Abstract
In dogs with symptomatic or asymptomatic leishmaniosis, Leishmania infantum appears to induce a mixed Th1/Th2 immune response that in the sick dog may eventually result in tissue damage via different pathomechanisms, notably granulomatous inflammation (eg, nodular dermatitis, osteomyelitis), immune complex deposition (eg, glomerulonephritis), and/or autoantibody production (eg, polymyositis). This is a compensatory but detrimental mechanism generated mainly because of the insufficient killing capacity of macrophages against the parasite in the susceptible dog. Clinical disease is typically exemplified as exfoliative and/or ulcerative dermatitis, with or without nasodigital hyperkeratosis and onychogryphosis, glomerulonephritis, atrophic myositis of masticatory muscles, anterior uveitis, keratoconjunctivitis sicca, epistaxis, and/or polyarthritis, appearing alone or in various combinations. The pathogenesis of these clinical conditions has recently been highlighted, to a greater or lesser extent. The usually subclinical conditions expressed as chronic colitis, chronic hepatitis, vasculitis, myocarditis, osteomyelitis, orchiepididymitis, and meningoencephalomyelitis, though uncommon, are of pathologic importance from a differential point of view. The leading cause of death among canine leishmaniosis patients is chronic proteinuric nephritis that may progress to end-stage kidney disease, nephrotic syndrome, and/or systemic hypertension. However, even the asymptomatic proteinuria, when profuse, may be a serious problem because it predisposes to arterial thromboembolism and eventually contributes to the deterioration of the body condition.
Canine leishmaniosis (CanL) due to Leishmania infantum is a systemic infectious disease with zoonotic potential that is quite common among the canine population in many parts of the world. 137 According to some polymerase chain reaction–generated data, the prevalence of CanL may reach up to 67% in selected canine populations, especially among asymptomatic dogs that far outnumber those with overt or symptomatic disease. 76,139 The evolution of Leishmania infection—notably, the potential self-elimination of the parasite and the development of an asymptomatic or symptomatic state—is the result of an interaction among the vector (eg, repeated infectious bites), parasite (virulence), and host (eg, genetic background, immune response, coexisting diseases). Infected dogs are typically allocated into susceptible, in which the unrestricted parasite multiplication leads to organ damage and dysfunction, and resistant, which eventually eliminate the parasite and remain clinically normal; however, there are some symptomatic dogs that are self-cured, either clinically or parasitologically, as well as supposedly resistant dogs that eventually become sick when their cellular immunity is compromised for various reasons. 128
General Pathomechanisms
In the susceptible dog, the innate or nonspecific immune system is evaded by Leishmania parasites via several mechanisms, such as the remodeling of phagosomal compartments in which they reside and the interference with signaling pathways. 124 The major role against the parasite is played by the specific immune response, although the activation of macrophages to eliminate Leishmania parasites by T-cell-secreted IFN-γ is still a matter of debate, at least in the dog. 10 However, the influence imposed by the balance between Th1 and Th2 cellular immune response is crucial for the evolution and outcome of natural CanL, where a mixed response appears to prevail in both asymptomatic and sick dogs. 9,106 Protective immunity in CanL is most likely mediated by the action of TNF-a, IL-2, and IFN-γ cytokines secreted by activated T cells to upregulate the antileishmanial activity of macrophages through nitric oxide production that is responsible for the parasite killing by apoptosis. 