Abstract
Borrelia burgdorferi is the causative agent of Lyme disease, which is mainly characterized by lameness in dogs. More than 95% of naturally infected dogs are asymptomatic or subclinical; however, in experimental studies, histologic synovial lesions are consistently observed in asymptomatic dogs inoculated with B. burdgorferi. This study investigates the ability of a synovial histopathologic scoring system, clinicopathologic data, and polymerase chain reaction (PCR) testing to differentiate between B. burgdorferi–infected and uninfected dogs. Eighteen 18-week-old beagles were subject to challenge with B. burgdorferi–infected wild-caught ticks (Ixodes scapularis), and 4 uninfected dogs served as controls. Infection was confirmed by serology (ELISA) and PCR amplification of B. burgdorferi–specific DNA of skin biopsies taken at the tick attachment site. A synovial scoring system from human medicine was adapted and implemented on postmortem synovial samples to discriminate infected and noninfected animals. Application of this system to elbows and stifles with a cumulative joint score cutoff > 4 showed a sensitivity of 88.2% and a specificity of 100%, with a positive likelihood ratio of infinity and a negative likelihood ratio of 0.12. Complete blood count, serum biochemistry, urinalysis, urine protein:creatinine, urine PCR, synovial and lymph node cytology, and synovial PCR were evaluated but were not reliable indicators of clinical disease.
Keywords
Lyme disease is a tick-borne disease that incidentally affects dogs and humans. The disease is caused by Borrelia burgdorferi, a small gram-negative spirochete bacterium. 10,25 In the northeastern United States, where the disease is endemic, the main vectors are the hard ticks Ixodes scapularis and Ixodes pacificus. 3
Confirmation of active or subclinical infection is challenging under experimental and natural conditions. Approximately 95% of naturally exposed dogs are asymptomatic, even though up to 90% of dogs are seropositive in endemic areas. 4,11,17,19,20 Although numerous clinical entities have been associated with Lyme disease in dogs, 18 the only disease successfully reproduced under experimental conditions is a self-limiting oligoarthropathy in young puppies. 3,29 In immunocompetent dogs experimentally infected with B. burgdorferi, lameness and synovial swelling are inconsistent and usually observed in only a minority of dogs. However, in a recent study, microscopic synovial lesions were consistently found in all experimentally infected animals, regardless of the presence of clinical signs. 30
The Centers for Disease Control and Prevention standard tests for the diagnosis of Lyme disease in humans are ELISA and immunofluorescence assay. 5 Newer serologic assays used in dogs and humans directed against a synthetic C6 peptide derived from the VlsE antigen have higher sensitivity and a high correlation with natural infection, 1,23 though false negatives still occur. 16
In experimental settings, polymerase chain reaction (PCR) testing has taken the place of bacterial isolation for confirmation of infection, due to its higher sensitivity. Although culture of skin punch biopsies soon after experimental infection are often positive, 26,27 culture of blood, urine, cerebrospinal, or synovial fluid is generally unrewarding for confirmation of infection. 1,9,18,22,24,26 PCR tests have detected B. burgdorferi DNA in both fresh and formalin-fixed, paraffin-embedded tissues (bone marrow, connective tissue fascia, kidney, lung, lymph nodes, meninges, myocardium, pericardium, skeletal muscle, spleen, synovium, and urinary bladder) 6,7,26 but cannot distinguish active infection from residual DNA. 7,23 Immunohistochemical evaluation using formalin-fixed, paraffin-embedded tissues can confirm both natural and experimental canine infection, 7,15 but PCR and immunohistochemistry tests do not always agree. 7 PCR testing of buffy coat preparations or peripheral blood has extremely poor sensitivity in humans and dogs and is not recommended for this reason. 1,28 To our knowledge, PCR testing of synovial fluid has not been evaluated in dogs, but in humans synovial fluid PCR tests have a sensitivity of 80% to 85% in detecting B. burgdorferi DNA when clinical signs are present. 1,22,24
It is challenging to confirm the presence or document the severity of Lyme infection because of the frequent discrepancies between laboratory results and the clinical or pathologic evidence of disease, the lack of readily recognizable clinical signs in adult dogs, and the high prevalence of subclinical infection.
However, in a recent study, even asymptomatic experimentally infected dogs were found to have synovial lesions. 30 Therefore, we hypothesize that histologic scoring of synovial lesions would be a useful method for evaluating the severity of the disease and provide standardization among studies.
In this study, we investigated an experimental canine model of Lyme disease, with the goals of establishing a standardized synovial histologic grading system to confirm infection and evaluate the severity of synovial lesions in B. burgdorferi–infected dogs. The synovial scoring system was compared to other routine clinical laboratory tests, including urine protein:creatinine ratio (UPC), complete blood count (CBC), and synovial and lymph node cytology.
Materials and Methods
Study Design
Twenty-two 18-week-old commercially purchased purpose-bred Beagles (Covance Research Products, Cumberland, Virginia) were used in this study, 18 of which were subject to tick challenge and 4 remained uninfected to serve as controls. Dogs were seronegative for Lyme antigen based on ELISA performed at a commercial diagnostic laboratory (Cornell Veterinary Diagnostic Laboratory, Cornell, New York) prior to tick exposure. Ticks were provided by a commercial supplier (Tick Encounter Resource Center, Kensington, Rhode Island), and the reported infection rate with B. burgdorferi was 66%. Twelve male and 13 female ticks were placed on clipped skin of the left shoulder in a secured plastic capsule as previously described. 2,14,30 Ticks were allowed to feed until detachment (about 4 to 7 days). Animals were euthanized, and necropsy examinations were performed at 90 days postinfection (dpi) (3 exposed and 1 control dogs), 120 dpi (3 exposed and 1 control), and 190 dpi (12 exposed, 2 controls). All procedures performed on the animals were in compliance with protocols approved by the Merial Institutional Animal Care and Use Committee.
