Abstract
We describe the clinicopathologic features of an arthritis outbreak in sheep induced by small ruminant lentivirus (SRLV), linked to the presence of a new SRLV isolate phylogenetically assigned to caprine arthritis encephalitis virus–like subgroup B2. Thirteen SRLV seropositive Rasa Aragonesa adult ewes were selected from 5 SRLV highly infected flocks (mean seroprevalence, 90.7%) for presenting uni- or bilateral chronic arthritis in the carpal joint. A complete study was performed, including symptomatology, histopathology, immunocytochemistry, immunohistochemistry, in situ hybridization, and microbiology. The carpus was the joint almost exclusively affected, with 10 sheep (76%) showing a moderate increase in carpal joint size (diameter range, 18–20 cm; normal range, 15–16 cm) without signs of locomotion problems and with 3 ewes (23%) showing severe inflammation with marked increase in diameter (21–24 cm), pain at palpation, and abnormal standing position. Grossly, chronic proliferative arthritis was observed in affected joints characterized by an increased thickness of the synovial capsule and synovial membrane proliferation. Microscopically, synovial membrane inflammation and proliferation and hyperplasia of synoviocytes were observed. More positive cases of SLRV infection were detected by immunocytochemistry of articular fluid than of bronchoalveolar lavage fluid. Immunohistochemistry and in situ hybridization also detected positive cells in the subsynovial connective tissue, lung, mediastinal lymph node, mammary gland, and mammary lymph node. All animals were negative for the presence of Mycoplasma or other bacteria in the articular space. The present outbreak likely represents an adaptation of a caprine virus to sheep. Our results underline the importance of the arthritis induced by SRLV in sheep, a clinical form that might be underestimated.
Keywords
Visna-Maedi virus (VMV) and caprine arthritis encephalitis virus (CAEV) cause chronic inflammatory diseases in sheep and goats. 18,23 These viruses have been classically considered as 2 different biological entities, but they show a high similarity, particularly in gag and pol genes. 13 Phylogenetic analysis of proviral sequences has demonstrated natural sheep-to-goat transmission 26,29,32 and also natural goat-to-sheep transmission. 11,25,33 Therefore, VMV and CAEV are considered at present as a single genetic group named small ruminant lentiviruses (SRLVs), which include VMV (VMV-like, SRLV subgroup A) and caprine arthritis–encephalitis virus (CAEV-like, SRLV subgroup B). 27
SRLV can produce 4 main clinical forms: pulmonary, nervous, mammary, and articular. Clinicopathologically, the pulmonary form is characterized by dyspnea, interstitial pneumonia, and cachexia; the nervous form causes ataxia and lymphohistiocytic encephalitis and/or myelitis; the mammary form produces interstitial mastitis and reduces milk production; and, finally, the articular form is characterized by lameness, enlarged carpal joints, chronic proliferative arthritis, and synovitis. 20 The pulmonary, mammary, and nervous forms are the most common in sheep, whereas the nervous and articular forms are more usually observed in goats. 38
In both sheep and goats, the articular form involves the presence of enlarged joints, mainly affecting the carpal but also the tarsal joint and, rarely, other locations, such as the atlantal bursa and the nuchal ligament. 14,19,21 Macroscopically, hardening of the periarticular tissue appears with synovial membrane proliferation with frequent erosions of the articular surfaces. 20 Microscopically, the synovial membrane presents papillary tissue projections into the articular space, with synoviocytes appearing hyperplasic and sometimes detached. The subsynovia and connective tissue are infiltrated by mononuclear cells, mainly composed by plasma cells. In the most severe cases, cartilage and bone destruction can be observed. 9,14,21
Sheep can develop arthritis as a consequence of SRLV infection, 9,19,22 but arthritis seems to be the least frequent clinical form of visna-maedi in sheep. In Spain, the most common forms caused by SRLV infection of sheep are pulmonary, mammary, and nervous, 3,16 with an apparent lack of observations on the arthritic form. However, an enzootic VMV outbreak showing arthritis has been detected in Spanish sheep in flocks with a high VMV seroprevalence. Isolate No. 496 was obtained from this outbreak; its viral genome was fully sequenced and assigned to SRLV genotype B2. The biological properties of isolate No. 496 indicated that it was highly replicative in sheep cells. Specifically, it was rapid in ovine synovial and sheep choroid plexus cells and rapid/high in skin sheep fibroblasts. 11 However, the clinicopathologic features and disease severity of the affected sheep in this outbreak have not been determined.
