Abstract
The objective of this work was to comparatively study the tissue tropism and the associated pathology of 2 autochthonous small ruminant lentivirus (SRLV) field strains using an experimental infection in sheep through the bone marrow. Fifteen male, SRLV-free lambs of the Rasa Aragonesa breed were inoculated with strain 697 (nervous tissue origin, animals A1–A6), with strain 496 (articular origin, animals B1–B6), or with uninfected culture medium (C1–C3). Clinical, serologic, and polymerase chain reaction (PCR) evaluations were performed periodically. Two lambs from each infected group and a control animal were euthanized at 134, 273, and 319 days postinfection. Tissues were analyzed by gross and histopathologic evaluation; immunohistochemistry for CD3, CD4, CD8, CD68, and FoxP3 cell markers; lung morphometric evaluation; and tissue proviral quantification by PCR. All infected animals became positive either by enzyme-linked immunosorbent assay and/or PCR, with group B lambs showing the highest serologic values and more consistently positive PCR reactions. Group A lambs showed representative lung lesions but only mild histopathologic changes in the central nervous system (CNS) or in carpal joints. Contrarily, group B lambs demonstrated intense carpal arthritis and interstitial pneumonia but an absence of lesions in the CNS. Proviral copies in tissues were detected only in group B lambs. Experimental infection with these SRLV strains indicates that strain 496 is more virulent than strain 697 and more prone to induce arthritis, whereas strain 697 is more likely to reproduce encephalitis in Rasa Aragonesa lambs. Host factors as well as viral factors are responsible for the final clinicopathologic picture during SRLV infections.
Keywords
Small ruminant lentiviruses (SRLVs; ie, Visna/maedi virus [VMV] and caprine arthritis-encephalitis virus [CAEV]) are worldwide viruses that cause persistent and progressive diseases in sheep and goats. 4,8,13,37 Small ruminant lentiviruses are transmitted mostly by air and milk, 26 and they produce lesions in different host target organs, most frequently lung, mammary gland, central nervous system (CNS), and carpal joints. 42 The tissue tropism appears to vary among SRLV strains according to incompletely understood factors that involve host-pathogen interactions. 5,9,38 The main target cells for SRLV replication are those of the monocyte-macrophage lineage, 30,52 and virus replication is induced when maturation into macrophages and dendritic cells occurs. As an inherent property of retroviruses, SRLVs have a strong ability to mutate, and the resultant antigenic drift produces new strains of the virus, 43 which may account in part for the different patterns of clinical disease. 3
Upon infection by SRLV, animals may remain asymptomatic carriers for life, and only a proportion of seropositive animals will develop clinical signs. The earliest signs observed in most infected sheep are loss of body condition and increasing tachypnea and dyspnea that is caused by different degrees of interstitial pneumonia. Respiratory signs can sometimes be associated with an indurative mastitis and a decrease in milk production in the postpartum period. 13 Although less often described, sheep can also be affected by arthritis, presenting unilateral or bilateral enlargement of the carpal joint, with clinically mild lameness. 38 Sheep can also be affected by the neurological form of the disease, often resulting in hind limb weakness, ataxia, hypermetria, or paralysis, leading to recumbency. 6,7,13 In goats, SRLVs usually produce the nervous or the articular forms, 23 although interstitial pneumonia can also be observed. 16
Antibody responses can emerge as early as 2–4 weeks postinfection and tend to fluctuate during the first 6 months of infection. 24 This response is critical because early serologic diagnosis and culling are the main control measures applied in SRLV control programs. 41 Neutralizing antibodies can take as long as 2 years to appear and can partially control virus infection. 27 However, mutations of viral epitopes may randomly occur and contribute to widened tropism and disease progression. 27,28 Small ruminant lentivirus infection can also alter macrophage function, which may favor immune dysregulation. 19
At necropsy, VM-affected animals may have voluminous lungs that do not collapse, a characteristic that is usually accompanied by chronic lymphadenitis of the mediastinal lymph nodes. 15 Arthritic animals have thickening and fibrosis of the articular capsule, with articular cartilage erosion and ulceration in severe cases. 38 Microscopic lesions are characterized by mononuclear cell infiltrates and lymphoid follicle formation in target organs. In addition, joints have synoviocyte proliferation, thickening of synovial membrane, fibrosis, and mineralization. 38 Immunohistochemistry reveals the presence of a high number of macrophages and CD8+ T cells and B cells in affected joints, with increased proportions of B cells in lesions as infection progresses. 49 The CNS presents a chronic lymphocytic meningoencephalomyelitis, usually accompanied by demyelination and mononuclear infiltration of the choroid plexus with variable hyperplasia of lymphoid follicles. 15
Successful experimental infections with SRLVs have been performed using viral strains isolated from natural field infections 25,48 using different inoculation routes such as intravenous, intratracheal, intracerebral, intra-articular, or conjunctival. 25,32,40 However, more studies are needed to evaluate tissue/organ tropism upon experimental infection with SRLVs to enhance our understanding of the pathogenesis of the different clinical forms. The main objective of this work was to study comparatively the tissue tropism and the associated pathology of two autochthonous SRLV field strains of different tissue origin using an experimental infection in sheep via the bone marrow route and to determine if SRLV lesions could be produced in this experimental model.
Materials and Methods
Animals
Fifteen 3-month-old, Rasa Aragonesa breed, male lambs were obtained from officially accredited VMV-free flock. 37 Animals came from the same lambing season, and only 1 lamb per ewe was selected. No information on the progenitor male was available, and therefore, these experimental animals might share some degree of relationship. Before experimental infection, an enzyme-linked immunosorbent assay (ELISA; Elitest MVV/CAEV, Hyphen Biomed, France) as well as different polymerase chain reaction (PCR) tests (see below) were serially performed in the lambs and the ewes from which they were derived, in order to assess and confirm the SRLV-negative status of each individual animal. The animals were housed at the University of Zaragoza and provided with an acclimation period of 2 months before experimental infection. The complete experimental procedure was approved and licensed by the Ethical Committee of the University of Zaragoza (ref: PI09/10). The care and use of animals were performed according to the Spanish Policy for Animal Protection RD1201/05, which meets the European Union Directive 86/609 on the protection of animals used for experimental and other scientific purposes. Castration was performed 2 weeks after animal arrival; 1 month later, a full clinical examination and a complete blood count were performed in all the animals with results within normal ranges, ensuring their suitability for the experiment. The animals were then homogeneously distributed, based on weight, for experimental infection in 3 groups: group A (A1–A6, 6 animals) and group B (B1–B6, 6 animals) and a control group (C1–C3, 3 animals). To avoid the risk of cross-infection during the experimental period, all groups were housed separately and under isolation.
Experimental Infection and Clinical Follow-up
Two autochthonous SRLV strains, 697 18 and 496, 17,38 previously isolated from cases of encephalitis in Assaf sheep and arthritis in Rasa Aragonesa sheep, respectively, were used. Briefly, strain 697 was subcultured in ovine skin fibroblasts, and strain 496 was subcultured once in blood-derived macrophages from different Rasa Aragonesa sheep, 14 with both cell types obtained from confirmed SRLVs-free lambs. Strain 697 culture supernatants were titrated by measuring the activity of the reverse transcriptase (RT) produced in the supernatant, using a commercial kit (HS-Lenti RT activity, Cavidi, Uppsala, Sweden), 17 and those from strain 496 were titrated by limiting dilution. 44 Culture supernatants were filtered and stored in aliquots containing 106 TCID50/mL at −80°C until used in experimental infection.
