Abstract
Ependymal tumors are reported rarely in domestic animals. The aims of this study were to examine the clinical and pathologic features of ventricular and extraventricular ependymomas and subependymomas in 18 domestic cats examined between 1978 and 2011. Parameters examined included age, sex, breed, clinical signs, and macroscopic and histopathologic features. The mean age of affected cats was 9 years, 4 months; median age, 8.5 years. There were 8 female and 4 male cats, and 6 cats for which sex was not recorded. Breeds included 10 domestic shorthaired, 2 domestic longhaired, 1 Persian, and 1 Siamese. Clinical signs included altered mentation or behavior, seizures, circling, propulsive gait, generalized discomfort, and loss of condition. The tumors often formed intraventricular masses and usually arose from the lining of the lateral or third ventricles, followed by the fourth ventricle, mesencephalic aqueduct, and spinal cord central canal. Three tumors were extraventricular, forming masses within the cerebrum and adjacent subarachnoid space. Histologically, 15 tumors were classified as variants of ependymomas (classic, papillary, tanycytic, or clear cell) and 3 as subependymomas. Tumors were generally well demarcated; however, 6 ependymomas focally or extensively infiltrated the adjacent neural parenchyma. Characteristic perivascular pseudorosettes were observed in all ependymomas; true rosettes were less common. Some tumors had areas of necrosis, mineralization, cholesterol clefts, and/or hemorrhage. This cohort study of feline ependymal tumors includes subependymoma and primary extraventricular ependymoma, variants not previously described in the veterinary literature but well recognized in humans.
Keywords
The most recent World Health Organization (WHO) classification system for tumors of the nervous system in domestic animals recognizes 2 neoplasms arising from the ependyma: ependymoma (including classic and papillary subtypes) and anaplastic ependymoma. 20 Ependymomas are uncommon glial tumors arising from the lining of the ventricles and the central canal of the spinal cord. Typically, these neoplasms form a solid well-defined mass that bulges into the ventricle or the central canal, although some forms, especially high-grade ependymomas, infiltrate the adjacent neuroparenchyma. Key histological features include the formation by the neoplastic cells of true rosettes, ependymal canals, and pseudorosettes. Ultrastructurally, neoplastic cells typically retain ependymal features such as cilia, blepharoplasts, and microvilli at the apical surface, junctional complexes at the lateral surface, and absence of basal lamina.13,23 Recognized human ependymal tumors include ependymoma and its variants: classic/conventional, papillary, tanycytic, cellular and clear cell subtypes (WHO Grade II), myxopapillary ependymoma (WHO grade II), anaplastic ependymoma (WHO Grade III), and subependymoma (WHO Grade I). 23 WHO Grade I tumors are those with low proliferative potential and possibility of cure following surgical resection alone; WHO Grade II tumors are generally infiltrative, with higher likelihood of recurrence, despite low proliferative activity; WHO Grade III is assigned to tumors with histological evidence of malignancy; and WHO Grade IV is reserved for cytologically malignant, mitotically active, necrosis-prone neoplasms that are often associated with rapid pre- and postoperative progression and a fatal outcome. 24 Subependymoma is not presently recognized in the WHO classification system for domestic species; 20 in humans, the histogenesis of this neoplasm is not definitively known; however, the neoplasm is thought to arise from subependymal glial precursors. 23
Few reports, mostly of single cases, describe ependymal neoplasms in the domestic cat,1,9,16,37–39 including one tanycytic subtype. 25 Ependymomas are reported occasionally in the other nonhuman species, including primates, 31 dogs,3,26,33,40 rats, 18 cattle,7,32 horses,4,12,15 sheep, 6 deer,22,30 and rabbits. 19 In this study, the clinical, macroscopic, histopathological, and immunohistochemical features of tumors of the ventricular lining or the immediate subependymal zone and 3 extraventricular ependymomas were reviewed in 18 cats.
Materials and Methods
Ventricular and extraventricular tumors with ependymal or subependymal differentiation from 18 domestic cats examined between 1978 and 2011 were collated. Cases comprised biopsy samples or postmortem specimens received by the Diagnostic Laboratory, College of Veterinary Medicine at Cornell University or the Cornell University neuropathology group (usually brain only). Parameters recorded included age, sex, breed, clinical signs, and macroscopic and histopathological features of the tumors.
