Abstract
Bovine viral diarrhea virus (BVDV) continues to be of economic significance to the livestock industry in terms of acute disease and fetal loss. Many of the lesions relating to BVDV infection have been well described previously. The virus is perpetuated in herds through the presence of calves that are persistently infected. Relationships between various species and biotypes of BVDV and host defenses are increasingly understood. Understanding of the host defense mechanisms of innate immunity and adaptive immunity continues to improve, and the effects of the virus on these immune mechanisms are being used to explain how persistent infection develops. The noncytopathic biotype of BVDV plays the major role in its effects on the host defenses by inhibiting various aspects of the innate immune system and creation of immunotolerance in the fetus during early gestation. Recent advances have allowed for development of affordable test strategies to identify and remove persistently infected animals. With these improved tests and removal strategies, the livestock industry can begin more widespread effective control programs.
Keywords
Introduction
Bovine viral diarrhea virus (BVDV) is an economically important pathogen that belongs to the genus Pestivirus of the family Flaviviridae. 1 Bovine viral diarrhea virus causes bovine viral diarrhea (BVD), which was first reported as a transmissible disease in 1946. 112 Since that time, BVD has been reported worldwide. 46,50,128,136,147 Two genotypes, BVDV type 1 and BVDV type 2, are further classified as cytopathogenic (cp) or noncytopathogenic (ncp) based on in vitro cell culture characteristics. 1,55,71,84,126
Modulation of the innate immune response in the fetus by ncp BVDV is of key importance in establishing persistent infection with BVDV. Understanding the interplay of BVDV proteins and cellular regulation of the innate immune system helps explain the development of persistent infection. Although acute BVDV infections are often subclinical or produce only mild clinical signs, they induce lymphopenia and a range of effects on the immune response, which exacerbate secondary bacterial or viral infections. 19,79,89,117,119 BVDV infects a wide variety of cell types but has a predilection for cells of the immune system such as monocytes/macrophages, dendritic cells (DCs), and lymphocytes. 19,61,90 The consequences of infection include the death of these cell populations as an extreme event or more subtle effects on cytokine expression and synthesis of costimulatory molecules. 18,26,61,119 The changes in cytokine production by immune or nonimmune cells affect both innate and adaptive immunity. 108
The persistently infected (PI) animal serves as the reservoir of BVDV and a source of infection. Recent advances allow better detection of PI animals and thus improve the possibility of enhanced control.
Pathogenesis
Two genotypes, BVDV type 1 and BVDV type 2, are identified as distinct species within this genus, with further classification as cp and ncp based on in vitro cell culture characteristics and genetic differences. 1,55,71,84,126 Genotype 1 is considered the Pestivirus-type species and is reportedly the most prevalent genotype. 1,44,56
There is evidence of immunologic cross-protection among some strains of BVDV, including different genotypes. 30,45,77,78 However, another study has shown incomplete immunologic cross-protection between isolates and genotypes of BVDV. 53 Type 2 genotypes are generally considered more virulent, causing severe disease, including thrombocytopenia in some cases. 16,109 There is more likely a spectrum of virulence within each Pestivirus species, and those spectra overlap. For example, an outbreak in Spain was presumed to have been the result of BVDV type 2 infection by veterinarians directly involved in the outbreak, 148 but later testing showed the cause to be a type 1b isolate, indicating that virulence of isolates can cross the lines of what are perceived as low- and high-virulence BVDV genotypes. One study demonstrated no difference in the frequency of respiratory or enteric disease (or both) between genotypes 1 and 2. 56 Other reports describe high- and low-virulence isolates of type 2 BVDV. 29,90 A type 1b isolate, New York 1, has been shown to be virulent and can cause leukopenia, severe lymphoid depletion of thymus, mesenteric lymph nodes, and Peyer patches as well erosions and ulcers in the gastrointestinal tract. 19 Thus, although type 2 BVDV is generally thought to be more virulent, type 1 BVDV can cause the same manifestations of disease.
