Abstract
In situ detection of ovine progressive pneumonia virus (OPPV) and the phenotypic identification of the cells that harbor OPPV have not been described for the OPPV-affected tissues, which include lung, mammary gland, synovial membranes of the carpal joint, and choroid plexus of the brain. In this study, the authors first developed a single enzyme-based automated immunohistochemical (IHC) analysis for detection of OPPV capsid antigen (CA) on OPPV-affected tissues, using 2 anti-CAEV CA monoclonal antibodies, 5A1 and 10A1, and 2 enzyme-based IHC systems. Out of 10 naturally and persistently OPPV-infected ewes, OPPV CA was detected in intercellular regions of the carpal synovial membrane of 1 ewe, in cells resembling alveolar macrophages and pulmonary interstitial macrophages in lung tissue of 3 ewes, and in mammary alveolar cells of 1 ewe. Furthermore, dual enzyme-based automated IHC analyses revealed that OPPV CA was predominantly detected in CD172a- or CD163-positive alveolar macrophages of the lungs and mammary gland. That anti-inflammatory (CD163) and downregulatory (CD172a) types of alveolar macrophage harbor OPPV CA leads to the possibility that during persistent infection with OPPV, the host alveolar macrophage might serve to limit inflammation while OPPV persists undetected by the host adaptive immune response in the lung and mammary gland.
Keywords
The small ruminant lentiviruses (SRLVs), including ovine progressive pneumonia virus (OPPV), maedi-visna virus (MVV), and caprine arthritis-encephalitis virus (CAEV), are phylogenetically similar and are part of the Retroviridae family, which includes human immunodeficiency virus (HIV). 54 Clinical signs of SRLV include dyspnea, firmness of the udder and/or reduced milk production (mastitis), swollen joints and lameness (arthritis), cachexia, and ataxia. Clinical signs can vary in animals; therefore, the reference standard for evaluating the extent of disease caused by SRLVs is postmortem histological assessment of lung, mammary gland, carpal synovial membrane, and choroid plexus. 7,11,13 Definitive diagnosis of SRLV infection cannot be made on the sole basis of histopathology; as such, serological (enzyme-linked immunosorbent assay [ELISA]) and/or molecular (polymerase chain reaction [PCR]) diagnostic tests are used to establish the presence of infection. 15
The SRLVs are considered immunopathological diseases, meaning that subsequent immune responses to the virus are thought to contribute more to the pathology of the disease than to the virus alone. 12 The histological progression of SRLV-induced pathology in the tissues is thought to follow a general pattern whereby there is increased lymphoid follicle development and mononuclear and plasma cell infiltration, as followed by edema and necrosis of tissues in severe cases. In MVV-infected sheep, more MHC class II–positive cells and CD8-positive cells are observed in the bronchoalveolar lavage fluid (BALF) than in uninfected sheep. 35 In addition, CAEV-infected goats experiencing arthritis show increasing numbers of CD4-, CD8-, MHC class II–, and CD45R-positive/CD5-negative mononuclear cells in their synovial membranes compared to those of CAEV-infected goats not experiencing clinical arthritis. 53 Combined, these studies indicate that there is mononuclear and plasma cell infiltration in the affected tissues of lung and carpal synovial membranes of SRLV-infected goats and sheep.
Detection of SRLV antigens in situ with immunohistochemistry or immunocytochemistry gives a snapshot of the local immune cell environment of these SRLVs. Several single-label immunohistochemical (IHC) analyses have been developed to detect OPPV, MVV, or CAEV proteins. SRLV capsid antigen (CA) has been detected in lung, bronchoalveolar lavage cells, mammary gland, lymph nodes, carpal synovial membrane, heart, liver, lymphoid tissue associated with the third eyelid, bone marrow, and the central nervous system, 4,5,8,10,19,21,37,51 with the mouse monoclonal antibodies (MAbs) 3F 37 generated to OPPV 85/34, 1A7 19 generated to MVV, and VPM70 49 generated to MVV EV1 CA or a rabbit polyclonal antibody 51 raised to a recombinant CAEV T9 CA protein. MVV CA has also been detected in tissue sections of kidney, gastrointestinal tract, and lung 2,46,47 of sheep, with the anti-CAEV-63 CA MAb 5A1. 38
Successful detection of SRLVs in tissues depends on a number of factors, including testing of several different antigen retrieval conditions and whether the antibody will perform on formalin-fixed, paraffin-embedded tissue sections even after antigen retrieval. Besides these obstacles to overcome, an important factor to consider is amino acid sequence similarity between the SRLV found in the natural host and the SRLV used to generate monoclonal or polyclonal antibodies. Unfortunately, the mouse hybridoma cell line for the anti-OPPV 85/34 MAb 3F no longer existed when we initiated this study (J. DeMartini, personal communication). The anti-MVV MAb 1A7 was generated to a European MVV; the anti-MVV MAb VPM70 was generated to MVV EV1 CA; and the anti-CAEV-63 CA MAbs 5A1 and 10A1 were generated to CAEV-63 CA. Because our prior research revealed that the envelope gene from US OPPV strains has higher amino acid sequence identity to US CAEV strains than does the European MVV strains, the anti-CAEV CA MAbs 5A1 and 10A1 were utilized to examine OPPV CA in the tissues of sheep of US origin (note that the envelope gene encodes the surface envelope glycoprotein and is one of the most variable genes of the SRLV genome). 26
Only a couple of studies have identified the phenotype of cells that harbor SRLV antigen. MVV gp135 was found to colocalize with rabbit anti-sheep alveolar macrophage polyclonal sera in monocyte-derived macrophages from blood. 20 Another study showed MVV p15 colocalization with the macrophage marker, VPM32, on leukocytes from BALF. 34 To our knowledge, immune cell phenotyping of OPPV-infected cells in affected tissues of naturally OPPV-infected sheep has not been described. Such studies may allow for the possibility of identifying one unique immune cell phenotype common to all affected tissues, as well as a better understanding of SRLV pathogenesis and persistence.