At 5 months old, animals from groups A and B were experimentally infected with the selected SRLV strains (697 and 496, respectively), and group C (control group) was inoculated with the same volume of noninfected culture medium. A dose of 106 TCID50/animal was used for both strains. The animals were sedated with xylazine and placed in lateral recumbency. The inoculum was deposited in the bone marrow of the right humerus, by injection into the trochanteric fossa. 45 Animals were reinfected at 62 days postinfection (dpi) using the same protocol.
The animals were observed daily, and a clinical follow-up was performed every 2 days for the first 15 days and then weekly until the end of the experiment at 319 dpi. In all clinical observations, special attention was given to the target organs typically affected by SRLV. To evaluate the articular changes in the live animals, the circumference and mediolateral diameter of the carpal joints were measured with a tape and a caliper, respectively.
Serologic, Blood PCR, and Hematologic Studies
Animals were sampled weekly to perform the following tests: (1) complete blood count, performed using an automated analyzer; (2) ELISA for detection of SRLV serum antibodies (Elitest MVV/CAEV, Hyphen Biomed); and (3) PCR analyses using 3 different primer pairs that targeted gag and long-terminal repeat (LTR) genetic regions. 46 Specifically, PCR for the gag gene yielded amplicons of 510 base pairs (bp; PCR named C/O) 17 or 800 bp (PCR named GAG). 20 Long terminal repeat (PCR named LTR) PCR amplicons were of approximately 300 bp (depending on the strain). 17 Each reaction mix included Reaction Buffer 1× (BioTools, Jupiter, FL), 2 mM MgCl2 (BioTools), 240 μM of each dNTP (Applied Biosystems, Foster City, CA), 20 pmol of each primer, 0.02 U/μl of Taq DNA polymerase (BioTools), and 1 μg of sample DNA to a final volume of 25–50 μl. PCR consisted of 45 cycles of denaturation at 95°C for 1 minute, annealing (55–60°C) for 1 minute, and elongation at 72°C for 1 min/Kb of amplified DNA, followed by a final elongation step at 72°C for 10 minutes.
Pathologic Studies
Animals were euthanized at 134, 273, and 319 dpi. Five animals were selected for each time point: 2 animals from each infected group (A and B) and 1 animal from group C. Since animals did not show relevant clinical signs, infected animals were chosen for slaughter according to their SRLV ELISA scores, such that at least 1 animal from groups A and B was strongly and consistently seropositive.
At necropsy, changes were evaluated with special attention to the target tissues, and samples of all organs were fixed in 10% formalin. Samples from lung, brain, cerebellum, brain stem, spinal cord, carpal joint capsule, and mediastinal and prescapular lymph nodes were also fixed with zinc fixative for immunohistochemistry. 11 For PCR studies, samples from the same tissues were collected in RNAlater solution (ThermoFisher, Waltham, MA) and stored at −80°C. Swab samples were collected from all carpal joints for both standard aerobic culture and Mycoplasma spp detection by PCR (EXOone Mycoplasma ovipneumoniae Basic qPCR kit, EXOPOL Labs, Zaragoza, Spain).
Fixed tissues were paraffin embedded, sectioned at 4 μm, and stained with hematoxylin and eosin. Intensity of lesions in target organs was estimated independently by 2 pathologists according to the following semiquantitative score: (1) –, negative; (2) +, mild; (3) ++, moderate; and (4) +++, severe.
To evaluate the degree of interstitial pneumonia, a quantitative study of the alveolar space was performed. For each animal, the area of open alveoli (air-filled space) was evaluated as a percentage of the total area, in 10 consecutive high-power fields (HPFs; 40×) using image analysis software (NIS-Elements BR Analysis, Nikon, Tokyo, Japan). For statistical purposes, animals from each group were considered together, irrespective of the dpi when necropsy was performed.
To evaluate the inflammatory process generated by the infection, an immunohistochemistry (IHC) study was carried out in the lung and mediastinal lymph node of all the animals and in carpal joints of the animals demonstrating arthritis. Antibodies and concentrations used are detailed in Table 1. All the IHC antibody-based procedures used the avidin-biotin-peroxidase complex (ABC) technique with diaminobenzidine as chromogen (Vector, Burlingame, CA) and Meyers hematoxylin for counterstaining. The above-mentioned image analysis software was used to evaluate and quantify the inflammation. The presence of CD3+ and CD68+ cells in the pulmonary interstitial infiltrate was measured as the mean of positive cells counted in 10 consecutive HPFs. In both hyperplastic lymphoid follicles and peribronchiolar infiltrates, the percentage of immunolabeled cells in comparison to cells with lymphoid morphology for the antibodies specified in Table 1 was estimated and the mean of all animals per group calculated. In carpal joints, the mean of CD3+, CD4+, and CD8+ T lymphocytes together with the expression of FoxP3+ cells was established by studying 10 HPFs in areas with inflammatory infiltrate, whereas CD68+ cells were semiquantitatively estimated, as indicated above.
Details of the Antibodies Used for Immunohistochemistry.
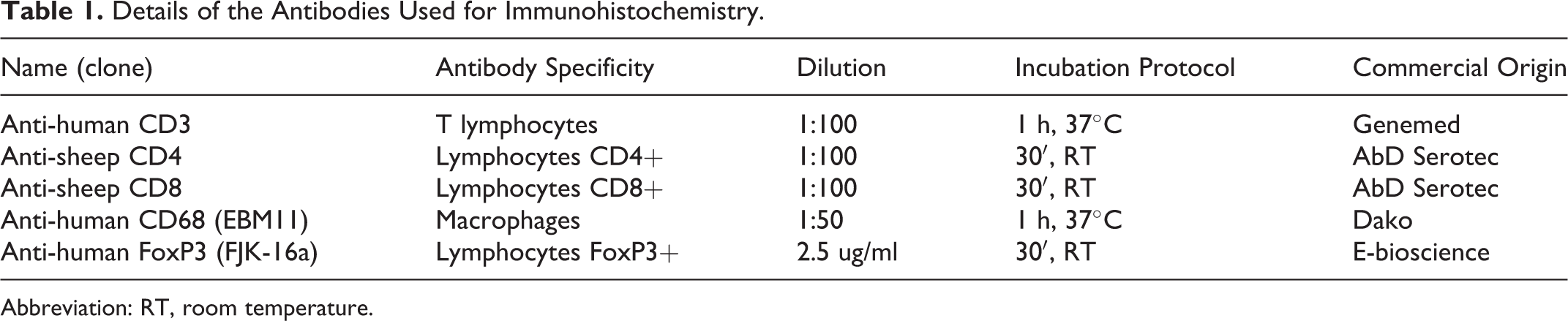
Abbreviation: RT, room temperature.
Viral Identification and Quantification in Tissues
Detection and quantification of viral DNA was carried out by classical and quantitative PCR (qPCR) respectively, using DNA extracted from the following tissues: prescapular, retropharyngeal, and mediastinal lymph nodes; lung; brain; bone marrow; spinal cord; and carpal joint synovium and capsule. DNA from tissues was obtained by disrupting 10 mg of tissue and incubating in ATL buffer and proteinase K, followed by extraction with the DNeasy Blood & Tissue kit (Qiagen, Germany). Amplicons derived from classical PCR using primers for gag (GAG and C/O) and LTR as specified previously 17,20 were visualized in agarose gels, purified, and cloned into shuttle plasmids (pGEMT easy) for external sequencing (Stab Vida, Caparica, Portugal).