Cattle are the natural host of the type species of pestiviruses, 1 but infection by BVDV has been demonstrated in numerous other species. 5,43,58,63,76,145 Persistent infection with BVDV is documented in alpacas, mouse deer, mountain goats, white-tailed deer (WTD), sheep, and goats. 5,8,43,106,131,145 Virus distribution among tissues in persistently infected sheep, WTD, and mountain goats is similar to that seen in cattle. 72,106,115,127,131 The fact BVDV can infect a variety of other species that are in natural contact with cattle poses a risk for reinfection of pestivirus-susceptible cattle populations. 83
Fetal infection may result in immunotolerance and lifelong persistent infection. 36,95,98,99 PI cattle with BVDV were considered immunotolerant based on the fact they were seronegative for BVDV. 98 It has been shown that persistent infection does not result from cp BVDV infection; rather, infection with ncp BVDV in early gestation will cause persistent infection. 22,99 Persistently infected animals can develop severe disease with widespread lesions of the alimentary mucosal surfaces and lymphoid tissues, referred to as mucosal disease (MD). Mucosal disease is the result of persistent infection with a ncp strain of BVDV followed by subsequent postnatal infection with a cp strain of a closely related or homologous virus. 92,97,99 Homologous cp BVDV in an animal persistently infected with ncp BVDV is thought to be the result of mutation of that ncp BVDV. Induction of MD in cattle PI with BVDV was not induced by administration of adrenocoticotropic hormone, supporting the idea that MD was not a consequence of glucocorticoid responses. 85
Clinical signs of acute transient postnatal infection of animals during the era when BVDV infection was emerging were reported to be temperatures as high as 42°C, diarrhea, ulceration of the muzzle and oral cavity, and leucopenia. 112 Few or no clinical signs were detected in other infected animals. 112 This very closely resembles clinical signs and lesions of animals with MD that were seronegative for BVDV. 111 Acute transient postnatal infection remains one of the most important manifestations of BVDV infection. 29,52,89,94 Highly virulent strains of BVDV can produce lesions similar to those seen in cases of MD, such as severe and widespread ulceration of the oropharynx, larynx, and esophagus and hemorrhagic enteritis. 52,73,94,141 Observed clinical signs consisted of inappetence, lethargy, and reduced milk yield, with a spectrum of severity.
Abortion had been noted in several herds as BVDV was first described and continues to be an economically important aspect of BVDV infections in the present day. 13,44,112 Fetal loss due to in utero BVDV infection has been well recognized and is often determined by the gestational age at the time of infection. 28,100 Hormonal imbalances play a role in fetal loss, as there are elevated prostaglandin levels in the dam during BVDV infection that result in lysis of the corpus luteum (the main source of progesterone in the cow). 28
