Abstract
The respiratory system is well defended against inhaled bacteria by a dynamic system of interacting layers, including mucociliary clearance, host defense factors including antimicrobial peptides in the epithelial lining fluid, proinflammatory responses of the respiratory epithelium, resident alveolar macrophages, and recruited neutrophils and monocytes. Nevertheless, these manifold defenses are susceptible to failure as a result of stress, glucocorticoids, viral infections, abrupt exposure to cold air, and poor air quality. When some of these defenses fail, the lung can be colonized by bacterial pathogens that are equipped to evade the remaining defenses, resulting in the development of pneumonia. This review considers the mechanisms by which these predisposing factors compromise the defenses of the lung, with a focus on the development of bacterial pneumonia in cattle and supplemented with advances based on mouse models and the study of human disease. Deepening our understanding of how the respiratory defenses fail is expected to lead to interventions that restore these dynamic immune responses and prevent disease.
As metabolism requires absorption of oxygen and elimination of carbon dioxide, the high metabolic rates of mammals have forced an expansion of ventilatory capacity and surface area for gas exchange. This, along with the need to keep the gas exchange region moist and covered only by a simple epithelium, creates a vulnerability in the system with respect to protection from inhaled gases, particles, and pathogens. Some components of the respiratory defenses have been recognized for many years: entrapment of particles in the mucus that lines the airways, cough and mucociliary clearance to eliminate these trapped particles, the bactericidal and immunomodulatory effects of resident alveolar macrophages and recruited leukocytes, antibody and complement as soluble mediators of bacterial recognition and elimination, and helper T cells for activation of macrophages. More recent advances in the understanding of respiratory defenses include the diversity of innate immune proteins and peptides within the epithelial lining fluid of the lung, the Duox/lactoperoxidase system, the role of T helper (Th)–17 cells, the polarization of macrophage functions, and improved understanding of the extracellular and intracellular signals that regulate the system. 45,54,84,127,154 Finally, an important conceptual advance is the recognition of extensive cross-talk or integration between these components of the respiratory defenses, such that the system is considered to operate as a layered series of defenses with extensive interrelationships between the layers. Clearly, gas exchange is critical for life, and an advanced system has evolved to sustain this function.
It might seem incredible that such an integrated system could fail, yet pneumonia caused by opportunistic bacterial pathogens is common and important in all domestic animal species and in humans. The bovine respiratory disease (BRD) complex is a paradigm for the interactions among predisposing factors and causative pathogens. BRD occurs as shipping fever pneumonia of feedlot cattle, enzootic pneumonia of dairy and veal calves, and pneumonia of adult cows but also affects cattle in other production systems. Direct causes of respiratory disease in cattle include the bacterial pathogens Mannheimia haemolytica, Bibersteinia trehalosi, Histophilus somni, Pasteurella multocida, Mycoplasma bovis, and Trueperella (formerly Arcanobacterium) pyogenes, as well as the viral pathogens bovine herpesvirus 1 (BHV-1), bovine respiratory syncytial virus (BRSV), bovine parainfluenza 3 virus (BPIV-3), and bovine coronavirus (BCV). 28,65 Other infectious agents—including Dictyocaulus viviparus and Mycobacterium bovis—cause pneumonia in cattle but are not included in the present review. It is notable that most bacterial pathogens causing pneumonia in cattle can also be detected in healthy animals.
Risk factors for the development of bacterial pneumonia in cattle are well recognized and include infection with bovine viral diarrhea virus (BVDV) or the viruses listed above, abrupt weaning, transportation, sale through auction barns, mixing of social groups, low body weight of recently purchased beef calves, adverse climatic conditions including precipitation and rapid change in environmental temperatures, and poor air quality in calf barns. 10,30,31,108,130,197 Comparable risk factors predispose humans to bacterial pneumonia, including the effects of viral infection, psychological stress, and concurrent diseases. 13,36
Thus, BRD can be considered analogous to human community-acquired pneumonia, most frequently caused by Streptococcus pneumoniae (pneumococcus), Haemophilus influenzae, or Mycoplasma pneumoniae. As an animal model, BRD cannot compare to mouse models with respect to genetic manipulation, ease of experimentation, and the wide availability of reagents. Nonetheless, cattle offer advantages in understanding the pathogenesis of opportunistic infection in the lung: experimental infections are feasible and well characterized, their large size facilitates sampling, certain interventions can be inexpensively tested on large groups of animals, cattle and humans share certain immunologic (eg, interleukin 8) and anatomic features (eg, bronchial glands, cartilaginous airways) that differ from rodents, and, most important, the natural disease is of high prevalence and involves infection of outbred animals with evolutionarily host-adapted pathogens. BRD is clearly not equivalent to bacterial pneumonia of humans, yet the findings in mice, cattle, and humans each contribute to an improved general understanding of these disease processes.