The aim of this work was to describe the clinicopathologic features in an enzootic outbreak of sheep arthritis in flocks naturally infected by the SRLV B2 genotype, compare them with previously reported SRLV sporadic arthritis in sheep, 9,19,21 and update the knowledge of this process in the light of currently available research methods. This is the first report where an ovine arthritis outbreak linked to a known SRLV genetic type is clinicopathologically analyzed.
Materials and Methods
Animals, Serology, and Tissue Sample Collection
Animals for this study were selected from 5 SRLV-infected flocks (A–E; Table 1) that constituted a total of 3152 sheep with a mean visna-maedi seroprevalence of 90.7% (range, 85%–96%), according to a commercial recombinant indirect ELISA (Elitest, Hyphen Biomed, France). 31 The annual incidence of adult animals with chronic nonresponsive arthritis was in the range of 0.5% to 2% among these flock, as reported by field veterinarians and owners. These flocks were geographically distributed in an area around the longitudes 41° 47′ 31.83′′ N and the latitudes 0° 09′ 26.07′′ W. The study of 24 additional flocks (15 355 sheep) in that area demonstrated a VMV mean seroprevalence of 83.7%. 24
Clinicopathologic Examination of 13 Ewes Showing Chronic Arthritis Linked to a B2 Genotype Small Ruminant Lentivirus.
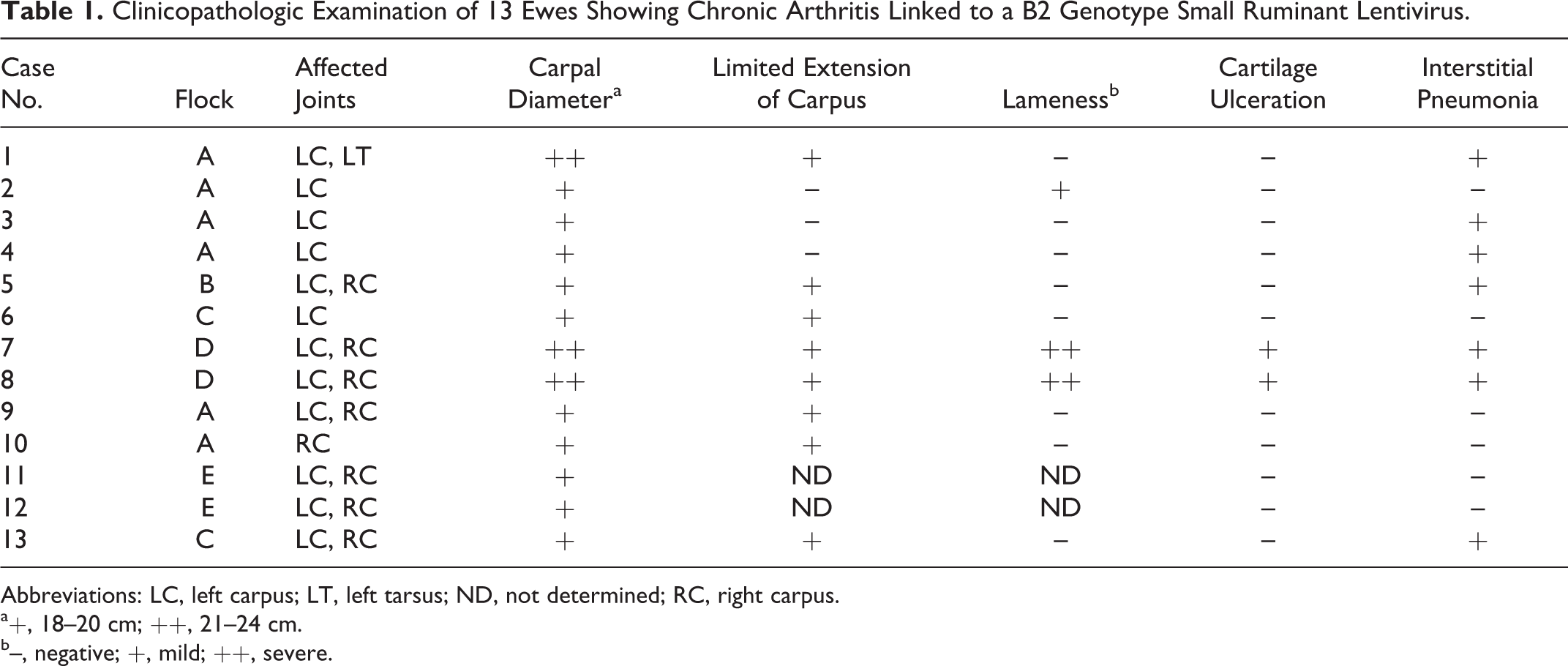
Abbreviations: LC, left carpus; LT, left tarsus; ND, not determined; RC, right carpus.