The lesions associated with acute BVDV infection were described in the first reports. 112,144 Those lesions consisted of erosions or ulcers in several tissues along the gastrointestinal tract, ranging from oronasal (Fig. 1) erosions and ulcers, through the pharynx, larynx (Fig. 2), esophagus (Fig. 3), forestomachs, abomasum, and small and large intestines. Catarrhal enteritis (Fig. 4) was described. 121 Marked multifocal lymphoid depletion, leaving karyorrhectic debris in germinal centers, can be seen. 144 Few lymphocytes are infected in Peyer patches compared with the extent of lymphocyte destruction, suggesting an indirect process is involved in depletion of lymphocytes. 19,117 In acute infections, there can be lymphatic nodules in Peyer patches that remain unaffected. 80 Thymus is markedly depleted of lymphocytes as the result of BVDV infection 110 and can be so severe as to leave just a collapsed stroma with few hemorrhages and scattered lymphocytes. 19 Some lymphoid lesions were mild despite the presence of large amounts of viral antigen in calves infected with low-virulence virus. 91 Viral antigen is widely distributed throughout lymphoid tissues such as the palatine tonsil, thymus, and Peyer patches. 19,80,88,111 BVDV antigen is demonstrated in interdigitating cells in lymphoid tissues during both acute and persistent infection. 12,19
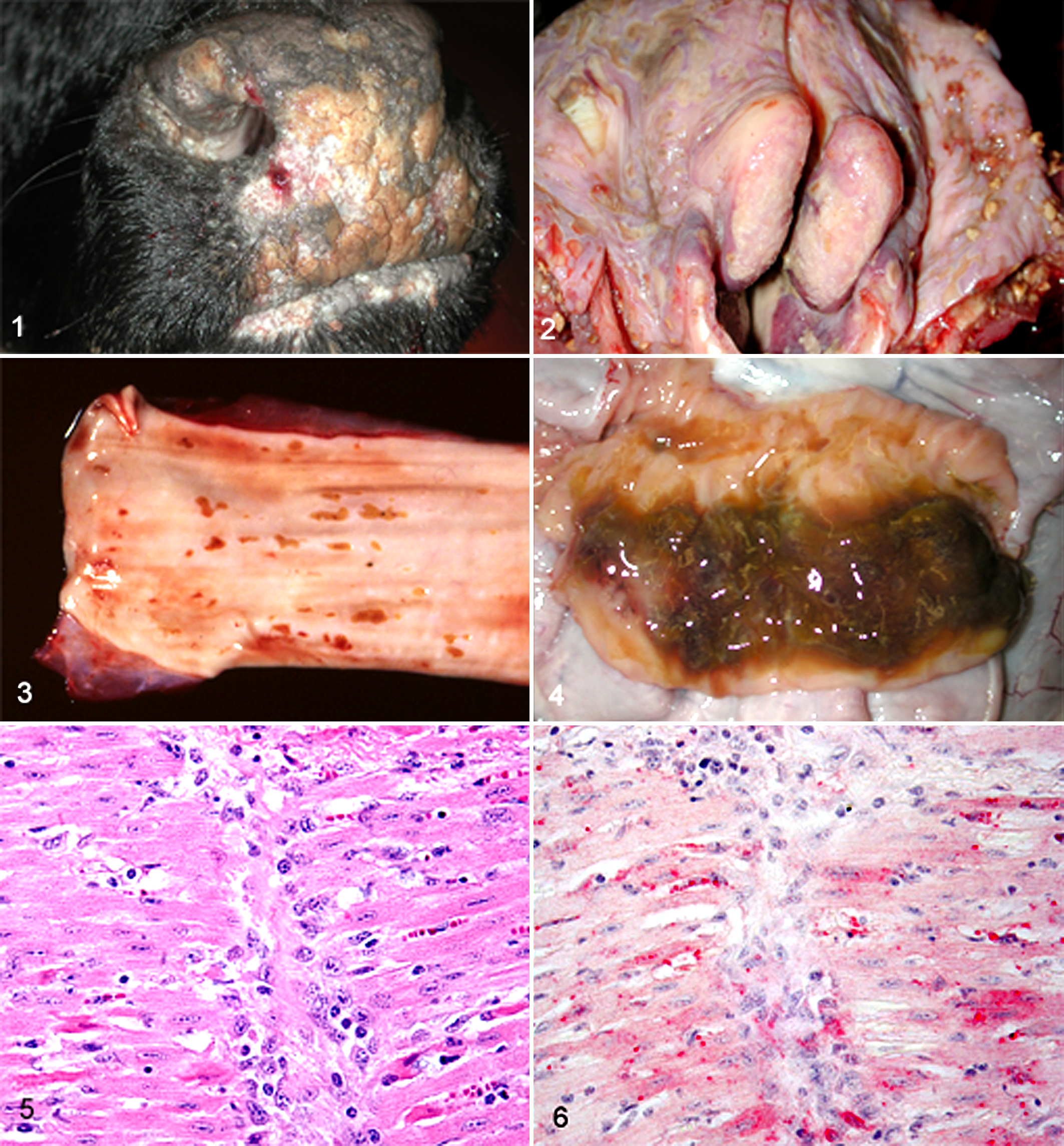
Comparison of clinically normal PI cattle with calves acutely (transiently) infected by the same virus was reported. 89 Viral antigen was reported in all tissues of the PI calves and was not associated with lesions. In the acutely infected animals, viral antigen was found mainly between days 5 and 9 postinoculation. Viral antigen was noted mainly in lymphoid tissues but not bone marrow. Other tissues were infected in only a few of the animals on various days during the 5- to 9-day period. Clinical signs were mild in these calves, and there was no thrombocytopenia. Lymphoid depletion was mild at day 3 postinfection, with increasing severity until day 9, and by day 13 there was evidence of repopulation of lymphoid tissues. This was consistent with what others have reported. 19,77,80 Severe fibrinoid necrosis of blood vessel walls was seen in lymph nodes and heart of 1 calf on day 13 postinoculation. 89 Myocardial necrosis has been seen in 8-month-old calves with BVDV antigen associated with the lesion (personal observation) (Figs. 5, 6), although it was not known if these were PI or acutely infected animals.
