysozyme was varying, and the staining appeared granular and confined to the cytoplasm. The immunolabeling of pigment-laden macrophages was generally weaker compared to Iba1 and A1AT.
Cytoplasmic A1AT immunolabeling was observed in macrophages of the skin and lymph nodes. However, only few macrophages labeled positive in the normal lymphatic tissue.
In the mammary gland tissue, small pigmented cells, which were located basally in the glandular epithelium, expressed melan A. Likewise, the pigmented cells present in the trabecular sinuses in lymph nodes from these sows and from the 2 Duroc crossbreeds with no melanomas expressed melan A.
Melan A–reactive cells were not observed in the lymph nodes from the 16 Yorkshire-landrace crossbreeds.
In the pigmented maculae, the oval to spindle-shaped cells expressed melan A, PNL2, and S100, whereas the larger, round to polygonal cells were immunoreactive for Iba1, lysozyme, and A1AT (Figs. 7 –10). In general, more melanocytes labeled positive for melan A and PNL2 than for S100.
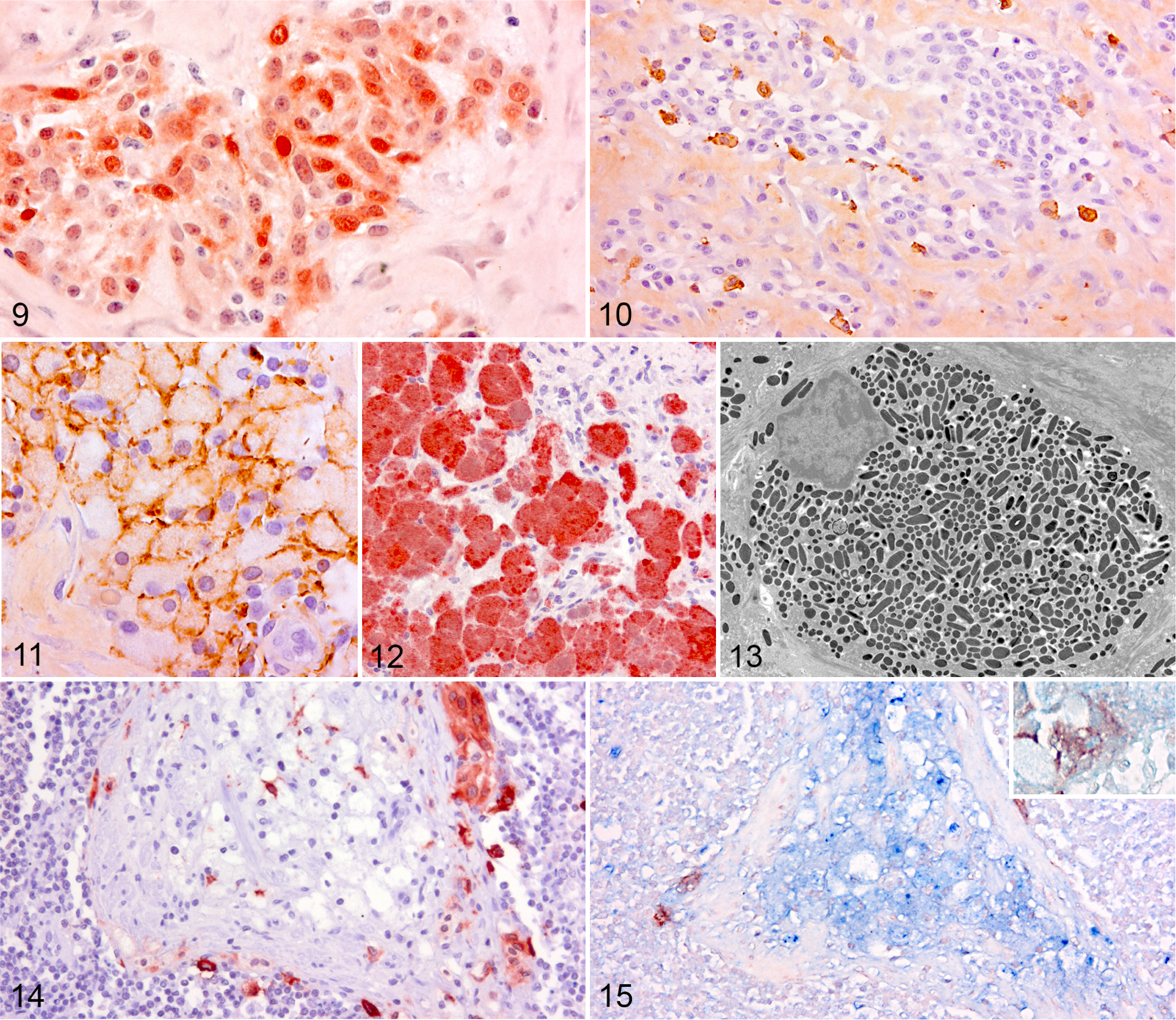
In the raised tumors, only few oval to spindle-shaped cells in the superficial dermis expressed melan A and PNL2, and the major part of the pigmented lesion was composed of Iba1-, lysozyme-, and A1AT-immunoreactive macrophages arranged in variable-sized nests (Fig. 11). In 4 of the raised lesions, however, the pigmented lesion was mainly composed of a mixture of spindle-shaped cells and larger round to polygonal cells showing moderate to strong immunoreactivity for melan A and PNL2 (Fig. 12). The large cells in the lesions were S100 negative. The round to polygonal melan A– and PNL2-positive cells in these melanomas sometimes contained large pigment granules and were histomorphologically indistinguishable from macrophages but negative for lysozyme and Iba1. However, weak A1AT immunolabeling was seen in some of these cells. Ultrastructural examination of the large cells revealed that they were packed with stage III and IV melanosomes, and no lysosomal bodies or compound melanosomes characteristic of melanophages were recognized (Fig. 13).