66,113 Recent research has revealed that the number of apoptotic cells within the inflammatory infiltrate is directly related to parasitic load and the severity of the clinical picture in leishmanial dogs. 148 The infected macrophages are also lysed by CD8+ cytotoxic T cells in a histocompatibility complex-restricted process that can be suppressed in the symptomatic dogs with a high parasitic load. 33,112 The opposite may be seen in the asymptomatic disease, with low parasitism, in which the CD8+ T lymphocytes usually prevail. 64,121 The majority of Leishmania-infected dogs express a cell-mediated immune response exemplified by a strong in vitro lymphocyte proliferation and positive skin reaction to intradermal leishmanin injection (Montenegro test). 19,81 With the progression of the disease, these responses are eventually blunted, along with decreased intracellular killing of Leishmania amastigotes by macrophages and neutrophils. 9,16 The usually prolonged incubation period, which may extend from approximately 3 months to 7 years after an infectious sandfly bite, may explain the inconsistent and variable transition from a resistant to a susceptible state. 10 Among other factors, susceptibility and resistance to CanL are also influenced by genetics, as has been shown by the polymorphisms and mutations of the Slc11c1 gene (solute carrier family 11, member a1), the association of its haplotype TAG-8-141 with Boxer breed predisposition to CanL, and the link between symptomatic CanL and the DLA-DRB1 genotype, considered a major histocompatibility complex class II allele in the dog. 7,119,125,126 Apart from the Boxer, some other breeds (eg, the Cocker Spaniel, Rottweiler, and German Shepherd) are more susceptible to develop symptomatic CanL in contrast to the Ibizan Hound, in which the clinical disease is rather rare. 55,132,138
In sick leishmanial dogs, depletion of T lymphocytes in the lymphoid organs is falsely compensated by an exuberant B-cell proliferation and activity that—along with that of plasma cells, histiocytes, and macrophages—may explain the generalized lymphadenomegaly, splenomegaly, and typically polyclonal hyperglobulinemia, which are considered the clinical hallmarks of the disease. 9,10 CanL-associated hyperglobulinemia is not protective but detrimental, either directly or indirectly, via the generation of autoantibodies (eg, immune-mediated thrombocytopenia), antihistone antibodies (eg, glomerulonephritis), and/or circulating immune complexes (eg, arthritis) generated in profuse amounts. 28,59,79 The latter are subsequently deposited in various tissues and organs, resulting in glomerulonephritis, vasculitis, uveitis, myositis, and polyarthritis, which predominate over the pathologic and clinical picture of CanL. 73,128 In cold weather, cryoglobulins may also be generated, which precipitate in the blood vessels of the extremities and result in ischemic necrosis. 10 The aforementioned humoral and cellular immune changes may explain why the symptomatic disease can range from a mild papular dermatitis with brisk cellular response to severe nephropathy, leading to chronic renal failure and cachexia. 29 Despite the impressive progress already made, it is still unclear in what way age, sex, nutrition, host genetics, concomitant diseases, multifactorial immunosuppression, parasitic load, virulence of Leishmania strains, and mode of transmission can influence the polarity of clinical manifestations in CanL. 137
The protean clinical picture of the disease is the result of complex interaction between the parasite and the genetic and immunologic background of the host. 80 Although it is well known that different Leishmania species cause different diseases in the dog (L. braziliensis, cutaneous or mucocutaneous disease; L. infantum, generalized disease or more precisely a mixture of cutaneous, ocular, myoskeletal, and visceral lesions), there is no clear evidence that the various strains (zymodemes) of the latter parasite can explain the subtle clinical diversity seen between the Mediterranean and Brazilian CanL. 6,31,73 Regardless of the subtype of the disease, the marked humoral response and the ensuing immune complex deposition in the target organs of the susceptible dog (eg, renal glomeruli, anterior uvea, synovial membrane, vascular wall), as well as the excessive granulomatous inflammation (eg, liver), are most likely a fruitless compensation to cellular immune insufficiency against the parasite, constitute the mainstay of CanL pathogenesis, and explain its wide clinical spectrum observed. 