Thus, the patterns of distribution of viral antigen differ in acute transient postnatal infection, persistent infection, and MD. 88 In acute transient postnatal infection, there is highly variable antigen distribution with marked depletion of lymphoid follicles in several tissues. 80,88 In PI, there is widespread distribution of viral antigen, including skin and salivary glands, but only mild or locally restricted depletion of lymphoid tissues. 111 In MD, widespread distribution of viral antigen including the salivary glands, severe depletion of all lymphoid tissues, and severe lesions in the gastrointestinal tract are observed. 88
Special Pathology
In addition to the gastrointestinal tract and lymphoid tissues, BVDV antigens have been demonstrated in a variety of tissues, including the cerebellum of mid-gestation fetuses, blood vessel walls, integument, and placenta from aborted fetuses. 48,110,111,120,123 Hypoplastic lesions of the cerebellum, affecting all folia, are described in a calf whose dam was infected as early as 79 days of gestation, and lesions of cavitation, folial degeneration, and granule cell depletion were present in calves from dams infected later in gestation up to 150 days. 21 Neurotropic strains of ncp BVDV, which caused multifocal meningoencephalitis with neuronal degeneration and necrosis, have been reported. 14,47,76,104 In experimental infection of lambs and calves, low- and high-virulence strains differ in the resulting clinical and pathological findings, as well as in the amount of viral antigen present. 76,80,91 Following experimental infection of pregnant cows at 75 days of gestation, viral antigen was widespread in the brain of the fetuses, particularly in nuclei in the thalamus. These findings were similar to natural infection. 103,104 Lesions of leukomalacia and infiltration of the meninges and neuropil by macrophages and neutrophils were seen. 11,104 The fetal brain is infected via infiltrating microglial precursor cells (ameboid glial cells), and vasculopathy occurs around the time of viral invasion. This is followed by microvascular loss (presumably by necrosis), which either leads to or is concurrent with microcavity formation or cysts that persist and may enlarge over subsequent weeks. 10 It appears neuronal infection is abortive, as viral antigen appears restricted to the Golgi region of most infected neurons. 10
Ocular lesions in fetuses 17 to 22 days after experimental mid-gestational infection of cows consisted of mild to moderate retinitis, mononuclear cuffing of inner retinal vessels, proliferation of pigment epithelium, and choroiditis. 20 Retinal atrophy was observed in newborn calves that were infected in mid-gestation. 20 Congenital cataracts have been reported in calves with BVDV infection, but it is unclear as to the circumstances of the infections relative to early or late gestation or characteristics of the infecting BVDV. 151
Bovine viral diarrhea virus has been associated with growth retardation lattices of long bones, described as sclerotic lines of metaphyseal bone parallel to the physis. 142 Experimental infection of pregnant cows at 75 days of gestation resulted in altered metaphyseal bone density, first detected at 192 days of gestation. 149 It was hypothesized that in the primary spongiosa, a lesser amount of residual intercolumnar cartilage was resorbed before osteoid formation commenced. 149 However, there was no evidence of reduced numbers of osteoclasts compared with postnatally infected calves, in which osteoclasts were not seen in primary or secondary spongiosa. 135
Chronic postnatal infections exist in animals in “immunoprivileged” sites such as the testicle and ovary. Aside from the impact of BVDV on fetuses in terms of congenital defects and abortions, acute BVDV infection can have a direct impact on reproductive performance. 59 As a result of acute infection, there can be prolonged testicular infection and shedding of virus in semen for as long as 5 months after infection. 59 BVDV has been demonstrated in cells lining seminiferous tubules. 59 In the ovaries of PI heifers, BVDV has been demonstrated in cumulus cells, oocytes in all stages of maturation, and ovarian stroma. 51 Oophoritis, characterized by the presence of virus-infected macrophages, was seen following acute infection of heifers. 66 Follicular growth in ovaries from acutely infected heifers was impaired during the 2 subsequent estrous cycles after acute infection. 65
Persistent Infection
Persistent infection is induced by infection of the fetus with ncp BVDV early in gestation (40–125 days) before development of the humoral immune system. 36,98,99 To persist, both the innate and adaptive immune responses are averted by the virus.