This review focuses on the mechanisms by which the respiratory defense system is compromised by factors that predispose to bacterial pneumonia. Specific considerations include the effects of these predisposing factors on bacterial colonization of the nasal cavity; effects on the epithelium of the lower respiratory tract, including mucociliary clearance, receptor expression, and signal transduction; effects on the production and function of host defense molecules and antibody in the epithelial lining fluid; and effects on leukocyte responses, including alveolar macrophages and neutrophils. The nature of these defenses is mentioned only superficially, as these are already well described. 45,54,132,154 This analysis is supplemented where possible with comparative information from the human disease or from animal models, along with a synopsis of how the bacterial pathogens themselves have virulence attributes that allow them to evade the respiratory defenses (Fig. 1).
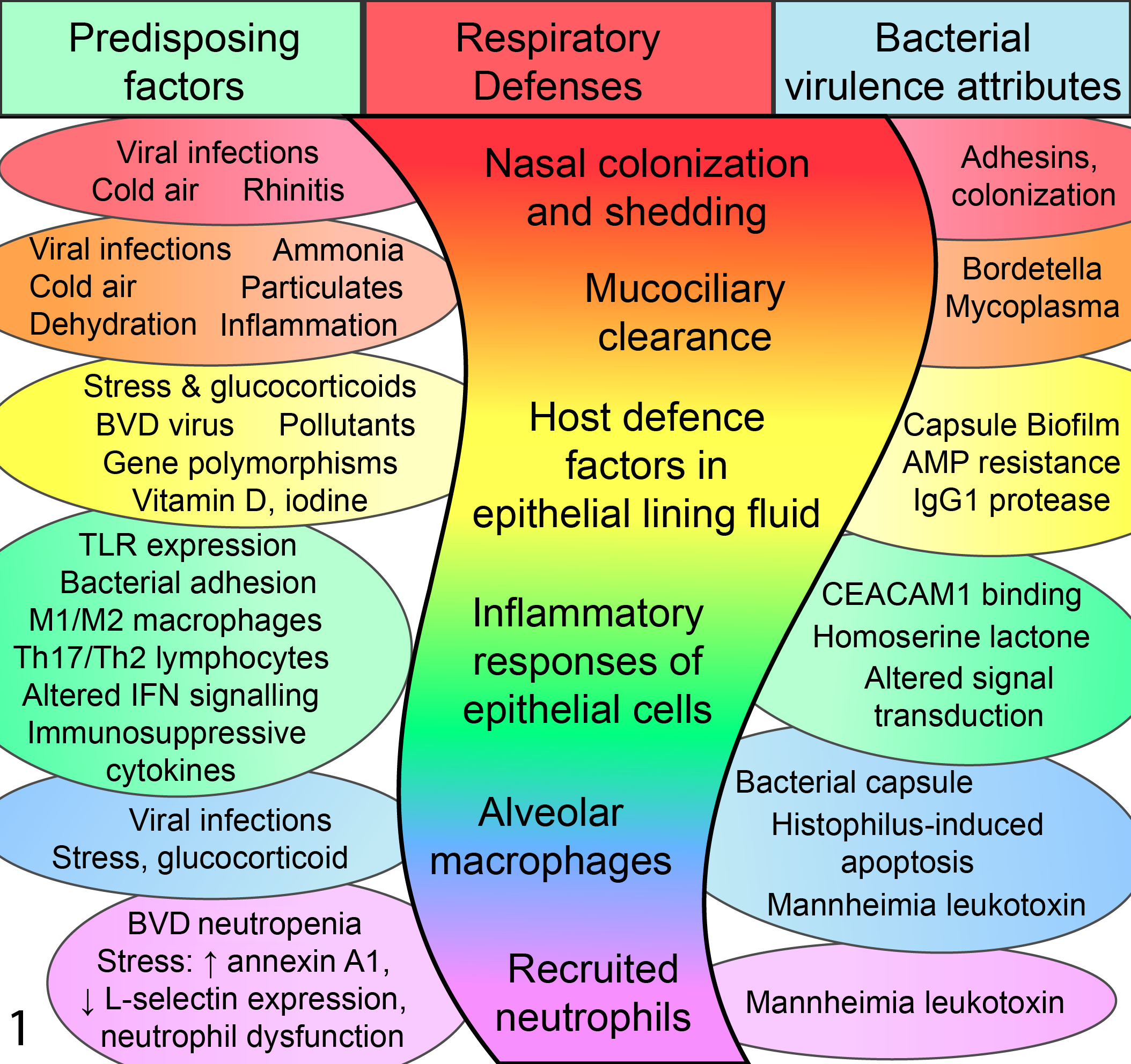
Mechanisms of failure of the respiratory defenses. The center column illustrates the layered arrangement of the respiratory defenses. At left is a partial summary of causes and mechanisms whereby each of these respiratory defenses is compromised by factors that predispose to bacterial pneumonia. At right is a partial summary of how bacterial pathogens further contribute to failure of the lung defenses. BVDV, bovine viral diarrhea virus; IFN, interferon; TLR, Toll-like receptor.
Effects of Predisposing Factors on Bacterial Colonization of the Nasal Cavity
Most discussions of innate immunity in the respiratory system focus on the lungs, yet the upper respiratory tract clearly plays an important yet poorly understood role in the pathogenesis of bacterial pneumonia. M. haemolytica is known to colonize the nasal mucosa 61 and tonsil 60 of clinically normal calves but is not uniformly present. Indeed, the number of these bacteria isolated from swabs of the nasal cavity increases as calves move from the farm of origin through auction barns and subsequently to feedlots. This increase in pathogen load is accompanied by a shift in the predominant serotype: serotype A2 is more frequently isolated from nonstressed pastured cattle, whereas the more pathogenic serotype A1 is more often isolated from stressed cattle soon after arrival to feedlots. 64
