Abstract
Swine dysentery is classically associated with infection by Brachyspira hyodysenteriae, the only current officially recognized Brachyspira sp. that consistently imparts strong beta-hemolysis on blood agar. Recently, several strongly beta-hemolytic Brachyspira have been isolated from swine with clinical dysentery that are not identified as B. hyodysenteriae by PCR including the recently proposed species “Brachyspira hampsonii.” In this study, 6-week-old pigs were inoculated with either a clinical isolate of “B. hampsonii” (EB107; n = 10) clade II or a classic strain of B. hyodysenteriae (B204; n = 10) to compare gross and microscopic lesions and alterations in colonic mucin expression in pigs with clinical disease versus controls (n = 6). Gross lesions were similar between infected groups. No histologic difference was observed between infected groups with regard to neutrophilic inflammation, colonic crypt depth, mucosal ulceration, or hemorrhage. Histochemical and immunohistochemical evaluation of the apex of the spiral colon revealed decreased expression of sulphated mucins, decreased expression of MUC4, and increased expression of MUC5AC in diseased pigs compared to controls. No difference was observed between diseased pigs in inoculated groups. This study reveals significant alterations in colonic mucin expression in pigs with acute swine dysentery and further reveals that these and other microscopic changes are similar following infection with “B. hampsonii” clade II or B. hyodysenteriae.
Introduction
Brachyspira spp. are Gram-negative spirochetes that have historically been distinguished by their strength of beta-hemolysis on blood agar plates and biochemical reactions including indole cleavage and hippurate hydrolysis. 2,9,22 Classically, Brachyspira hyodysenteriae and Brachyspira pilosicoli isolates have been associated with clinical disease in swine 14 where B. hyodysenteriae is the causative agent of swine dysentery (SD). 18 Clinical isolates of B. hyodysenteriae generally have positive indole and negative hippurate reactions and generate strong beta-hemolysis that is enhanced around slits in blood agar (ring phenomenon). 22 Historically, such reactions allowed for differentiation of B. hyodysenteriae from weakly beta-hemolytic Brachyspira spp. that can be present in the porcine colon including B. intermedia, B. murdochii, B. innocens, and B. pilosicoli.
Not all strongly beta-hemolytic, ring phenomenon positive isolates are identified as B. hyodysenteriae by PCR. Rather, several strongly beta-hemolytic Brachyspira spp. have been recently reported including atypical isolates of B. intermedia, 7 “Brachyspira sp. SASK30446,” 16 and the proposed novel species “Brachyspira suanatina” 35 and “Brachyspira hampsonii.” 6 Recently, isolates of “B. sp. SASK30446” induced clinical disease and lesions similar to B. hyodysenteriae in mice 2 and pigs 3,38 following oral inoculation. Comparison of nox gene sequences from isolates with sequences available in GenBank reveals that “B. sp. SASK 30446” sequences are nearly identical (≥99% identity) over the region compared to clade II isolates of “B. hampsonii.” A representative isolate of this clade, EB107, is the subject of the study reported here.
Clinical SD is characterized by a mucohemorrhagic diarrhea with gross lesions restricted to the large intestine that typically include variable mucosal thickening, hemorrhage, fibrinonecrotic exudate, and variable but often abundant mucus. Histologically, crypt lumens are frequently distended with mucus, there is moderate to severe neutrophilic infiltration in the lamina propria, and spirochetes can often be visualized within crypt lumens and mucus-producing goblet cells by silver staining.
The mucus layer is an important structural feature of the colonic mucosal surface that is responsive to environmental factors in the gut lumen. 8,23 The protein component of mucus is composed of large glycoproteins called mucins, 20 which play an important role in innate mucosal defense. 13 Mucins can provide both a barrier and reporting function and are divided into 2 subgroups: gel-forming mucins and cell surface mucins. 20,34,41 Gel-forming mucins, including mucin 5 AC (MUC5AC), are extracellular and secreted by goblet cells while cell surface mucins, including mucin 4 (MUC4), are membrane-tethered with a short, cytoplasmic tail and extensive extracellular domain. 20 MUC4 is thought to have a largely anti-adhesive role 5 and may provoke a response intended to maintain the integrity of the epithelium. 21 MUC5AC is thought to inhibit adherence and invasion of colonic epithelial cells 33 and instill resistance to enteric nematode infections. 19 Despite the abundant mucus typical of SD, the chemoattractant nature of B. hyodysenteriae to mucus, 24,30 and the importance of mucus bilayer in intestinal disease, the histochemical and immunohistochemical characteristics of mucin-secreting colonic goblet cells have not been extensively evaluated in pigs with SD.
The objectives of the study described here were threefold: (1) to describe the time to onset of clinical SD and the duration of viable spirochete shedding in pigs inoculated with either “B. hampsonii” (EB107) or B. hyodysenteriae (B204), (2) to describe gross lesions and compare histologic lesion severity and distribution within the colons of pigs that develop SD following infection with EB107 versus B204, and (3) to characterize the biosynthetic responses in mucin-secreting colonic goblet cells in pigs with acute clinical SD following infection with either EB107 or B204.
Materials and Methods
Animals
All procedures were approved by the Institutional Animal Care and Use Committee of Iowa State University. Twenty-six 6-week-old crossbred pigs were obtained from a commercial source with no known previous history of Brachyspira-associated disease and were PCR negative for Lawsonia intracellularis upon arrival. Pigs were separated into 3 groups with 6 control pigs and 10 pigs per inoculated group. All pigs were ear tagged for individual identification and swabbed rectally to test for the presence of Brachyspira spp. by microbial culture 7 days prior to inoculation. Each group of pigs was maintained in a separate room to prevent any contact between groups. Pigs were acclimated to these groups and to the facility for 1 week. During this period and throughout the study, pigs were fed a nonmedicated, corn and soybean diet nutritionally complete for their age.
Bacterial Strains, Growth Conditions, and Preparation of Inocula
Media, isolates, and challenge inocula were prepared as previously described. 3 Isolates used in this study were obtained from the culture collection at the Iowa State University Veterinary Diagnostic Laboratory. The “B. hampsonii” clade II isolate (EB107) was previously used in a pig inoculation experiment 3 after being recovered from a clinical case of SD in 2011 and was 9th through 12th passage at the time of inoculation. The B. hyodysenteriae B204 isolate was originally recovered from a clinical case of SD in 1972 and was 9th through 10th passage at the time of inoculation.
For isolation of Brachyspira spp., individual rectal swabs were collected at 5, 7, 9, 12, 15, and 19 days post inoculation (DPI) and at necropsy. Swabs were plated onto selective agars within 6 hours of collection and incubated as previously described. 3 Specifically, swabs were plated onto trypticase soy agar with 5% defibrinated bovine blood (TSA); CVS selective agar containing colistin, vancomycin, and spectinomycin; and BJ selective agar containing pig feces extract, spiramycin, rifampin, vancomycin, colistin, and spectinomycin. Plate media used in this study were prepared in-house and passed the quality assurance standards of the Iowa State University Veterinary Diagnostic Laboratory. An anaerobic environment was provided by a commercial system (BD GasPak EZ Anaerobe Container System, BD Diagnostic Systems, Sparks, MD) and plates were incubated at 41°C ± 1°C. Agar plates were observed for growth at 2, 4, and 6 DPI. Mucosal scrapings collected at necropsy were plated and evaluated as described previously and were also plated onto MacConkey’s agar and brilliant green agar with novobiocin in addition to tetrathionate broth enrichment subcultured to brilliant green with novobiocin and XLT4 to test for the presence of Salmonella spp.