Type I interferons (IFNs) are important mediators of the innate immune responses to viruses, and BVDV interference with type I IFN signaling has been implicated as a contributing factor in the establishment of PI. 32,133,137 Macrophages infected with ncp BVDV (of relevance to persistent infection) fail to induce type I IFN, whereas macrophages infected with cp BVDV (of relevance to mucosal disease) do release type I IFN. 2,3,31,32,118,133 Similarly, infection of 60-day bovine fetuses with ncp BVDV did not result in detectable IFN-α/β in amniotic fluid, whereas IFN-α/β was detected in amniotic fluid following infection with cp BVDV. 17,32
Viral double-stranded and single-stranded RNA fragments are able to stimulate type I IFN production. 96 Double-stranded RNA is able to stimulate production of type I IFN in a RIG-I–independent manner. 96,146 RIG-I detects double-stranded RNA during replication of positive sense single-stranded RNA, which leads to activation of interferon regulatory factor 3 (IRF-3). 150 In addition, Toll-like receptor (TLR) 3 binds to double-stranded RNA and activates IRF-3. 150 Viral RNA is recognized by pattern recognition receptors such as TLRs. 87 Cells infected with cp BVDV contain more viral double-stranded RNA (a replication intermediate of many RNA viruses) than cells infected with ncp BVDV. 96 The greater amount of dsRNA produced by cp BVDV has been postulated to contribute to stimulation of IFN production via RIG-I– and TLR-3–mediated activation of IRF-3. 96 Conversely, the BVDV glycoprotein Erns is able to inhibit IFN expression, and the RNAse activity of Erns is required for this inhibition. 96 Erns lacks a typical transmembrane anchor and is not tightly bound to cellular membranes, 49 so this RNAse activity of Erns is able to degrade extracellular single- and double-stranded RNA. Since these are triggers of type I IFN production, this is one way that Erns prevents type I IFN induction even in uninfected cells. 96
The BVDV glycoprotein Npro has been shown to be essential in blocking induction of type I IFN. Involved in the IFN-regulatory cascade are IRF-3 and IRF-7. IRF-3 is depleted by the viral protein Npro through proteasomal degradation. 6 The combined effect of Npro and Erns is necessary to adequately block IFN production and establish PI. Infection of fetuses with Npro- or Erns-deleted mutants of ncp BVDV alone was not sufficient to block establishment of fetal infection. Mutants of ncp BVDV containing both deletions blocked fetal infection in vivo. 101
Additional work has conversely demonstrated there is activation of the innate response in dams of calves in the first trimester as a result of ncp BVDV infection, 139 but this is only partially effective in curtailing viral replication, and an adaptive response is required to eliminate infection. Upregulation of interferon-stimulated gene 15kD (ISG15) has been observed in the blood of pregnant cattle during the early stage of ncp BVDV infection both in early and middle gestation. 137,138 By day 45 postinoculation, ISG15 had returned to baseline in both the PI and transiently infected (TI) cows in PI mothers, using quantitative reverse transcription polymerase chain reaction (qRT-PCR). Further work has shown that ncp BVDV has effects on several families of chemokines. 27 There is upregulation of type I IFN in maternal blood of calves infected with ncp BVDV at day 175 of gestation. 138 Leukocytes from these dams with a PI fetus showed downregulation of T-cell receptor signaling and of CXCR4 (the receptor for the lymphocyte chemoattractant, stromal cell–derived factor 1). 15,123,138 It is these features of the BVDV proteins Erns and Npro that are thought to enable the virus to avoid the innate immune system in the early gestation fetus.