Infection with B. burgdorferi was confirmed in tick-exposed dogs by culture and PCR testing of skin punch biopsies taken at 83 dpi as previously described, 7,14,27 as well as by ELISA, as this was sufficient time to induce an immunoglobulin G antibody response. 2 Before humane euthanasia, serum samples were obtained for testing for antibodies to B. burgdorferi, Anaplasma phagocytophilum, Dirofilaria immitis, and Ehrlichia canis with a commercial kit (C6 SNAP 4Dx, Idexx Laboratories, Westbrook, Maine).
At 90 and 120 dpi, EDTA whole blood, serum, urine, lymph node aspirates, and synovial fluid were collected from all remaining dogs under sedation (medetomidine, 0.02 mg/kg intramuscular; butorphanol, 0.2 mg/kg intramuscular). Carprofen (2.2 mg/kg by mouth) was administered upon recovery from the effects of sedation. The following tests were conducted on each dog at days 90 and 120 dpi: CBC, serum biochemistry panel, urinalysis, UPC, PCR testing of urine for B. burgdorferi. Cytologic examination of synovial fluid from the left elbow was performed at 90, 120, 160, and 180 dpi. When lameness and obvious joint swelling were noted on clinical examination, synovial fluid for cytology was collected from the affected joints regardless of the sampling schedule or location; in addition, PCR testing was performed if sufficient fluid was available. Cytology of the left popliteal lymph node was performed at 90 and 120 dpi.
Clinical Observation
After tick exposure, all dogs were observed daily for clinical signs of disease, including lameness, fatigue, or malaise. Rectal temperatures were recorded weekly for all dogs and daily when an animal exhibited signs of disease. Criteria for early humane euthanasia included nonweightbearing lameness, lameness lasting more than 3 days, severe depression, inability to stand, > 10% dehydration, body temperature < 36.6°C or > 40.5°C for more than 2 consecutive days, weight loss > 20%, or if the animals were moribund. Dogs were humanely euthanized by intravenous injection of 65 mg/kg pentobarbital solution (Fatal-Plus) after sedation with 0.2 mg/kg of butorphanol and 0.02–0.04 mg/kg of dexmedetomidine administered subcutaneously.
Confirmation of Infection Status
Infection with B. burgdorferi was defined by a positive PCR result and at least 1 positive serology test (ELISA or sensory nerve action potential [SNAP]). ELISA was performed by the Cornell University Animal Health Diagnostic Center (Ithaca, New York), while PCR, bacterial culture, and SNAP testing was performed by the University of Georgia Athens Veterinary Diagnostic Laboratory (Athens, Georgia).
Necropsy Examination and Tissue Collection
For the first 8 dogs, tissues harvested for histopathology included both elbows, stifles, shoulders, carpi and tarsi, and representative samples of trachea, lung, liver, heart, pericardium, grossly enlarged lymph nodes, thymus, small and large intestine, pancreas, skin near the site of tick attachment and from the contralateral shoulder, tonsils, and kidneys. Based on the preliminary results in the 8 dogs, tissue sampling for the remaining 14 dogs was restricted to elbows, stifles, shoulders, lymph nodes, kidneys, and pericardium. Tissues were placed in 10% buffered neutral formalin for at least 72 hours before processing.
To prepare samples of the major joints, the proximal and distal shafts of the bones were sawed to isolate the unopened joints, and an approximately 5-mm incision was made in the joint capsule to allow penetration of the fixative before the intact joints were immersed in formalin. After fixation, the joints were opened and evaluated for abnormalities of the articular cartilage, and synovial tissue was harvested for histopathology and scoring. To assess the distribution of histologic lesions within each joint, synovial tissues from specific areas were collected. For the shoulder joint, synovial tissue was collected from the cranial joint surface that partially encloses the tendon of the biceps brachii and from both medial and lateral sides of the shoulder joint. Evaluation of the stifle included synovium from the lateral sides of the femoropatellar joint as close as possible to the base of the femoral trochlea and synovial tissue immediately adjacent to the patella, including the fat pad. As the elbow had minimal synovium that could be removed, all the workable tissue was collected. At least 2 cm2 of synovial tissue was examined per joint.