Real-time PCR was carried out with specific primers and probes for each of the viral strains under study. Specifically, strain 697 was quantified using primers Fw: 5′-ATTTACAAGCAGGGGGAAGAA-′3 and Rv: 5′-GCAGCTGCTGAAAGACTACTGA-3′, and an LNA-based probe from the Universal Probe Library (Roche, Basel, Switzerland) called UPL-697 (5′-TGGAAGGC-′3) was used. On the other hand, strain 496 was quantified using primers Fw: 5′-GGCCAGGAAATCCTATGTTAGT-′3 and Rv: 5′-CGTGACTGGTTCTGCATCTAT-′3 and a TaqMan probe 5′-FAMCTTTGCAGCAAGGCTGCTAGA-TAMRA′3. Control animals were analyzed by both PCR tests. Reaction mix consisted of 200 nM of each primer, 200 nM of 496 TaqMan, and UPL-697 with 1U of Taq (BioTools) in a final volume of 25 μl. The standard thermal profile (45 cycles of 95°C and 60°C) was performed in a 7900-HT Real Time machine (Applied Biosystems). Targeted genetic regions of the different strains were cloned previously in pGEM-T easy plasmid (Promega, Madison, WI) and then 10-fold diluted as a standard in the qPCR. A standard curve of threshold cycle (Ct) versus plasmid copy number was generated to infer the quantity of viral DNA in test samples.
Statistical Analysis
The nonparametric Mann-Whitney and Kruskall-Wallis tests were used for comparison of the ELISA results, alveolar space values, and IHC expressions.
Results
Antemortem Findings
Animals were screened for antibodies against SRLV and for the presence of virus by 3 different diagnostic PCRs prior to experimental infection, showing consistent negative results upon retesting. Following inoculation of viral strains 697 (group A) and 496 (group B) via the bone marrow, the infection was detected in all animals either by ELISA and/or PCR. The serologic response (ELISA optical density [OD] ratios) developed over time in both experimental groups. Group B lambs always showed a higher and earlier serologic response when compared with group A animals, even after reinfection at 62 dpi. Moreover, the serologic response of group B was significantly higher from 117 to 163 dpi (P < .05). The control group (group C) remained ELISA negative throughout the experimental period. All the group B animals were serologically positive at the time of necropsy (Fig. 1; individual data are given in Suppl. Figs. 1–3).
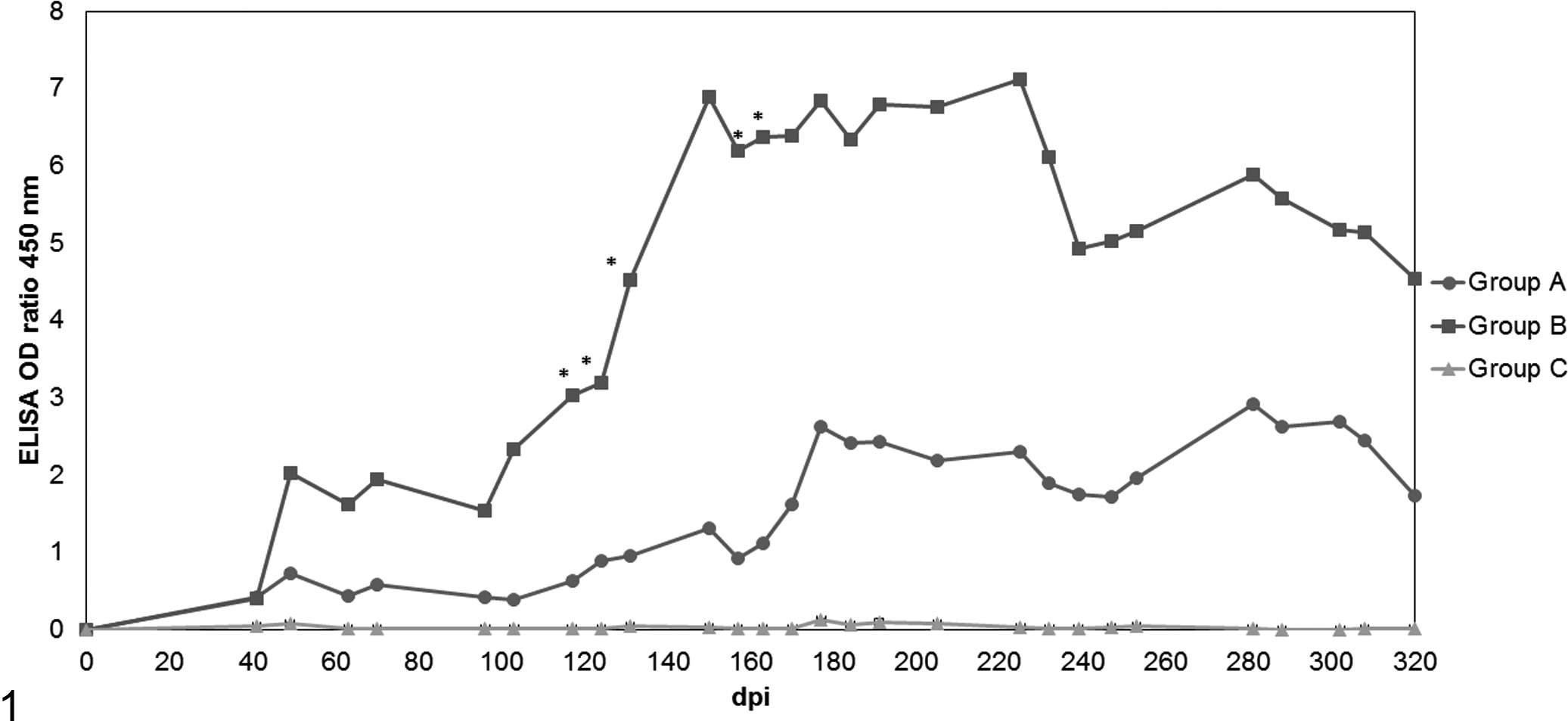
Development of serum antibody titers (enzyme-linked immunosorbent assay optical density ratio mean values) over the course of 319 days postinfection with small ruminant lentivirus. Group A: viral strain 697 of nervous origin. Group B: strain 496 of articular origin. Group C: control mock-infected group. *Statistically different from group A (P < .05).
All blood parameters analyzed were within the normal range in all animals during the experiment. Nevertheless, mean leukocyte values were higher (albeit not significantly) in infected groups compared with the control group. In contrast, mean erythrocytes and hematocrit and hemoglobin values were (nonsignificantly) lower in infected groups.
The PCR tests on blood samples were positive in 11 of 12 animals from groups A and B, whereas control sheep (group C) were consistently SRLV negative (Table 2). Evidence of infection was confirmed within the first month of exposure to the virus in some animals from both infected groups, although PCR reactivity fluctuated throughout the study period. The most sensitive PCR primers targeted the gag gene (GAG and/or C/O), being positive by 125 dpi in all 6 lambs infected with virus strain 496 and 4 of the 6 lambs infected with virus strain 697. In contrast, the LTR PCR was least sensitive, particularly in animals exposed to the 697 strain (Table 2). After reinfection at 62 dpi, these differences in PCR performance were maintained. The specific viral strain present in infected animals always corresponded to the original inoculated strain, as revealed by PCR amplicon length (which differs between strains 697 and 496) and by direct sequencing of GAG amplicons.