a+, 18–20 cm; ++, 21–24 cm.
b–, negative; +, mild; ++, severe.
Thirteen Rasa Aragonesa adult ewes (Nos. 1–13) were selected from the 5 flocks, as they presented uni- or bilateral chronic arthritis, mostly affecting the carpal joint. Serum VMV-specific antibody detection was tested by the above-mentioned test. Sheep were also tested for the presence of Mycoplasma spp antibodies (Mycoplasma Agalactiae ELISA, Institut Pourquier, Montpellier, France).
A complete clinical examination with special emphasis in the locomotion system was carried out, with the exception of Nos. 11 and 12. At necropsy, SRLV target tissues were sampled, including joints, lung, mammary gland, central nervous system, and mediastinal and mammary lymph nodes. Articular fluid (n = 10) and bronchoalveolar lavage fluid (BALF; n = 13) were also collected as previously described. 1,17 All procedures were carried out under license approved by the Ethic Committee for Animal Experiments from the University of Zaragoza.
Histopathology, Immunocytochemistry, and Immunohistochemistry
Tissue samples were fixed in 10% buffered formalin for 48 hours and Bouin’s solution for 6 hour. Formalin-fixed tissues were used for histopathologic evaluation with hematoxylin and eosin staining and for in situ hybridization (ISH) studies. Bouin’s solution tissue samples were washed several times in 70% ethanol and used in immunohistochemistry (IHC). All tissue samples were embedded in paraffin wax and 4-μm sections obtained. Articular fluid and BALF were processed for cytocentrifugation, fixed in 4°C acetone, and kept at –80°C for further immunocytochemistry (ICC) studies.
IHC was performed using the Envision system (DAKO, Glostrup, Denmark). Briefly, antigen retrieval was carried out by treatment in a pressure cooker with citrate buffer pH 6 for 3 minutes, and endogenous peroxidase was blocked in 3% H2O2 solution for 5 minutes. An additional protein blocking solution (DAKO) was used to eliminate nonspecific reactions. Slides were incubated with undiluted monoclonal antibody VPM70 30 to the capsid viral protein p25 for 1 hour at 37°C, followed by incubation with Envision Plus (DAKO) for 30 minutes. After color development with diaminobenzidine solution, sections were counterstained with Mayer hematoxylin. ICC studies were performed with the same protocol used for IHC, with the exception of the antigen unmasking that was not used in cytocentrifuges.
The presence of Mycoplasma spp was also investigated by IHC in carpal joint tissues from all the animals with the polyclonal antibody 295 against Mycoplasma agalactiae and with the monoclonal antibody 5A10 against Mycoplasma bovis. 2
In Situ Hybridization
Samples of joint, lung, and mammary gland were tested by ISH as previously described 5 with a viral DNA probe. Briefly, a plasmid containing the VMV gag gene (435 bp) derived from ovine lentivirus strain 85/34 8 was used for the probe construction. The forward 5′-GGAATTGTCTATGGGCATTG-3′ and reverse 3′-TCAAAATCCTCGGACACAAG-5′ oligonucleotides were used for polymerase chain reaction amplification, which was done in the presence of digoxigenin to obtain a labeled probe with the commercial kit Dig-High Prime (Roche Diagnostics SL, Basel, Switzerland).