Histologic Evaluation
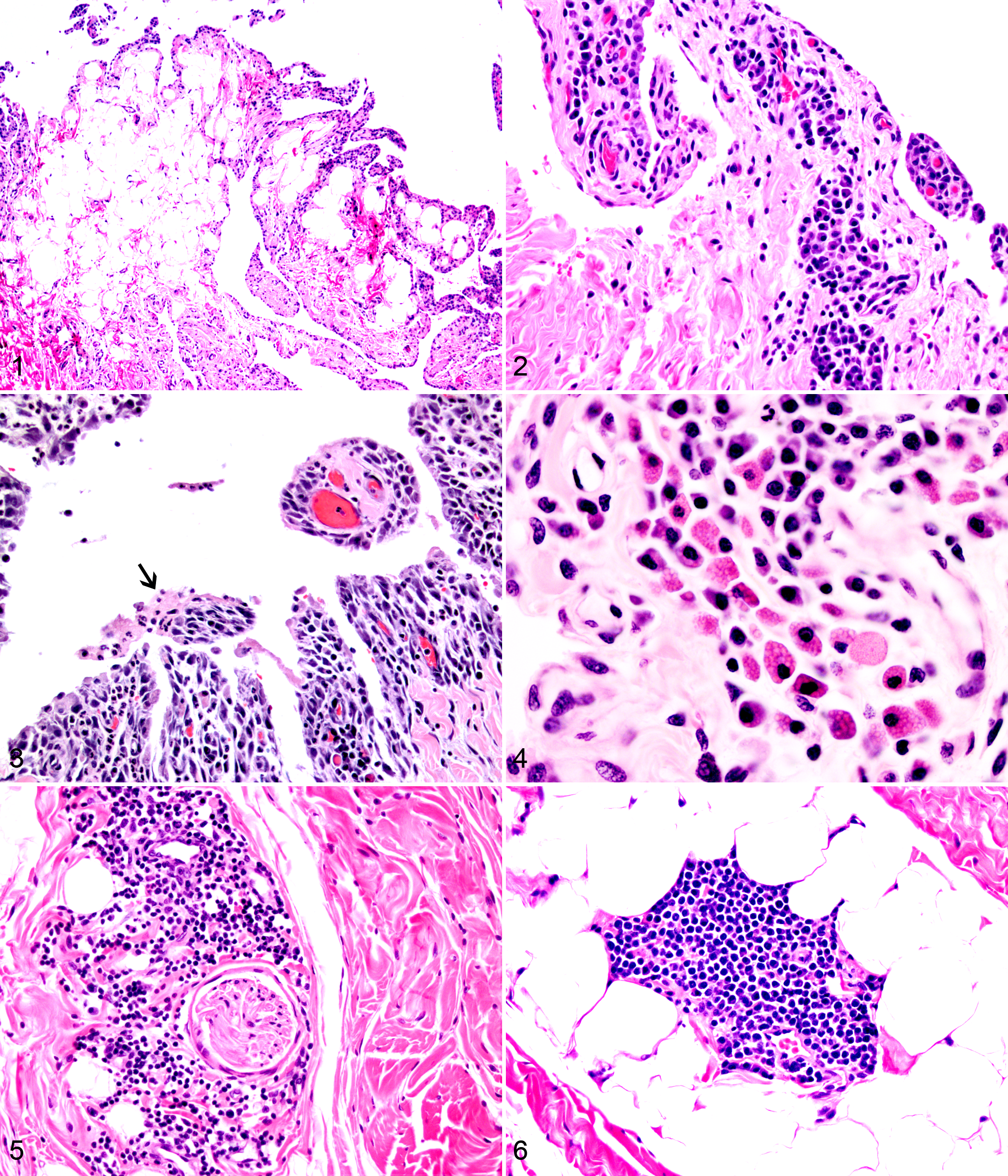
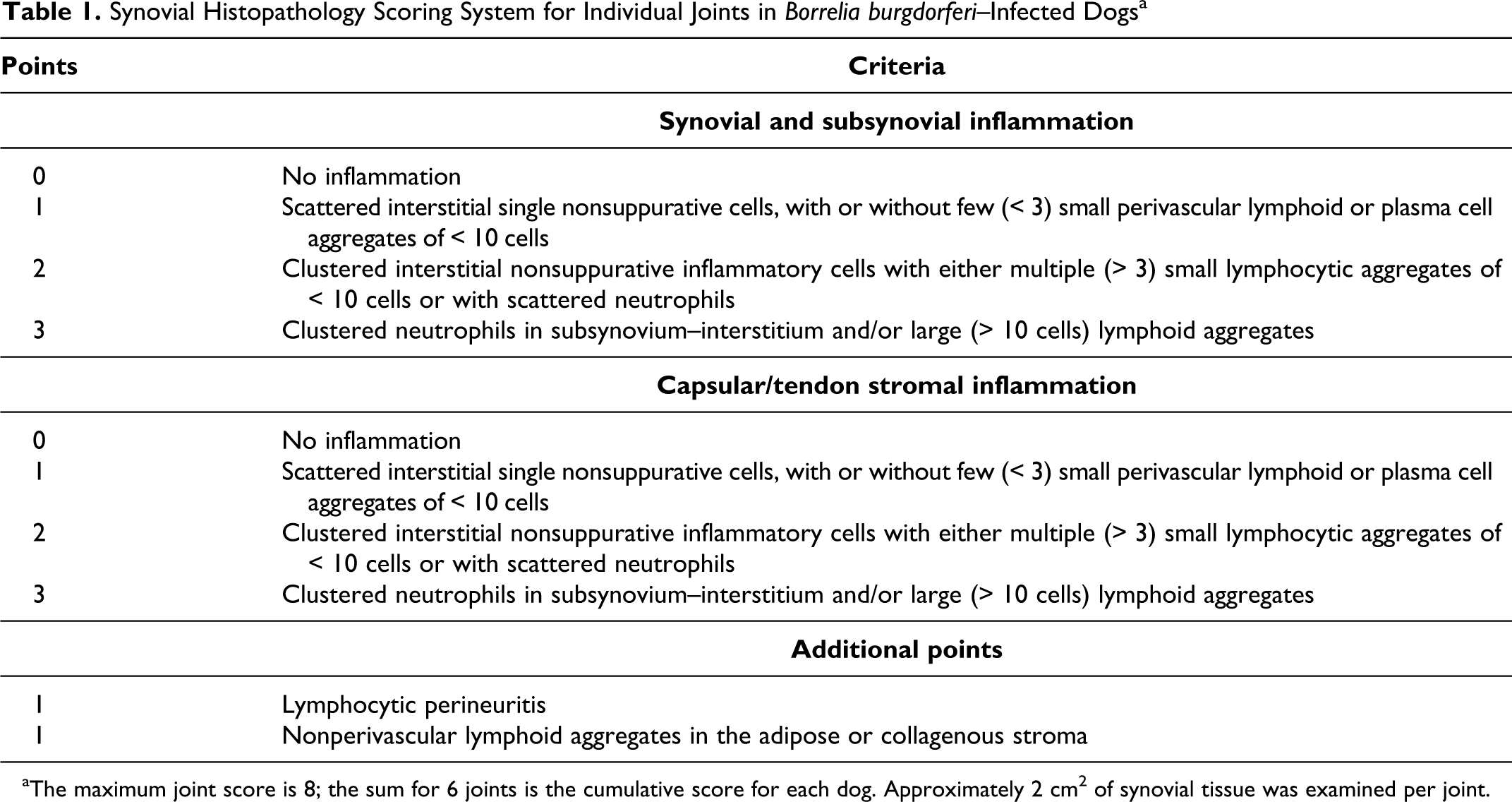
Formalin-fixed tissues were routinely processed to produce 3-μm thick sections that were stained with hematoxylin and eosin. All slides were independently evaluated by 2 pathologists (P.M.K., L.S.), and representative slides were reviewed by a third (E.W.U.) without knowledge of animal identification or infection status. Synovial lesions were graded using a classification scheme based on that for chronic synovitis in humans 13 but adapted to the findings in the first 8 dogs of this study (Table 1; see also Supplemental Figs. 1 –6 of Appendix A, available online at http://vet.sagepub.com/supplemental) since synovial hyperplasia was not identified in any animals. Unlike the human version, the adapted canine scoring system added points for follicle formation and lymphoid perineuritis; thus, the maximum individual joint score was 8. The final cumulative score for each dog was the sum of the findings in 6 joints (right and left shoulders, elbows, and stifles), which made the maximum possible cumulative score 48. At least 2 cm2 of synovial tissue was examined per joint.
Left stifle; canine; dog No. 3. Normal villous synovium corresponding to a synovial score of 0. HE.
Synovial Histopathology Scoring System for Individual Joints in Borrelia burgdorferi–Infected Dogs a
aThe maximum joint score is 8; the sum for 6 joints is the cumulative score for each dog. Approximately 2 cm2 of synovial tissue was examined per joint.
Synovial Fluid
Synovial fluid was collected under sedation from the left elbow and from other joints if these presented with effusion or if fluid could not be harvested from the left elbow. Synovial aspirates were evaluated for overall cellularity, and a 100-cell differential count was performed if sufficient cells were present and there was minimal blood contamination. Aspirates with blood contamination were excluded from analysis. Joints were classified as inflamed if there was ≥ 20% or more neutrophils, borderline if they had 8% to 19% neutrophils, and noninflamed if there were < 8% neutrophils. When available, fluid from more than 1 joint was evaluated from each dog; if any joint was inflamed, the dog was classified as having an inflamed synovial cytology. If sufficient synovial fluid was collected, an aliquot was submitted for PCR testing to a commercial laboratory (University of Georgia Athens Veterinary Diagnostic Laboratory, Athens, Georgia).