Viral infection may also play a role in these changes, as most viruses that predispose to bacterial pneumonia replicate in the nasal cavity before reaching the lung. 210 In calves experimentally infected with M. haemolytica, subsequent challenge with BHV-163 or BPIV-361 leads to greater number of M. haemolytica in nasal swabs, longer duration of bacterial shedding in nasal secretions, reactivation of shedding in quiescent infections, 62 and increased severity of clinical signs. 63,224 The effect of these viral infections seems unique to nasal populations of M. haemolytica, since comparable changes in tonsillar populations are not observed. 60 Similar to the effect of viruses, exposure to colder environmental temperatures also increases the prevalence of nasal colonization with M. haemolytica. 94,96 Thus, viral infection and exposure to cold air seem to selectively trigger M. haemolytica serotype A1 to colonize and grow to greater numbers in the nasal cavity.
The mechanisms of this effect in cattle are not known, but findings in a mouse model may be relevant. Hemagglutinin (H)–3 subtypes of influenza virus replicate to high titer in the middle ear of mice, and the ensuing inflammatory response promotes replication of S. pneumoniae at this site. 181 This effect is dependent on hemagglutinin subtype, with greater replication of H3 than H1 subtypes in the middle ear, 181 and on anatomic site, with no differences between hemagglutinin subtypes in the nasopharynx. 133 Although influenza in mice is not equivalent to viral infections of the cattle, the concept that virus-induced inflammation can trigger bacterial replication may be informative.
M. haemolytica can be cultured from samples of tracheal air of recently weaned and transported cattle, at levels ranging from 67 to 441 colony-forming units/m3, 74 and M. haemolytica is isolated from bronchoalveolar lavage fluid of healthy calves within 2 weeks of arrival to the feedlot but is rare at later times. 25 Paired M. haemolytica isolates from nasal swabs and transtracheal aspirates had the same genotype in 77% of cases (based on pulsed field gel electrophoresis), with significant agreement between the presence and absence of M. haemolytica in these 2 sites. 200 Thus, the presence of M. haemolytica in the nasal cavity likely influences the likelihood of bacteria reaching the lung, although the number of M. haemolytica in the nasal cavity and trachea is not well correlated. 74 Together, these findings indicate that weaning, transport, viral infections, and cold air stimulate colonization of the nasal cavity by bacterial pathogens, and this in turn challenges the lung by inhalation of infected droplets. In the context of M. haemolytica in cattle, the mechanisms of these effects remain unknown, but they represent a promising target for prevention of this disease.