SRLVs are found in cells of the monocyte/macrophage lineage. 20 After initial assessment of a few antimacrophage MAbs on sheep tissues, we chose to analyze CD163 and CD172a in tissues affected by OPPV. CD163 is a member of the group B scavenger receptor cysteine-rich superfamily, which binds haptoglobin–hemoglobin (Hp-Hg) and free hemoglobin (Hg) and is considered a marker of an anti-inflammatory phenotype. 31,32,36,50 The main function of CD163 is as an anti-inflammatory molecule by binding to Hp-Hg with high affinity and to free Hg with low affinity, by facilitating endocytosis of Hp-Hg and Hg, and by degrading oxidative and reactive forms of heme. More recently, human CD163 has been found to participate as a surface receptor for recognition of Gram-positive and Gram-negative bacteria, and it has been described as part of the receptor for porcine reproductive and respiratory syndrome virus. 9,18 In humans, rats, or mice, CD163 has been detected on macrophages in lung, lymph nodes, thymus, spleen, skin, bone marrow, liver, synovial membrane, and brain. 17
CD172a or SWC3 is part of the signal-regulatory protein alpha family, 30 and it binds to CD47 via its immunoglobulin domain. The binding interaction with CD47 on leukocytes is thought to protect CD172a-positive macrophages from elimination. 3 CD172a also binds lung surfactant proteins A and D, resulting in a down-regulating signal for the macrophage. 29 CD172a is involved in down-regulating macrophage functions; that is, phosphorylation of tyrosine residues in inhibitory motifs of the cytoplasmic domain of CD172 leads to an association with tyrosine phosphatases (SHP-1 and SHP-2) that negatively regulate signal transduction cascades. 1 CD172a is expressed in monocytes/macrophages, dendritic cells, and neurons of humans, rats, cattle, and pigs. 52 CD172a has been detected in sheep lung, skin, lymph cells, lymph node cells, monocytes and granulocytes of peripheral blood leukocytes, and lymphoid follicular areas. 22,24,40,43
In this study, we first developed 2 enzyme-based automated single IHC analyses utilizing MAbs 5A1 and 10A1 to detect OPPV CA in situ in the lung, mammary gland, carpal synovial membrane, and choroid plexus of ewes naturally and persistently infected with OPPV. Furthermore, we developed automated dual IHC analyses to codetect OPPV CA in situ with the cluster-of-differentiation (CD) markers for macrophage/dendritic cells: CD163 and CD172a.
Materials and Methods
Animals
Ten naturally infected ewes (7 to 9 years of age) were defined as OPPV infected as measured by a commercially available serological based diagnostic test, CAEV cELISA (VMRD Inc, Pullman, WA), and the presence of ovine progressive pneumonia provirus in peripheral blood leukocytes. 25,28 These 10 OPPV-infected sheep had been infected for at least 3 years before euthanasia. Three 6-year-old OPPV-negative ewes were used as uninfected specific pathogen-free ewes and were defined by a negative cELISA test result and undetectable ovine progressive pneumonia provirus in peripheral blood leukocytes. Both OPPV-infected and uninfected sheep had scrapie-resistant genotypes or came from flocks known to be free of scrapie. Procedures used on the ewes were performed in accordance with the Institutional Animal Care and Use Committee at Washington State University.
Western Blot Analysis
Field isolate OPPV LMH17 was produced in goat synovial membrane cells using previously published methods. 25 Western blot analyses of OPPV LMH17 using MAbs 5A1 and 10A1 was conducted as previously described. 16
Tissue Processing and Histological Assessment of Tissues
The following were collected immediately postmortem, fixed in neutral buffered 10% formalin for at least 24 hours, and processed routinely: right and left cranial, middle, and right and left caudal lung lobe; right and left udder glandular and streak canal tissue of the mammary gland; right and left synovial membranes of the carpal joint; and the fourth ventricle region of the brain containing the choroid plexus. Sequential 3-μm-thick paraffin sections were individually placed onto SuperFrost/Plus slides (Fisher Scientific, Pittsburgh, PA), dried overnight at room temperature, and heated for 35 minutes at 56°C in a dry oven. Thereafter, slides were either deparaffinized in Clearite or xylene, rehydrated through a series of alcohols and water for histological assessment, or processed by the automated Benchmark Staining Module (Ventana Medical Systems, Inc, Tucson, AZ) for IHC analysis as follows. Processing of tissue sections for histological assessment and the scoring system for histological assessment of OPPV-related lesions in the tissues have been described elsewhere. 27
Single IHC Analyses
An indirect biotin–streptavidin (BS) horseradish peroxidase (HRP) 3-amino-9-ethylcarbazole (AEC; BS-HRP-AEC) system and an indirect alkaline phosphatase red (AP-Red) system were used for single IHC analyses. A basic AEC detection kit (Ventana Medical Systems, Inc) was used, and it included biotinylated secondary antibody, streptavidin-labeled HRP, and AEC. A procedure named BMK AEC Paraffin was developed on the Benchmark automated immunostainer and was followed per the steps listed under BS-HRP-AEC in Table 1 . This procedure defaults to specific times for conditioner 1, mild CCl, and standard CCl. In addition, a biotin-free UltraView Universal AP-Red detection kit (Ventana) on the Benchmark (Ventana) was used. A procedure named BMK ultraView Red Par was developed on the Benchmark automated immunostainer and was followed per the steps listed under AP-Red in Table 1. Briefly, the Benchmark automated system uses heat and proprietary chemical treatments to deparaffinize the tissue-thin sections and achieves antigen retrieval using cell conditioner solutions.