Lymph Node Cytology
Aspirates from popliteal lymph nodes were subjectively evaluated for an increase in the number of plasma cells, large lymphocytes, neutrophils, or eosinophils. Lymph nodes with an increased proportion of any of these cell types were classified as reactive.
Statistical Analysis
All tests were performed using Microsoft Excel 2007 (Microsoft, Redmond, Washington). An unpaired Student’s t-test was used to compare results between groups, with significance set at P < .05. After calculating sensitivity and specificity, a receiver operator characteristic (ROC) curve was used for evaluation of different cumulative score cutoffs for diagnostic joint combinations that discriminate between infected and uninfected dogs. A ROC curve graphs the sensitivity (or true-positive rate) on the y-axis and 1-specificity (or false-positive rate) on the x-axis. Without consideration of other factors, the best selected cutoff value is typically the one with the greatest area under the curve and is closest to the top left corner. Positive and negative likelihood ratios were also calculated. Differences were considered significant at P < .05.
Results
Confirmation of Infection Status
Tick attachment was successful in all 18 cases (a summary of the infection status and clinical signs is presented in Appendix B, available online at http://vet.sagepub.com/supplemental). Seventeen dogs exposed to ticks tested positive by PCR and ELISA and were therefore considered infected; B. burgdorferi was cultured in 8 of these dogs. One dog (case No. 5) exposed to ticks had negative results on all serologic and molecular tests to B. burgdorferi and was considered exposed but uninfected, yet it was positive for A. phagocytophilum, indicating that tick attachment was successful. Eleven infected dogs were also positive for A. phagocytophilum on the SNAP test. Two infected dogs (case Nos. 14 and 22) had negative results for B. burgdorferi based on the SNAP test, one of which (case No. 22) was euthanized early due to severity of clinical signs. Both these dogs had negative results for A. phagocytophilum infection. Interestingly, both these dogs were clinically lame and had histologic synovial lesions with very high scores. The 4 control dogs were negative on all tests. All dogs were negative for Dirofilaria immitis and Ehrlichia canis.