Double labeling for melan A and lysozyme was seen in a small number of perivascular cells, which were similar histomorphologically and ultrastructurally to those described above.
Independent of the histologic appearance of the melanocytic lesion in the skin and in the lymph node, the melan A– and PNL2-reactive cells in the pigmented lymph nodes were confined to the trabecular sinuses. Sometimes, small individual cells, which were weakly immunoreactive for melan A/PNL2, were seen in the peripheral lymphoreticular tissue, the deep cortex, and the trabeculae (Figs. 14, 15). The pigmented cells in the trabecular sinuses were negative for S100. The majority of heavily pigmented round and polyhedral cells aggregated in the trabeculae were lysozyme, A1AT, and Iba1 positive (Fig. 15). Likewise, the majority of the pigment-laden cells arranged separately or in clusters in the deep cortex and lymphoreticular tissue expressed the macrophage markers.
Discussion
Two morphologically distinct types of melanocytoma were observed: pigmented maculae and raised tumors. The maculae were composed of melan A–/PNL2-reactive spindle-shaped cells and only few macrophages. In contrast, nests of pigment-laden macrophages and sometimes lymphocytes constituted most of the cells in the raised tumors. Flatt et al observed a similar pattern in benign melanomas from miniature pigs and proposed a transition of melanocytic lesions from early heavily pigmented maculae to raised black tumors and subsequent progressive regression of the skin lesion. 13 That is, the raised tumors occur when pigment present in the dermis is phagocytized by macrophages causing an increase in volume and thereby an elevation of the epidermis. The transition of melanocytic lesions and the increase in tumor volume have also been observed in other studies, and these changes have been correlated with the age of the animals. 21,38 In present study, however, pigmented maculae and raised tumors were seen in all age groups.
Both the maculae and the raised tumors were associated with pigmentation of the regional lymph nodes, and melanocytes were detected by immunohistochemical staining for melan A and PNL2 in all lymph nodes. In diffusely and focally black lymph nodes, pigmentation was mainly caused by large aggregates of pigment-laden macrophages. The presence of these cells in conjunction with reduced numbers of melanocytes has been associated with the regression of cutaneous and metastatic melanomas. 8,38,50 Therefore, the relatively few melanocytes present in the pigmented lymph nodes may represent remnants of metastatic melanocytes. However, pigmented lesions in the internal organs, which are common in miniature pigs with malignant melanomas, 14,20,37 were not seen in the pigs in this study. Furthermore, the distribution and histomorphology of the melanocytes in the skin and lymph nodes were not indicative of malignancy.
In pigs, the cortical tissue with lymphatic nodules is centrally located in the lymph node, and afferent vessels penetrate via the trabeculae deep into this area to join the trabecular sinuses. 28 The pigmented cells expressing melan A and PNL2 were mainly confined to the trabecular sinuses, which suggests that the melanocytes arrive via lymphatics to regional lymph nodes. In humans, the “benign metastasis theory”—that is, nevus cells that enter dermal lymphatics and travel to assign residency in the draining lymph nodes—has been proposed to explain why benign nevus cells are sometimes found in the lymph nodes draining cutaneous nevi. 2,4,6 The benign metastasis theory also seems probable in pigs, when considering that the melan A–/PNL2-reactive cells in the trabecular sinuses were uniform, without signs of malignant behavior, and melan A–reactive melanocytes were also seen in trabecular sinuses of lymph nodes from pigs with no cutaneous melanocytic lesions.