Tissues from biopsy and postmortem specimens fixed in 10% neutral buffered formalin were processed for light microscopy by routine methods and embedded in paraffin. Sections, cut at 5 µm thickness, were stained with hematoxylin and eosin (HE). Further formalin-fixed, paraffin-embedded sections were mounted on lysine-treated slides and immunolabeled for reactivity to glial fibrillary acidic protein (GFAP, rabbit polyclonal, Dakocytomation) and pancytokeratin AE1/AE3 (mouse monoclonal cocktail, Dakocytomation), respectively, using a peroxidase-based method for indirect immunohistochemistry. 3,3′Diaminobenzidine (DAB) chromagen was used for visualization of bound antibodies. GFAP was used at 1:3000 without pretreatment and AE1/AE3 used at 1:100 after pepsin pretreatment. Appropriate positive and negative controls were included.
Results
Signalment and Clinical Features
Eighteen domestic cats were diagnosed with ependymal tumors in this study. The cats averaged 9 years, 4 months in age (median, 8.5 years; range, 4 - 12.5 years), and comprised 8 females, 4 males, and 6 cats for which sex was not recorded. Breeds included 10 domestic shorthair (DSH), 2 domestic longhair (DLH), one Persian, and one Siamese. Reported clinical signs in cats with tumors in the lateral or third ventricles or in an extraventricular location included altered mentation (n = 7), circling (n = 6), opisthotonus (n = 4), propulsive gait and/or head pressing (n = 5), pain and/or altered sensorium (n = 4), other abnormal behavior (n = 2), seizures (n = 2), and diminished or absent menace response (n = 4) (Table 1), primarily indicating prosencephalic localization. Other nonspecific findings included anorexia and loss of body condition. Caudal cranial fossa signs were reported in the single cat with an ependymoma in the fourth ventricle. Clinical information was not available for the cats with an ependymoma in the mesencephalic aqueduct or central canal.
Signalment, Clinical Features, and Results of Imaging for Feline Ependymomas and Subependymomas (n = 18).
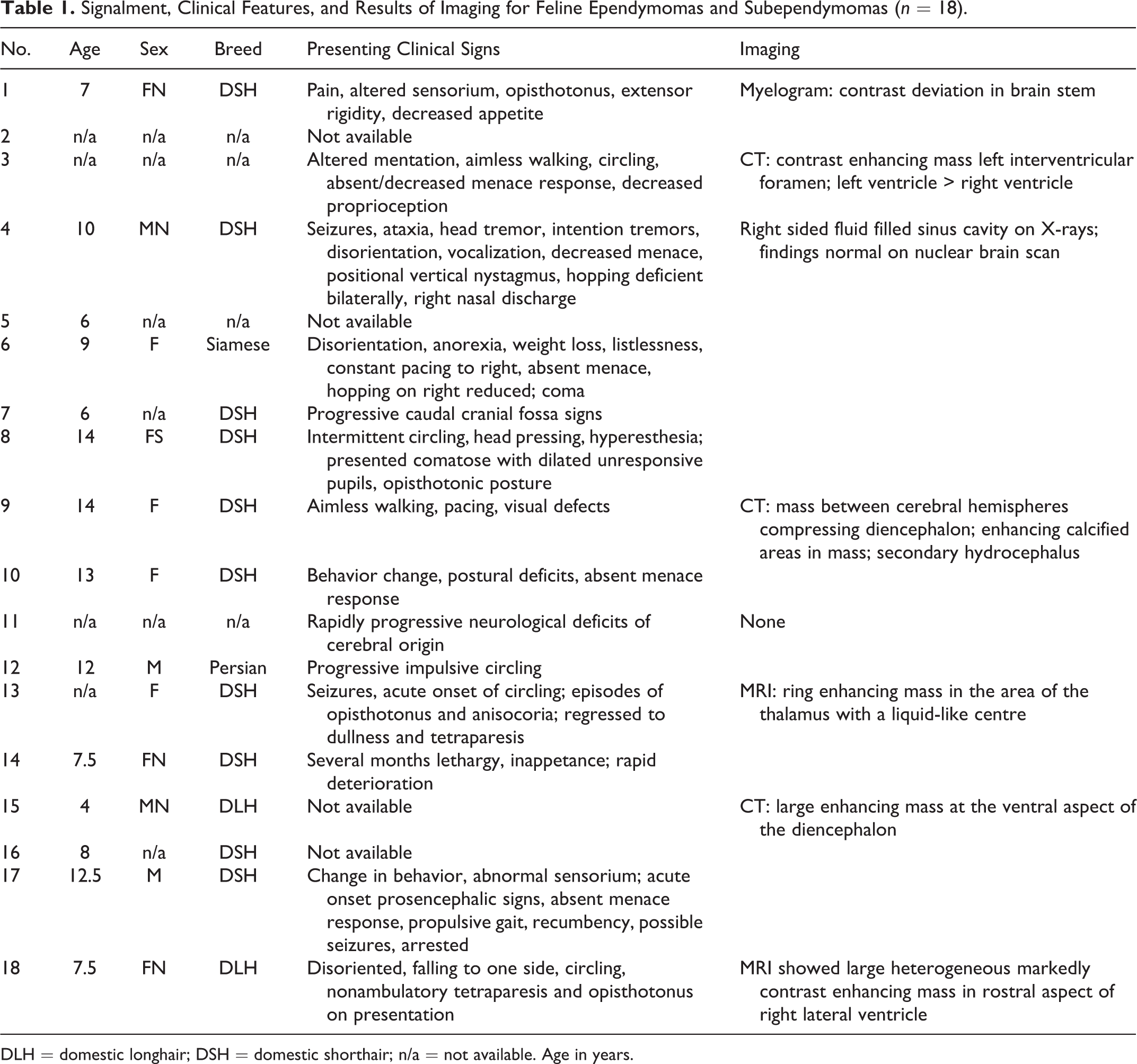
DLH = domestic longhair; DSH = domestic shorthair; n/a = not available. Age in years.