Sequence of Automated Events for Single and Dual Enzyme-Based Immunohistochemical Analyses a

a Single enzyme based: BS-HRP-AEC (biotin–streptavidin horseradish peroxidase 3-amino-9-ethylcarbazole) and AP-Red (alkaline phosphatase red). Dual enzyme based: HRP-DAB and AP-Red. All reagents are from Medical Systems, Inc (Tucson, AZ) unless otherwise noted. AP, alkaline phosphatase; HRP, horseradish peroxidase; Ig, immunoglobulin; IgG, immunoglobulin G; IgM, immunoglobulin M.
b Times used for CD163 and CD172a monoclonal antibodies, respectively.
c Richard-Allen Scientific.
Primary MAbs for the single IHC analyses were diluted in antibody diluent (Ventana) at the following concentrations: 5A1 (VMRD Inc) at 2 μg/ml (mouse anti-goat CA immunoglobulin G1[IgG1] 38 ), 10A1 (VMRD Inc) at 2 μg/ml (mouse anti-goat CA IgG1 38 ), DH59B (VMRD Inc) at 2.5 μg/ml (mouse anti-bovine CD172a IgG1 14 ), and MCA1853 (Serotec, Inc, Raleigh, NC) at 2.5 μg/ml (mouse anti-human CD163 IgG1 1 ). MAb 99.97.6.1 (VMRD Inc) (mouse anti-bovine prion peptide IgG1 44 ), used at 2.5 μg/ml, and mouse negative control (Ventana) were used as MAb isotype–negative controls.
Dual IHC Analyses of CD 163 or CD 172 a and OPPV CA
Automated dual IHC analyses used biotin-free UltraView Universal DAB detection kit followed by UltraView Universal AP-Red detection kit (both from Ventana Medical Systems, Inc). A procedure named BMK IHC DS uDAB-uRed was employed on the Benchmark IHC/ISH staining module and was followed per the steps listed under HRP-DAB and AP-Red in Table 1. Four protocols were generated for dual IHC analyses of CD163 and OPPV CA (ie, the CD163 protocols): (1) CD163 (DAB first series) and OPPV CA (red second series), (2) CD163 (DAB first series) and isotype control MAb (red second series), (3) isotype control MAb (DAB first series) and OPPV CA (red second series), and (4) isotype control MAb (DAB first series and red second series). In addition, 4 protocols were generated for dual IHC analyses of CD172a and OPPV CA (ie, the CD172a protocols): (1) CD172a (DAB first series) and OPPV CA (red second series), (2) CD172a (DAB first series) and isotype control MAb (red second series), (3) isotype control MAb (DAB first series) and OPPV CA (red second series), and (4) isotype control MAb (DAB first series and red second series). Primary MAbs used in the first and second series of the dual IHC analyses were diluted in antibody diluent (Ventana). Final concentrations of MAbs were as follows: DH59B at 10.0 μg/ml, MCA1853 at 2.5 μg/ml, 99.97.6.1 at 2.0 μg/ml, and 5A1 at 2.0 μg/ml.
Results
Detection of OPPV CA With Western Blot and IHC Analyses
Initially, to check the specificity of the anti-CAEV CA MAbs 5A1 and 10A1 for naturally occurring OPPV strains, a Western blot analysis was performed on a field isolate, OPPV LMH17, originating from colostrum cells of a naturally infected ewe, and the OPPV WLC1 strain. The analysis confirmed that both mouse MAbs 5A1 and 10A1 recognized a single OPPV protein band of approximately 28 kDa, the apparent molecular weight of CA for both strains of OPPV, whereas an isotype control did not recognize any protein bands (data not shown).
Based on Western blot analyses, development of automated single immunohistochemistry analyses ensued with MAbs 5A1 and 10A1 and 2 enzyme-based indirect IHC detection systems. Overall, OPPV CA was detected in situ with 2 enzyme-based IHC detection systems in 4 of 9 sheep with moderate to severe histological lesions in the lung, mammary gland, or the carpal synovial membrane (animal Nos. 1, 5, 7, and 9; see Table 2 ). OPPV CA was not detected in any affected tissue from 1 ewe with mild histological lesions and was not consistently detected in tissues from 5 ewes with moderate to severe histological lesions. OPPV CA labeling was not detected in leukocytes or other cells composing the choroid plexus in any of the ewes.