Blood PCR Reactivity Targeting Gag (Gag and C-O) and LTR Genomic Regions of SRLV in Group A (697 Strain; A1–A6), Group B (496 Strain; B1–B6), and the Control Group (C1–C3).
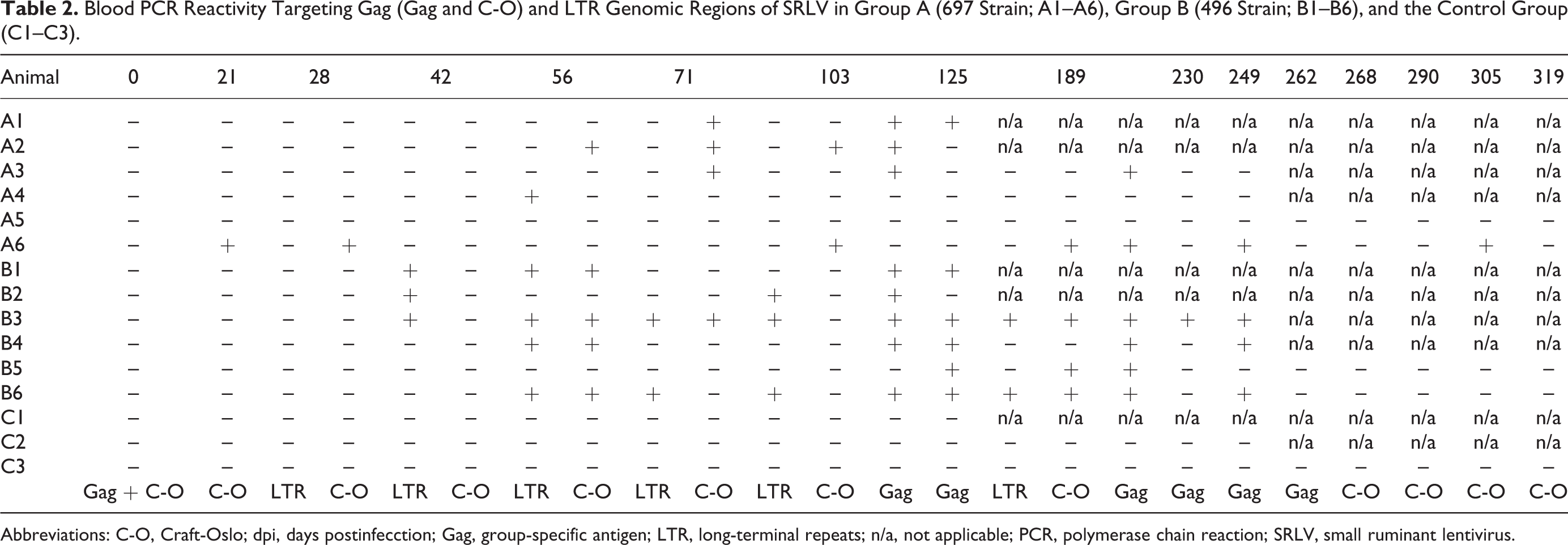
Abbreviations: C-O, Craft-Oslo; dpi, days postinfecction; Gag, group-specific antigen; LTR, long-terminal repeats; n/a, not applicable; PCR, polymerase chain reaction; SRLV, small ruminant lentivirus.
Animals A4 and A5 infected with strain 697 presented controversial results regarding their infection status. Animal A4 consistently showed seronegative results, but PCR revealed the presence of virus by 56 dpi. Conversely, animal A5 was seropositive by 177 dpi (Suppl. Fig. 1), but it was consistently negative using the 3 PCR assays employed (Table 2).
During the experiment, animals B1, B2, B5, and B6 presented bilateral enlargement of the carpal joint (Table 3) and a mild increase in size of the corresponding prescapular lymph node. However, there were no statistically significant differences in either circumference or diameter between group B and the control group throughout the experimental period. Animals of groups A and C did not present clinical signs suggestive of SRLV infection.
Macroscopic Findings in Group B Lambs, Infected With SRLV Strain 496 Originating From a Case of Arthritis.
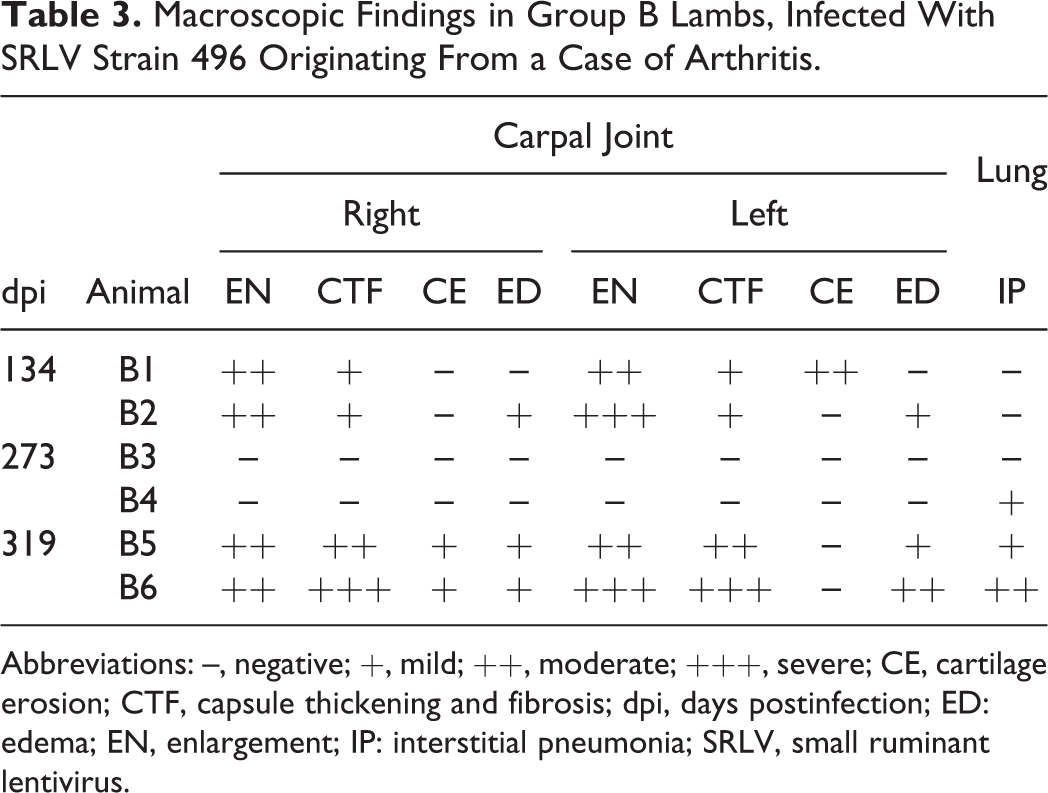
Abbreviations: –, negative; +, mild; ++, moderate; +++, severe; CE, cartilage erosion; CTF, capsule thickening and fibrosis; dpi, days postinfection; ED: edema; EN, enlargement; IP: interstitial pneumonia; SRLV, small ruminant lentivirus.