Clinical Observations
Of 17 infected dogs, 5 (29.4%) displayed signs of lameness prior to joint aspiration. Of these, 4 (23.5%) developed lameness in only 1 limb, while 1 dog was lame on 2 limbs. Three of these dogs had lameness on the left front limb, which is the limb closest to the site of tick attachment. Of the 5 clinically affected dogs, only 2 showed swelling of the affected joint, and these 2 dogs were the only ones to develop anorexia. In both groups, animals were not febrile, although infected dogs had significantly higher overall mean temperatures (P < .001) over the course of the study. None of the dogs developed weight loss. One dog was humanely euthanized early at 164 days due to severity of lameness, fever, and prolonged anorexia. All other dogs were humanely euthanized as scheduled.
Necropsy
All tick-exposed dogs had enlarged peripheral lymph nodes, especially the prescapular and left axillary, although uninfected dogs had some degree of peripheral lymphadenomegaly. Tick attachment sites were characterized by 2 to 4 erythematous crusty cutaneous lesions (5 mm in diameter) on the left shoulder. Incidental lesions were each observed in one dog—namely, tarsal gland hyperplasia and a subcutaneous lipoma in the region of the left ventral thorax.
Synovial Histopathology and Scoring
The joints in uninfected dogs were mostly normal (Fig. 1) with a few areas of minimal lymphocytic inflammation consistent with a histologic grade of 1. Lesions in infected dogs consisted mostly of perivascular accumulations of plasma cells and lymphocytes within the subintimal space immediately supporting the synovial layer (Fig. 2). Neutrophils were uncommon, usually individual or scattered, and rarely accumulated in small groups. Fibrin was infrequently present, found in only 7 joints out of all evaluated (7 of 132, 5.3%), and was usually localized in small aggregates present in association with neutrophils and in joints with larger plasma cell clusters (Fig. 3). Mott cells were found in only 2 joints—the right elbow of case No. 10 and the left shoulder of case No. 12—and one of these was the only slide with extensive fibrin (Fig. 4). Minimal synovial edema was noted in 2 joints.
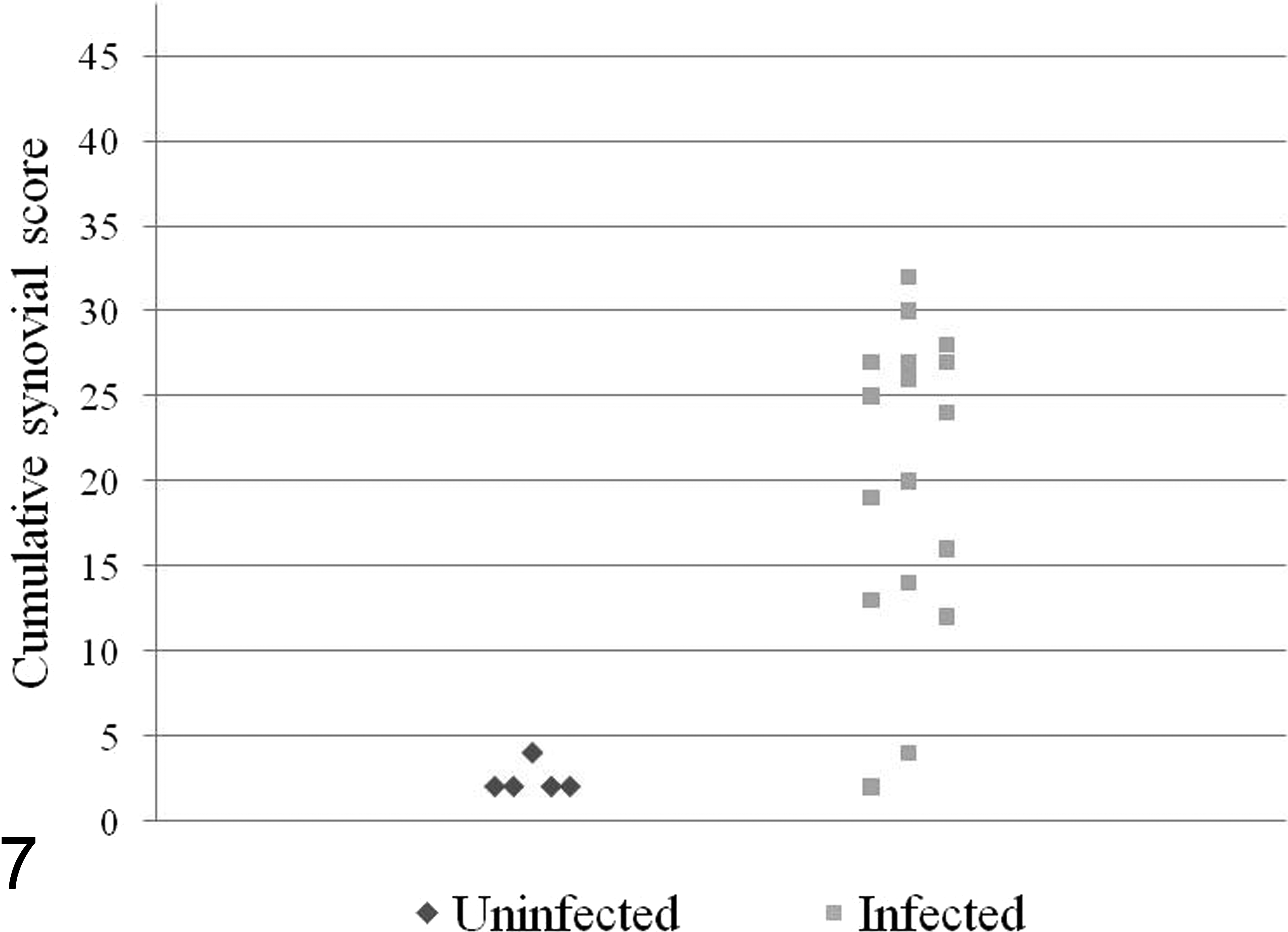
Histogram of` cumulative synovial scores from dogs experimentally infected with Lyme disease
Inflammation was generally more common in the villous synovium but was also frequent in the adipose synovium. Neither synovial erosions nor synovial hyperplasia was observed. The sections of stifle joint containing fat pad had minimal lesions, but no other geographic differences in lesion distribution were found.