10 In the typical CanL case, history and physical examination may reveal anorexia or increased appetite, lethargy, emaciation, cachexia (Fig. 1), peripheral lymphadenomegaly, exercise intolerance, skin lesions, temporal muscle atrophy, splenomegaly, polyuria/polydipsia, epistaxis, ocular lesions, onychogryphosis, lameness, and vomiting/diarrhea, which appear alone or, more often, in various combinations. 13,22,73 The variability and nonspecificity of clinical signs in CanL make the list of differentials quite extensive, thus necessitating a preliminary investigation for Leishmania infection in the majority of the cases admitted in the endemic areas of the disease. The situation becomes more complicated because the clinical diversity of CanL may also be generated by other vector-borne organisms that flourish in the same geographic areas and may infect any leishmanial dog. 122 This is far more common in dogs with an outdoor lifestyle and in those not routinely treated with ectoparasiticides. 136 Indeed, several epidemiologic and clinical studies have shown that leishmanial dogs can be coinfected by some infectious or parasitic diseases, such as monocytic ehrlichiosis (Ehrlichia canis), granulocytic anaplasmosis, rickettsiosis, bartonellosis, babesiosis, hepatozoonosis, and dirofilariasis. 70,87,97 –99,111,122,128,130,141,143 Because of the clinical and clinicopathologic similarities among all these diseases, including CanL, a cautious diagnostic approach is advised to avoid, as much as possible, reaching erroneous conclusions and witnessing subsequent therapeutic failures in the asymptomatic leishmanial dogs bearing low anti-Leishmania antibody titers. 137
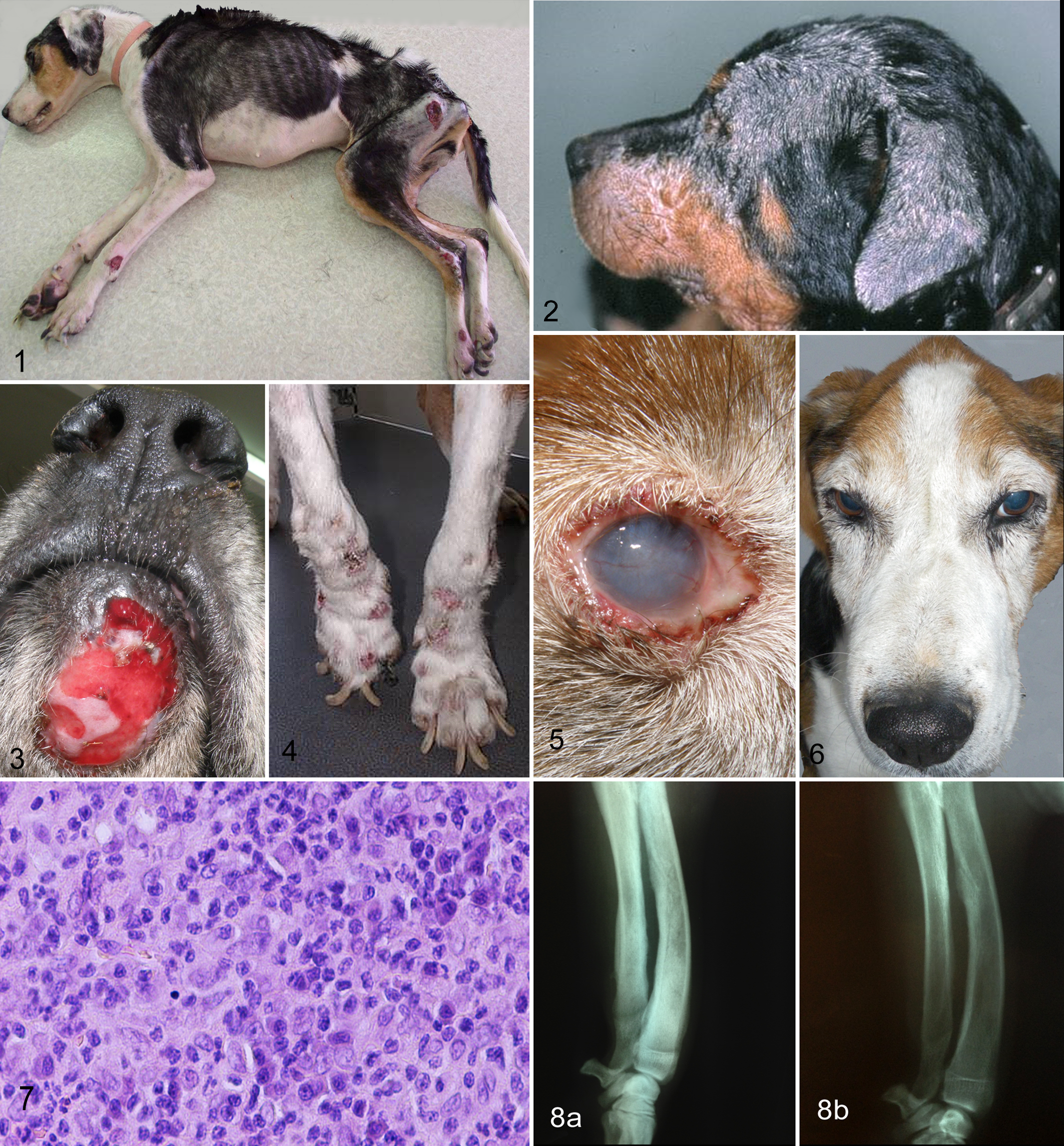
On gross postmortem, apart from the generalized lymphadenomegaly, splenomegaly, and perhaps hepatomegaly, other lesions in the form of light-colored nodular granulomas (skin, liver, kidneys), erosions/ulcerations (oral/nasal/gastrointestinal mucosa, mucocutaneous junctions, skin, synovial membrane), and petechiae/ecchymoses on mucosal and serosal membranes may be seen in some dogs. 10 Joint and bone lesions, usually localized in the appendicular skeleton, are easier to demonstrate on radiographs than on postmortem. 4 The histopathologic picture of CanL in the numerous affected tissues and organs is dominated by pyogranulomatous to granulomatous inflammation and/or immune-mediated injury, imposed by immune complex deposition or autoantibody generation. 43,84 Whereas immune complex deposition is the mechanism for the induction of glomerulonephritis, pyo-to granulomatous inflammation—characterized by infiltration and/or proliferation of macrophages, histiocytes, lymphocytes, and plasma cells—is mainly responsible for the histologic picture demonstrated in lymph nodes, spleen, bone marrow, liver, intestine, bone, genital organs of male dogs, and mucosae. 3,35,38,43,50,72,108,120,153 It is possible that both the aforementioned pathomechanisms contribute equally to the generation of the microscopic lesions observed in the skin, muscle, synovial membrane, and eye structures. 107,109,140,147 In most cases, the granulomatous infiltrate is admixed with a lymphoplasmacytic and/or lymphohistiocytic and occasionally with neutrophilic infiltrate, especially in the early stages of the disease. 3,74,111