Animal Inoculation
Pigs were inoculated with an agar slurry as previously described 3 with slight modification: pigs received 3 doses of inoculum (100 ml/dose) administered via gavage 24 hours apart with each administration preceded by a 12- to 18-hour fast. A 5-g sample of each inoculum was reserved from which 1 gram was vortexed for 45 seconds in tubes with 9 ml of sterile PBS and a few glass beads. A standard plate count procedure was performed by titration of 1 ml of the vortexed sample into 9 ml and carried out to 10–9. The dilution series was plated on trypticase soy agar with 5% defibrinated bovine blood and incubated anaerobically for 6 days with plates being observed on days 2, 4, and 6. Brachyspira spp. grew confluently on the more concentrated dilutions, but discrete colonies were observed from the more dilute plates. Colonies were counted after 6 days incubation to obtain the inoculum titer in colony-forming units (CFU) per ml. Pigs in the B204 group received 5 × 105 CFU/ml, 8 × 105 CFU/ml, and 4 × 105 CFU/ml on DPI 0, 1, and 2, respectively. Pigs in the EB107 group received 7 × 105 CFU/ml, 1 × 106 CFU/ml, and 1 × 106 CFU/ml on DPI 0, 1, and 2, respectively. Pigs in the control group received a sham inoculum consisting of agar material from non-inoculated culture plates prepared in the same manner as for the Brachyspira inocula.
Molecular Identification
The subpassaged isolates used to prepare the inocula were verified to species by PCR assays targeting nox gene sequences as previously described (B. hyodysenteriae, B. pilosicoli, and B. intermedia; 39 B. murdochii and B. innocens; 46 and “B. sp SASK30446” 3 ). All necropsy isolates were confirmed to species by either direct sample PCR on mucosal scrapings or from primary cultures of mucosal scrapings in cases where mucosal scrapings were negative by PCR.
Animal Observations and Necropsy
Throughout the study period, investigators were blinded to the molecular identification of the inoculum. Following inoculation, animals were observed at least twice daily for feed consumption, availability of adequate water, and clinical illness. Fecal consistency was determined daily, and each pig received a score based upon the following system: 0 if normal, 1 if soft but formed, 2 if semisolid, and 3 if liquid to watery with an additional 0.5 point added each for the presence of discernible mucus and/or blood. Animals were euthanized by barbiturate overdose 24 to 48 hours after the first observation of diarrhea with blood and mucus or at the termination of the study 21 DPI. At necropsy, the entire intestinal tract was observed for gross lesions and the full length of the cecal and colonic lumens were exposed and evaluated for the presence and distribution of luminal mucus, mucosal hemorrhage, and fibrinous exudate. Tissue samples were collected and placed in 10% neutral buffered formalin and included representative sections of jejunum, ileum, cecum, base of the spiral colon (section of the centripetal spiral colon that connects with the cecum), apex of the spiral colon, and a site-independent section of colon representative of overall lesion severity or midsection of the centrifugal spiral colon if no lesion was present. After 24 hours of fixation, tissue samples were transferred to 70% ethanol and processed routinely for histopathology. Samples were also collected from each pig for microbial culture and direct sample PCR and included a rectal swab and colonic mucosal scrapings.
Histopathology
The investigator was blinded to individual pig numbers and group identification. Sections were cut to 4 µm and stained routinely with hematoxylin and eosin. Sections of jejunum and ileum were evaluated for the presence of any lesions unrelated to Brachyspira infection. Sections of cecum and spiral colon were evaluated for the presence of ulceration, hemorrhage, and neutrophilic infiltration of the lamina propria. Neutrophils were counted in ten 40× fields and the mean for each section of the cecum, base of the spiral colon, apex of the spiral colon, and lesion or midsection of the spiral colon was determined. Ulceration was scored as follows: 0 if no ulceration, 1 for focal ulceration spanning 1 to 3 crypts, 2 for focal ulceration spanning 3 to 5 crypts, and 3 for focal ulceration spanning more than 5 crypts. An additional 0.5 was added for multifocal ulceration or 1 for multifocal ulceration spanning more than 5 crypts. The lamina propria and lumen were evaluated for hemorrhage, and each anatomic location was assigned a score based upon the most severe lesion in that tissue section. Scores were determined as follows: 0 if <5 red blood cells (RBC), 1 if 6 to 10 RBC, 2 if 11 to 20 RBC, 4 if 21 to 50 RBC, and 5 if >51 RBC were observed with an additional 1 point added to the score for more than 3 foci of hemorrhage within a single histologic section of a given anatomic location. Hemorrhage scores at all locations were combined to form a composite score. Mucosal thickness was measured in each section using a standard eyepiece micrometer and measurements were taken from an area where the crypts were perpendicular to the mucosal surface with intact epithelium. The mean of 3 measurements was calculated for each section of the cecum, base of the spiral colon, apex of the spiral colon, and lesion or midsection of the spiral colon for each pig.
Immunohistochemistry
Primary antibodies for MUC4 and MUC5AC that have previously been shown to have specificity in pig large intestine 25 were used for immunohistochemistry (IHC) (Zymed Laboratories, Invitrogen Corporation, Carlsbad, CA). Sections (5 μm) of the apex were cut onto positively charged slides, placed in an incubator at 57°C for 2 hours, and deparaffinized through graded alcohol. Endogenous peroxidase was inhibited by application of 3% hydrogen peroxide for 20 minutes followed by 3 rinses with ultrapure water. Slides were processed for antigen retrieval by microwaving in Tris/EDTA (pH 9.0) for 1 minute followed by a steam chamber for 20 minutes. Slides were then incubated with 1:10 dilution of goat serum and PBS buffer for 20 minutes at 37°C to saturate nonspecific protein binding sites. All primary antibodies were diluted 1:50 in PBS containing 0.1% Tween 20. Slides were incubated with primary antibodies overnight at 4°C in a humidified chamber. After washing 2 times in PBS, a 5 minute PBS bath, and 2 more rinses with PBS, sections were incubated with a conjugated secondary antibody (Mouse-on-Farma HRP-Polymer, Biocare Medical, Concord, CA) for 1 hour at 37°C and then washed as described for the primary antibody. Slides were subsequently incubated with a commercial chromagen (NovaREDTM, Vector laboratories, Burlingame, CA) for 5 minutes at 37°C followed by rinsing with ultrapure water. Finally, the sections were counterstained with Shandon’s hematoxylin, placed in Scott’s Tap water for 1 minute, rinsed with ultrapure water, dehydrated through a graded alcohol series, and mounted.
Histochemistry
Sections (4 µm) of the apex were placed onto positively charged glass slides, deparaffinized, and treated with a combination of high-iron-diamine and Alcian blue (pH 2.5) (HID/AB) staining. Sections were placed in high-iron-diamine solution for 18 hours at 37°C, rinsed with water, placed in Alcian blue solution for 5 minutes, rinsed with water, dehydrated, and mounted.