Macroscopic Appearance and Lesion Distribution
Most tumors formed an intraventricular mass and were not obvious in the intact brain (Table 2; Figs. 1–3). Tumors arose from the walls of the lateral ventricles (n = 7, Fig. 1), third ventricle (n = 5), fourth ventricle (n = 1, Fig. 2), mesencephalic aqueduct (n = 1), or central canal (n = 1). These tumors were generally well demarcated and expanded the ventricle. Three tumors were extraventricular, involving the subarachnoid space and adjacent cerebrum. On the basis of available surgical reports and neuroimaging (n = 2) or postmortem examination (n = 1, Fig. 3), all 3 extraventricular tumors lacked any obvious connection with the ventricular system.
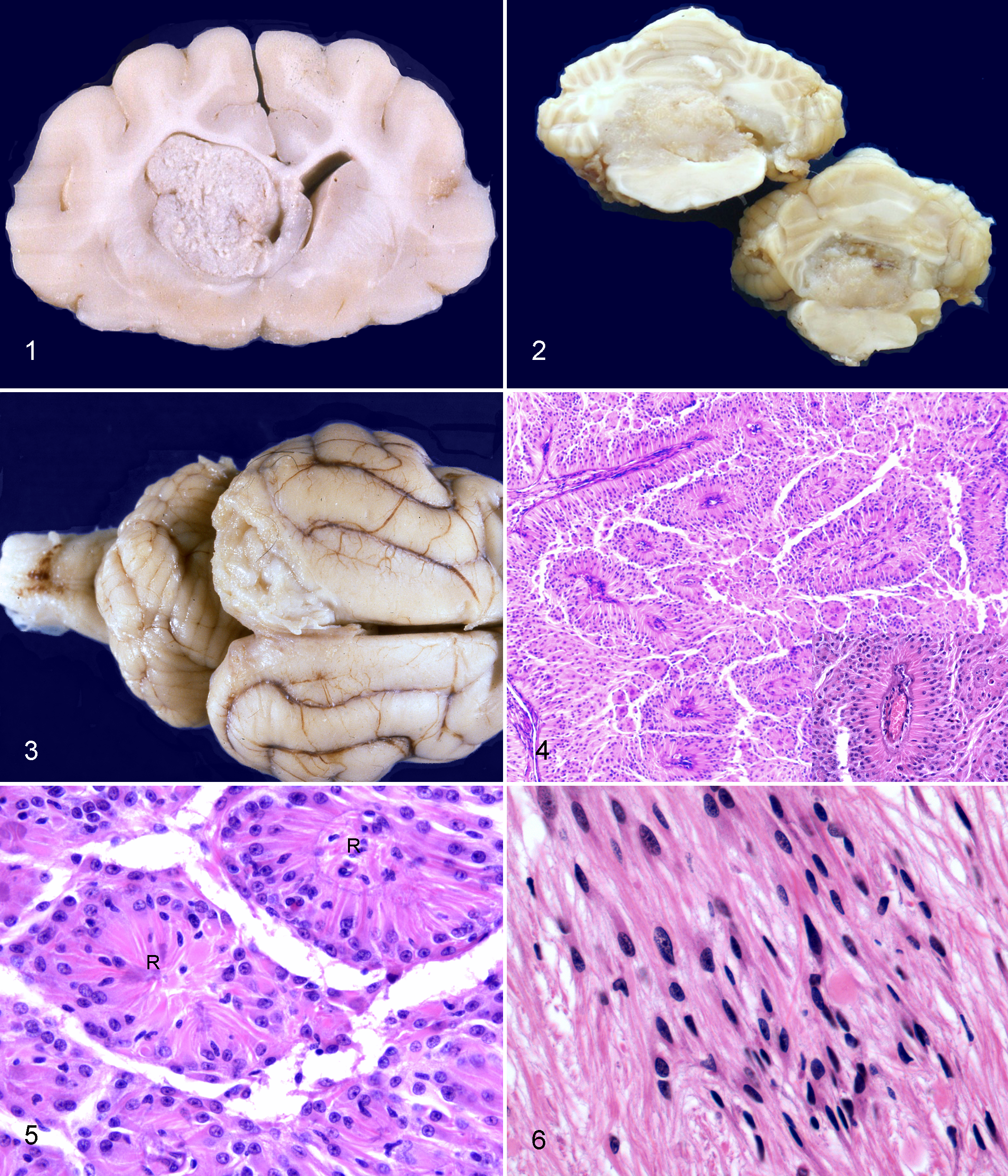
Brain; cat no. 3. A well-demarcated, pale gray to tan mass with irregular surface fills the rostral aspect of the left lateral ventricle. This ependymoma arises at the left interventricular foramen and compresses the caudate nucleus, corpus callosum, and fornix.