Postmortem Findings
Four animals from group B presented macroscopic lesions compatible with chronic proliferative arthritis that consisted of enlarged carpal joints with synovial capsule thickening, fibrosis, cartilage erosion, and edema (Table 3, Figs. 2–4). Voluminous and heavier lungs, compatible with interstitial pneumonia, were mostly seen at 319 dpi in animals from group B (Figure 5), whereas only animal A6 showed a mildly voluminous appearance of the lung (data not shown).
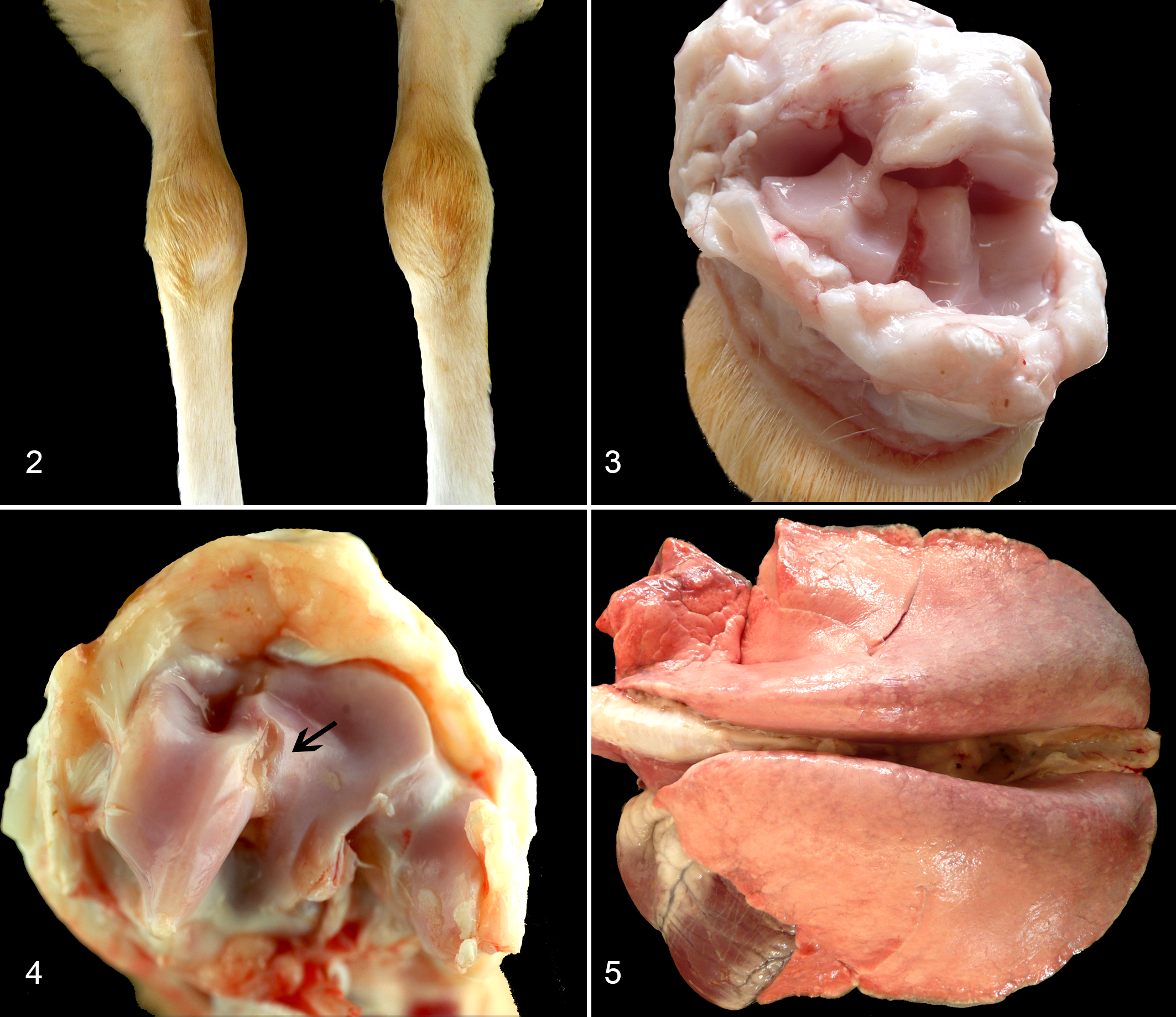
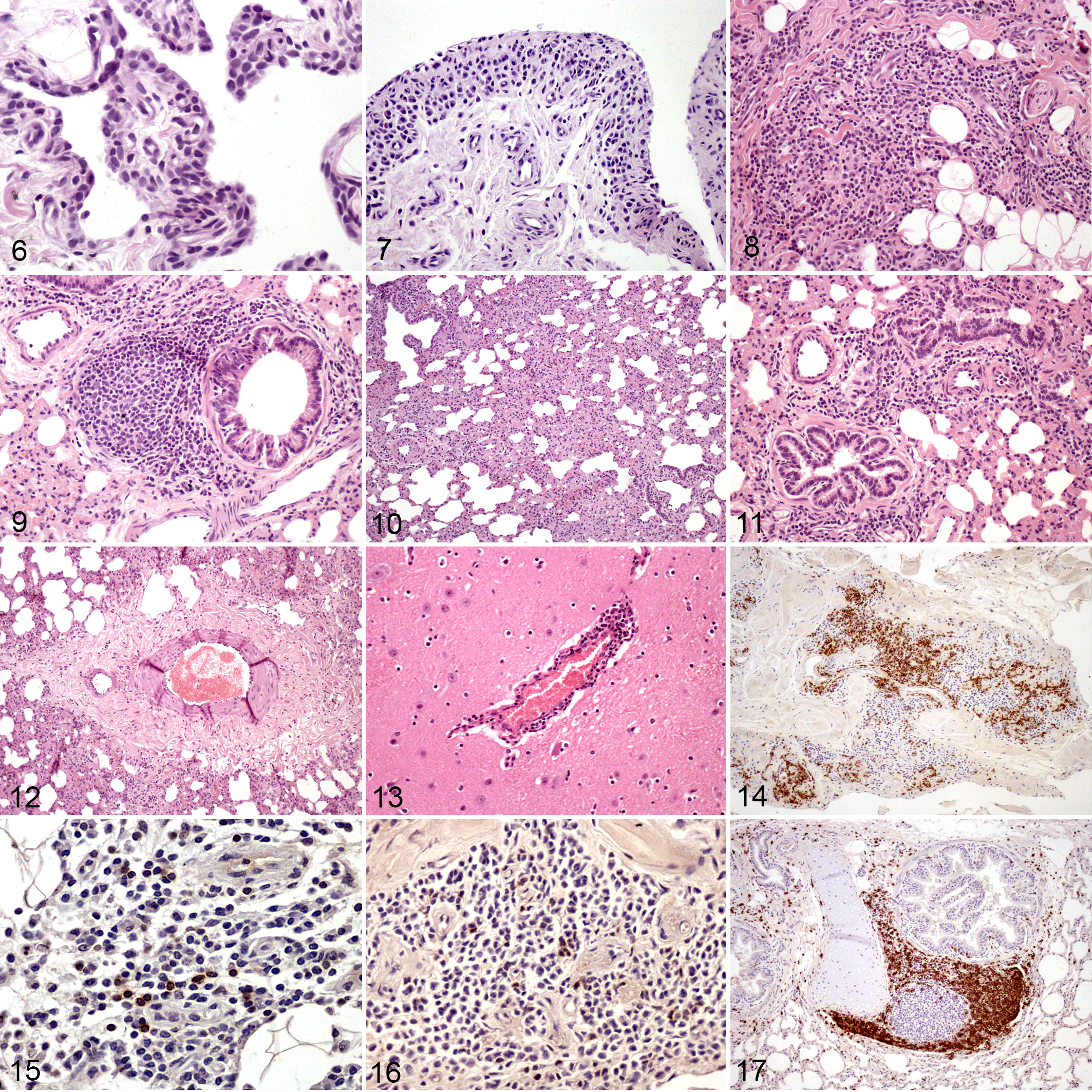
Microscopic pathology results in carpal joints were observed in all group B animals and only a few, scattered articular lesions were detected in group A animals. These articular lesions were characterized by papillary synoviocyte proliferation with synovial membrane thickening, multifocal mononuclear inflammatory infiltrate, and diffuse fibrosis (Figures 6–8). Regarding the lung, both groups exhibited microscopic lesions, although they were more abundant and developed earlier in group B. Pulmonary microscopic lesions consisted of an interstitial infiltrate of mononuclear inflammatory cells with increased thickness of alveolar septa, hyperplasia of lymphoid follicles, and lymphocytic peribronchiolitis (Figures 9–11). In addition, a severe perivascular fibrosis was observed in large pulmonary arteries from some animals (Figure 12). The mediastinal lymph node showed cortical hyperplasia in nearly all infected animals and in the prescapular lymph node in all group B animals. Regarding lambs A4 and A5, which presented controversial PCR and ELISA results (see above), no lesion related to SRLV was found in animal A4, but animal A5 showed moderate interstitial pneumonia and peribronchiolitis. Results are detailed in Table 4.