Image Analysis
Quantitative analysis of histochemical and immunohistochemical assays was performed using a commercial software program (Photoshop CS4, Adobe Systems Inc., San Jose, CA). Five representative 40× images were captured for each slide where each image had the same number of total pixels. The number of pixels representing positive staining was recorded for each image and the average number of pixels representing positive staining was calculated per slide.
Fluorescent in Situ Hybridization
An oligonucleotide probe specific for 23 S rRNA of Brachyspira spp. (SER1410) 1 was used under hybridization conditions as previously described. 4 A genus-based probe was used to ensure the evaluator remained blinded to the molecular identification of the spirochete present in the section. Five 40× fields from each lesioned section of spiral colon were evaluated for localization (within epithelial cells, goblet cells, and crypt lumens) and quantification (few, moderate, many) of spirochetes.
Statistical Analyses
A commercial statistical software package(JMP Pro 10, SAS Institute, Cary, NC) was utilized to perform all analyses. A one-way analysis of variance (ANOVA) was used to detect significant differences in mean neutrophilic inflammation, ulceration score, mucosal crypt depth, and composite hemorrhage score with a Tukey-Kramer or Steel-Dwass adjustment for multiple comparisons when a residual plot demonstrated equal or unequal variance, respectively. Following logarithmic transformation, HID/AB staining (sulphated mucins), MUC4 expression, and MUC5AC expression was analyzed using an ANOVA with Tukey-Kramer adjustment. In all circumstances, values of P ≤ .05 were considered significant and means are reported with standard error of the mean.
Results
Animal Observations
Loose stools with blood and mucus were first observed 5 DPI in the “B. hampsonii”–inoculated group (EB107) and 8 DPI in the B. hyodysenteriae–inoculated group (B204). The average DPI to the onset of clinical SD for the EB107 group (mean ± SEM = 12.2 ± 5.36) was similar to the B204 group (11.7 ± 2.60). Fecal scores and timing of euthanasia for each pig are summarized by inoculation group in Table 1.
Summary of Fecal Scores, Brachyspira Culture Results, and Timing of Euthanasia.a
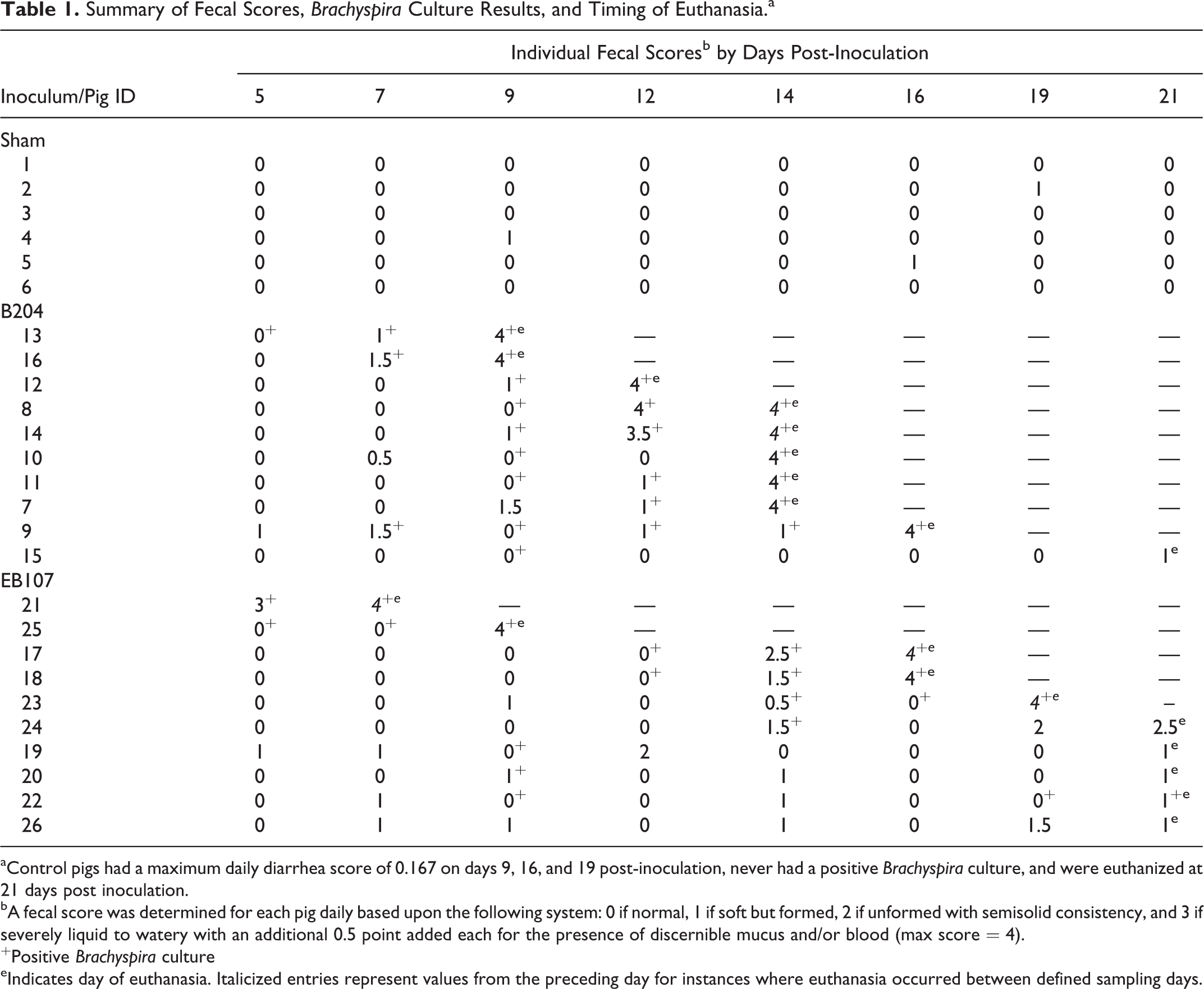
aControl pigs had a maximum daily diarrhea score of 0.167 on days 9, 16, and 19 post-inoculation, never had a positive Brachyspira culture, and were euthanized at 21 days post inoculation.
bA fecal score was determined for each pig daily based upon the following system: 0 if normal, 1 if soft but formed, 2 if unformed with semisolid consistency, and 3 if severely liquid to watery with an additional 0.5 point added each for the presence of discernible mucus and/or blood (max score = 4).
+Positive Brachyspira culture
eIndicates day of euthanasia. Italicized entries represent values from the preceding day for instances where euthanasia occurred between defined sampling days.
Microbial Culture
All preinoculation fecal samples were negative for Brachyspira spp. The days with positive Brachyspira culture for each pig are reported in Table 1, and a summary of the percentage shedding and PCR results by group are summarized in Table 2. Shedding was first detected 5 DPI for both the EB107 and B204 groups. The average period of positive culture to the development of clinical SD was shorter for the EB107 group (2.6 days ± 1.5) than the B204 group (3.2 days ± 2.5).