Brain; cat no. 15. Area of clear cell differentiation in a papillary ependymoma. Tall columnar to polygonal cells have clear cytoplasm and small round nuclei. Rosettes (R) are present. HE.
Variety, Location, and Histological and Immunohistochemical Features of Feline Ependymomas and Subependymomas (n = 18).
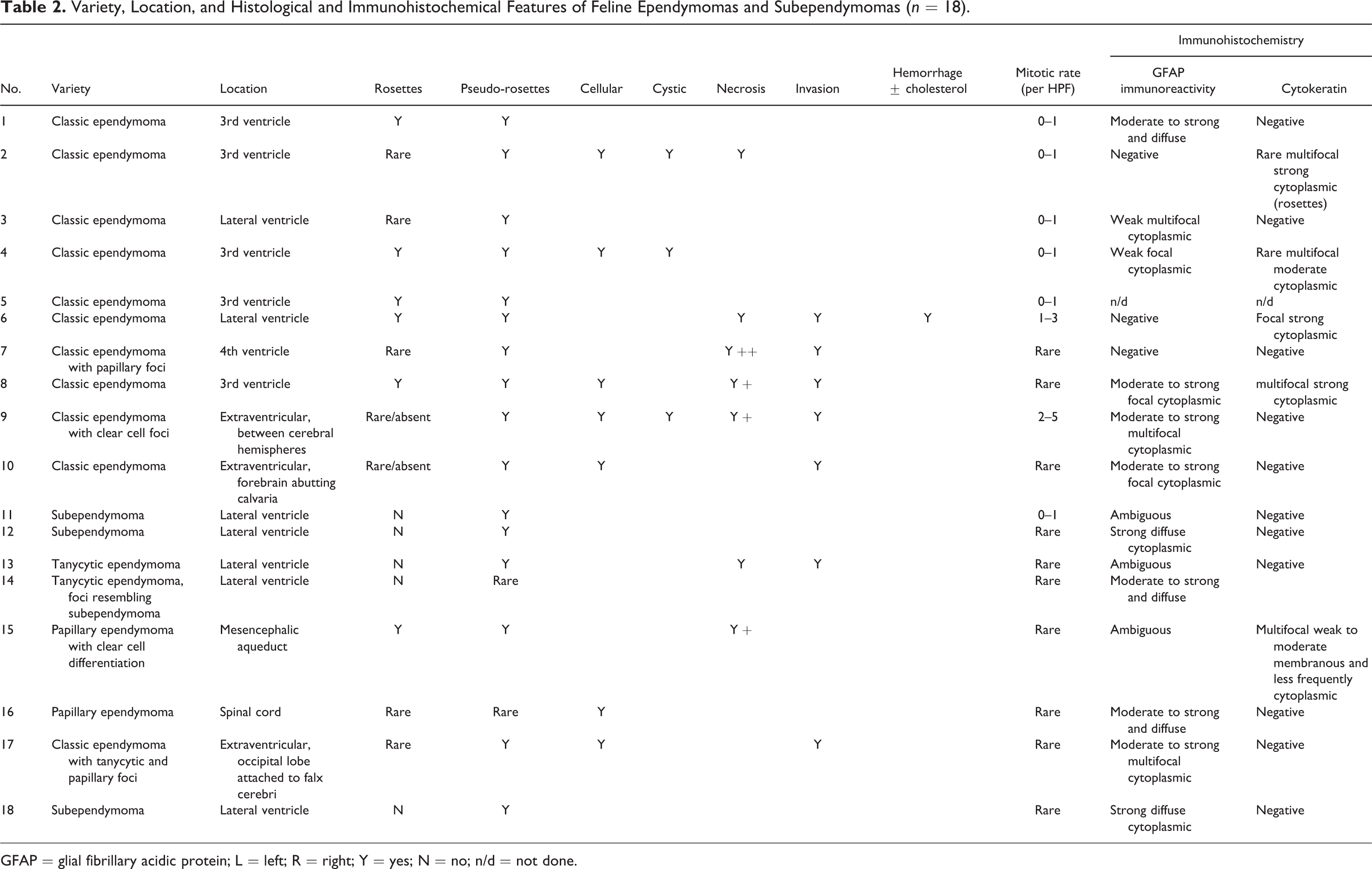
GFAP = glial fibrillary acidic protein; L = left; R = right; Y = yes; N = no; n/d = not done.