Ovine Progressive Pneumonia Virus Capsid Antigen Detection in 10 Infected Sheep
a Tissue with moderate to severe histological lesions: L, lung; MG, mammary gland; CS, carpal synovial membranes (all formalin fixed, paraffin embedded).
b MAb, monoclonal antibody. Two plus signs(++) indicate more labeling using one MAb over the other MAb based on alkaline phosphatase red immunohistochemical analyses.
c Capsid antigen–positive tissue based on alkaline phosphatase red immunohistochemical analyses.
d Capsid antigen–positive tissue based on biotin-streptavidin horseradish peroxidase 3-amino-9-ethylcarbazole immunohistochemical analyses (MAb 5A1).
Specific Findings
OPPV CA detected in lungs, mammary gland, and carpal synovial membranes of persistently infected sheep
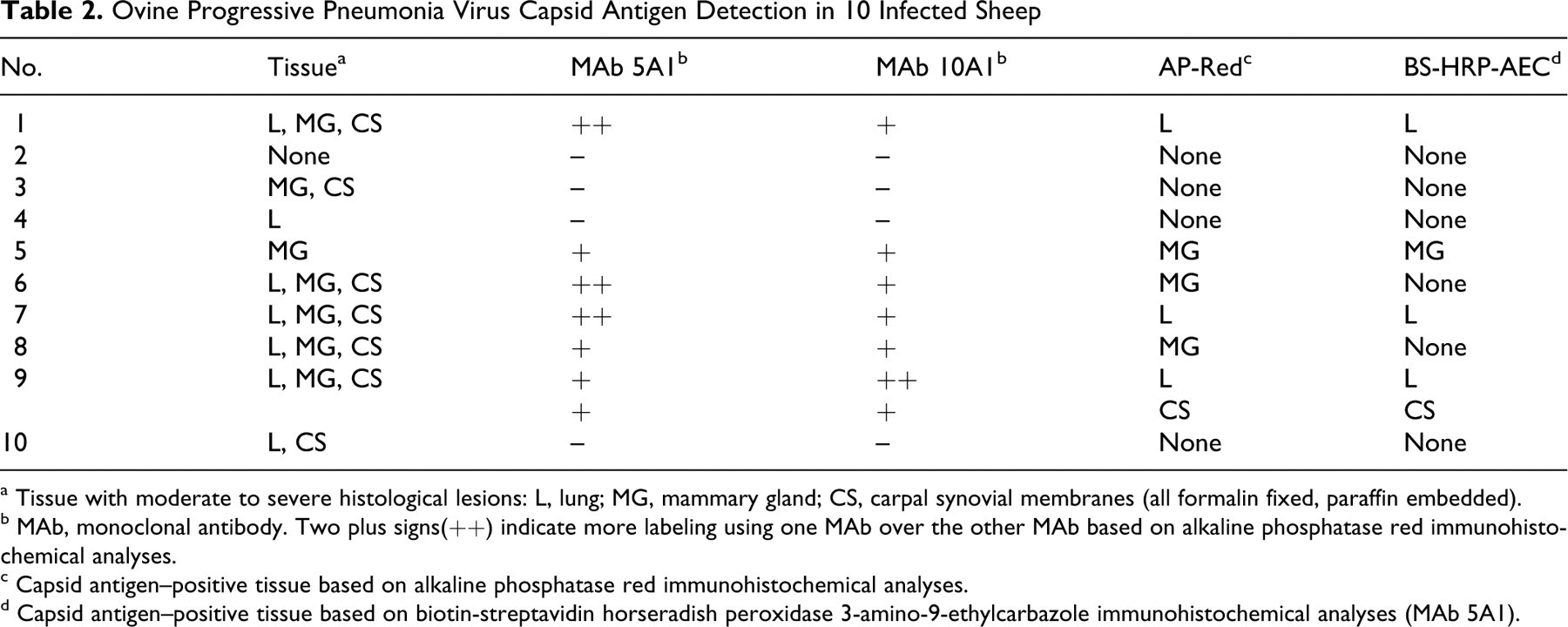
Of those ewes (animal Nos. 1, 7, and 9) with OPPV CA labeling in the lung, OPPV CA labeling was predominantly observed within cells resembling alveolar macrophages (Fig. 1A, arrowheads), occasionally in pulmonary interstitial macrophages and between cells in peribronchiolar lymphoid follicular areas, but there was no labeling in the same cells or areas when an isotype control MAb was used (data not shown). OPPV CA was not detected in any of the tissues collected from the specific pathogen-free or uninfected OPPV sheep (representative lung tissue shown in Fig. 1B). To verify that the labeling observed in the lung was due to the presence of OPPV CA, we evaluated MAb 5A1 on lung sections using another enzyme-based indirect IHC detection system on the automated immunostainer. There was similar labeling in cells resembling alveolar macrophages of lung from an OPPV-infected sheep based on the BS-HRP-AEC system as compared to the AP-Fast Red system, whereas there was no labeling in the lung of the OPPV-negative sheep or in the lung of the OPPV-infected sheep based on the isotype control MAb on the HRP-AEC system (data not shown). The degree of OPPV CA labeling in the lung alveolar macrophages based on MAb 5A1 in the OPPV-infected sheep was comparable between the AP-Red and BS-HRP-AEC systems. However, the AP-Red kits had an increase in nonspecific background as the expiration date of the kit neared.