Positive Brachyspira Culture and PCR Results in Feces and Tissue Following B. Hyodysenteriae (B204) or “B. hampsonii” (E107) Challenge.
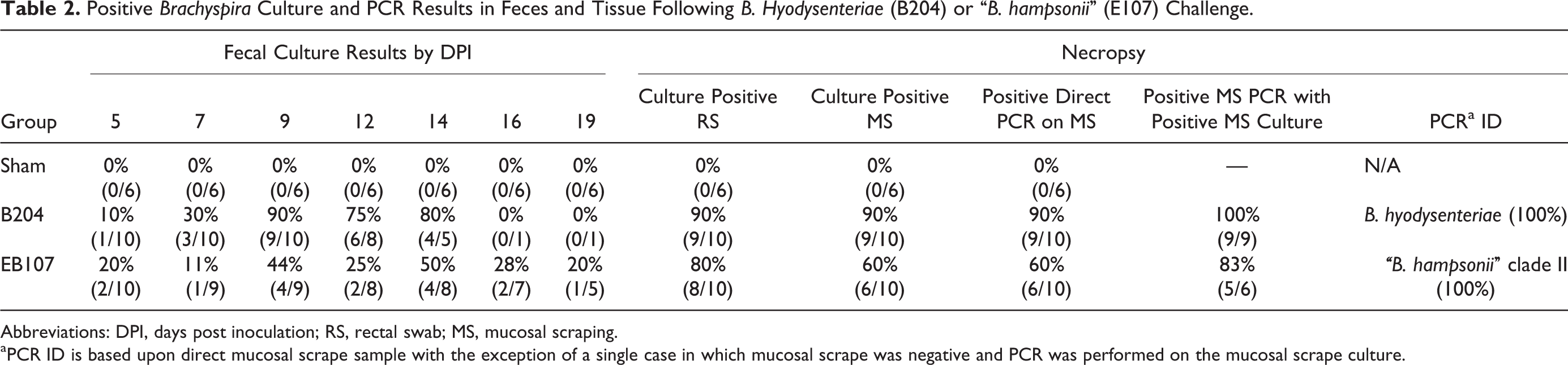
Abbreviations: DPI, days post inoculation; RS, rectal swab; MS, mucosal scraping.
aPCR ID is based upon direct mucosal scrape sample with the exception of a single case in which mucosal scrape was negative and PCR was performed on the mucosal scrape culture.
Gross Pathology
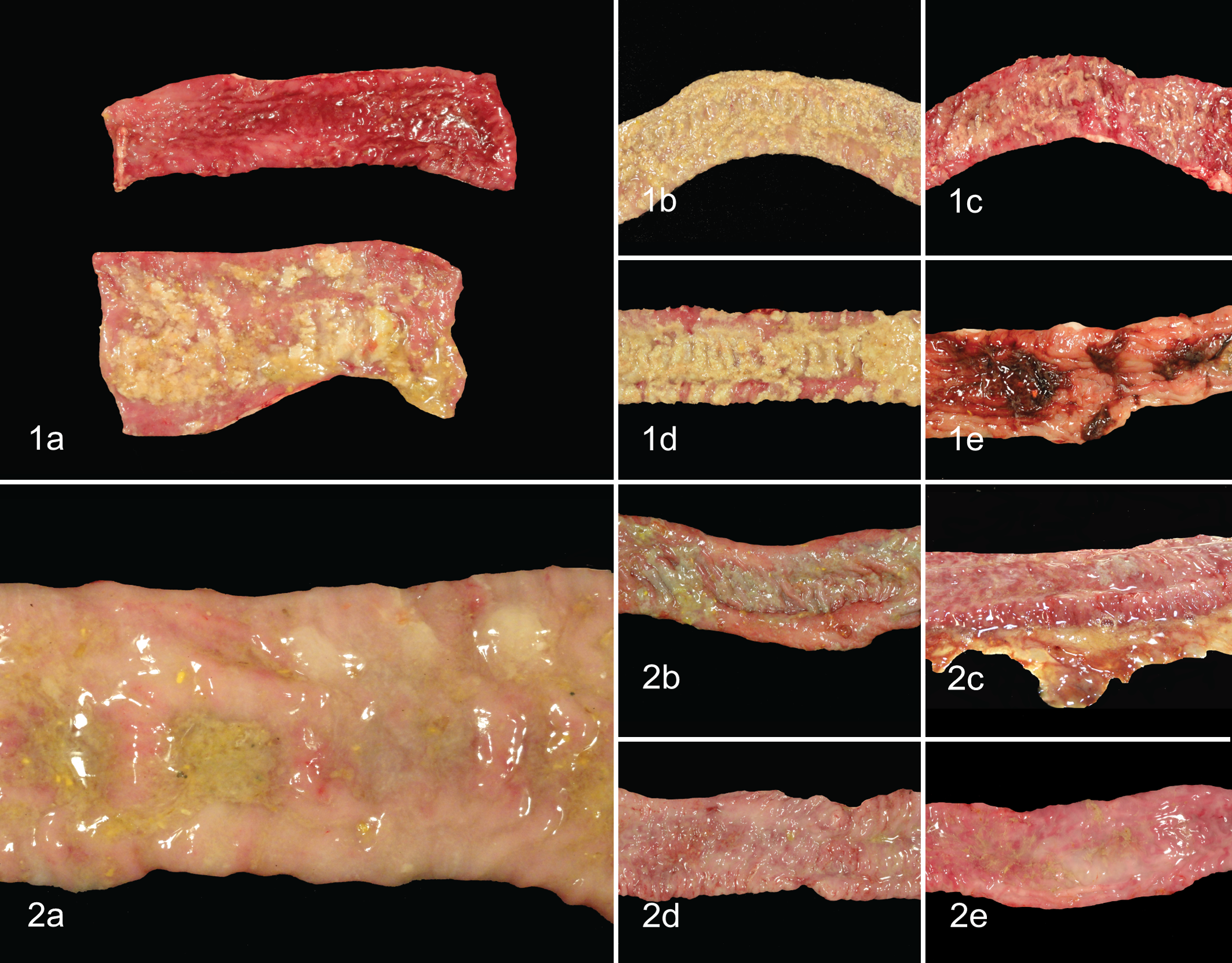
Gross lesions observed in each individual group are summarized in Table 3. When present, gross lesions in the intestinal tract were limited to the cecum and large intestines but were most common in the centripetal, apex, and centrifugal portions of the spiral colon. Lesions consisted of variable degrees of fibrinous exudate on the luminal surface; mucosal thickening, mucosal congestion, and hemorrhage; and excessive luminal mucus. Representative lesions from each inoculation group are displayed in Figs. 1 and 2. Cecal lesions were observed only in animals inoculated with B204 and were limited to those pigs in which fibrinous exudate was also present in the majority of the large intestine. Fibrinous exudate was more common in those animals in the B204 group that developed SD (8/9; 89%) than those in the EB107 group that developed SD (1/5; 20%). Of those inoculated with EB107 that developed SD, the frequency of moderate to severe luminal mucus was greater (5/5; 100%) than the frequency of those inoculated with B204 that developed SD (6/9; 67%) while the frequency of moderate to severe mucosal hemorrhage was more common in those inoculated with B204 that developed SD (5/9; 56%) than those inoculated with EB107 that developed SD (1/5; 20%). No gross lesions were observed in the sham-inoculated group.