The thicker collagenous stroma of the fibrous joint capsule and closely applied fibrous capsule of the tendon sheaths had mostly perivascular plasma cells, lymphocytes, and multifocal areas in which fibroblasts and endothelial cells were hypertrophied. Less commonly, neutrophils were also present. Additionally, clusters of lymphocytes surrounded small nerves in the fibrous layer of the joint capsule (Fig. 5) or formed solitary follicular-like aggregates within the adipose or connective tissue, sometimes adjacent to vessels (Fig. 6).
A histogram summarizing scoring results are presented in Figure 7 (with details provided in Appendix C, available online at http://vet.sagepub.com/supplemental). Based on the grading scheme for synovial lesions, infected dogs had an average cumulative score of 20.4 ± 8.9 (range, 2–32), while uninfected dogs had an average score of 2.4 ± 0.89 (range, 2–4). Two infected dogs had extremely low cumulative scores (scores of 2 and 4), while the next-lowest cumulative score was 12; if those 2 dogs are excluded, the average score of remaining infected dogs (n = 15) is 22.7 ± 6.5. Though there was overlap in cumulative scores between infected and uninfected dogs, 15 of 17 infected dogs (88.2%) had synovial scores of ≥ 12, and all 5 uninfected dogs had cumulative synovial scores of ≤ 4. There was no difference in average cumulative score between A. phagocytophilum–positive and A. phagocytophilum–negative infected dogs.
In descending order, the most severe lesions were observed in the left elbow, left shoulder, right elbow, right shoulder, left stifle, and right stifle, but this varied between dogs, and some joints in infected dogs had scores of 0. As such, evaluation of a single joint was not a reliable indicator of infection. To maximize the efficiency of the histopathologic evaluation, different combinations of joint scores were evaluated to determine if < 6 joints could be used to differentiate between infected and uninfected dogs. The joint combinations evaluated and detailed in Appendix C were as follows: all 6 joints, all left joints (elbow, shoulder, and stifle), all right joints (elbow, shoulder, and stifle), left elbow and left shoulder, left and right elbow, left elbow, left shoulder and right elbow, and both left and right elbows and stifles.
ROC curves were plotted and evaluated for different cumulative score cutoffs for these joint combinations as well as the 6 individual joints. This analysis showed that evaluation of synovium from both elbow and stifle joints (4 joints) provided the highest combination of sensitivity and specificity (the corresponding ROC curve and source data are presented in Supplemental Figure 7 of Appendix D, available online at http://vet.sagepub.com/supplemental). According to this system, dogs with a cumulative score > 4 in all 4 joints (left and right elbows and stifles) were classified as infected, while dogs with a score ≤ 4 in all 4 joints were considered uninfected. At this cutoff, the sensitivity was 88.2%, the specificity 100%, the positive likelihood ratio was infinity, and the negative likelihood was 0.12 for confirmation of infection status.
Histopathology of Other Tissues and Lymph Node Cytology
All the tick-exposed dogs had marked lymph node hyperplasia that appeared to be more severe in the lymph nodes closest to the site of tick attachment. However, some degree of lymph node hyperplasia was also observed in all control dogs. All the tick-infested dogs showed focal lymphoplasmacytic and histiocytic periadnexal and perivascular to interstitial chronic dermatitis, with lymphoid follicle formation, erosion, and serocellular crusting near the site of tick attachment. In all infected dogs, multifocal lymphocytic perineuritis was observed surrounding small nerves in the dermis and subcutis in these areas of tick attachment. Cutaneous perineuritis was not observed with the only dog that was exposed to ticks but was uninfected. Control dogs, not exposed to ticks, did not show any cutaneous changes.
Lymph node aspirates from infected and uninfected dogs showed reactive changes characterized by increased numbers of eosinophils and plasma cells. In total, reactive changes were detected in lymph node aspirates from 11 of 17 (64.7%) infected animals at 90 dpi and 5 of 15 (33.3%) infected animals at 120 dpi. In controls animals, the same reactive changes were observed in 2 of 5 (40%) at 90 dpi and 1 of 4 (25%) at 120 dpi. The rate of reactive nodes was not statistically significant between infected and uninfected groups.
