Abstract
A high prevalence of leishmaniosis has been reported from an increasing number of domestic and wild mammals around the world. In Australian macropods, Leishmania spp infection has been occasionally described in its cutaneous form only. The purpose of this report is to present a case of fatal visceral leishmaniosis in a captive Bennett’s wallaby in Madrid, Spain, which was investigated by detailed macroscopic, histologic, and immunohistochemical examinations.
Leishmaniosis is a zoonotic parasitic disease caused by an intracellular protozoan of the genus Leishmania (family Trypanosomatidae). Members of the genus are parasites of humans, dogs, and other mammals. This intracellular parasite of the mononuclear-phagocytic system is transmitted by blood-sucking sandflies (Phlebotomus spp). The parasite reproduces itself within the host’s macrophages, causing an intense inflammatory reaction consisting of mononuclear immune cells.6,7 Leishmania infantum is a zoonotic agent that is endemic in Mediterranean countries. L. infantum is the species usually implicated in both animal and human visceral leishmaniosis, a severe systemic form of disease characterized by progressive wasting due to involvement of multiple organs, such as spleen, liver, lymph nodes, bone marrow, kidney, and skin.6,8,9 The clinical presentation of visceral leishmaniosis varies widely, depending on both the organ(s) affected and the extent of functional impairment caused by the infection. Diagnosis of leishmaniosis may be challenging, particularly if the disease appears subclinical. 2
Reports of leishmaniosis in Australian macropods describe only skin lesions.3,7 This short communication describes macroscopic, histopathologic, and immunohistochemical features associated with visceral infection by Leishmania in a Bennett’s wallaby (Macropus rufogriseus rufogriseus) that died suddenly in a zoo in Madrid, Spain.
Pathologic Findings
A captive 2-year-old female Bennett’s wallaby was found dead in its cage. No previous clinical signs were observed. The animal had been housed with others of its species, none of which showed clinical signs suggestive of disease. At necropsy, external examination yielded normal results. The spleen was dark brown to black, 2 times or more normal size, with numerous granulomas, occasionally coalesced, which measured 0.5 to 2 cm in diameter. The liver was uniformly enlarged and dark brown containing white spots and small granulomas (< 0.5 cm in diameter). Pulmonary edema and generalized hyperemia were also observed.
Samples of liver and spleen from the wallaby were submitted in 10% neutral buffered formalin for histopathologic analysis. The referred material was routinely processed and embedded in paraffin wax. Sections 3 μm thick were stained with hematoxylin and eosin (HE), with Giemsa, and by the periodic acid–Schiff method for examination under light microscopy. Histologically, the spleen contained multifocal, variable-sized granulomas with abundant necrosis, cellular debris, and mineralization. Marked lymphoid hypoplasia affecting mainly the periarteriolar lymphatic sheaths was observed. Macrophages located in the splenic marginal zone, within the lymphoid follicles, and surrounding the arterioles contained a high number of cytoplasmic round to oval, 1- to 2-μm-wide, and 2- to 4-μm-long organisms, which had a round basophilic nucleus and distinct bar-shaped paranuclear kinetoplast (Fig. 1). These organisms appeared to reside within cytoplasmic parasitophorous vacuoles. Organisms were accentuated with Giemsa staining but not with periodic acid–Schiff staining. Morphologic features were compatible with Leishmania amastigotes. Occasionally, the lymphocytes were virtually missing, being replaced by plasma cells and parasite-containing macrophages. The red pulp was enlarged with sinus areas diffusely occupied by large macrophages heavily laden with intracytoplasmic organisms, numerous plasma cells, and megakaryocytes. Proliferation of the sinus endothelial cells was also seen. In the liver, a heavy infiltrate composed mainly of macrophages was evident in portal areas and in small foci between hepatocytes (Fig. 2). Macrophages contained numerous amastigotes. Variable numbers of lymphocytes and plasma cells were also observed in the hepatic infiltrates. Megakaryocytes were occasionally observed in the hepatic parenchyma. Capillaries and medium-sized blood vessels in the spleen and liver contained homogeneous acidophilic microthrombi, consistent with disseminated intravascular coagulation.