Necropsy Lesions in Pigs That Developed Clinical SDFollowing B. Hyodysenteriae (B204) or “B. Hampsonii” (E107) Inoculation.
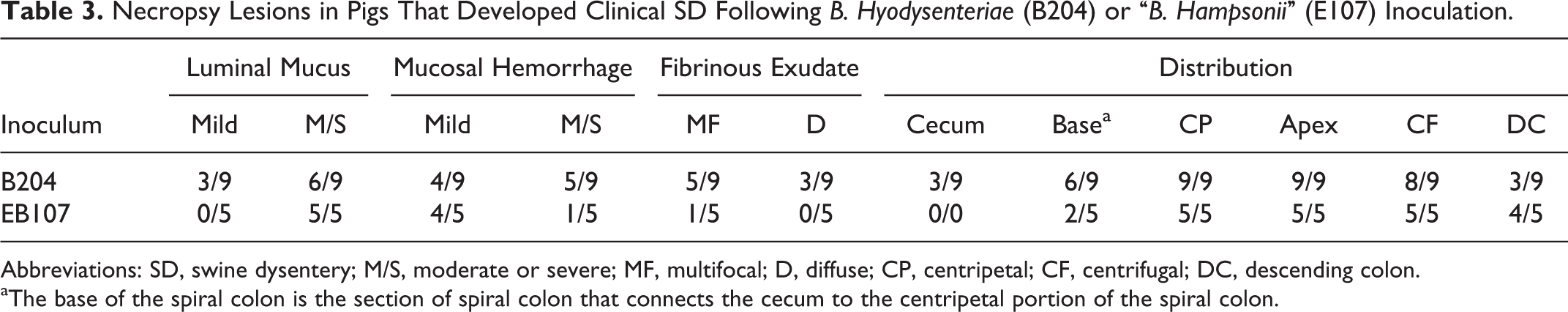
Abbreviations: SD, swine dysentery; M/S, moderate or severe; MF, multifocal; D, diffuse; CP, centripetal; CF, centrifugal; DC, descending colon.
aThe base of the spiral colon is the section of spiral colon that connects the cecum to the centripetal portion of the spiral colon.
Histopathology
Site-Independent, Representative Lesion
Microscopic evaluation of site-independent, lesioned sections from diseased pigs or midsections of the spiral colon in sham-inoculated pigs is summarized in Fig. 3. A significant difference was observed in neutrophilic inflammation (P < .03), crypt depth (P < .03), and ulceration (P < .0001) between each infected group and the sham-inoculated group. However, a significant difference in the mean composite hemorrhage score was only identified between the B204-inoculated group that developed disease (7.33 ± 2.73) and the sham-inoculated group (0.50 ± 0.50, P = .005) and not between the EB107-inoculated group that developed disease (7.80 ± 4.92) and sham-inoculated group (P = .07).
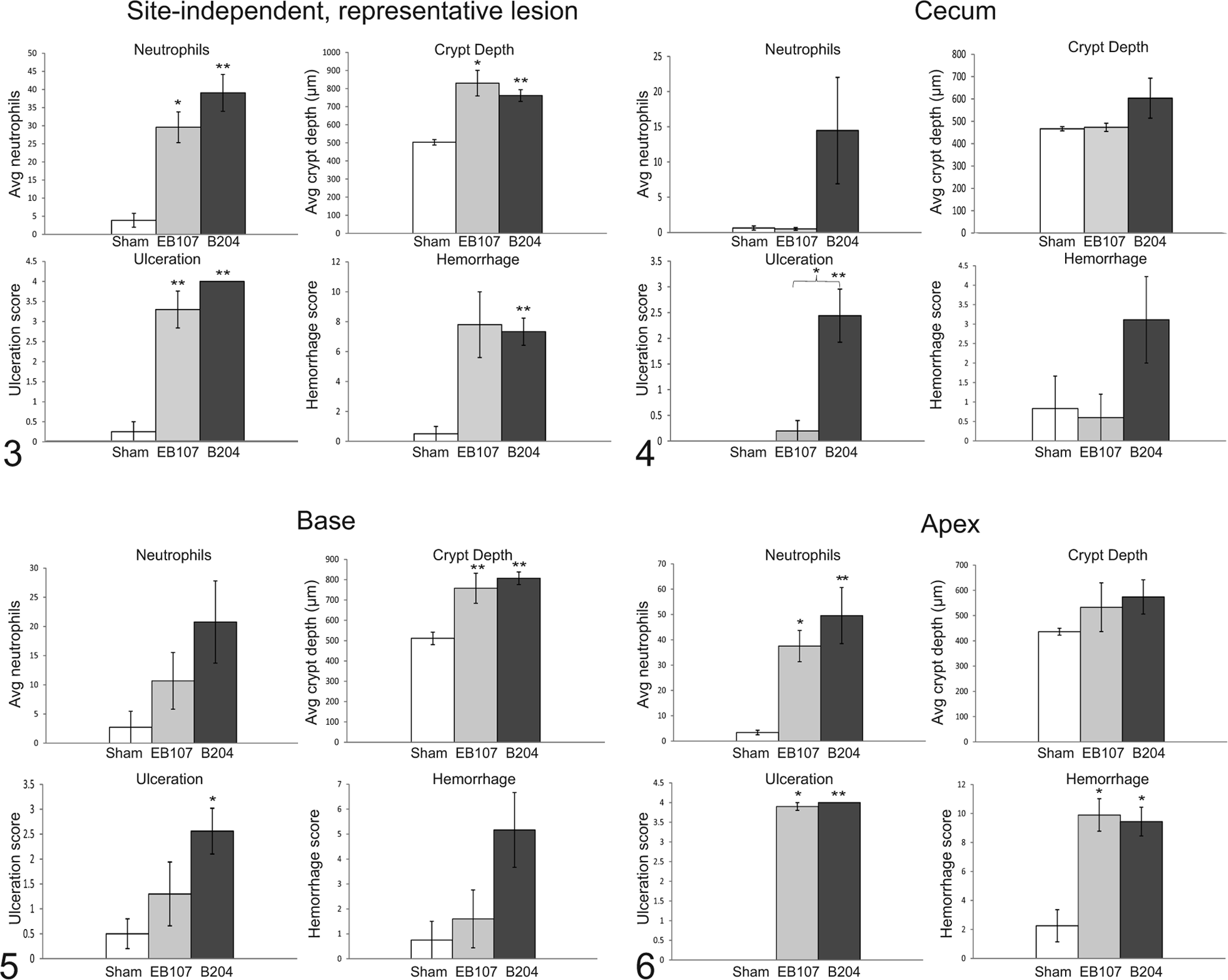
Cecum
Cecal histologic evaluation is summarized in Fig. 4. Briefly, in the cecum there was a significant difference in cecal ulceration score between the B204-inoculated group that developed disease (2.44 ± 0.52) versus those in the EB107-inoculated group that developed disease (0.20 ± 0.20) and the sham-inoculated group (0.00 ± 0.00) (P = .02 and P = .008, respectively).