Epithelial Cell Responses in the Lower Respiratory Tract
Effects of Predisposing Factors on Clearance of Inhaled Particles
Pulmonary clearance depends on deposition of inhaled particles, quality of the mucus layer and periciliary layer, ciliary beat frequency, and coordinated function of the ciliary apparatus. Altered mucus quality is well recognized in cystic fibrosis. Several explanations are proposed to link the causative CFTR mutation to excessive secretion of tenacious mucus and subsequent airway obstruction: direct effects of the abnormal electrolyte conductance on physicochemical qualities of the secreted mucus, reduced fluid secretion or increased absorption by airway epithelial cells leading to dehydration of the mucus layer and thinning of the periciliary layer, and excessive mucus secretion with goblet cell hyperplasia as a consequence of airway inflammation. 57,89,106,168 The viscosity of the mucus interferes with clearance, leading to airway obstruction, colonization with bacteria, and impaired migration and bactericidal activity of neutrophils. 128
The shrinkage or weight loss experienced by recently weaned and transported cattle is partly attributed to dehydration. Although dehydration is a potential cause for reduced thickness of the periciliary layer and increased tenacity of airway mucus, this has not been studied in cattle.
Ciliary dysfunction is a consequence of viral infection in cattle, 224 and BHV-1, BRSV, BPIV-3, and BCV are the important viruses infecting airway epithelial cells. Mucociliary clearance in normal calves eliminates 75% of aerosolized M. haemolytica from the lung within 2 hours of exposure and 90% by 4 hours. 114 BRSV infection causes loss of cilia as early as 1 to 2 days postinfection (DPI), with cellular necrosis at about 4 to 7 DPI, hyperplasia with undifferentiated or immature cells at 7 DPI, and repair by 10 DPI. 21,26,27,189 Reduced pulmonary clearance is documented at 7 DPI, taking 50% more time to clear ovalbumin after BRSV infection, 67 and retention of as much as 90% of inhaled cobalt particles after BPIV-3 infection. 184 In these respects, respiratory viral infections in cattle are comparable to human infection or mouse models of influenza. 158 Despite these abnormalities in airway epithelial cell morphology and function, delayed mucociliary clearance has been considered less important than defective bactericidal activity in the pathogenesis of bacterial pneumonia. 224
The effect of cold air on mucociliary clearance has been studied in ruminants. Calves held at 3°C compared with 17°C had a 24% reduction in nasal mucus velocity. 47 Similarly, for bovine and sheep trachea studied ex vivo, reducing the air temperature from 37°C to 34°C or 30°C induced a marked reduction in ciliary beat frequency and mucus transport, with ciliary clumping and reduced height of the epithelium observed at 30°C. 47,100,186,187 Since cold-adapted animals are not at great risk of pneumonia, the mechanisms of adaptation to cold air and the detrimental effects of whole-body hypothermia 155 also merit consideration.
Particulates and xenobiotics are known to affect mucociliary clearance. Inhaled particles are likely relevant to reduced air quality in intensively housed calves and swine, but the effect on ciliary function is not straightforward: swine barn dust extracts increased ciliary beat frequency in primary cultures of bovine bronchial epithelial cells, whereas they quelled the enhancing effects of sympathetic stimulation with a β-agonist. 221 Elevated ammonia levels are associated with reduced ventilation and use of solid partitions in crowded calf barns in the autumn and winter and are thus another potential reason for susceptibility to pneumonia. Chronic exposure to ammonia is reported to cause ciliary dysfunction, degeneration of nasal epithelium, and rhinitis in mice, 211 as well as exacerbate the lesions of mycoplasmal infection in rats, 19 but its effects have not been studied in cattle. Although the above conditions are associated with increased frequency of respiratory disease, 20,49,108,126 ammonia levels in 1 study were inversely correlated with BRD prevalence. 118 Indeed, a study of swine suggests that organic particulates may be of more importance than ammonia in explaining the effects of poor air quality on growth and feed conversion. 137 Cigarette smoke, 212,220 nitrogen dioxide, 46 and sulfite 34,142 —although not of relevance to BRD—are other xenobiotics causing reduced ciliary function.