Rehydrated sections were sequentially incubated with 5 mM levamisole, 0.2 N HCl, 25 μg/ml of proteinase K, and 1 M triethanolamine–acetic anhydrous 0.25%. Prehybridization was done at 37°C via the “hybridization cocktail 50% formamide” (Amresco, Solon, OH, USA). The labeled probe was added at 25 ng/μl and the slides heated at 95°C in a humidity chamber. Hybridization was carried out overnight at 37°C, and slides were washed twice in a stringency solution containing 50% formamide at 53°C. After blocking, slides were incubated for 1 hour with a 1:500 antidigoxigenin alkaline phosphatase–conjugated antibody (Anti-digoxigenine-AP Fab Fragments, Roche Diagnostics SL, Basel, Switzerland) at room temperature and washed. Substrate solution (5-bromo-4-chloro-3-indolyl phosphate nitroblue tetrazolium) was applied to the sections for 2 hours in the dark. Slides were counterstained with nuclear fast red. Positive controls for IHC and ISH consisted of tissues from a serologically positive sheep showing lesions consistent with the presence of SRLV infection. Negative controls consisted of tissue sections from 2 uninfected sheep.
Microbiological Studies
A swab was introduced at necropsy, under sterile conditions, into the articular space of both carpal joints for microbiological analysis by culture in blood and MacConkey agar medium, erysipelas selective medium, 34 and Mycoplasma medium. 10
Results
Serologic, Microbiological, and Clinical Findings
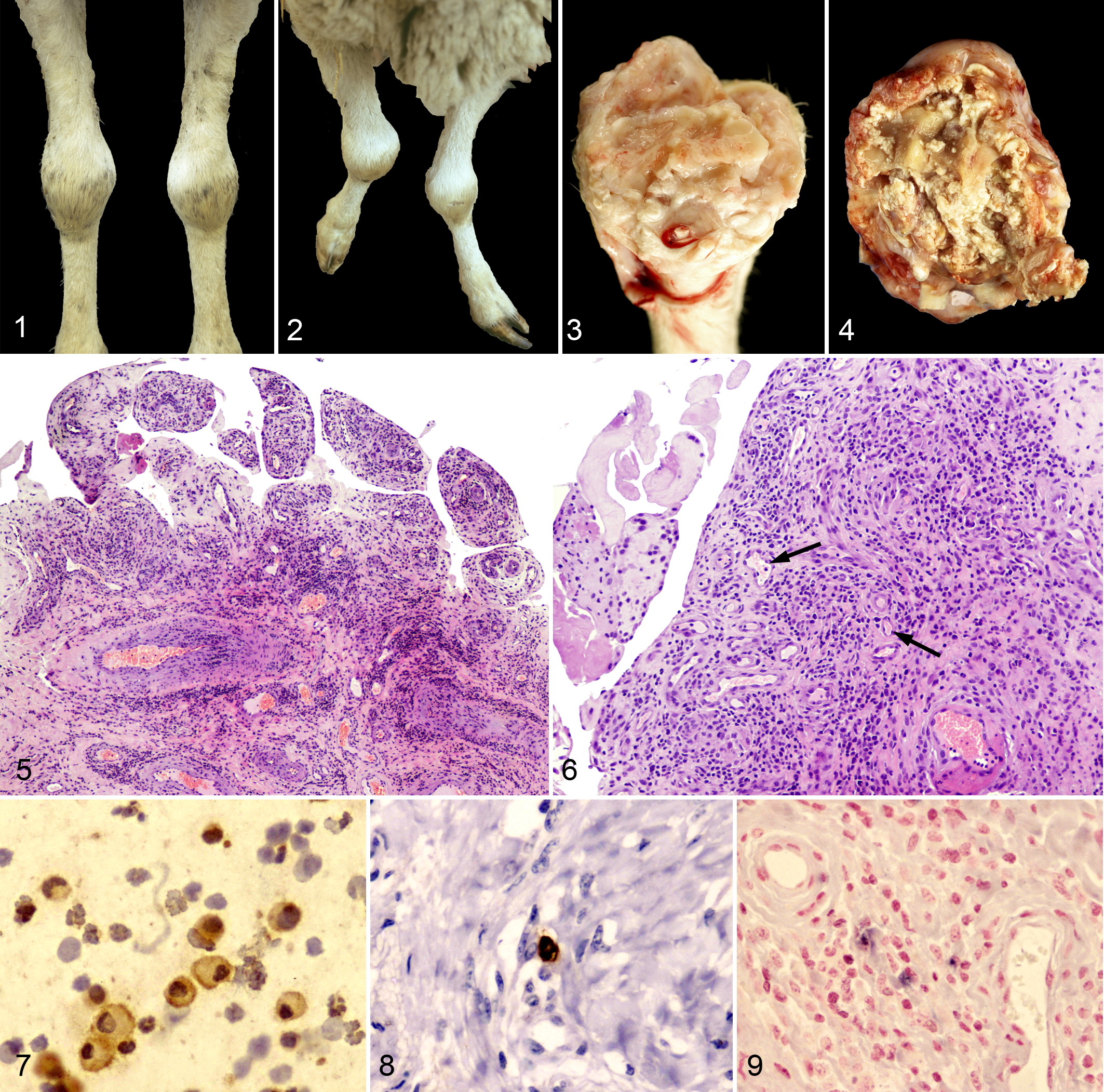
All 13 arthritic animals were seropositive for VMV, whereas antibodies against Mycoplasma were not detected in any sheep. Moreover, Mycoplasma were not detected in any of the joint samples analyzed by microbiological and immunohistochemical means. Clinical findings are summarized in Table 1. The carpus was the joint almost exclusively affected in all 13 animals, whereas only 6 cases (Nos. 1–4, 6, 10) showed unilateral and asymmetrical affection. Ten sheep (76%) showed a moderate increase in carpal joint size (diameter range, 18–20 cm; normal range, 15–16 cm) without signs of acute inflammation or conspicuous locomotion problems (Fig. 1). Only 3 ewes (23%) showed severe inflammation with a marked increase in diameter (range, 21–24 cm), pain at palpation, and abnormal standing position (Fig. 2); severe lameness was observed in only 2 of those animals. In any case, a limited extension movement in the carpal joint was the most remarkable finding in 8 of 11 ewes (72%). No other clinical signs were noted.
Pathologic Findings