Clinical Pathology
CBC and serum biochemistry panel revealed minor changes consistent with the young age of the dogs, such as occasionally elevated alkaline phosphatase. No other significant abnormalities or differences between groups were found. Urine was collected from 16 dogs (14 infected, 2 uninfected) at 90 dpi and from 17 dogs (14 infected, 3 uninfected) at 120 dpi. Urinalysis results were within normal limits at both time points in infected and uninfected dogs. In 8 of 17 (47.0%) infected dogs, the UPC was higher at 120 dpi than 90 dpi; however, these values were still within normal limits (< 1), and these changes were not statistically significant.
Synovial Cytology
Synovial fluid was successfully collected from 16, 14, and 12 dogs at 90, 120, and 180 dpi, respectively, and results are shown in Table 2. At all time points, diagnostic aspirates from uninfected dogs were not inflamed. Over the course of the study, only 8 of 17 (47.0%) infected dogs had an inflamed or borderline aspirate. The average cumulative histologic score for dogs with at least 1 inflamed or borderline aspirate was 22.1 ± 10.1, while the score for those with consistently noninflamed aspirates was 18.4 ± 7.6, which was not statistically significant between groups.
No. of Lyme Disease–Infected Dogs With Synovial Cytology
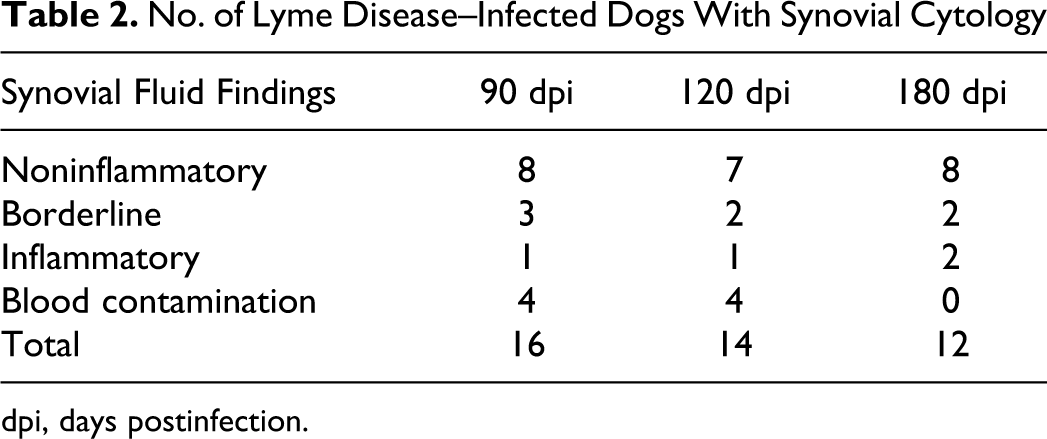
dpi, days postinfection.
PCR Testing of Urine and Synovial Fluid
PCR testing was performed on urine for 16 dogs (14 infected, 2 uninfected) at 90 dpi and for 17 dogs (14 infected, 3 uninfected) at 120 dpi. Collecting sufficient synovial fluid for PCR proved challenging, and synovial fluid from only 4 joints of 3 clinically lame infected dogs were tested by PCR. All PCR results were negative.
Discussion
This article describes clinical pathology, histopathology, and molecular features of dogs experimentally infected with B. burgdorferi, particularly focusing on the synovial histopathology, and presents a method for standardizing the evaluation of a canine model for Lyme borreliosis. Standardization of this animal model is necessitated by the inconsistency of clinical signs and because synovial lesions are found in symptomatic and asymptomatic infected animals. Therefore, the purpose of this study was to develop a standard system for synovial tissue collection and grading joint histopathology. In addition, other minimally invasive clinicopathologic parameters were evaluated.
The technique for collection of synovial tissue—through the isolation and fixation of intact joints and a small hole piercing the capsule to permit entry of formalin within the articular space—allowed for simple and consistent collection of tissue. After fixation and exposure of the entire joint, orientation was straightforward and allowed sampling from specific areas and easy evaluation of the articular surfaces. With one exception, we did not find significant histologic differences in the different topographic region of the synovium, and we concluded that the lesions were evenly distributed throughout the villous and adipose synovium of the joints. The only region that was regularly uninformative was the synovium and fat pad around the patella. In ongoing studies (data not published), the patellar region did occasionally contain the most severe stifle lesions, and inclusion of this sampling site may be helpful.
Overall, histopathologic lesions were mild to moderate, and the predominant cells were plasma cells and lymphocytes, associated with plumped and prominent fibroblasts with basophilic cytoplasm and coarsely to finely stippled chromatin, and, rarely, scattered neutrophils. Small lymphocytic aggregates were occasionally observed. Fibrin exudation and Mott cells were rare. These findings are less severe and only partially consistent with those of a recent article 30 that described experimental Lyme disease in dogs. In that report, numerous dogs—especially when lameness was present—showed neutrophilic and fibrinous synovitis associated with disruption of the synovial layer, whereas subclinical dogs had mainly lymphocytic synovitis characterized by numerous lymphoid follicles. However, in the reported study in which severe lesions were observed, 30 almost all the Beagles were infected at 6 weeks of age, whereas the dogs in our experiment were infected at 18 weeks. This suggests that the age at which the animals are experimentally infected with B. burgdorferi might play a role in the development of the clinical signs and lesions in canine models of Lyme disease.