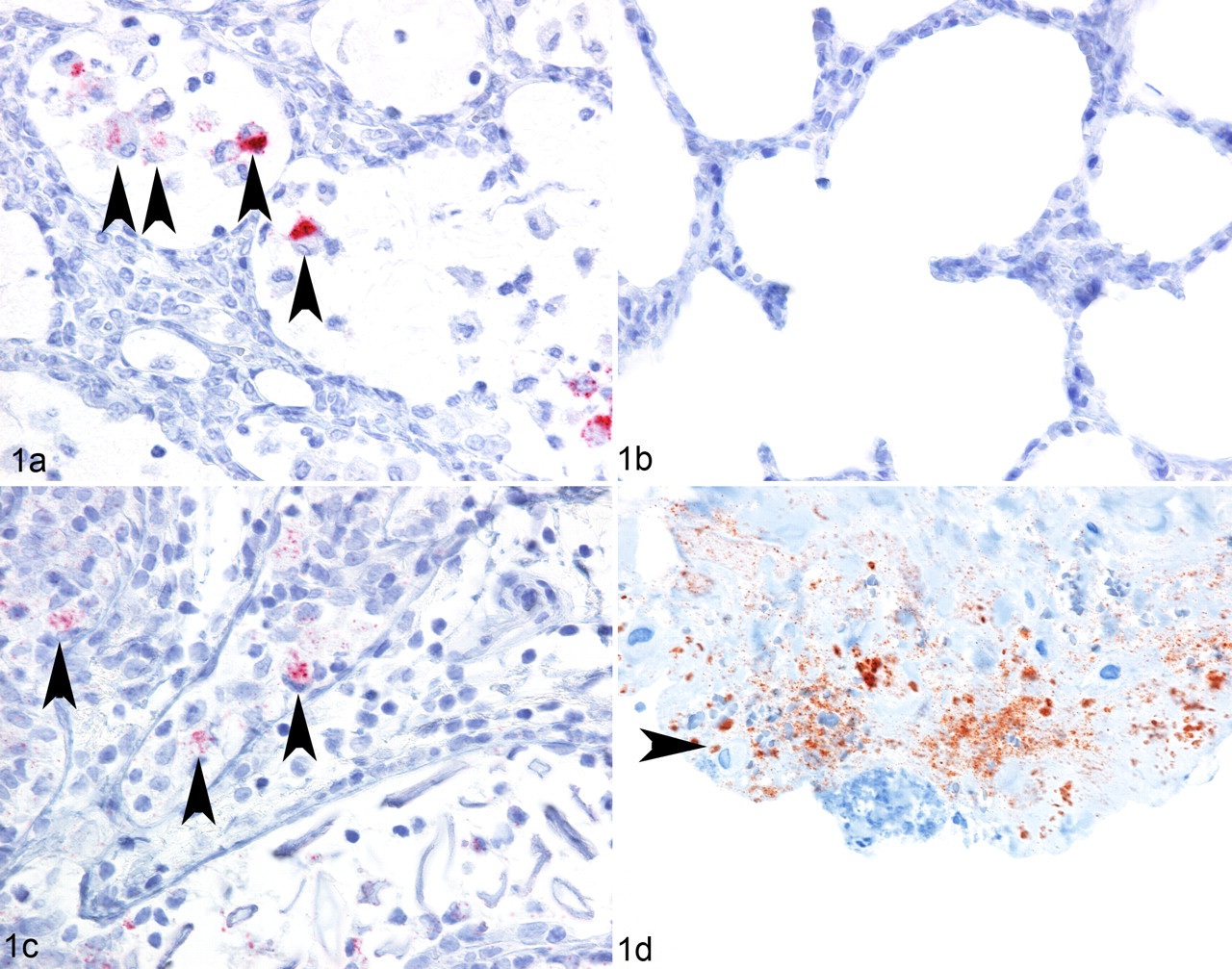
Single enzyme-based automated IHC detection of OPPV CA in formalin-fixed, paraffin-embedded lung, mammary gland, and carpal synovial membrane tissue sections from OPPV-infected and uninfected ewes. A, representative cranial lung section from an OPPV-infected ewe labeled with anti-CAEV CA MAb 5A1 and based on the AP-Red detection system. Arrowheads show alveolar macrophages positive for OPPV CA (red). B, representative cranial lung section from an uninfected ewe labeled with anti-CAEV CA MAb 5A1 based on the AP-Red detection system (no red). C, mammary gland section from an OPPV-infected ewe labeled with anti-CAEV CA MAb 5A1 and based on the AP-Red detection system. Arrowheads indicate OPPV CA in cells resembling macrophages in alveoli (red). D, carpal synovial membrane section from an OPPV-infected ewe labeled with anti-CAEV CA MAb 5A1 based on the BS-HRP-AEC detection system. Arrowhead indicates a cell resembling a macrophage with labeling of the adjacent extracellular space (red). OPPV, ovine progressive pneumonia virus; CA, capsid antigen; MAb, monoclonal antibody; CAEV, caprine arthritis-encephalitis virus; AP-Red, alkaline phosphatase red; BS-HRP-AEC, biotin–streptavidin horseradish peroxidase 3-amino-9-ethylcarbazole.