Histopathology
Histological features are detailed in Table 2. Fifteen tumors were classified as variants of ependymoma, and 3 as subependymoma. Seven tumors focally or extensively infiltrated the neural parenchyma. Tumors replaced the ventricular lining with focal retention of the junction between ependymoma and the nonneoplastic ependymal lining. All 3 subependymomas were adjacent to the right lateral ventricle (nos. 11, 12 and 18).
Eleven tumors (nos. 1–10, 17) had features of classic ependymoma (Fig. 4) with moderate to high cellularity, characteristic ependymal canal formation, pseudorosettes (Fig. 4 inset), and true rosettes (Fig. 5). Rosettes were formed by tall columnar cells with small basal nuclei or with a pseudostratified nuclear arrangement. In these cases, the neoplastic cells not forming canals or rosettes were smaller and low columnar, cuboidal or polygonal.
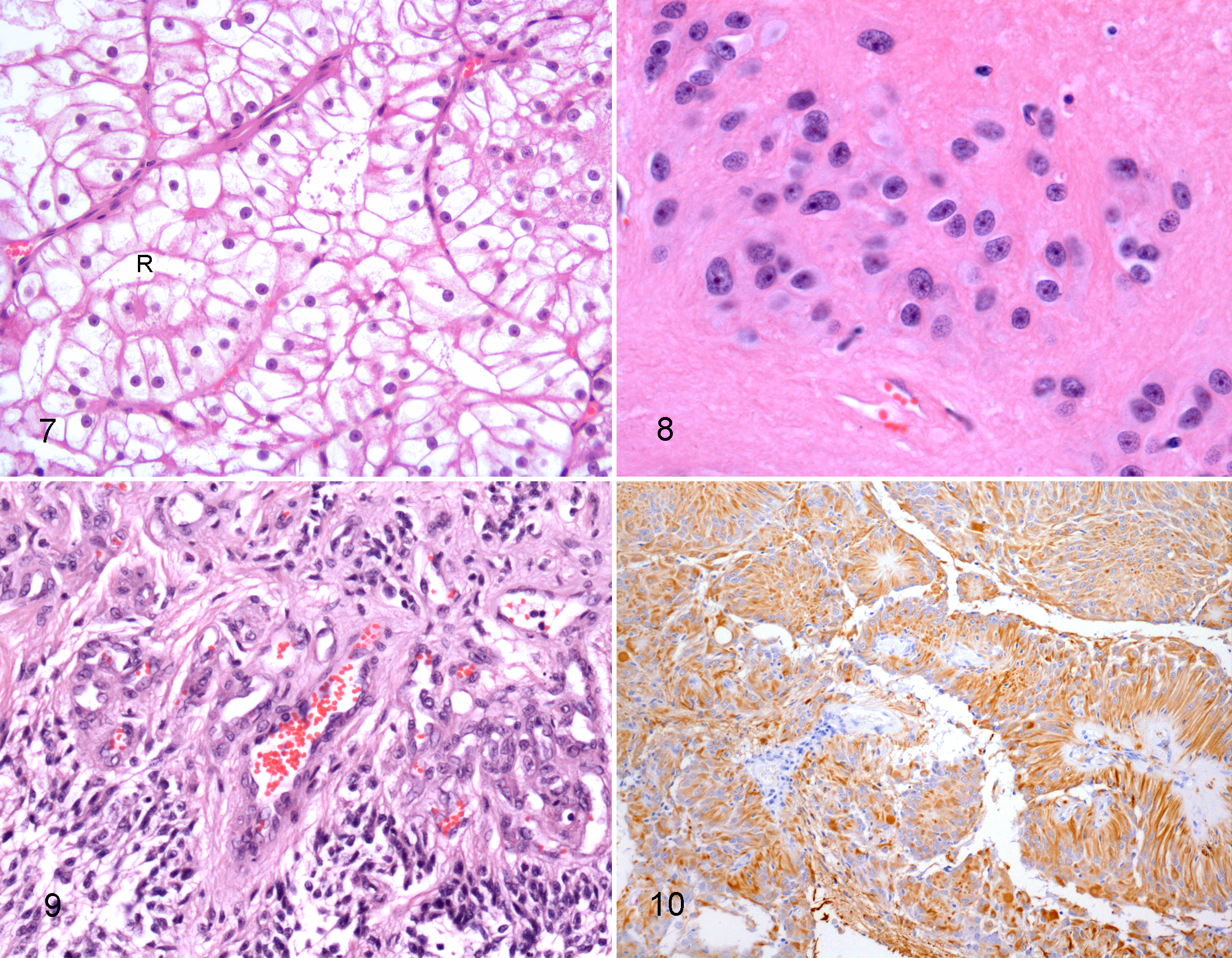
Many ependymomas also had either diffuse or focal tanycytic (n = 3), papillary (n = 4), cellular or solid (n = 6), microcystic (n = 3), and/or clear cell (n = 2) morphology. In some cases, multiple patterns were observed in the same tumor. Four ependymomas were classified as tanycytic (n = 2) or papillary (n = 2) variants because these patterns predominated (Table 2). Tanycytic ependymomas were characterized by elongated cells with delicate fibrillar processes forming bundles and fascicles of variable width and cell density (Fig. 6). Neoplastic cell nuclei were large to very large, fusiform to ovoid, and vesicular with prominent nucleoli. Two neoplasms, classified as tanycytic ependymomas, were within the lateral ventricles (Table 2). In the 2 papillary ependymomas, tall columnar cells formed papillary arrangements along delicate fibrovascular stroma; one papillary ependymoma also had clear cell differentiation (no. 15, Fig. 7). In ependymomas with clear cells, the smaller rounded cells with a clear perinuclear halo resembled oligodendroglioma; one was in the mesencephalic aqueduct.