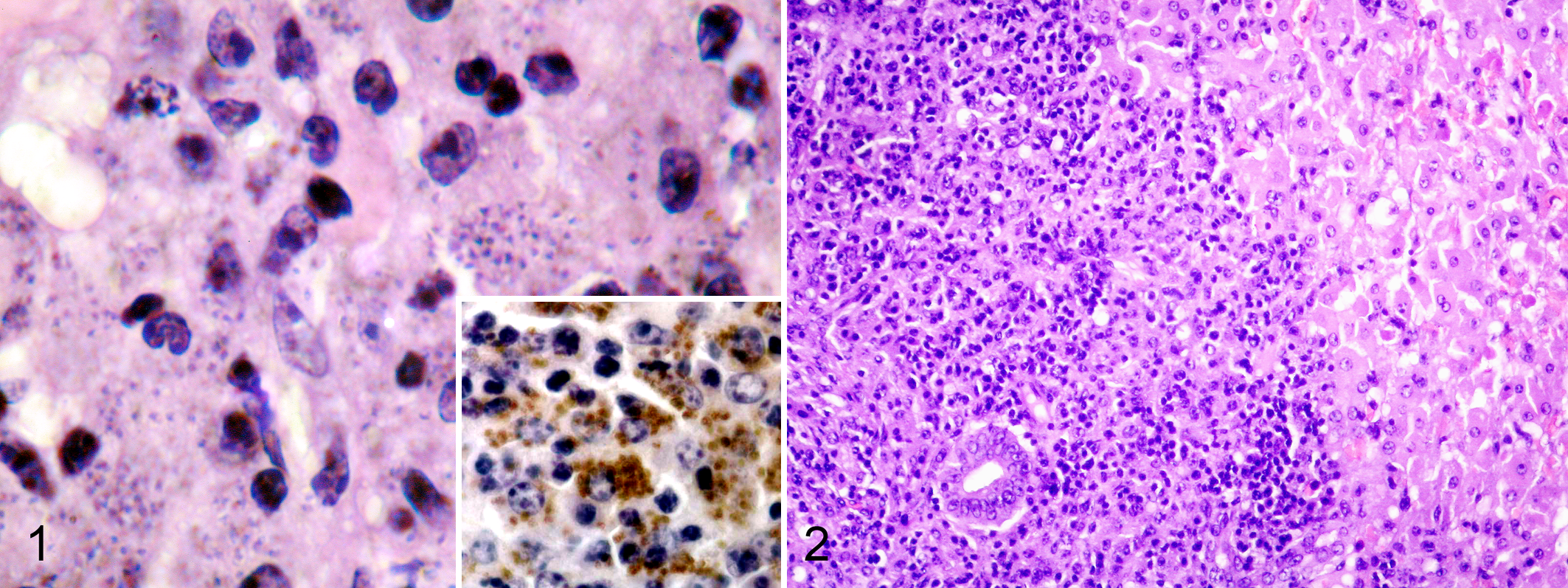
The etiologic diagnosis and identification of parasitized macrophages was greatly facilitated by the immunohistochemical detection of Leishmania antigens. The avidin–biotin–peroxidase complex method (Vector Laboratories, Burlingame, CA) was used. Sections 3 μm thick were labeled with a rabbit polyclonal antibody specific for L. infantum (Departamento de Anatomía y Anatomía Patológica Comparadas, Facultad de Veterinaria, Universidad de Murcia, Spain 4 ). Labeling was detected by incubation with the liquid DAB+Substrate Chromogen System (Dako, Carpinteria, CA). The slides were counterstained in Mayer’s hematoxylin. A sample of skin from a dog with leishmaniosis was used as a positive control. Intracytoplasmic organisms showed a strong positive labeling for the L. infantum antibody in both the spleen (Fig. 1) and the liver.
Discussion
Leishmaniosis includes 3 diseases: cutaneous leishmaniosis, mucocutaneous leishmaniosis, and visceral leishmaniosis. 9 In Australian macropods, clinical signs have been limited to skin lesions consistent with cutaneous leishmaniosis. Australian marsupials seems well adapted to infection, remaining asymptomatic or exhibiting only mild skin lesions that may persist for years.3,7 There are no previous descriptions of visceral leishmaniosis in macropods. The clinical signs of visceral leishmaniosis in the dog are chronic debility, often recurrent oculonasal discharge (with some crusting of the nose), and recurrent diarrhea. There may be mild enlargement of lymph nodes, and the spleen is always enlarged to some extent in the visceral form 9 but at any particular time may not attract clinical attention as with the case presented herein.
The animal died in a state of shock, reflected in large numbers of megakaryocytes in liver and spleen and the presence of disseminated intravascular coagulation. Histologic depletion of lymphocytes accompanied by proliferation of parasitized macrophages has been reported in visceral leishmaniosis associated with defective T-cell-mediated immunity. Factors such as stress or immunosuppressive agents may induce such an impaired immune response.1,4,9 In the animal present herein, lymphoid depletion was evident in the spleen. In addition, females were usually stressed by the males of the colony. Although lymphoid depletion at present case may be the result of an immunologic dysfunction allowing active multiplication and dissemination of the parasite to various organs, unfortunately only samples from liver and spleen were collected for histopathologic exam, and no definitive conclusions can be done regarding the competency of the immune system in this animal. Lymphoid depletion in the spleen could be both a cause and a consequence of systemic parasitism.
The domestic dog is the only reservoir host of leishmaniosis of major veterinary importance6,9; domestic cats might be the secondary reservoir hosts of L. infantum in Southern Europe. 4 Moreover, a high prevalence of Leishmania spp has been reported from an increasing number of domestic and wild mammals around the world, including Spain.5,6,8 Australian macropods are reported to be a reservoir host of Leishmania spp in Australia’s Northern Territory.3,7 Whether captive wallabies function as a reservoir of Leishmania spp in an endemic region such as Spain remains to be elucidated. However, as macropods are not abundant in Spain, it seems likely that their role in amplifying the organism as a reservoir or incidental host would be limited.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