Besides in the lung, OPPV CA labeling was observed in the lymphoid follicles of the mammary gland in 3 OPPV-infected sheep (animal Nos. 5, 6, 8) based on MAb 5A1 in the AP-Red system and was observed within and adjacent to alveolar cells (Fig. 1C, arrowheads) in 1 OPPV-infected sheep (animal No. 5). The lymphoid follicle labeling was punctate and intercellular. When the isotype control MAb was used on the mammary gland of the 3 OPPV-infected ewes, labeling was absent (data not shown). However, when MAb 5A1 was used with BS-HRP-AEC IHC system, OPPV CA labeling remained in the alveolar cells of animal No. 5, but the OPPV CA labeling in the lymphoid follicles in mammary gland of animal No. 6 and animal No. 8 was absent (data not shown). Because of this discrepant result with the 2 different enzyme-based IHC systems, we excluded the mammary streak canal from animal Nos. 6 and 8 from further IHC analyses. When the isotype control MAb was used in the BS-HRP-AEC system, there was no background labeling in the mammary streak canal (data not shown).
In 1 animal (No. 9), OPPV CA labeling surrounded collagen fibers in the carpal synovial membrane (Fig. 1D). In contrast, there was no labeling using the isotype control MAb or in the carpal synovial membrane of an uninfected control (data not shown). In addition, we evaluated the AP-Red IHC system using MAb 5A1 on the same carpal synovial membrane and observed a similar pattern of labeling as compared to that of the BS-HRP-AEC IHC system.
OPPV CA and CD163/CD172a detected in alveolar lung macrophages of persistently infected sheep
Because cells resembling macrophages were positive for OPPV CA in both single IHC analyses, we first tested and titrated a few macrophage-specific and dendritic cell–specific MAbs on sequential formalin-fixed, paraffin-embedded tissues using the AP-Red IHC system. Two such markers, CD163 and CD172a, were chosen to be further evaluated using dual IHC analyses on sections of lung, mammary gland, and carpal synovial membranes owing to their consistent labeling and performance on formalin-fixed, paraffin-embedded tissue sections.
Automated dual IHC analysis was developed with the HRP-DAB IHC system as the first series and with the AP-Red IHC system as the second series with a denaturation step in between the two. When anti-CD163 MAb was used in the first series and CAEV CA MAb 5A1 in the second, OPPV CA (red) was detected in CD163-positive (brown) cells resembling alveolar macrophages based on their location and amorphous nucleus (Fig. 2A, arrowhead). On occasion, an alveolar macrophage would have OPPV CA labeling on one side of the cell and CD163 on the other. When anti-CD172a MAb was used in the first series (brown) and anti-CAEV CA MAb in the second (red), OPPV CA was detected in CD172a-positive cells resembling alveolar macrophages (Fig. 2B, arrowhead). Interestingly, there was a relative increase in the numbers of CD163- or CD172a-positive macrophages in areas where OPPV CA and interstitial septal thickening were present, as compared to (1) areas without OPPV CA and with less interstitial septal thickening in the same section (data not shown) or (2) areas without OPPV CA and with thicker alveolar septal areas of an OPPV-uninfected control (compare Figs. 2A, 2C; Figs. 2B, 2D). Based on the isotype control MAb for the first series (DAB or brown) and the CAEV CA MAb 5A1 in the second (red) of the dual IHC analyses, there was no brown labeling except for some light brown labeling of a few blood vessels, and OPPV CA labeling (red) was predominantly found in cells resembling alveolar macrophages (data not shown). When the isotype control MAb was used in both the first and second series, there was no brown or red labeling in any part of the lung tissue except for a few blood vessels that labeled a very light brown (data not shown).