Acute Infection
Little has been published regarding innate immunity to BVDV in the postnatal animal. Generally, the innate response to BVDV in the postnatal animal is not considered unique among viral infections. However, apparent differences exist between high-virulence (HV) and low-virulence (LV) isolates as well as between cp BVDV and ncp BVDV. Infection of 7-month-old calves with a high-virulence ncp BVDV isolate has been shown to upregulate interferon α and β messenger RNA (mRNA) levels in tracheobronchial lymph nodes in the HV group but not in the LV group. 114 Type I IFN genes in spleens of both HV and LV compared with control calves were upregulated. 114
Important differences occur between ncp BVDV and cp BVDV in the induction of apoptosis. Cytopathic BVDV, but not noncytopathic (ncp) BVDV, has been shown to induce apoptosis in vitro in embryonic bovine turbinate cells. 68,134,154 Infection of several types of cultured cells with the cp, but not with the ncp, biotype of BVDV triggers the induction of apoptosis. 2 Expression of the NS3 protease of cp BVDV is unable to inhibit TLR-3– and RIG-I–dependent activation of IFN-β, thus allowing apoptosis to occur in cp BVDV infections. 57
Whereas the viral protease NS3 is continually expressed by cp BVDV-infected cells and is thought to be important in inducing apoptosis, this protein is expressed only early in cellular infection or not at all in ncp BVDV infections. This difference may partially explain the pathologic findings of cell death observed in mucosal disease resulting from cp BVDV infection, whereas such lesions are not observed in calves persistently infected with ncp BVDV.
There are conflicting reports regarding apoptosis due to ncp BVDV infection. The intrinsic pathway is induced, at least in vitro, by ncp BVDV by overexpression of apoptotic protease-activating factor 1 and increasing caspase 9 activity. 67 Further evidence supporting the intrinsic pathway as a mechanism for apoptosis during acute BVDV infection is a reduction in the number of tumor necrosis factor–α (TNF-α)–expressing cells in the thymus of calves experimentally inoculated with a type 1 ncp BVDV. 122 Noncytopathic BVDV can prime bone marrow–derived macrophages for production of reactive nitrogen in response to stimulation, 3 and reactive nitrogen (nitric oxide) can induce the extrinsic pathway of apoptosis. 24
BVDV can induce apoptosis of lymphocytes through the RNase activity of Erns. 25,70,132 In cells infected with ncp BVDV, the NS2/3 protein is cleaved in the early stages of cellular infection, to release the proapoptotic viral protein NS3. That cleavage is then reduced, resulting in the presence of predominantly uncleaved NS2/3 in later stages of infection. 84 The viral protein NS3 induces apoptosis via activation of caspase 8 and the cytochrome c release-dependent caspase 9, thus inducing both extrinsic and intrinsic pathways. 140 Induction of moderate amounts of caspase 8 is associated with apoptosis of T-lymphocyte–rich interfollicular areas and marked apoptosis of B lymphocytes in lymphoid follicles during infection of ileal Peyer patches. 116 This finding suggests a role of the extrinsic pathway of apoptosis in gut-associated lymphoid tissue (GALT).