Histopathological Findings in Target Organs in Group A (697 Strain; A1–A6), Group B (496 Strain; B1–B6), and the Control Group (C1–C3).
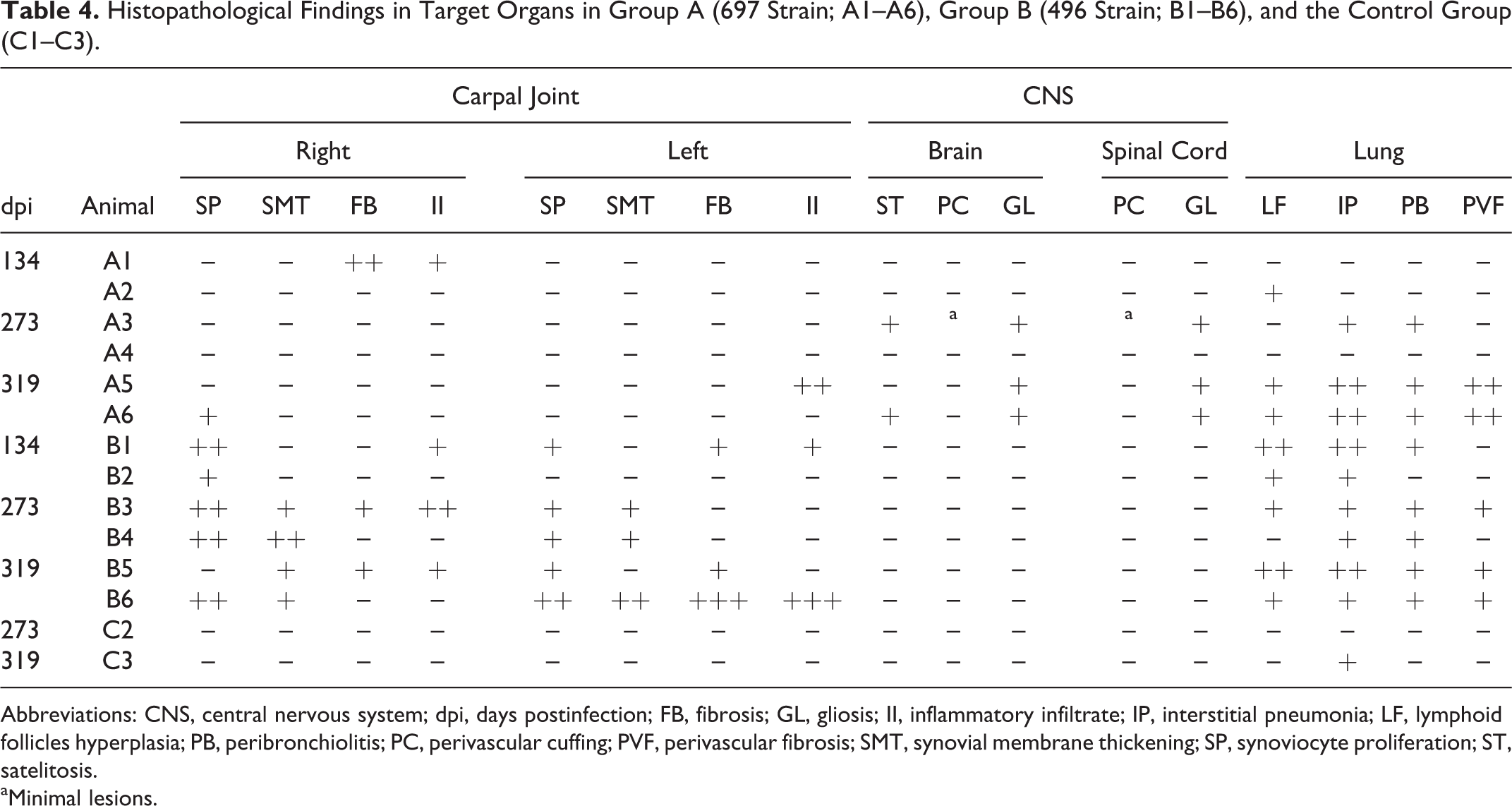
Abbreviations: CNS, central nervous system; dpi, days postinfection; FB, fibrosis; GL, gliosis; II, inflammatory infiltrate; IP, interstitial pneumonia; LF, lymphoid follicles hyperplasia; PB, peribronchiolitis; PC, perivascular cuffing; PVF, perivascular fibrosis; SMT, synovial membrane thickening; SP, synoviocyte proliferation; ST, satelitosis.
aMinimal lesions.
The pulmonary alveolar space (area of open air-filled space as a percentage of total area) was 63.56 ± 8.33 (mean ± standard deviation) in the control group (group C), 54.57 ± 15.87 in group A, and 49.16 ± 10.10 in group B. A significant reduction of the alveolar space was observed in group B when compared with group A (P < .05) and also between both infected groups and the control group (P < .01). The reduction of alveolar space did not seem to intensify along the study period within each group. Finally, CNS lesions were scant and seen only in group A animals. They were characterized by mild satellitosis, gliosis, and perivascular cuffing (Fig. 13). None of the animals from group B exhibited any detectable microscopic lesion in the CNS.
The control animal euthanized at 134 dpi (C1) showed a moderate to severe interstitial pneumonia not related to SRLV, as no serologic response or presence of virus was demonstrated. Therefore, lamb C1 was eliminated from the experiment, and the corresponding results are not shown. Swabs from carpal joints were negative for all types of bacteria according to standard culture and Mycoplasma spp. PCR.