OPPV CA and CD163/CD172a detected in alveolar mammary macrophages of persistently infected sheep
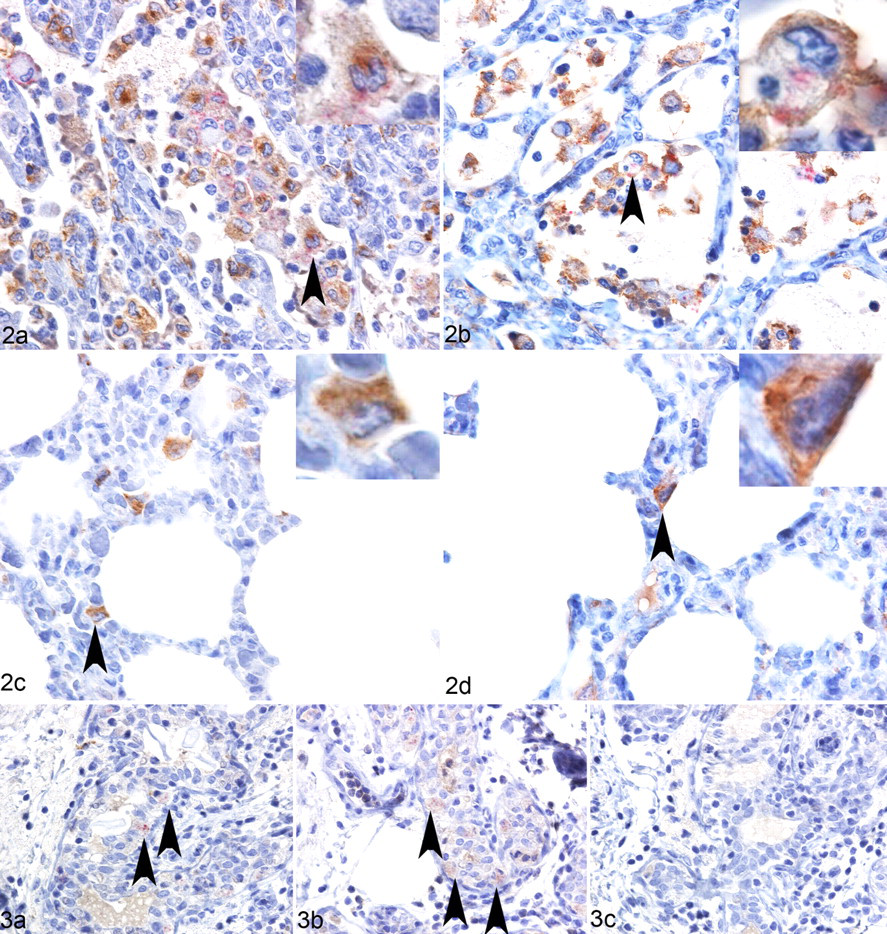
Based on the single IHC results, the mammary gland of animal No. 5 was further examined by dual IHC analyses with the anti-CAEV CA MAb 5A1 and the anti-CD163 or anti-CD172a MAbs. Baesd on anti-CD163 MAb or anti-CD172a MAb in the first series (brown) and the isotype control MAb in the second (red), intense CD163 labeling was detected mostly in cells resembling macrophages in and between alveolar and ductal areas of the mammary gland and in cells of lymphoid follicles (data not shown). Based on anti-CD163 MAb in the first series (brown) and anti-CAEV CA MAb in the second (red), a few cells resembling macrophages of the alveoli and possibly ducts were positive for OPPV CA and CD163 (Fig. 3A, arrowheads). Based on anti-CD172a MAb in the first series (brown) and anti-CAEV CA MAb in the second (red), cells resembling macrophages of the alveoli were positive for OPPV CA and CD172a (Fig. 3B, arrowheads). Qualitatively, there were more CD172a- or CD163-positive cells in the OPPV CA–positive mammary gland of animal No. 5 as compared to the mammary gland of the OPPV-negative control (data not shown). Based on the isotype control MAb in the first series (brown) and the MAb 5A1 in the second (red), OPPV CA was detected in cells that resembled macrophages within the alveoli of the mammary gland (red); furthermore, based on an isotype MAb control for both the first and second series, there was slight brown labeling in the ducts (much less than that observed in Figs. 3A, 3B), which could not be eliminated (Fig. 3C).
Absence of OPPV CA and CD163/CD172a in the same cells of the carpal synovial membranes
CD163 was found in a few cells resembling synoviocytes, fibrocytes in the connective tissue, and macrophages of the carpal synovial membranes in uninfected and OPPV-infected sheep based on the AP-Red IHC system (data not shown). However, CD172a was mostly confined to cells resembling macrophages (amorphous nucleus) and some synoviocytes based on the AP-Red IHC system (data not shown). Although we could not show on a repetitive basis that OPPV CA colocalized with CD172a or CD163 in specific cells of the carpal synovial membrane, we did observe OPPV CA labeling and moderate to severe histologic lesions in the tissue in close proximity to these CD172- or CD163-positive cells (data not shown).
Discussion
This study is the first to show that OPPV CA is found in CD172a- or CD163-positive alveolar macrophages in the lung. This finding is consistent with previous findings from others showing that MVV p15 colocalizes with VPM32-positive leukocytes from BALF of MVV-infected sheep and that MVV gp135 colocalizes with rabbit anti-sheep alveolar lung macrophage sera in monocyte-derived macrophages. 20,34 To our knowledge, there is no reference for the specificity of VPM32, although others have stated that this MAb is thought to bind to Fc receptors on macrophages. 42 In addition, our study is the first to show OPPV CA within CD172a- or CD163-positive alveolar macrophages of the mammary gland. The presence of OPPV CA in alveolar macrophages of the mammary gland is consistent with previous research where viral RNA and CA have been found in sheep mammary cells with the morphology of macrophages. 6,8,23,39 These combined results suggest that CD163- or CD172a-positive alveolar macrophages acquired OPPV CA by phagocytosis of CA, whole virus, or virus-infected cells and/or by receptor-mediated endocytosis of whole virus. HIV can infect cells using receptor-mediated endocytosis via dynamin; thus, OPPV could have a similar way of infecting alveolar macrophages. 41 Regardless of how the virus infects, our phenotypic observations, in combination with prior morphological and anatomical studies, show that provirus, viral RNA, and CA are found in lung alveolar macrophages, which suggests that these cells are productively infected.