Inflammation and sex hormones may also affect ciliary function, but this is based on only scant evidence. Using in vitro systems, acute exposure to inflammatory cytokines increased ciliary beat frequency, while this was reduced with chronic exposure to interleukin 8. 4,163,179 In vitro ciliary beat frequency was inhibited by progesterone treatment but restored with estrogen, 90 a preliminary finding of potential relevance to feedlot cattle treated with growth promotants. Thus, reasons for failure of mucociliary clearance in cattle include infection with viruses and cold exposure, with more poorly characterized contributions from dehydration, exposure to organic particulates or noxious gases in the barn environment, and chronic airway inflammation.
In addition to the above predisposing factors, several bacterial pathogens are known to impede ciliary function, both by secretion of bacterial toxins and by the physical effect of adhering to cilia. Ciliostatic effects are important in the pathogenesis of M. pneumoniae in humans and mouse models, as well as Mycoplasma hyopneumoniae in swine. Ciliostasis is also induced by infection of bovine tracheal explants with Mycoplasma dispar but not with certain other BRD-associated mycoplasmas, including M. bovis and Acholeplasma laidlawii. 5,86,107 Cilia-associated respiratory bacillus isolates from cattle cause loss of cilia from tracheal explants, but this pathogen is a minor contributor to BRD. 81,141 Bordetella bronchiseptica adheres to canine cilia, causes ciliostasis and reduced clearance of particles within 1 hour of infection, stimulates mucus secretion by 4 to 8 hours, and later induces epithelial cell cytotoxicity at 36 hours from secretion of tracheal cytotoxin. 7,15 Therefore, ciliary dysfunction is a well-recognized effect of several bacterial pathogens but is not known to occur with any of the bacteria causing BRD.
Effects of Predisposing Factors on Receptors on the Surface of Airway Epithelial Cells
Toll-like receptors (TLRs) 1, 2, 4, 5, and 6 are cell surface receptors recognizing bacterial products, 154 and their genes are expressed in bovine airway epithelial cells (Kawashima et al 97 and unpublished data). Various factors that predispose to bacterial pneumonia are known to alter expression of these receptors. TLR2 expression in airway epithelial cells is decreased in patients with chronic obstructive pulmonary disease, and TLR2–/– mice have defective lactotransferrin (lactoferrin) production following bacterial challenge. 218 On the other hand, TLR2 expression and subsequent functional responses are increased by treatment of airway epithelial cells with tumor necrosis factor (TNF)–α, interferon (IFN)–γ, or corticosteroid. 216 TLR4 expression in nasal epithelium and cultured airway epithelial cells is reduced by corticosteroid or exposure to cigarette smoke, with subsequent adverse effects on antimicrobial peptide production. 120 Human respiratory syncytial virus induces TLR3 expression in airway epithelial cells, with increased cytokine responses when these cells are then stimulated with TLR3 agonists. 76 Thus, altered TLR expression in airway epithelium is 1 mechanism whereby viral infections, corticosteroids, and inflammatory cytokines alter the susceptibility to bacterial infection.
Bacterial adhesins bind to host cell ligands, and altered expression of these ligands affects colonization of the lung by pathogens. BHV-1 is reported to promote adhesion of M. haemolytica to airway epithelial cells in vitro, 41,66 and similar effects are well studied in human virus-bacterial interactions. Human influenza A virus, respiratory syncytial virus, and parainfluenza virus enhance adhesion of S. pneumoniae and H. influenzae to cultured bronchial epithelial cells by at least 2 mechanisms: altering the cell-surface expression of receptors for bacterial adhesins (eg, platelet activating factor receptor, which binds both the lipooligosaccharide of H. influenzae and phosphorylcholine in the cell wall of S. pneumoniae), as well as cleavage of host sialic acid by influenza viral neuraminidase, thus exposing receptors on epithelial cells. 9,39 Whether bovine viruses induce similar changes remains unknown.