Immunohistochemistry Findings
The IHC study of joints was limited to group B, as it was the only one consistently affected by arthritic lesions. CD3-, CD4-, and CD8-positive T lymphocytes were present in affected lambs (Figs. 14–16) and composed 32.37% ± 15.99%, 18.41% ± 8.7%, and 13.23% ± 7.94% of the leukocytes, respectively (mean ± standard deviation). CD68+ cells were more abundant in joints with severe lesions (Table 4).
Regarding the interstitial infiltrate in the lung, the percentage of CD3-positive cells was significantly greater in group B compared with control (P < .01). Moreover, the percentage of CD68-positive cells was significantly greater in both infected groups compared with the control group (P < .001). Regarding immunolabeling in both lymphoid follicles and the peribronchiolar infiltrate of infected groups, none differed significantly (Table 5), and comparison with the control group was not possible as control animals did not show these lesions. In infected groups, the peribronchiolar infiltrate was predominantly CD3+ (Fig. 17). FoxP3-positive cells were few (less than 1 lymphocyte/HPF), and it was not analyzed further.
Immunohistochemical Characterization of the Inflammatory Infiltrate in Lungs Following Infection With SRLV.
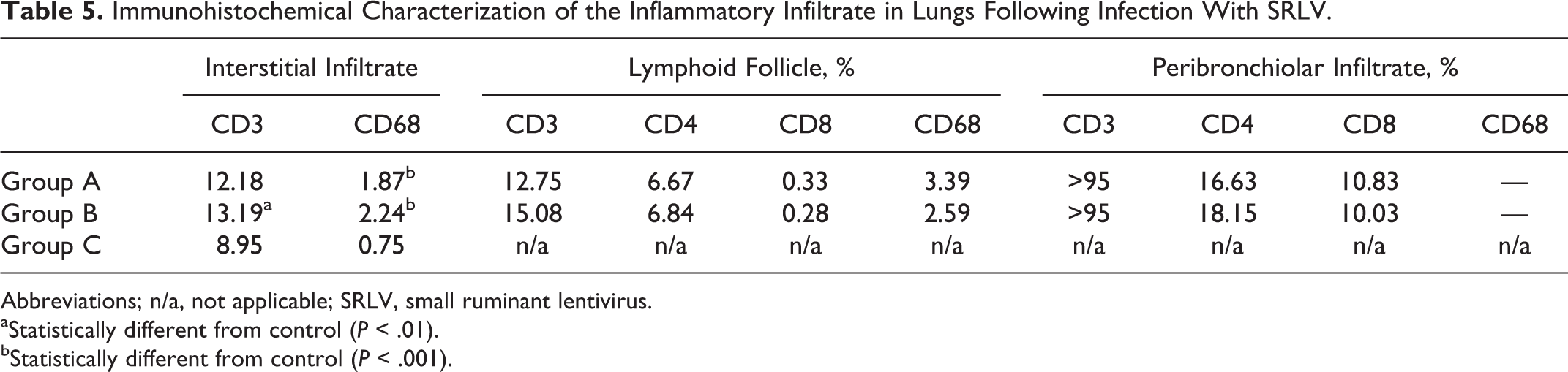
Abbreviations; n/a, not applicable; SRLV, small ruminant lentivirus.
aStatistically different from control (P < .01).
bStatistically different from control (P < .001).
Viral Quantification in Tissues
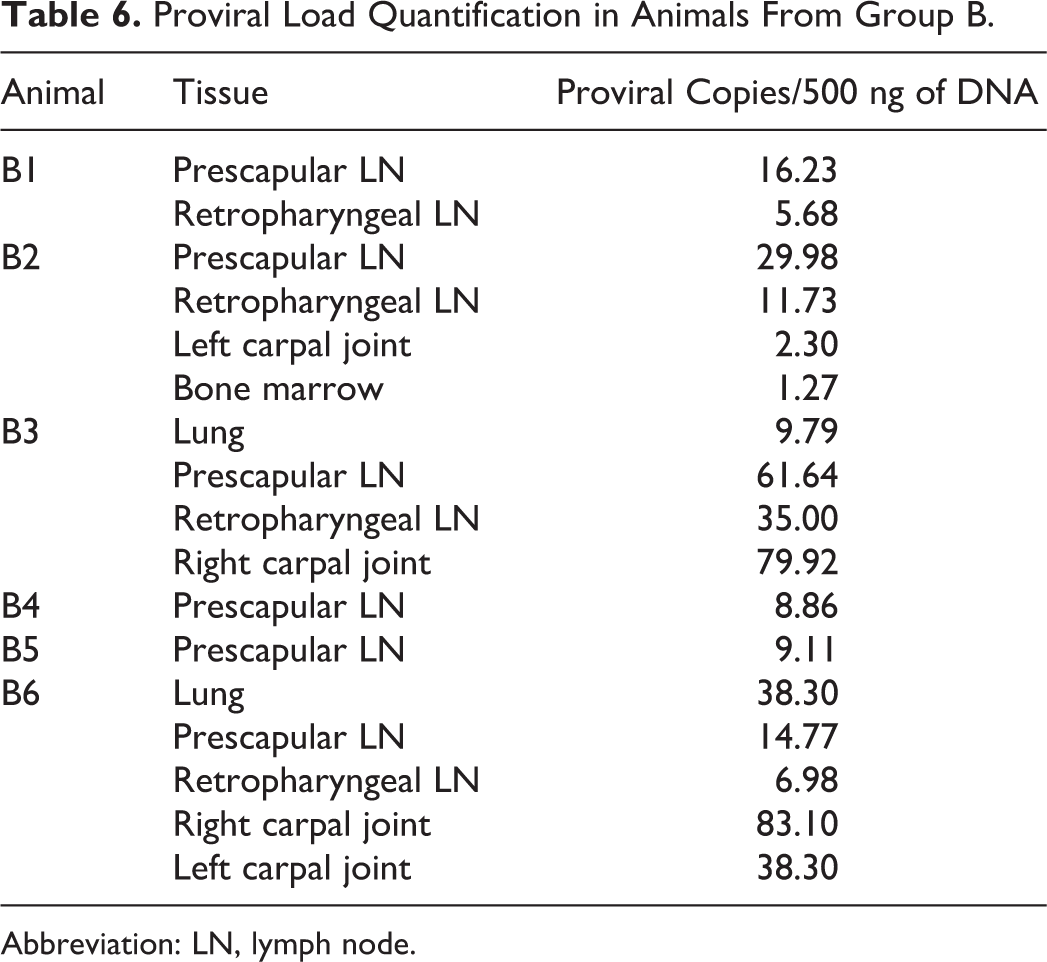
SRLV proviral sequences were detected only in target tissues of group B animals, with values ranging from 1 to 80 copies per 500 ng of tissue DNA (Table 6). Real-time PCR did not detect SRLV proviral sequences in animals from group A and controls.
Proviral Load Quantification in Animals From Group B.
Abbreviation: LN, lymph node.
Discussion
In this work, we comparatively studied the pathology and tissue tropism of two autochthonous SRLV strains after inoculation via the bone marrow. In addition, we determined if SRLV lesions could be produced in this experimental model. Previous experimental infections have used other routes mainly directed to target organs or blood. 25,33,40,48 Results of this work confirm the usefulness of the bone marrow route as a tool for experimental SRLV infection in sheep and for studying viral tropism as most infected animals in this study showed a marked serologic response and pathologic changes resulting from SRLV infection.