Pseudorosettes were observed in all ependymomas; however, true rosettes were less common (n = 6), and often most evident in invasive variants. True rosettes were rare or absent in all 3 tanycytic ependymomas. Eight tumors had areas of necrosis and mineralization with cholesterol clefts and hemorrhage in one.
Tumors from nos. 11, 12 and 18 were classified as subependymomas. These tumors were quite different, formed by discrete clusters of glial cells within a dense fibrillary matrix (Fig. 8). The neoplastic cells had round to ovoid hypochromatic nuclei and central discrete nucleolus. True rosettes were not observed; however, tumor cells did form pseudorosettes.
Three ependymomas were located extraventricularly in the neocortex and subarachnoid space. Histologically, extraventricular ependymomas infiltrated the superficial to deep neocortex and the subarachnoid space extensively. All 3 were classic ependymomas; 2 also had focal clear cell, papillary or tanycytic features (Table 2). One extraventricular ependymoma (no. 17) had small proliferations of capillaries adjacent to areas of neuropil invasion that were reminiscent of the “glomeruloid” vessels of high-grade gliomas (Fig. 9).
Immunohistochemistry
Fourteen of the 15 ependymomas and all 3 subependymomas were investigated for immunoreactivity using antibodies against GFAP and pancytokeratin. Positive GFAP immunoreactivity of variable intensity and distribution was observed in the neoplastic cells of 9/14 ependymomas. Immunoreactivity was moderate to strong and diffuse in nos. 1, 14 and 16 (Fig. 10); moderate to strong and focal or multifocal in nos. 8, 9, 10 and 17; and weak focal or multifocal in nos. 3 and 4. Three classic ependymomas (nos. 2, 6 and 7) were diffusely negative for GFAP. In 2 neoplasms (nos. 13 and 15), GFAP staining was difficult to interpret, confounded by nonspecific binding of the chromagen. Occasionally, neural tissue adjacent to the neoplasm contained GFAP-immunoreactive astrocytes, interpreted as peritumoral astrocytosis.
Five of 14 ependymomas (nos. 2, 4, 6, 8, and 15) had rare patchy or multifocal cytoplasmic, or rarely membranous, positive immunoreactivity for pancytokeratin. Four of the 5 were classic ependymomas; one was a papillary variant. The remaining 9 ependymomas were diffusely negative for pancytokeratin.