Another theory is that melanocyte progenitor cells, which normally migrate from the neural crest to the epidermis, prematurely end their migration in the lymph nodes rather than in the skin. 2,4,6 This theory would explain the presence of melanocytes in lymph nodes from pigs devoid of melanocytic cutaneous lesions.
As described in both slaughter pigs and miniature pigs, 8,21,38,40 regression characterized by heavy infiltration of macrophages and sometimes lymphocytes was present in the majority of the examined melanocytomas. Porcine tissue macrophages are heterogeneous and show differences in morphology and antigenicity, among others. 7 Three macrophage markers were therefore applied in present study, of which Iba1 had not been tested on porcine tissue before. The Iba1 expression pattern was comparable to what has been reported in mouse, rat, and human tissues, and in contrast to lysozyme and A1AT, strong immunolabeling was present in normal macrophages and in the pigment–laden macrophages. Iba1 expression was seen in lymphocytes but not in melanocytes, and this antibody therefore proved useful in differentiating macrophages from melanocytes in the skin and lymph nodes.
In 4 melanocytomas, cells histomorphologically indistinguishable from the pigment-laden macrophages labeled positive for the melanocyte markers melan A and PNL2. In addition, double labeling for melan A and lysozyme was seen in some perivascular cells with similar morphology. Some of the large cells also labeled positive for A1AT but were consistently negative for Iba1. The immunoreactivity of melan A and PNL2 in porcine melanomas has never been examined. However, melan A 4,6,16,31,42 and PNL2 1,9,16,44,45,48 are considered specific markers for melanocytes in human and dogs, whereas lysozyme and A1AT are reported to label not only histiocytes but also melanocytes in humans. 22,41 Ultrastructurally, the cells contained no compound melanosomes, which were seen in melanophages. The cells were, however, stuffed with stage III and IV melanosomes but lacking stage I and II melanosomes, which are indicative of melanin synthesis. 11,34 The presence of almost exclusively late-stage melanosomes in porcine neoplastic melanocytes has also been reported in other studies 10,46 and is typical for terminally differentiated melanocytes. 20
Greene et al used porcine melanoma cells, which had been transplanted into severe combined immunodeficient mice, to show that the melanoma cells were capable of differentiating into large, pigmented, macrophage-like cells. 20 The large macrophage-like cells were negative for S100, 20 which was also characteristic of the melan A–/PNL2-reactive macrophage-like cells in present study. In vitro studies of human melanoma cells have also shown that small melanoma cells with little or no pigmentation may differentiate into larger and heavily pigmented cells with little replicative activity. 3,17,26 The melan A–/PNL2-reactive macrophage-like cells observed in some of the melanocytomas might be terminally differentiated neoplastic melanocytes with little replicative activity. Thus, differentiation of neoplastic melanocytes may be an important mechanism of regression of melanocytic lesions in the Sinclair miniature pig 20 and in slaughter pigs.
Another possibility is that the melan A–/PNL2-reactive macrophage-like cells are hybrids between melanoma cells and macrophages. Melanoma–macrophage hybrids have been generated in vitro, and when these cells were implanted in mice, they showed a higher metastatic potential compared to the original melanoma cell line. 39 In contrast to present study, melanosome-containing autophagosomes were seen ultrastructurally in the melanoma–macrophage hybrids. 39
In summary, porcine normal and neoplastic melanocytes express melan A and PNL2. Neoplastic melanocytes may resemble melanophages, and immunohistochemistry with antibodies to melan A, PNL2, and different macrophage markers was therefore required to differentiate neoplastic melanocytes from melanophages. Melan A–reactive melanocytes confined to the trabecular sinuses were present in lymph nodes from pigs with melanocytomas, from sows with melanosis uberis, and from pigs with dark skin and no melanocytic lesions. These cells most likely reflect melanocytes, which incidentally have been transferred to the lymph nodes via regional lymphatics, or melanocyte progenitor cells, which have prematurely ended their migration in the lymph nodes. In addition, immunohistochemical examination revealed that pigmentation of regional lymph nodes in slaughter pigs with melanocytomas was mainly caused by aggregates of melanophages in the trabecular connective tissue, deep cortex, and peripheral lymphoreticular tissue.
Footnotes
Acknowledgements
We are grateful to the staff at Danish Crown, Skærbæk, Ringsted, Herning, Horsens and Blans, Denmark, for help in collecting samples. We thank Betina Gjedsted Andersen and Dennis Brok, Department of Veterinary Disease Biology, and Hanne Mølbak Holm, Department of Veterinary Clinical and Animal Sciences, Faculty of Health and Medical Sciences, University of Copenhagen, for technical assistance. A special thanks to Prof Björn Rozell, Faculty of Health and Medical Sciences, University of Copenhagen, for critically reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