The frequently detected peripheral lymphadenomegaly and splenomegaly are the result of lymphoid reactive hyperplasia. 61 However, the ensuing peripheral lymphadenomegaly may be reduced to normal or even hypoplastic levels in the advanced stages of the disease, especially in the nephritic dog. 73 This observation coincides with the notion that in asymptomatic Leishmania-infected dogs, lymph nodes are hyperplastic, but when the disease turns symptomatic, their cortex often atrophies. 61 Splenomegaly is usually not severe enough to be palpated on physical examination, in contrast to what is commonplace with other splenomegalic diseases (eg, ehrlichiosis, babesiosis, lymphoma, splenic torsion) 73 ; spleen enlargement is the result of proliferation and/or infiltration of immune cells and the associated hyperplasia of white and red pulp, along with changes in the microvasculature. 12,43,102 The ineffective immune response of the spleen seems to increase the survival rate of Leishmania parasites, thus explaining the frequently noticed high parasitic load. 41,78 Another organ bearing a high number of amastigotes is bone marrow, the granulomatous inflammation of which goes parallel with lymphoplasmacytic hyperplasia, erythroid and megakaryocytic hypoplasia and/or dysplasia, increased myeloid/erythroid ratio, and erythrophagocytosis, which may contribute to the frequently noticed anemia and thrombocytopenia of leishmanial dogs. 50
The number of parasites in the smears made from peripheral blood of Leishmania-infected dogs are regularly low, in contrast to what has been observed in cultures and by applying molecular techniques. 1,32,48,56,83 In particular, the amastigote load varies in the skin of the symptomatic dogs, either lesional or normally looking, but seems to increase in the preferential feeding sites of sandflies, where Leishmania parasites can be seen in the skin of the muzzle in as many as 70% to 100% of the biopsies, with the aid of immunohistochemistry or immunofluorescence. 107,129,136
Fine-needle aspiration cytology, either direct or ultrasound/endoscopy guided (liver, spleen, colon), is a practical and inexpensive technique to obtain a quick parasitologic diagnosis via the demonstration of amastigotes in various tissues. However, the search for amastigotes by cytology would be unrewarding due to the low or moderate numbers of detectable parasites, even in dogs with symptomatic CanL. 94 Nodular lesions, especially in the skin, exhibit the highest sensitivity compared to other type of lesions (eg, ulcerations, papules, exfoliative dermatitis). 5,104 Histopathology with hematoxylin and eosin is a valuable diagnostic tool in doubtful cases, although the identification of amastigotes is more difficult than with Giemsa cytology. 34,129 Granulomatous inflammation has been associated with higher cutaneous parasitism compared to the other types of dermatitis and regardless of its pattern. 39 To diagnose CanL, the presence of amastigotes should be combined with compatible microscopic lesions, although the opposite can also be true. In the endemic areas of the disease, all those dogs diagnosed as cutaneous sterile pyogranuloma/granuloma syndrome, kerion infection leproid granuloma, acral lick dermatitis, or reactive histiocytosis should be investigated for L. infantum infection with the aid of immunohistochemistry, immunofluorescence, or molecular techniques. 24,37,51,127,131 Immunohistochemistry has a higher sensitivity and specificity than histopathology, but problems arise in those dogs bearing a low parasitic load and because of the confusing effect of background staining. 15,142 In doubtful cases, an appealing alternative could be the application of direct immunofluorescence. 129 Finally, polymerase chain reaction and in situ hybridization may be used, even in paraffin-embedded blocks, because of their higher specificity and sensitivity. 34,89,151
Organ-Specific Pathomechanisms