CD-163-positive macrophages have an anti-inflammatory function 50 ; so, the result that CD-163 and OPPV CA colocalize in the same alveolar macrophage suggests that infected macrophages may limit inflammation. Other studies have shown that HIV-1-infected monocyte-derived macrophages maintain cell surface CD163 in vitro but at a lower level of expression compared to that of uninfected monocyte-derived macrophages, suggesting that HIV infection dampens the anti-inflammatory function of these macrophages. 45 We did not observe any obvious difference in the CD163 labeling intensity between OPPV CA-positive and CA-negative macrophages. Because the IHC assay is not quantitative, further studies need to be conducted to observe whether OPPV causes a measurable decrease in CD163 expression in alveolar macrophages.
Although we could not demonstrate OPPV CA in CD172a- or CD163-positive cells in the carpal synovial membrane, there was intercellular OPPV CA labeling in the connective tissue (data not shown) consistent with previous data on the distribution of CAEV RNA and OPPV CA in carpal synovial membranes of goats and sheep. 8,33 The absence of clearly CD163- or CD172a-positive cells colocalizing with OPPV CA in the synovium suggests that the intercellular and connective tissue CA deposition may be residual from cell lysis of macrophages or other synovial cells.
In our study, we found that there were more CD172a- or CD163-positive macrophages in the vicinity of OPPV CA-positive macrophages and areas of severe histological lesions in the lung as compared to areas without OPPV and lesions or uninfected controls. CA-negative animal Nos. 4 and 10 showed less CD163 or CD172a labeling in areas of moderate to severe lung lesions than did CA-positive animal Nos. 1, 7, and 9 (data not shown). This suggests that CD172a- or CD163-positive cells are recruited more heavily to OPPV-infected cells. From a virus perspective, this is a way to infect newly recruited macrophages in local areas of inflammation and OPPV. Once the virus has infected the anti-inflammatory phenotype (CD163) or down-regulatory phenotype (CD172a), this might allow evasion and persistence in the host.
One important finding of this study is that histological assessment of tissues is more sensitive than IHC analyses based on the anti-CAEV-63 CA MAbs 5A1 and 10A1. Nine ewes had moderate to severe lesions in at least one of the OPPV-affected tissues; however, only 4 ewes demonstrated OPPV CA in OPPV-affected tissues. This result is congruent with other reports that used the anti-OPPV 85/34 CA MAb 3F, anti-MVV CA MAb 1A7, and anti-MVV CA guinea pig sera. 8,19,23 There are several possible underlying mechanisms for the reduced sensitivity of OPPV CA detection in situ, as compared to histological assessments. One possible reason is that the 2 MAbs do not detect all ovine progressive pneumonia CA viral variants among all the ewes in IHC analyses. However, in this study, both 5A1 and 10A1 reacted similarly at 28 kDa on Western blot analyses to 2 phylogenetically distinct OPPV strains (data not shown): the Idaho OPPV strain LMH17 and OPPV WLC1. In addition, MAbs 5A1 and 10A1 radio-immunoprecipitate visna virus, OPPV WLC1, CAEV-63, and CAEV-Co, and MAb 5A1 binds to visna virus, OPPV WLC1, CAEV-63, and CAEV-coinfected cells using indirect immunofluorescence. 38 Because there was no discrepant labeling between the 2 different anti-CAEV CA MAbs among the ewes (see Table 2), this result suggests that MAbs 5A1 and 10A1 bind OPPV strains found in US sheep. Another possible mechanism for the reduced sensitivity of OPPV CA detection in situ is that host adaptive immune responses reduce the amount of viral antigen in affected tissues, but histologic lesions arising from inflammatory responses increase over time.
A final possible mechanism is that some component of viral infection other than CA causes the associated pathological changes in the affected tissues. Links have been made between the presence of provirus in the lung of naturally OPPV-infected sheep or the amount of provirus in tissues and peripheral blood mononuclear cells of experimentally CAEV-infected goats and the degree of histological lesions in tissues. 7,13,48 Furthermore, a statistically significant correlation was observed between the amount of provirus in lung alveolar macrophages and adherent peripheral blood mononuclear cells of naturally infected sheep and the degree of histological lung lesions. 55 More recently, we showed that peripheral blood leukocyte ovine progressive pneumonia provirus levels correlate significantly with histological lesion scores in any of the affected tissues. 27 These studies together suggest that tissue and peripheral blood provirus contribute to histopathology in the affected tissues. Potential mechanisms for why peripheral blood and tissue provirus levels correlate with the extent of histopathology need to be further explored in the future.
In summary, this study showed that OPPV CA was found in CD163- and CD172a-positive alveolar macrophages of the lung and mammary gland using automated IHC methods. Furthermore, phenotypic and functional analyses of these macrophages will aid in the understanding of viral pathogenesis and cellular control of the virus. In addition, this study provided automated single IHC analyses useful for detecting OPPV CA in formalin-fixed, paraffin-embedded sections of lung, mammary gland, and carpal synovial membranes of sheep. These automated IHC analyses for detection of OPPV CA will be an additional postmortem diagnostic tool for detecting sites of virus replication in situ and possibly locating viral sources of transmission.
Footnotes
Acknowledgements
We thank Nicholas Durfee and Liam Broughton for technical assistance and Dr Timothy V. Baszler for the initial reading and critiquing of this article. We also thank Emma Karel, Lori Fuller, and Duane Chandler for animal handling.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
This work was funded under USDA-ARS No. 5348-32000-029-00D.