Base of the Spiral Colon
Histologic evaluation of the base of the spiral colon is summarized in Fig. 5. At the base of the spiral colon there was a significant difference in mucosal crypt depth between the B204-inoculated and EB107-inoculated groups that developed disease and the sham-inoculated group (P = .0003 and P = .005, respectively). A significant difference in the base ulceration score was only observed between the B204-inoculated group that developed disease and the sham-inoculated group (P = .016).
Apex of the Spiral Colon
Histologic evaluation of the apex of the spiral colon is summarized in Fig. 6. At the apex of the spiral colon there was a significant difference in neutrophilic inflammation (P < .03, all analyses), ulceration score (P < .04, all analyses), and composite hemorrhage (P < .03, all analyses) score between each isolate-inoculated group and the sham-inoculated controls.
Immunohistochemistry
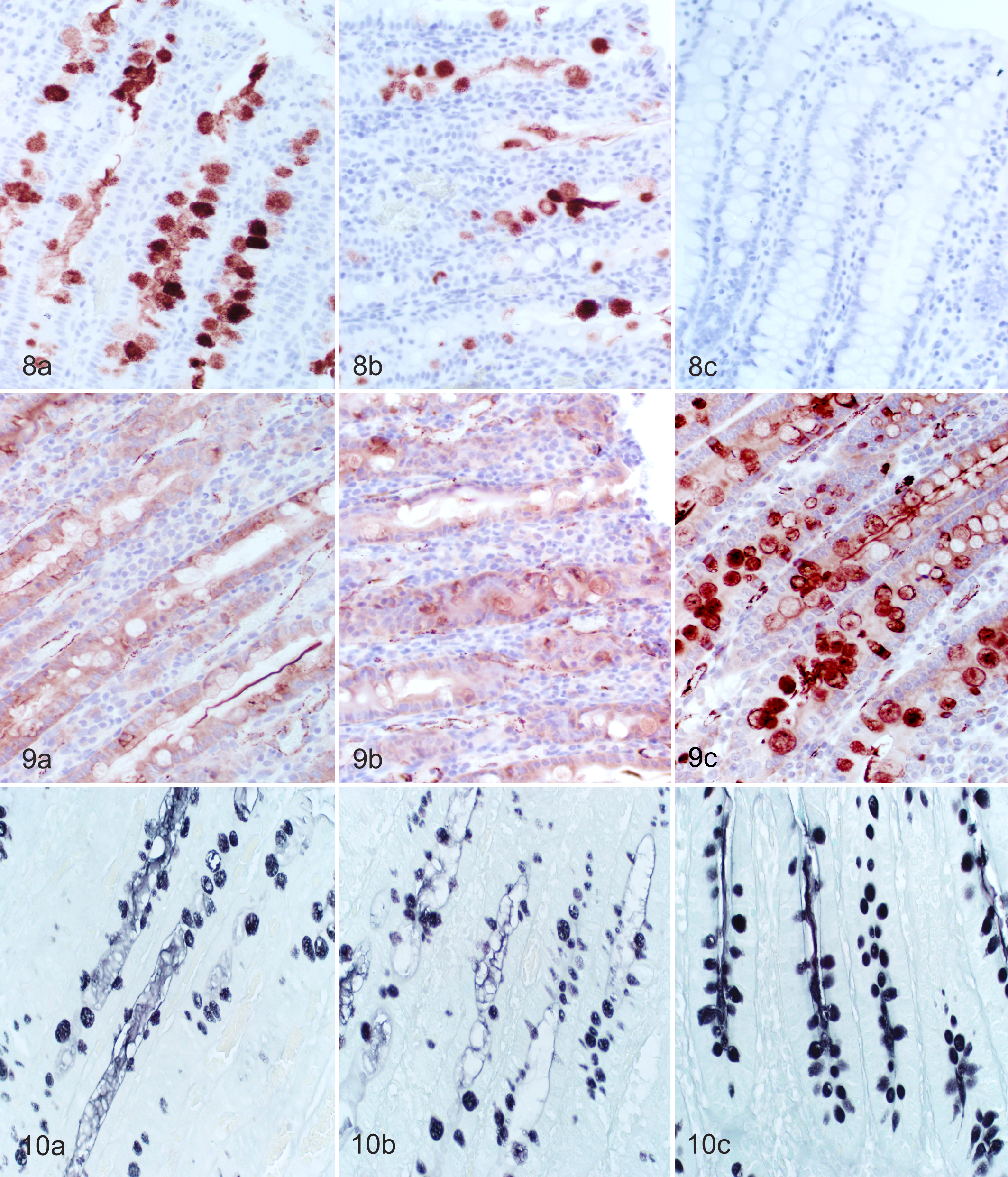
Image analysis results of mucin immunohistochemistry are summarized in Fig. 7. There was a significant decrease in MUC4 expression between the B204-inoculated and the EB107-inoculated groups that developed SD and controls (P < .0001 and P < .0001, respectively). The decrease in MUC4 expression was concurrent with a significant increase in MUC5AC expression in pigs that developed SD in the B204-inoculated and the EB107-inoculated groups compared to controls (P = .0005 and P = .007, respectively). Representative photomicrographs of tissue sections used for image analysis are displayed in Fig. 8 and Fig. 9.
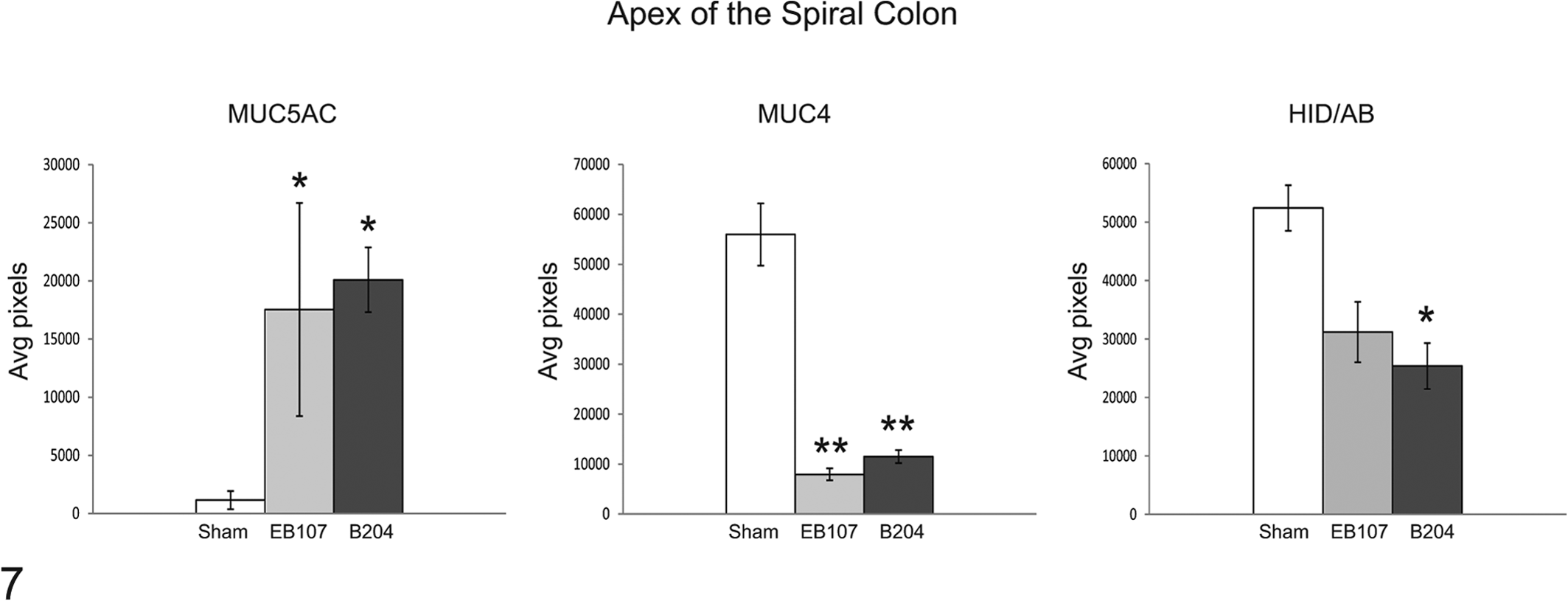
Means ± SEM for image analysis of immunohistochemical and histochemical stains on sections taken from the apex of the spiral colon. *P < .01. **P < .0001.