The main owner complaints to admit sick leishmanial dogs for veterinary consultation have been associated with the skin, renal and/or ocular disease, as well as epistaxis. 73 Skin lesions are perhaps the most common clinical finding, occuring in 80% to 90% of the admitted cases. 73,135 However, the rate of cutaneous disease is possibly even higher, as it has been shown in the biopsies obtained from the clinically normal skin of sick leishmanial dogs. 107,136 Common dermatologic entities associated with CanL have all been described, such as exfoliative dermatitis (which can be alopecic, erythematous/hyperchromic, generalized, regional, or localized; Fig. 2), ulcerative dermatitis (Fig. 3), onychogryphosis (Fig. 4), and papular dermatitis,. 45,74,104 Other, less common, and in some instances ill-defined cutaneous manifestations of CanL include pustular dermatitis, nasal depigmentation, nasodigital hyperkeratosis, paronychia, panniculitis, acral lick dermatitis, alopecia areata or pemphigus foliaceous–like disease, and erythema multiforme. 13,36,60,73 The simultaneous presence of more than one type of skin lesions on the same animal could be attributed to the vulnerability of different areas of the skin to mechanical trauma and/or vascular changes. 43,52 The aforementioned common lesions are typically generated by pyogranulomatous inflammation that targets different structures of the skin and, less often, by immune complex deposition. 128 The presence of parasites in the skin is believed to originate from hematogenous dissemination, with the exception of nodules and papules at the site of Leishmania promastigote inoculation by sandflies. 136 Interestingly, amastigote density has been positively linked to specific areas of the skin (eg, ear pinnae), local cytokine (eg, IL-10, TGF-1) and chemokine milieu (eg, CCD, CC14), and circulating immune cells. 64,90,91 Within the inflammatory infiltrate, the predominating macrophages can be epithelial, vacuolated, or apoptotic. 11,148 Typical cutaneous microscopic lesions are consistent with orthokeratotic or parakeratotic hyperkeratosis, acanthosis, crusting, and ulceration in the epidermis and with inflammation of the dermis and occasionally panniculus, appearing in various histologic patterns (eg, interstitial, lichenoid, nodal, diffuse, periadnexal). 45,74 The periadnexal pattern in particular can be confused with the granulomatous form of idiopathic sebaceous adenitis. 63 However, the observation of sebaceous adenitis in clinically suspicious CanL cases (eg, exfoliative dermatitis), which is not escorted by dermal inflammation, makes the diagnosis of CanL far less possible, even in the areas where the disease is endemic. 11,107 Cutaneous ulcers that have been attributed to local trauma and/or vasculitis are usually localized on the ear margins, pressure points, limbs, and mucocutaneous junctions. 73,74 The focal or multifocal nodular form, characterized by severe granulomatous dermatitis and a high amastigote number, may indicate either inefficient or strong cellular immunity by the host. 13,45,47 This should not be confused with the papular dermatitis that is the result of sandfly bites and has been linked to host resistance; this is based on low antibody titers, positive leishmanin skin test, and a favorable outcome. 104 The histopathology of onychogryphosis, which is clinically characterized by hypertrophy and increased curvature of the claws, has basically revealed a mild to severe lichenoid and interface mononuclear dermatitis, with actual absence of parasites, thus opposing a long-standing hypothesis. 71,115 This clinical entity, as well as the CanL-associated nasodigital hyperkeratosis, may represent a localized variety of the usually coexisting exfoliative dermatitis (excessive buildup of stratum corneum). The pathogenesis of the less common atypical lesions is still obscure and contradictory, perhaps because of the paucity of relevant cases and the wide spectrum of differentials. Finally, the cutaneous lesions of CanL may be overshadowed by coexisting (eg, sarcoptic mange, hypothyroidism, atopic dermatitis, nodular and/or ulcerative neoplasms, systemic or subcutaneous mycoses) or complicating skin diseases, such as bacterial pyoderma, dermatophytosis, Malassezia dermatitis, and demodicosis. 13,18,21,27,44,74,96,141