All affected animals showed chronic proliferative arthritis in the macroscopically affected joints, characterized by an increased thickness of the synovial capsule with synovial membrane proliferation (Fig. 3). Articular fluid was always scarce, with the exception of sheep No. 1. In severe cases (Nos. 7 and 8), arthritis was observed with erosion and ulceration of articular cartilage (Fig. 4, Table 1). Lung lesions compatible with interstitial pneumonia and chronic lymphadenitis caused by VMV were observed in 7 sheep (54%; Table 1).
The microscopic lesions observed in joints are summarized in Table 2. Chronic proliferative arthritis was characterized by synovial membrane proliferation and synoviocyte hyperplasia with villi-like structure formation, protruding toward the articular space (Fig. 5). Multifocal to diffuse inflammatory infiltrates, mostly characterized by lymphocytes but also plasma cells and macrophages, were located at the subsynovial connective tissue, in the surrounding adipose tissue, and around blood vessels (Fig. 6). Clusters of plasma cells and binucleated or multinucleated syncytia were detected in the connective tissue of the proliferated synovial membrane. Diffuse fibrosis of the capsule and other structures was always found, and in severe cases, tissue necrosis was frequently observed, sometimes with dystrophic calcification and cartilaginous and/or osseous metaplasia (Table 2). In addition, degeneration of arterial tunica media and presence of thrombi within blood vessels (Nos. 7, 12, 13) were also observed in the affected joints. In 2 animals (Nos. 10 and 13), nonamyloid hyaline material was observed in the subsynovial connective tissue.
Histopathologic Examination of the Carpal Joints of 13 Ewes Showing Chronic Arthritis Linked to a B2 Genotype Small Ruminant Lentivirus.

Abbreviations: C, cartilaginous; L, lymphocytes; M, macrophages; O, osseous; PC, plasma cells; PI, perivascular infiltrates; SH, synoviocyte hyperplasia; SMP, synovial membrane proliferation; VF, villous formation.
a–, negative; +, mild; ++, medium; +++, severe.
In all lungs with macroscopic lesions compatible with VMV infection, chronic lymphocytic interstitial pneumonia with hyperplasia of lymphoid follicles and smooth muscle hyperplasia was also found. In the mammary gland, lesions compatible with VMV infection were observed in 10 ewes (76.9%; case Nos. 1, 2, 4, 6–8, 10–13), consisting of mild multifocal to diffuse mononuclear inflammatory interstitial infiltrates composed mainly of lymphocytes and macrophages with hyperplasia of lymphoid follicles around acini and lactiferous ducts.