Histochemistry
Image analysis results for mucin histochemistry are summarized in Fig. 7 with representative photomicrographs of tissue sections used for analysis displayed in Fig. 10. There was a decrease in the amount of sulphated mucins at the apex of the spiral colon in pigs that developed SD; however, this difference was only significant between the B204-inoculated group that developed disease (25,374.6 ± 3,923.0) and the sham-inoculated group (52,401.5 ± 6,236.3, P = .0023) and not between the EB107-inoculated group that developed disease (31,192.0 ± 5,162.1) and sham-inoculated group (P = .054).
Fluorescent In-Situ Hybridization
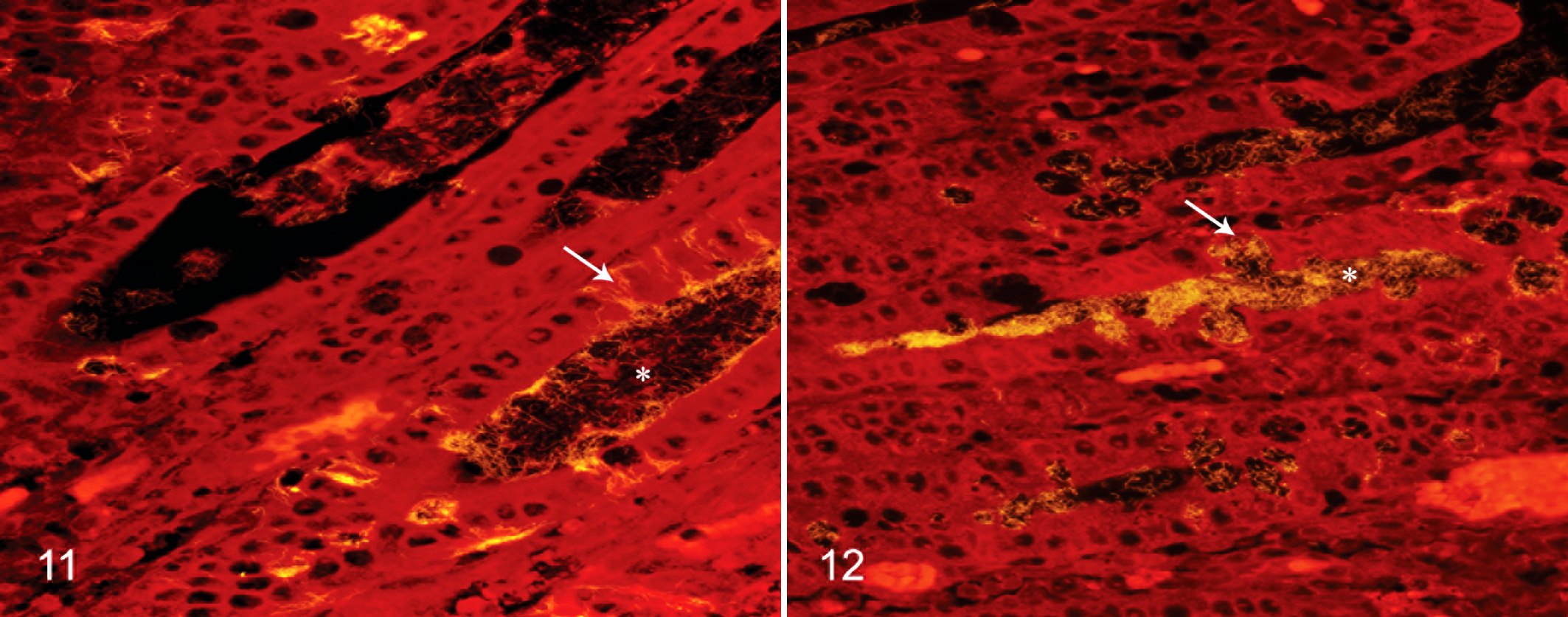
Both B204 and EB107 were observed within epithelial goblet cells (Figs. 11 and 12); however, moderate to high numbers of intracellular spirochetes were only observed in sections from B204-inoculated pigs. Both B204 and EB107 were observed in sparse to large numbers within multifocal crypt lumens throughout affected sections.
Molecular Identification
All isolated spirochetes and inocula were positive only for the appropriate Brachyspira sp. in all testing. When positive, direct PCR assays performed on mucosal scrapings revealed the presence of the species inoculated (Table 2) and only a single mucosal scraping was negative by direct PCR assay but positive by microbial culture.
Discussion
Recent work in C3H/HeN mice 2 and pigs 3 has suggested that lesions associated with infection by clinical isolates representative of clades I and II of “B. hampsonii” are similar to, if not indistinguishable from, B. hyodysenteriae infection. Findings of another recent study using a Canadian isolate of “B. hampsonii” clade II provide further support that lesions in inoculated pigs are similar to those associated with B. hyodysenteriae infection. 38 In the present study, multiple gross and microscopic parameters were compared and revealed minimal differences in lesion characteristics, severity, or distribution between pigs infected with either B. hyodysenteriae or “B. hampsonii” and provide unequivocal support for “B. hampsonii” as an agent of SD.
The average DPI to the onset of clinical SD and the average period of shedding prior to the development of clinical SD was similar between the 2 Brachyspira spp. tested. The duration between inoculation and initial observation of grossly detectable fecal blood and/or mucus in the present study was similar to previous studies. 3,26,38 With regard to preclinical spirochete shedding, previous reports have described the appearance of spirochetes in the feces 1 to 4 days before observable diarrhea with B. hyodysenteriae 26 and rarely more than 2 days before the onset of mucoid or hemorrhagic diarrhea with “B. hampsonii” clade II. 38 While this period of preclinical shedding is consistent with a majority of pigs in the present study, a single pig in the B204-inoculated group had 4 consecutive positive cultures spanning an 8-day period prior to the development of diarrhea, suggesting the potential for prolonged shedding period and horizontal exposure prior to disease recognition.