Of the 3 subependymomas (nos. 11, 12 and 18), 2 had strong diffuse GFAP immunoreactivity of tumor cells. The other had focal ambiguous GFAP immunoreactivity, in which case it was not possible to determine if labeling was within reactive astrocytes or the neoplastic ependymal cells. All 3 subependymomas were negative for pancytokeratin.
Discussion
The pathological features of ependymal and subependymal tumors in 18 domestic cats presented with neurologic disease over a period of 33 years are reviewed in this study. Seven ependymomas were included in a series of feline intracranial tumors; the median age of affected cats was 8.1 years (range, 5.0–13.2 years), the sex distribution was 4 castrated males and 3 spayed females, and breeds included DSH and DLH. 39 All cats were presented with neurological signs, usually altered consciousness and seizures. Nonspecific clinical signs of lethargy and anorexia were noted in one cat. An ependymoma of the third ventricle was reported in a 10-year-old spayed female cat with behavioral changes and central blindness. 37 Ependymoma has also been reported in the fourth ventricle of a 3-year-old neutered male DSH 16 and a 4.5-year-old spayed female Siamese cat; 25 both cats had clinical signs of central vestibular disease.
Ependymomas may affect any age group in humans, but infratentorial tumors occur predominantly in children, whereas supratentorial tumors affect adults as well as pediatric patients. 23 Spinal cord ependymomas are a major manifestation of neurofibromatosis type 2. 5 No sex predilection is apparent in people.29,34 This study indicated a mild bias toward female cats, but a greater sample size is required to determine if this is significant. Too few cases of feline ependymoma have been studied to assign age predilections to particular variants, or to the location at which they arise. Most feline ependymomas in this study were intraventricular masses arising from the wall of the lateral or third ventricles, with only single cases from the fourth ventricle, mesencephalic aqueduct and central canal. Similarly, the lateral and third ventricles were the most common sites in previous studies, with only single case reports of tumors in the fourth ventricle.9,16,25,37,39 It therefore seems that the vast majority of feline ependymal tumors occur in adult cats in a supratentorial location. Notably, 3 tumors in this study had an extraventricular location within the cerebrum and adjacent subarachnoid space. Extraventricular ependymal tumors have not been described previously in nonhuman species.
Although most tumors were well demarcated masses that expanded into a ventricular space, 7 focally or extensively infiltrated the adjacent neural parenchyma. Based on their predominant cellular differentiation, 4 variants of ependymoma were identified (classic, tanycytic, clear cell and papillary). In addition, some tumors contained foci of secondary patterns. Characteristic perivascular pseudorosettes were observed in all ependymomas; however, true rosettes were less common. Ependymomas that infiltrated the adjacent parenchyma were considered to have a malignant phenotype. Interestingly, cellular pleomorphism in the infiltrative tumors was frequently low, and these tumors frequently had numerous well-differentiated true rosettes. In one particularly invasive extraventricular ependymoma, invasion of the neocortex and corona radiata was associated with proliferation of capillaries, reminiscent of “glomeruloid vessels” of high-grade gliomas. In addition, that tumor had a higher mitotic rate and regions of necrosis. Microvascular proliferation, invasiveness, high mitotic rate, and necrosis are salient features of anaplastic ependymoma in humans (WHO grade III); therefore, this feline tumor can be classified as an extraventricular anaplastic ependymoma.
Three feline tumors in this study were histologically consistent with subependymoma. In human medicine, subependymoma is a slow-growing low-grade tumor typically found attached to a ventricular wall, and is composed of neoplastic glial cell clusters in an abundant fibrillary matrix. 23 The histogenesis of this neoplasm is not known; proposed cells of origin include the subependymal glia, progenitors of astrocytes of the subependymal plate, ependymal cells, or a mixture of astrocytes and ependymal cells. 23 The tanycytic variant of ependymoma was included in the differential diagnosis for subependymomas in this study. Histologically, tanycytic ependymoma was differentiated from subependymoma by cellular characteristics of finer fibrillary and more distinct cytoplasm and elongated, bipolar slender nuclei. Subependymoma nuclei were rounded to ovoid and clustered in a dense matrix. One neoplasm, which was classified as tanycytic by the authors and by consulted colleagues in medical neuropathology, had focal areas with features that were more typical of subependymoma, highlighting the difficulty in distinguishing these 2 tumors. Subependymoma was distinguished from other glial tumors (eg, astrocytoma) by its location adjacent to the ventricular lining and bulging into the ventricular lumen, clusters of isomorphic nuclei in a dense fibrillary matrix, and by the formation of pseudorosettes. An astrocytoma is more likely to be intraparenchymal and less likely to bulge into the ventricle. Microscopically, the characteristic feature of subependymoma is the uneven cell distribution. Unlike astrocytoma, in which neoplastic cells are fairly uniformly dispersed within the mass, subependymoma cells are arranged in clusters that are separated by the process-rich fibrillary matrix.