ICC, IHC, and ISH Findings on VMV-Infected Animals
Table 3 summarizes ICC, IHC, and ISH results on the 13 ewes under study. Immunocytochemical analysis of the articular fluid revealed numerous SLRV-positive cells in 6 of 10 animals (60%; Table 3), with these cells being morphologically consistent with macrophages (Fig. 7). SRLV-positive macrophages were found in BALF from only 2 of 13 sheep (Nos. 2 and 8). On IHC, the antigen retrieval process carried out in severely damaged articular tissue was very problematic, and only 5 animals yielded conclusive results. Of these 5 animals, 3 showed positive cells in the subsynovial connective tissue by IHC (Fig. 8), and 6 of the 12 ewes tested by ISH were positive (Fig. 9). In lung, 5 of the 11 analyzed animals were positive by IHC, but ISH allowed the detection of an increased number of positive results (9 of 11). Only 5 of the 12 mediastinal lymph nodes analyzed by IHC were positive (Table 3). In the mammary gland, a similar number of positive results (about 50%) were observed by IHC or ISH. Finally, the mammary lymph node showed a relevant proportion of SRLV labeling by IHC (7 of 11).
ICC, IHC, and ISH Results in Target Tissues From 13 Ewes Showing Chronic Arthritis Linked to a B2 Genotype Small Ruminant Lentivirus.

Abbreviations: ICC, immunocytochemistry; IHC, immunohistochemistry; IN, inconclusive results; ISH, in situ hybridization; NT, not tested.
Discussion
In this work, we describe the clinicopathologic features of a SRLV-induced arthritis outbreak in sheep, linked to the presence of a new SRLV isolate assigned phylogenetically to CAEV-like subgroup B2. In previous work, partial B2 genotype SRLV sequences were obtained from 6 of the animals used in the present study (Nos. 2, 4–6, 8, 10), and viral isolation from a further sheep (No. 3) confirmed the outbreak to be caused by a B2 genotype SRLV. 11 Given the similar pathologic features among them, the geographic clustering, and the presence of SRLV-infected goats in the 5 flocks studied, it is assumed that this pathology represents an outbreak caused by a interspecies transmission of SRLV between goats and sheep.
SRLV-induced arthritis was first described in the United States, 9,21,22 and it has been observed in several European countries, mostly as incidental cases related to individual flocks. 1,12,36 However, this is the first description where sheep lentiviral arthritis is observed within a geographically related group of flocks in a highly infected area, as the individual SRLV seroprevalence mean was higher than 80%. Moreover, incidence of clinical cases of SRLV arthritis in that area has been estimated at a remarkable annual rate of 1% to 2% per flock, thus constituting an arthritic outbreak. Visna-maedi is a prevalent infection in certain Spanish areas and causes a high incidence of the respiratory, nervous, and mammary forms, but the arthritis has never been reported. 3,24 There might be several reasons to explain the lack of detection of the arthritic form. First of all, the clinical findings in SRLV-induced arthritis in sheep in the present work are generally mild, as only 3 of 13 animals (23%) showed lameness and, consequently, the increase in size of the carpal joint is frequently overlooked. If observed, the “big knee” syndrome is often attributed to bacterial infections, mostly to infection by Mycoplasma spp, 4 an attribution that has not been demonstrated in our work. The erroneous assumption of Mycoplasma or other bacterial infections can partly explain why such a prevalent SRLV form has not been previously detected in our region. It is therefore possible that this form is also present—but undetected—in other SRLV-infected flocks of sheep.
The occurrence of this form is most likely related to the type of virus and to interspecies transmission of SRLV between goats and sheep. Sequences already obtained in a previous work from animal Nos. 2, 4, 8, and 10, included in the present article (gag: Genbank FJ187802–FJ187812; LTR: Genbank FJ187813–FJ187820), and unpublished viral sequences of animal Nos. 5 and 6 confirmed the assignment of the infecting viruses causing the outbreak to the B2 phylogenetic group within SRLVs. Additionally, a representative strain of the outbreak was isolated from sheep No. 3 (strain 496), fully sequenced (Genbank FJ195346) and characterized, reinforcing the B2 linking of the infecting virus, 11 closer to other CAEV-like SRLVs than to classical VMV-like lentiviruses. This is in contrast with other previous isolates in Spain that belonged to A and D VMV-like groups. 12,29 In the local semi-intensive production system, a certain number of goats are always kept in the majority of flocks admixed with sheep to feed orphan lambs or lambs from ewes with mastitis. Interestingly, the genome of isolate 496 was CAEV-like B2 except at the integrase region of the pol gene, where it was VMV-like. Possibly, an originally goat SRLV could have been transmitted to sheep, likely by the respiratory route 35 or by milk, adapted to the new host by acquiring the integrase VMV region, an adaptation that helped in further integration of the viral genome into the host DNA and the cross-species barrier transgression by the virus, resulting in a pathology similar to that observed in CAEV. This adaptation and subsequent spread could have been favored by the high viral in vitro replication demonstrated for the strain 496. 11 In this context, another favoring factor would have been the high viral pressure present in flocks (as indicated by the level of seroprevalence), but to the best of our knowledge, it could not explain the arthritic affection alone.