Renal disease, a common entity and occasionally the sole clinical manifestation of CanL, may progress from asymptomatic proteinuria to nephrotic syndrome and/or end-stage kidney disease (uremic syndrome), and it is the principal cause of death among the affected leishmanial dogs. 29,73,116,118 Since renal disease plays a pivotal role in the prognosis and outcome of these animals, it has been adopted as a major criterion in the clinical staging of CanL by Leishvet. 137 In most cases, the only indication of renal involvement is asymptomatic proteinuria, which, due to its severity, may contribute to muscular wasting and cachexia and occasionally lead to pulmonary thromboembolism. 72,73 CanL nephropathy, though a multifactorial process, is actually the result of immune complex deposition within the structures of glomeruli, secondary to persistent antigenemia and circulating immune complexes generation, eventually culminating in glomerulonephritis. 8 High serum concentration of circulating immune complexes has been associated with impairment of renal function. 79 Interestingly, an increased probability for the generation of glomerulonephritis has been linked to moderate or strong production of antihistone antibodies; 59 T lymphocytes and adhesion molecules may also contribute to renal damage, observed in CanL. 30 Renal histopathology of these nephritic dogs almost always reveals glomerulonephritis, usually mesangioproliferative and membranoproliferative, and tubulointerstitial nephritis, whereas amyloidosis is a rare occurrence. 8,29,72,116,118 Tubulointerstitial lesions appear secondarily to glomerular pathology as well as to immune complex–mediated inflammation and the fibrosis of renal interstitium. 8,29,116,153 This proteinuric nephropathy progressively leads to excretory dysfunction or sometimes to decreased or increased glomerular filtration rate and to systemic hypertension that can establish a vicious cycle, thus amplifying the underlying glomerular pathology. 25,26
Ocular disease is quite common in CanL and can be the only or the main clinical manifestation in 3.7% to 16% of the affected dogs. 22,73,93,110 Ocular pathology in leishmanial dogs has revealed that the granulomatous and lymphoplasmacytic inflammatory infiltration of ocular tissues involves, in order of frequency, the conjunctiva, limbus, ciliary body, iris, cornea, sclera and iridocorneal angle, choroid, and the optic nerve sheath. 109 Anterior uveitis is perhaps the most common manifestation of ocular CanL, and regardless of its chronicity, is characterized by uveal and corneal edema, miosis, fibrin formation in the anterior chamber, and multiple nodules within the iris stroma (Fig. 5). 57,110 Posterior uveitis, usually escorting anterior uveitis, is less commonly diagnosed. Multifocal chorioretinitis is consistent with small hyperreflective foci, retinal detachment, and hemorrhage in the tapetal fundus. 57,93,110 Uveitis may have an immunologic or allergic basis similar to post-kala-azar leishmaniosis of humans and may result in secondary glaucoma and panophthalmitis with permanent loss of vision. 22,40,42,93,123 Of similar importance is keratoconjuctivitis sicca (KCS), which may also appear as a sole manifestation in some leishmanial dogs. 22,93,110 This common ocular entity is usually characterized by purulent and sticky ocular discharge, dry cornea, corneal ulceration, and neovascularization. It is assumed to result from the direct destruction of lacrimal and Meibomian glands due to granulomatous inflammation or obstruction of secretory ducts by the affected adjacent structures. 100 In the same study, meibomian glands were the most commonly affected, thus highlighting the possibility of a qualitative KCS, recently confirmed by the results of a research project in our institution. 144 KCS may also be the result of tear hyposecretion following hypoesthesia of the damaged cornea. 86,152 Furthermore, cataract, granulomatous scleritis, and orbital cellulitis have been reported in a few CanL cases. 22,93,110 Apart from the frequently seen ulcerative, exfoliative, or nodular blepharitis, which is actually a skin problem, retinal detachment due to systemic hypertension and inflammation of intraocular, extraocular, and adnexal smooth and striated muscles are other pathologic entities that have been noticed in natural CanL cases. 25,101,110