In addition to the above direct effects of viral infection, the ensuing host inflammatory response may alter expression of receptors for bacterial adhesins on respiratory epithelial cells, potentially promoting colonization of the lung by bacteria. 39,180 Since bacterial peptidoglycans in barn dusts induce TLR2-dependent inflammatory responses in mouse lung, 159 these findings may have relevance to the pathogenesis of enzootic pneumonia in intensively housed dairy and veal calves. 108,137 Inflammation induced by short-term exposure to cold air 44 could have similar effects, although specific evidence is lacking.
Effects of Predisposing Factors on Epithelial Cell Signal Transduction
Respiratory epithelial cells have elaborate mechanisms to sense and eliminate viral infection, including recognition by TLR3, TLR7/9, RIG-I, MDA-5, and Nlrp3, leading to activation of type I interferon and induction of a range of antiviral responses. 174,208 Successful viral pathogens have evolved diverse mechanisms to evade these recognition and effector mechanisms, 205,208 and some of these evasion strategies alter the immuno-inflammatory response to bacterial infection of the lung.
Several bovine respiratory viruses dysregulate the expression of IFN-α and IFN-β. BHV-1 encodes the viral proteins ICP0 and ICP27, which block induction of IFN-β gene expression. 42 BRSV also appears to inhibit type I interferon responses. 190,193 In vitro infection of macrophages with BVDV has both stimulatory and inhibitory effects on these responses; the in vivo situation is further complicated by effects of virus strain and time after infection. 152,193 Although BVDV also infects airway epithelial cells, the effect on type I interferon responses in these cells is unknown. 3,37
This viral manipulation of host cells impairs specific aspects of pulmonary defenses against bacteria, as described in later sections, and also has broader effects on regulation of inflammation and immunity in the lung. For example, nuclear factor (NF)–κB signaling is essential for a protective immuno-inflammatory response to Pseudomonas aeruginosa in mice, 35 and viral infections are known to modulate this signaling pathway. 170,203 Similarly, viral infections affect RIG-I–, MDA-5–, and TLR3-induced signaling through the type I interferon pathway. 13,140 These effects seem to vary among different viruses, time points after infection, and details of the host cells analyzed. Consequently, it is difficult to make general inferences about how the virus-manipulated epithelial cells respond to inflammatory stimuli to induce innate defense proteins, vascular changes in inflammation, and recruitment and activation of leukocytes.
Some virus-induced defects in the host immuno-inflammatory response promote bacterial survival and proliferation in the lung, 178 but viral meddling with the host response may also exacerbate the immuno-inflammatory reaction to bacterial infection. For example, influenza virus infection induces glucocorticoid, IL-10, or transforming growth factor (TGF)–β, which are immunosuppressive factors that predispose to bacterial infection. 13,93 BRSV infection of lambs induces gene expression of IFN-β, IFN-γ, TNF-α, IL-8, and MCP-1, 190 and BHV-1 infection of bronchial epithelium elicits IL-1, IL-8, and TNF-α expression. 165 These findings are similar to the above-described influenza models in mice, but ovine BRSV infection differs from the mouse model in a reduced production of IFN-α, IL-10, and TGF-β. 65 Thus, the findings in mouse models of influenza-bacterial coinfection may not mirror the viral-bacterial synergy that occurs in BRD.
In addition to these immunosuppressive effects, mice coinfected with influenza virus and Legionella pneumophila have defective production of tissue-repairing growth factors that is associated with more severe damage to airway and alveolar epithelium and increased mortality. 92 Thus, consideration should be given not only to virus-induced failure of antibacterial immunity but also to defective repair and tolerance of injured tissues.