In our experimental cases, even when lymphocytic and plasmacytic inflammation was present, lymphoid follicles were not a prominent feature, and fibrin was infrequently observed. Mild to moderate deposition of fibrin was most commonly found in joints with higher scores (> 5) and where plasma cell infiltration was prominent. This could be consistent with an immune-mediated disease mechanism, where antigen–antibody complex deposition induces local inflammation and/or fibrin deposition. The presence of Mott cells, though rare, would support this.
This study found that selection of 4 easily harvested joints (elbows and stifles), rather than the original 6 joints (elbows, stifles, and shoulders), did not change the predictive value as determined with a ROC curve. Diagnostic tests must always balance false-positives and false-negatives, and the choice of a cutoff varies with the purpose of the test and the implications of misclassification. 8 In this study, the actual joint scores were skewed far to the right such that most scores of infected dogs were quite high and only 2 infected cases were below the cumulative joint-score selected cutoff > 4. In fact, the next-lowest infected dog cumulative joint score was 12. As such, it was not desirable to lower the cutoff even further to reduce false-negatives at the expense of creating false-positives.
The calculated positive likelihood ratio of infinity confirms that synovial histopathology is an excellent test to confirm Lyme disease in experimentally infected young Beagles. In this case, a negative likelihood value of 0.12 indicates this is a very good but not excellent test to confirm a negative disease status. Overall, a cutoff > 4 is an excellent indicator for confirming disease (positive likelihood ratio = infinity) and is a good but not excellent marker for negative disease status (negative likelihood ratio = 0.12). Additionally, the dogs that presented with lameness were those who had the highest final scores calculated in all the joints, suggesting that the proposed scoring system may be a good indicator of the severity of disease.
Histopathologic lesions were not found in any other examined tissue, aside from skin. All infected dogs showed some degree of lymphocytic perineuritis in the skin adjacent to the tick attachment site. Unfortunately, negative controls were not tick challenged; therefore, it could not be determined if these lesions were caused by infecting B. burgdorferi or simply by tick attachment. Perineuritis was occasionally observed in the synovial membrane of infected dogs; it has been observed in 1 other experimental study of Lyme disease in dogs; 30 and it is commonly described in human Lyme disease. 12 These findings and our observations suggest that cutaneous lymphoid perineuritis might be a feature of B. burgdorferi–associated infection.
The absence of renal lesions on histopathology correlates with urinalysis findings that showed UPC within normal limits. Therefore, neither UPC nor renal histopathology is recommended for verification of experimental infection status. This is consistent with what is reported in literature, in which experimental Lyme disease in dogs has failed to reproduce histologically identifiable renal disease, although UPC has not been measured. 3,23,30
Negative PCR findings on urine samples concur with findings in humans, which have shown that asymptomatic cases are rarely positive, though PCR testing has moderately high sensitivity during active disease. 21 Although PCR testing was performed on only 4 synovial aspirates, none were positive. These aspirates were all inflamed, as they were from dogs with clinical lameness and joint effusion. In human medicine, synovial PCR under these conditions has quite a high sensitivity; 1,24 therefore, the negative PCR results were unexpected. Although the small amount of synovial fluid collected in this study could have been used for PCR testing rather than cytology, PCR has lower sensitivity when arthritis is inactive in humans. 1,24 Whereas further evaluation of the utility of urine and synovial PCR is justified (especially closer to time of infection), neither test can be recommended to confirm infection based on these findings.
CBC and biochemistry have been evaluated only once in experimental Lyme disease, and as in the previous study, neither was useful in differentiating infected from uninfected dogs. 2 In this study, synovial fluid cytology proved to be quite specific but minimally sensitive; it did not correlate with histologic scores; and it was not considered to be a reliable method for verification of infectious status. Similarly, lymph node cytology was neither sensitive nor specific and is not recommended.
Only one dog that was exposed to ticks did not become infected; this dog also had a very low cumulative joint score. Although it is theoretically possible that none of the ticks to which this dog was exposed carried B. burgdorferi, it is more likely to be representative of the natural disease process, where a combination of infective strain and individual animal susceptibility affect outcome. It is interesting that 2 of the 4 clinically lame dogs were positive on the whole antigen ELISA but negative on the SNAP test for B. burgdorferi. According to the manufacturer, the SNAP test has a low false-negative rate, only 2% (sensitivity of 98%), but in our study, 2 of 17 (11.7%) experimentally infected dogs tested negative (sensitivity of 88.3%). The higher false-negative rate in this study may reflect differences between natural and experimental infection, disparity in genetic susceptibility of the dogs, or variability in the infectivity of the strain of organisms.
Overall, histopathologic scoring of the synovial membrane using the proposed grading scheme was a reliable method to confirm experimentally infected animals. Evaluating 4 easily harvested joints and using a cumulative joint score cutoff > 4 had a sensitivity of 88.2% and a specificity of 100% for diagnosis of infection, with a positive likelihood ratio of infinity and negative likelihood ratio of 0.12. Results are applicable only to similar populations under experimental conditions and cannot be generalized to naturally infected dogs, nor are lesions specific for Lyme disease. All other examined tests—including CBC, biochemistry, UPC, synovial and lymph node cytology, and PCR of synovial fluid and urine—were not useful to identify subclinical B. burgdorferi infection.
Footnotes
Acknowledgements
We thank Marsha Royston and Christopher Stephens at Merial for their technical assistance and dedicated care of the research dogs, as well as Rachel Steffens for her assistance with serologic testing.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this project was provided by Merial Limited, Athens, Georgia.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