The leading causes of epistaxis in CanL—which may be acute or chronic/recurrent, unilateral or bilateral, and sometimes severe enough to pose a threat to the animal’s life because of uncontrollable blood loss—are consistent with thrombocytopathy, increased serum viscosity due to hyperglobulinemia, and rhinitis, ulcerative or not 9,68,73,111 ; vasculitis may also be blamed in some cases and can be expressed as bleeding ulcerations on the nasal philtrum and/or nostrils. In the endemic areas of the disease, epistaxis must be differentiated from that caused by the also common monocytic ehrlichiosis and transmissible venereal tumor. 99 Of the other hemostatic abnormalities described in CanL, platelet aggregation abnormalities—leading to platelet dysfunction, thrombocytopenia, decreased coagulation factor activities most likely due to chronic hepatitis, and fibrinolysis—are the most remarkable. 23,95 Nevertheless, the various hemorrhagic clinical entities seen in CanL cases are more often associated with the inflammatory and/or ulcerative lesions of mucosae, rather than disorders of hemostasis. 42
Of the less common clinical conditions that have been linked or attributed to CanL, muscle disease has a leading edge. Masticatory muscle myositis (Fig. 6) is a rather common entity in CanL-afflicted dogs, which is progressive and may cause severe muscular atrophy but spares masticatory function, in contrast to the clinically similar idiopathic or immune-mediated disease. 73,117 This myopathy has been attributed to lymphoplasmacytic and/or granulomatous, endomysial, and perimysial inflammation and is followed by degenerative, necrotic, regenerative, and fibrotic changes in myofibers, most likely induced by the presence of Leishmania amastigotes and immune complex deposition upon the myofibers, in conjunction with the sporadic neutrophilic vasculitis and serum antimyofiber antibodies. 147 The skeletal muscle polymyositis of CanL—affecting mainly the appendicular musculature and causing progressive muscular weakness and atrophy, lameness, and exercise intolerance—although histopathologically identical to masticatory muscle disease, is actually subclinical, with only a few clinical cases having been reported so far. 69,105,147
Joint disease in CanL is consistent with an erosive or nonerosive mono- or polyarthritis that results from the lymphoplasmacytic to granulomatous inflammation (Fig. 7) secondary to synovial membrane affliction by Leishmania amastigotes and neutrophilic inflammation that follows the deposition of immune complexes. 128 Only 2 studies containing 26 and 38 leishmanial dogs have primarily addressed the problem of arthritis in this disease. 4,140 The uncommon occurrence of this orthopaedic disease in the everyday practice could be explained by its subclinical character and the predominance of its nonerosive type, witnessed in these 2 studies. Although arthritis is the leading cause of lameness in CanL, the possibility of other or additional causes, such as neuralgia, foot pad ulceration, polymyositis, and osteomyelitis, should also be of clinical concern. The latter orthopedic disease, which can also be caused by L. infantum, may lead to more serious locomotor problems due to its destructive nature, exemplified by the osteolytic and osteoproliferative lesions, especially on the radiographs of the long bones, and as a consequence of granulomatous inflammation (Fig. 8a, 8b). 4,17,146 However, a diagnostic dilemma may arise when these radiographic lesions are actually due to bacterial, mycotic, or neoplastic disease in an asymptomatic but Leishmania-seropositive dog.