This study also describes the clear cell variant of ependymoma in a domestic species, and provides additional evidence to substantiate the existence of the tanycytic variant in the domestic cat. 25 The clear cell variant arose from and effaced much of the mesencephalic aqueduct. It formed rosettes and pseudorosettes characteristic of ependymoma. Clear cell ependymoma may require differentiation from oligodendroglial neoplasms, central neurocytoma (reported in the spinal cord of a dog), 14 metastatic renal cell carcinoma, and clear cell variant of meningioma, 43 the latter of which has not yet been reported in domestic species. Clear cell ependymoma in humans is a rare variant typically reported in children and young adults. 8 The cat with clear cell variant was 4 years of age at the time of death, the youngest of the cohort.
The tanycytic variant, so called for the resemblance of the spindly, bipolar tumor cells to the tanycyte (specialized paraventricular cell),10,23 was observed as the predominant pattern or just focally in 6 ependymomas. This subtype has been reported in a cat. 25 All tumors with diffuse tanycytic morphology arose within the lateral ventricles. In contrast, the tanycytic variants in humans have a reported predilection for the spinal cord. The absence of true rosettes and vague delineation of pseudorosettes in feline tanycytic ependymomas in this study was consistent with findings in the human variant. 23 The differential diagnosis considered for tanycytic ependymoma included pilocytic astrocytoma and (in some cases) subependymoma. Four ependymomas had areas with papilliform features that required differentiation from choroid plexus papilloma, papillary meningioma, or metastatic papillary carcinoma. The ependymomas were distinguished by their papillary support that consisted predominantly of a glial (astrocytic) core, in contrast to the fibrovascular core of choroid plexus neoplasms.
Extraventricular ependymal neoplasms, specifically ependymoma (and its variants), anaplastic ependymoma, and subependymomas, have all been reported outside the ventricles and central canal.27,28,36 Extraventricular ependymomas are rare in humans and have not been reported in nonhuman species. Despite ependymal morphology, their histogenesis remains uncertain. Ependymal cell rests in the white matter at the angle of the ventricles have been proposed as the cell of origin.11,35 However, the random distribution of extraventricular ependymomas, rather than restriction to the angle of the ventricles, suggests an alternative mechanism. 11
Immunoreactivity of neoplastic cells was observed in 9/14 (64%) ependymomas for GFAP and in 5/14 (36%) for pancytokeratin. Labeling intensity and distribution varied and did not correlate with the tumor variant. Previous cases of feline ependymomas had immunoreactivity for GFAP and vimentin.1,25 Canine ependymomas have been both immunohistochemically positive and negative for GFAP expression. 40 Most (67–100%) human ependymomas express GFAP.2,17,21,36 Immunoreactivity in human tumors was strongest within cells of pseudorosettes closest to the vascular endothelium; expression in rosettes was more variable.2,17,36 Studies of human ependymomas also report variable expression of S100, vimentin, and epithelial membrane antigen, the latter particularly in high-grade or anaplastic ependymoma. 17 Ependymomas do not express neuronal antigens. In the current study, 2 of 3 subependymomas had strong diffuse GFAP immunoreactivity; all were negative for pancytokeratin. Human subependymomas have partial to diffuse GFAP immunoreactivity.41,42
In summary, this study provides a macroscopic, histologic, and brief immunohistochemical analysis of ependymomas and subependymomas in 18 cats. Extraventricular location and a clear cell variant are described for the first time in a domestic species, and further evidence is provided for the inclusion of tanycytic variant in the classification of feline ependymomas. Immunohistochemistry for GFAP and pancytokeratin resulted in variable labeling intensity and distribution without correlation between immunoreactivity and tumor subtype. The recognition of extraventricular ependymoma broadens the differential diagnosis of feline brain tumors.
Footnotes
Acknowledgements
The authors gratefully acknowledge the histology technicians of the College of Veterinary Medicine, Cornell University, and the Royal Veterinary College, London, for skilful production of histologic and immunohistochemical slides. Lucy Woolford’s residency program was generously supported by the Horse Trust, United Kingdom. We thank Dr Susan Morgello, Mt Sinai Medical School, New York, for assistance with one extraventricular tumor, and faculty at the Institute of Neurology, Queen Square, London. The neuropathology group at Cornell University included Drs John Cummings, Alexander de Lahunta, and Brian Summers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