Interestingly, it was demonstrated there is inactivation of caspase 9 during acute infection with ncp BVDV in GALT, suggesting the intrinsic pathway is not involved in apoptosis during ncp BVDV infection. 116 Further evidence that the intrinsic pathway is regulated during ncp BVDV infection is that reports of the apoptotic pathway regulator, Bcl-2, blocks the actions of the Bax gene in a balance between Bcl-2 and Bax. 23,38,102 The antiapoptotic Bcl-2 protein is upregulated by ncp BVDV, suppressing initiation of intrinsic apoptosis and cell death. 9 Overexpression of Bcl-2 in T-cell interfollicular areas may have an inhibitory effect on apoptosis, protecting T lymphocytes in those interfollicular areas. 116 Reduced levels of Bcl-2 in follicular areas during acute infection could be associated with apoptosis of B lymphocytes. 116 Oxidative stress occurs early in the interaction between cp BVDV and its host cell and may be a crucial event in the sequence leading to apoptotic cell death. 134 Hence, apoptosis is not required for the multiplication of the cp biotype of BVDV. 134
Adaptive Immunity, Vaccination, and Immunosuppression
Modified live and killed vaccines have been available for more than 50 years, yet the incidence of BVDV-induced disease remains significant, suggesting a need for improved vaccines. This may be due, in part, to the lack of proofreading function during replication of the single-stranded genome of BVDV and the resultant antigenic variability. 124 Neutralizing antibodies prevent disease following challenge with homologous virus, as well as prevent or ameliorate disease with challenge with heterologous virus. 23,30,53,54 There are 4 structural proteins of BVDV, but the E2 glycoprotein is the major target of neutralizing antibodies. 40,41 Newly designed vaccines have addressed the issues related to virulence of E2-expressing vaccine viruses by incorporating the E2 gene into nonvirulent vectors. 4,93 The E2 region of the genome has been used as one of the regions for classification of BVDV isolates due to its variability in the genome sequence. 7,125,148 Due to the antigenic variability, there is potential for a lack of cross-protection against wild-type BVDV viruses, even though cross-neutralization studies have shown reactivity to differing genotypes. 93 Since disease due to BVDV remains widespread, additional measures beyond vaccination are needed.
Resolution of postnatal infection is reliant on function of CD4+ T cells, 75 and one of the mechanisms for persistence is tolerance of these cells due to in utero infection. 34 It has been shown that as little as 1 amino acid difference in quasispecies is sufficient to stimulate an antibody response to homologous virus, demonstrating the specificity of the tolerance of the adaptive immune response. 34
Vaccines are not without their adverse effects on the host. In the event that there is PI in animals with BVDV in a group that is administered a closely related cp BVDV in the modified live vaccine virus, there is potential for the unfortunate occurrence of MD as a result of these combined infections. 125 Modified live vaccines for BVDV can retain some virulence and can result in lymphoid depletion in Peyer patches as well as alteration of neutrophil and lymphocyte functions after vaccination. 78,129 Vaccines can also be contaminated by virulent BVDV, resulting in PI in calves with BVDV after vaccination of pregnant animals. 113 Vaccine virus can also be shed after extra-label administration to pregnant animals and, in the proper circumstances, can result in PI calves. 113
Immunosuppression is an important feature of BVDV infection. Both monocytes and DCs are susceptible to infection by cp and ncp BVDV. 61 Monocytes but not DCs were killed by cp BVDV, while ncp BVDV infection reduced responses of CD4+ T cells to antigen. 61 Macrophages infected with ncp BVDV have downregulation of CC- and CXC-chemokine expression. 27 During acute infections, antigen-presenting cells are compromised, contributing to immune suppression. Marked depletion of CD4+ and CD8+ cells has been reported in the thymus. 18 In vivo infection with ncp BVDV reduced the number of major histocompatability complex (MHC) class II cells in Peyer patches and lymph nodes. 18 Infection of peripheral blood mononuclear cells with cp BVDV results in altered expression of proteins involved in adhesion, apoptosis, antigen uptake, processing, and presentation, thus compromising the response of antigen-presenting cells. 86 However, monocytes from calves with PI are nonetheless able to generate MHC class I–restricted and MHC class II–restricted responses to BVDV. 62
Contemporary Diagnostic Methods