The serologic response to the virus varied between infected animals and appeared within 6 to 25 weeks postinoculation, a period of time in accordance with that previously reported by others. 32,48 However, comparing both infected groups, ELISA mean score values (OD ratio) were higher in group B (strain 496 of articular origin) compared with group A (strain 697 of neurological origin) from 41 dpi until the end of the experiment, a finding that might indicate that group B was inoculated with a more virulent strain. 2 Despite this difference, all the animals in groups A and B were successfully infected, as demonstrated by ELISA and/or PCR techniques. The finding that sheep A4 was PCR positive (only at 56 dpi; Table 2) while showing consistently seronegative results and that animal A5 was seropositive (at 177 dpi; Suppl. Fig. 1) but consistently negative to the three PCRs employed is regarded as a further confirmation of the complementary diagnostic value of both methods in asymptomatic infections. It is known that in lentiviral infection, the serologic response and PCR results can be intermittent and modified during the course of infection. 47 The relatively low proportion of positive blood PCR tests could be attributed to the low viral load and the low proportion of infected circulating cells in blood, since 10% are monocytes and only about 1% of these carry proviral sequences in a successful infection. 36
In agreement with our ELISA results, previous studies using a peptide-based ELISA on the same animals also showed a higher and more consistent reaction in sheep infected with strain 496 compared with those infected with strain 697. 46 The antigen spectrum covered by a particular test should always be taken into account, and synthetic peptides designed on the basis of circulating strains may improve diagnosis. 12,46
When evaluated clinically, most animals from group B presented carpal enlargement, indicating that the original disease was reproduced experimentally. However, this enlargement was not statistically significant, likely because of the difficulty in standardizing the technique employed to measure the carpal joint. Nevertheless, necropsy confirmed the presence of a chronic proliferative arthritis, 38 and lesions in carpal joints from group B were the most remarkable pathology observed in this experiment (Table 4).
Both strains could reproduce lesions in target organs, but only strain 496 was efficient in reproducing the lesions triggered by the original isolate. Specifically, strain 496 was isolated from a case of arthritis, 17 and experimental infection caused severe arthritis, whereas strain 697 was isolated from a case of encephalitis 18 yet caused only minimal lesions in the CNS. Strain 496 did not cause demonstrable lesions in the CNS, and strain 697 caused minimal lesions in joints. Both strains were able to reproduce pulmonary SRLV pathology—the most common SRLV form in sheep 13 —and lung lesions were more abundant in group B (496 strain). Despite not having developed lesions in the CNS, animals from group A had a notable serologic response and showed severe lesions in lungs, proving the efficacy of experimental infection with strain 697. Therefore, strain 496 demonstrated its increased virulence when compared with strain 697 in this experimental model.
The alveolar space evaluation was demonstrated to be a useful tool to objectively evaluate the degree of interstitial pneumonia, showing that group B lambs had the most reduction in alveolar space. Our findings in carpal joints and lungs from group B animals further confirm that clinical forms may coexist in the same animal. 26,36,38 The perivascular fibrosis observed in lungs, a finding not often described in sheep, could be associated with the chronic pulmonary disease 1 caused by experimental infection with these SRLV strains. 21,34 For both infected groups, the severity of lesions in target organs seemed to increase over time. Finally, the lack of cross-infection between infected groups and the negative ELISA and PCR results obtained in the control group proved the appropriateness of the sanitary measures taken in this work.
The increased percentage of CD4+ T cells in arthritic lesions is compatible with previous studies on both sheep and goats. 49,50 FoxP3+ regulatory lymphocytes in target tissues did not seem to play a crucial role in lesion development in this study, since FoxP3+ cells were infrequent in affected tissues. Both groups of infected animals showed an elevated number of macrophages (CD68+ cells) in lungs, which is compatible with the role of macrophages as a Trojan horse for SRLVs. 29,35
The results suggest that strain 496 is more pathogenic than strain 697 since its inoculation in our experimental conditions produced higher serologic score values, earlier appearance of pulmonary lesions, increased severity of tissue damage, higher expression of inflammatory markers in pulmonary lesions, and more consistent and positive PCR reactions, and it was the only group in which proviral copies were detected in tissues. Actually, strain 496 is more virulent in vitro and has been classified previously as a rapid/high strain, 17 whereas strain 697 does not cause cytopathic effect in cellular culture and has been classified as slow/low. 18 The failure to reproduce significant CNS lesions by infection with strain 697 in the Rasa Aragonesa lambs might indicate a breed resistance to this form of disease or to this particular strain, as isolate 697 was obtained from a different breed (Assaf sheep) in which nervous disease was the major clinical and pathologic outcome. 7 Indeed, the observation of nervous disease in Rasa Aragonesa sheep is rare (L. Luján, personal observations), despite SRLV infection being endemic in the geographical area where animals of this breed are raised. 37 However, single cases of nervous disease linked to strain 697 infection have been described by our group in Rasa Aragonesa animals, 39 demonstrating the capability of this strain to cause CNS damage also in this breed. Experimental infection with strain 697 in Assaf sheep would definitively clarify this point in future studies.
Besides the above-mentioned host and virus factors, our results suggest a relationship between peripheral blood proviral copies and severity of lesions, as has been demonstrated in previous research. 22,51 Specifically, as observed in results from animals B3 and B6, there is a link between increased serologic score values, almost constantly positive PCR results in blood, severe lesions, and increased number of proviral copies in tissues. It is also known that CNS lesions caused by SRLV are favored by other concurrent infections, 7 a circumstance that was not present under our experimental conditions. SRLV-derived lesions are most likely immune mediated 10 ; thus, the low viral load in group A tissues may have hampered the development of strong immune responses and lesion development 31 or vice versa.
In conclusion, the experimental infection described in this work using 25 autochthonous SRLV strains by the inoculation into bone marrow has been demonstrated to be a useful tool for studying lentiviral pathogenesis, tissue tropism, and its associated pathology. In this Rasa Aragonesa SRLV experimental infection model, strain 496 was more prone to induce arthritis than strain 697 to reproduce encephalitis. This could be due to a higher virulence of strain 496 and/or to a higher resistance of this breed to strain 697. Therefore, our results seem to indicate that both host factors and viral factors are responsible for the final clinical and pathologic findings induced by SRLV infections. More studies are being carried out by our research team to confirm 496 and 697 SRLV tropism and their associated pathology using the intratracheal route in a longer duration of experimental infection.
Footnotes
Acknowledgements
We are indebted to Professor J. Badiola and team at the Centro de Investigación en Encefalopatías y Enfermedades Transmisibles Emergentes that performed all the ELISA tests. F. Vázquez and N. Ardanaz helped with the surgical experimental infection procedures, N. Insausti helped with sampling of experimental animals, and J. Molín provided help with figures and references; all are gratefully acknowledged. We also thank S. Becerra and R. Puyó for technical help. We would like to acknowledge the Servicio General de Apoyo a la Investigación-SAI, Universidad de Zaragoza, Spain.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Grants by the Ministry of Science and Innovation of the Spanish Government, ref. No. AGL2010-22341-C04-02 and AGL2010-22341-C04-01. P. Pinczowski was a PhD student funded by the Brazilian Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, ref. No. 202385/2011-6). R. Reina is supported by Spanish Ministry of Science and Innovation under a ‘Ramón y Cajal’ contract.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