Oral disease, witnessed mainly in the form of tongue nodules or papules or as multifocal to diffuse ulcerative glossitis and stomatitis, more often seen on the hard palate, is an interesting clinical aspect of CanL. These lesions have been attributed to the migration of infected macrophages to sites of microtrauma in the oral mucosa or to accidental chewing on infected sandflies. 49,75,108,149
Digestive disease in CanL is an uncommon to rare cause to admit leishmanial dogs and is mainly expressed as chronic hepatitis or chronic colitis, the subclinical form of which far outnumbers the clinical one. 3,46,120 In the liver, chronic granulomatous inflammation may involve the sinusoids, portal area, or capsule or become diffuse along with the presence of parasites that may also be seen within Kupffer cells and hepatocytes. 43,120 Hepatomegaly, which is far more often a pathologic than clinical finding, is the result of inflammatory infiltrate, hypertrophy/hyperplasia of resident cells, and possibly passive congestion. 43,62,120 Fibrogenesis in the liver of CanL cases has been positively correlated to parasitic load, clinical status of the infected dogs, and degenerative changes observed in the hepatic parenchyma. 62,88 CanL-induced hepatitis, although informative from a clinicopathologic point of view, is actually a subclinical condition (sensu stricto), as the cases that are admitted with liver failure are very few, at least according to our experience. 120 Nevertheless, in the endemic areas of the disease, it should be differentiated from the more common idiopathic chronic hepatitis and copper- or drug-associated hepatitis. Regarding the colonic disease, the colonoscopy of symptomatic dogs without historical or clinical evidence of chronic/recurrent colitis may reveal the typical lesions of the disease in approximately one quarter of the cases. 3 This observation, taken with the positive correlation between colonic parasitism and colonoscopic gross pathology also noticed in the same study, may justify the differentiation of CanL colitis from the idiopathic, food-induced, and parasitic disease. 46 Finally, in a recent study where the whole gastrointestinal tract of asymptomatic and symptomatic leishmanial dogs was examined histologically, the typical, though mild, lesions of the disease were demonstrated all over its length, along with a high parasitic load, which was more intense in the colon, regardless of clinical status. 114
The relationship between cardiopulmonary disease and L. infantum is rather unclear with its pathological conditions—notably, nonsuppurative myocarditis, fibrinous pericarditis, and pneumonia—being a rare occurrence. 53,135,145 The underlying pathology of CanL-associated meningoencephalomyelitis, which would explain the various neurologic signs observed (eg, seizures, painful and rigid neck, paraplegia), is consistent with a granulomatous and/or neutrophilic meningitis, central nervous system granulomas, spinal hemorrhages, vasculitis, or brain infarcts. 54,58,65,67,150
The increasing evidence regarding the coital vertical transmission of L. infantum worldwide has highlighted the clinical importance of genital disease in the leishmanial dog. 14,103,133 Although the clinical or subclinical disease and genital pathology of the male dog in the form of orchitis, epididymitis, chronic prostatitis causing hematospermia–teratozoospermia, penile granulomatous disease, and balanoposthitis have been indisputably linked to CanL, it seems that L. infantum does not have genital tropism in the bitch. 38,82,92,134 However, coital transmission of the parasite could be achieved by the neoplastic cells of transmissible venereal tumor, a common stray dog disease in many countries. 77 Indeed, the tissue of transmissible venereal tumor in leishmanial dogs usually harbors large numbers of amastigotes, although its histiocytic origin is still a matter of debate. 63,85 Nevertheless, a plausible explanation might be the immunosuppressive role of symptomatic CanL against malignancy. 85
Finally, the most plausible scenario for the diagnosis of some seemingly irrelevant diseases, such as sclerosing encapsulating peritonitis and acute hemorrhagic pancreatitis in leishmanial dogs, is a mere coexistence, at least in our opinion. The reason is that CanL is a progressive disease or a long-standing asymptomatic state, and the infected animals can be affected by any other condition in the same way and, perhaps, frequency as their noninfected cohorts. 2,20
Conclusion
In conclusion, the clinical variability and underlying pathology of CanL actually reflect the combined result of an inefficient cellular immunity that leads to compensatory granulomatous inflammation and an overactive humoral immunity, which eventually result in immune complex deposition on target tissues and/or generation of autoantibodies. Doubt is cast on the various atypical or rare clinical and pathologic conditions associated with CanL, since the cause-and-effect relationship has not always been clear. For a sound diagnosis, the visualization of Leishmania amastigotes in the cytologic and/or histologic slides of lesional tissues obtained from dogs with low antileishmanial antibody titer should be correlated with the typical microscopic and perhaps the clinical picture of the disease or, even better, with a positive response to antileishmanial medication.
Footnotes
Acknowledgments
We express our thanks to Drs Flora Kaltsogianni and Dimitra Psalla for their contribution to the photographic material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