Several techniques for the diagnosis of BVDV infections have been developed recently that have allowed affordable widespread testing of animals for BVDV infection. Those techniques have included both immunoenzymatic assays as well as PCR assays. Early immunohistochemical tests were based on cryosections of fresh tissues. 64,143,152 However, later techniques were developed, using formalin-fixed, paraffin-embedded (FFPE) tissues. 69 This was based on the monoclonal antibody directed against the Erns glycoprotein of BVDV, 41 which was shown to recognize all isolates of BVDV tested. 35 This same MAb was used in the development of an antigen capture enzyme-linked immunosorbent assay (ELISA). 130
The PI animal serves as a reservoir for BVDV and is a source of infection for herd mates. Control strategies have focused on identification and removal of PI animals from herds. Previously, PI animals were identified by repeated demonstration of virus in buffy coat cells or nasal or lacrimal exudate from the same animal. 98 These animals also remain seronegative for extended periods. Contrasted to this, acutely infected animals remain viremic for a relatively short period (generally less than 3 weeks) and seroconvert, producing neutralizing antibodies as early as 2 to 3 weeks after infection. 77,98,117 In acutely infected animals, virus is cleared from most tissues by day 9 postinfection, and these animals generally do not serve as a source of infection to herd mates. 89 Strategies to control BVDV infection in herds include vaccination and removal of PI animals. The prevalence of PI animals is reported to be between 1% and 2%, 74,153 so large numbers of animals need to be tested to identify those few that have PI with BVDV. As a result, it is important to be able to affordably identify the PI animal. Virus isolation was too slow and expensive for large-scale testing. The immunoperoxidase microplate virus isolation test was developed for serum, which greatly enhanced screening for PI animals. 39
In early work, skin was identified as a location of viral antigen, and subsequently immunohistochemistry on cryosections of skin biopsies was shown to reliably identify PI animals with BVDV. 111,143 Use of skin along with development of immunohistochemistry (IHC) on FFPE tissues enabled high-throughput testing for identification of PI animals. That is when the idea of using FFPE ear notch samples was developed (Brodersen, Proceedings 31st AABP Annual Meeting, 1998. Spokane, WA). Additional testing methodologies have used ear notches as the substrate—namely, antigen capture ELISA and PCR. 37,82,105,107 In large-scale outbreaks of BVDV with many PI animals, BVDV antigen is expressed in the skin of transiently infected animals for as long as 120 days. 37 In those situations, it is advisable to use different methodologies over an extended period to accurately identify which animals are PI and which are transiently infected. It is particularly important to perform additional testing on animals suspected of being PI when blood samples and nasal swabs are used during initial testing. 54 The current testing methodologies allow for implementation of BVDV control strategies. 42
Summary
BVDV is a pestivirus that can cause severe clinical disease. Major economic losses continue to result from reproductive losses and exacerbation of concurrent bacterial or viral infections. Of less clinical importance is severe clinical disease due to BVDV alone, either as a result of acute infection by highly virulent isolates or by development of mucosal disease in persistently infected calves. Disease due to BVDV infection can arise at any stage of life and is often dependent on the virulence of the infecting isolate.
Survival of BVDV in the population depends on the characteristic of persistently infecting its host. Persistent infection arises by the unique ability of the BVDV to survive by inducing immune tolerance in the bovine fetus through evasion of both innate and acquired immunity in utero. Adaptive immunity is avoided by immune tolerance through infection of the fetus prior to development and maturation of the adaptive immune system. The PI calf with BVDV then serves as a source of infection of herd mates and thereby leads to continuation of the viral species. Because of this unique and complex pathogen-host interaction, a voluminous body of research has been produced and summarized here as well as in other sources. 33,55,60,81 Control of BVDV infections in herds is dependent on identification of PI calves with BVDV and removal of those PI animals.
Footnotes
Acknowledgement
I wish to thank Dr. Clayton L. Kelling for his advise in composition of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