No difference in the sensitivity of detection was observed when comparing culture of either rectal or mucosal scraping swabs to direct PCR on colonic mucosal scrapings collected at necropsy in the B204 group with each sample being positive in all 9 pigs that developed clinical SD. Similarly, the 5 pigs that developed clinical SD in the EB107 group had positive rectal and mucosal cultures and appropriate molecular identification via PCR. Two pigs did not develop clinical SD but had either a positive rectal swab or a positive mucosal scraping culture at necropsy. Interestingly, a positive molecular identification via PCR occurred in the pig with the negative mucosal scraping culture while the pig with the positive mucosal scraping culture was PCR negative. Surprisingly, direct PCR assays for Brachyspira spp. from clinical specimens were no less sensitive than microbial culture in the current study, which is in contrast to a previous report. 3 This discordance may be due to differences in the severity of clinical disease and the time between disease onset and euthanasia for necropsy and sample collection. In the current study, pigs were euthanized within 48 hours of the onset of clinical SD and all had severe gross and histologic lesions with numerous Brachyspira detected by fluorescent in-situ hybridization. This is suggestive of a large population of infectious organisms that could be easily detected by either selective culture or direct PCR. Accordingly, it seems likely that in acute cases of SD either culture or direct PCR may be equally sensitive; however, in less fulminate cases or in surveillance situations, direct culture of either the colonic mucosa or feces may be of highest diagnostic value.
Given the abundant mucus typical of SD, the chemoattraction of B. hyodysenteriae to mucus, 24,30 and the importance of the mucus bilayer in intestinal health and disease, the histochemical and immunohistochemical characteristics of mucin-secreting goblet cells were evaluated in the present study. Evaluation of these cells at the apex of the spiral colon revealed a significant decrease in MUC4 with a significant concurrent increase in MUC5AC in pigs infected with either Brachyspira sp. compared to controls; however, a decrease in sulphated mucins was only statistically significant when comparing B204-inoculated pigs and controls. The lack of a statistically significant difference in sulphated mucins between EB107-inoculated pigs and controls may be due to the greater variability in sulphated mucin expression in those pigs that developed SD or the low total number of diseased pigs in this inoculation group or both. This is supported by a P-value that approaches significance (P = .054); however, further study is warranted to fully evaluate the effect of “B. hampsonii” infection on sulphated mucin expression.
The increased expression of MUC5AC, a gel-forming mucin important in inhibition of bacterial adherence and invasion, 33 observed in all infected pigs that developed SD in this study is likely associated with the inflammatory infiltrate present in the lamina propria as induced expression of MUC5AC is caused by proinflammatory cytokines including interleukin-1β (IL-1β), 11,33 interleukin-17(IL-17), 11 and neutrophil elastase. 10 The intense inflammatory infiltrate characterized by neutrophils and probable presence of proinflammatory cytokines pertinent to the differentiation of CD4+ T cells suggests the Th17 cells may be involved in acute SD. Th17 cells play an important role in host defense at mucosal surfaces against extracellular bacteria and have a unique link to neutrophils. 45
MUC4 is a membrane-tethered mucin likely important in epithelial cell renewal, differentiation, and signaling. 12,34 The decreased MUC4 expression in pigs with acute SD may result from TGF-β repression of the precursor cleavage of MUC4 thereby inhibiting MUC4 expression. 28 MUC4 is also expressed at significantly lower levels on the surface of the crypt epithelium of S. typhimurium–infected pigs compared with non-infected pigs. 25
Decreased expression of sulphated mucins constitutes the first step in mucin degradation, providing a rich source of nutrients including carbon for bacterial growth, 37 and improved growth of intestinal Brachyspira spp. has been observed following mucin addition to media. 40 Evidence of mucin degradation also suggests a mechanism by which pathogenic Brachyspira spp. may breach the protective mucus bilayer to access the underlying epithelial cells. 40 Mucin sulphatase activity has been documented in bacteria associated with most mucosal surfaces including the enteric genera Bacteroides and Prevotella; 36,43,47 However, in a recent comparison of the complete genome sequences of multiple Brachyspira spp., only a limited number of sulphatase genes were identified including 3 in B. pilosicoli 95/1000, 1 putative gene in B. murdochii 56-150T, and none in B. hyodysenteriae WA1. 44 Sequencing of additional species and strains is needed to better characterize the presence of sulphatase genes in Brachyspira spp. as well as further work to determine the activity of the encoded proteins. To the authors’ knowledge, sulphatase activity has not been described with either B. hyodysenteriae or “B. hampsonii,” suggesting the potential necessity of concurrent colonization with another bacterium capable of degrading sulphated mucins in order to induce the decreased expression of these mucins as observed in the present study. Indeed, the requirement for additional bacteria as part of the pathogenesis of SD has been demonstrated in gnotobiotic pigs. 17,29
It is well recognized that diet has a major impact on SD expression as feeding diets that are either highly digestible 32,42 or rich in inulin inhibits colonization by B. hyodysenteriae. 15 Sulphatase activity may also be directly impacted by feed composition resulting in a shift of the microbiome of the gut. A recent study revealed that pigs lack the terminal restriction fragments corresponding to the sulphatase-producing Prevotella group when fed cooked rice or cooked rice supplemented with either 10% potato starch or 20% wheat bran. 27 Another study found that pigs fed fructan-rich diets, similar to those shown to prevent SD, had a higher proportion of Bifidobacterium thermacidophilum subsp. porcinum and Megasphaera elsdenii possibly resulting in alterations in the intestinal environment. 31 Such changes may decrease the sulfphatase activity present in the gut inhibiting penetration of the protective mucus layer and disease initiation.
In summary, the results of the present study reveal that the time to onset of mucohemorrhagic diarrhea and the duration of spirochete shedding were similar for pigs infected with either “B. hampsonii” clade II or B. hyodysenteriae under the conditions of this report. Furthermore, significant alterations in colonic mucin expression were observed in pigs inoculated with either “B. hampsonii” or B. hyodysenteriae within 48 hours of the onset of mucohemorrhagic diarrhea, and these and other microscopic changes were not significantly different between infected groups, thus further supporting a role for either spirochete as a cause of SD. The specific alterations in colonic mucin expression observed suggest the potential importance of proinflammatory cytokines in the induction of differential mucin expression in acute SD as well as the potential need for concurrent colonization with a bacterium possessing sulphatase activity. Further studies exploring these potential relationships are warranted and may shed light on the specific host and microbial factors involved in the pathogenesis of swine dysentery.
Footnotes
Acknowledgements
The authors wish to thank Hallie Warneke for her help with the Brachyspira culture work and maintenance of the isolate collection at the ISU VDL; Diane Gerjets, Jennifer Groeltz-Thrush, and Toni Christofferson for preparation of the histologic sections; Deb Moore for her help optimizing the immunohistochemistry procedures; and Kate Sawyer and Marissa Rotolo, for their assistance with the animal inoculation and necropsy procedures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a grant from the Iowa Pork Producers Association and the National Pork Board (NPB #11-178).