The SRLV-induced arthritis observed in this work affected adult sheep only, and the clinicopathologic affection was similar to previous descriptions. 9,21 Lesions in carpal joints were either uni- or bilateral, with only 1 case being affected at the level of the tarsal joint. The age of the animals in our work ranged between 4 and 8 years old, apparently higher than ages previously described, 14 but this might also be a reflection of the lack of knowledge on the clinical evolution for these 13 individuals.
Macroscopically, lesions were similar to those previously-described. 21,37 The most remarkable gross changes were the proliferation of synovial membrane, the thickening and fibrosis of the articular capsule, and, in specific cases, a slight increase of the volume of intrarticular fluid. Erosion and ulceration of articular cartilages appeared in only the most severe cases. Microscopically, lesions were considered moderate and were characterized by proliferation and inflammation of the synovia, forming tissue projections protruding into the synovial space. No lymphoid follicles were observed, but this is a very unusual finding in this form, 36 except upon experimental infection as observed in goats. 6
Variable amounts of synovial fluid obtained from 10 animals and 6 samples showed VMV-infected cells by ICC, sometimes in relevant proportions. This does not agree with previous findings reporting a very low number of infected cells in the articular fluid. 15 This might reflect the rapid/high phenotype of the infecting CAEV-like B2 strain. Our data suggest that IHC of synovial fluid might be a simple, although of limited sensitivity, diagnostic approach that may be used to confirm the in vivo diagnosis of this Visna-Maedi form. Both IHC and ISH in tissues detected positive cells that were morphologically compatible with macrophages. 7 These techniques confirmed the presence of SRLV in sheep joints in most of the cases but had a limited sensitivity likely reflecting technical limitations or other difficulties in detecting genotype B sequences and/or antigens.
Other pathologic findings showed that 53.8% of animals had gross or microscopic lesions compatible with the pulmonary form of VMV whereas 76.9% showed lesions in the mammary gland, thus confirming that a single animal may show several forms of infection. This could indicate a polyclonal infection through multiple contacts with infected animals or a microevolution from a single viral strain within tissues. Phylogenetic relationships among SRLV sequences from different tissues in a genotype A infection showed compartmentalization by tissue as a result of a microevolution after infection with a single strain. 28
In conclusion, we have studied and described an outbreak of the arthritic form of SRLV in sheep in a highly infected area linked to infection by a CAEV-like subgroup B2, with a VMV-like integrase in its genome. This virus likely represents an adaptation of a caprine virus to sheep, maintaining the capacity to trigger arthritis—reproducing the main clinical finding in goats—upon the transgression of the species barrier. The clinicopathologic picture described shares features with previous descriptions in sheep. The data set obtained in our work links these findings exclusively to SRLV infection, not concurrent infection by Mycoplasma or other bacteria. Our results underline the importance of the arthritis induced by SRLV in sheep, a clinical form that might be underestimated and perhaps not properly diagnosed.
Footnotes
Acknowledgements
We thank Azoye Castro and the Unidad de Anatomía Patológica Veterinaria, Las Palmas de Gran Canaria University, for performing the immunohistochemical tests for Mycoplasma. We also thank S. Becerra and R. Puyó for technical support and J. Medina for professional help with the figures. We acknowledge the use of Servicio General de Apoyo a la Investigación-SAI, University of Zaragoza.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorshipm and/or publication of this article: This work has been partially funded by a grant of the Ministerio de Economía y Competitividad (AGL2010-22341-C04-02 GAN). E. Salazar was a PhD student funded by a grant from Gobierno de Aragón.