Bacteria that cause pneumonia are also known to modulate epithelial signaling pathways. For example, virulence factors of Moraxella catarrhalis and Neisseria meningitidis adhere to carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) on airway epithelial cells, and this interaction interferes with signaling through the TLR2 pathway. 78 N-acyl homoserine lactone is produced by P. aeruginosa and functions in quorum sensing, but this bacterial small molecule also inhibits I-κB kinase to interfere with NF-κB–induced stimulation of innate immune responses. 105 Commensal bacteria may play a similar role in pathogenesis: Streptococcus salivarius suppresses NF-κB responses in epithelial cells and thereby represses the production of neutrophil-recruiting chemokines when the epithelial cells are subsequently exposed to the pathogen P. aeruginosa. 38 Other examples involve the production of immunosuppressive cytokines, such as the cAMP-mediated expression of IL-10 that is triggered by the adenylate cyclase toxin of Bordetella pertussis. 78
Host Defense Molecules in the Epithelial Lining Fluid
Host defense molecules in the epithelial lining fluid of the respiratory tract are derived from the epithelium, macrophages and other leukocytes, and plasma. These factors function as opsonins, kill or limit the growth of microbes, protect tissues against oxidative and proteolytic injury, modulate the immuno-inflammatory response, and promote tissue repair. 77,132,154 Although it is helpful to classify innate defense factors into these functional categories, this is certainly a simplification because each of these molecules generally has multiple functions that contribute to the health of the respiratory system. Antibacterial molecules include defensins, cathelicidins, lactotransferrin, hypothiocyanite, and hypoiodous acid (products of the Duox/lactoperoxidase system); anionic antimicrobial peptides; and lipocalins. Opsonins include immunoglobulins (Ig), complement proteins, the surfactant proteins A and D, and pentraxins. Most of the above factors are immunomodulatory and contribute to tissue repair, as do annexins, odorant binding protein, Clara cell secretory protein, haptoglobin, and SPLUNC. 132,154,167 Despite the relatively short time since their discovery, much has been discovered of how these innate defenses fail by mechanisms including lack of production, degradation or impaired function, or resistance of pathogens to their effects.
Effects of Predisposing Factors on Production of Innate Defense Molecules
Expression of innate defense genes may be constitutive, inducible, or both. For example, human β-defensin (HBD)–1 is expressed in normal individuals, whereas HBD-2 as well as the bovine defensins tracheal and lingual antimicrobial peptide (TAP, LAP) are normally present at low levels, but their expression is stimulated by bacteria, inflammatory cytokines, or TLR agonists. In contrast, lactotransferrin is constitutively present but induced to higher levels by inflammation. 3,116,134,154,171 Factors that reduce the baseline or induce levels of these innate defense molecules are one mechanism by which predisposing factors lead to bacterial pneumonia.
Polymorphisms are present in the noncoding regions of bovine β-defensin genes that are associated with somatic cell count in milk, 11,139,196,217 and promoter polymorphisms in the lactotransferrin gene correspond to levels of this protein in milk and with somatic cell count. 12,87,144 Thus, genetic variations in these and upstream regulatory genes are a potential explanation for differing levels of innate defense molecules among individuals and are of potential utility with respect to genetic improvement of disease resistance. 113 Although human β-defensin genes have copy number polymorphisms that show some association with a variety of diseases, in silico analysis of the bovine genome has so far not revealed such polymorphisms in cattle (unpublished data).
Corticosteroids impair the lipopolysaccharide (LPS)–inducible expression of tracheal antimicrobial peptide expression in cattle 134 and of HBD-2 201 and HBD-3 in humans, 51 perhaps by interfering with TLR4/NF-κB signaling. Inhalation of corticosteroid presents little risk to otherwise healthy human asthmatics as they have few bacteria reaching the lung, but this is a risk factor for patients with chronic obstructive pulmonary disease (COPD) or community-acquired pneumonia, in which bacterial challenge of the lung is greater. 6,93,143,213 Since corticosteroid levels are elevated in calves that are stressed by disruption of social groups and transportation or by exposure to cold, 82,117,187 this is a plausible mechanism contributing to disease in calves exposed to these stressors. Weaning stress is also associated with lower levels of blood lactotransferrin in cattle, and glucocorticoids reduce secretion of lactotransferrin from human bronchial epithelial cells. 101,166 Thus, impaired secretion of innate defense factors from the airway epithelium is 1 way that corticosteroids and stress predispose to bacterial pneumonia.
Viruses can also cause failure of expression of innate defense factors. Noncytopathic BVDV is a major predisposing cause of bacterial pneumonia in feedlot cattle 164 and infects bronchial epithelial cells in vivo without causing morphologic change. 37 Infection of bovine tracheal epithelial cells with this virus does not affect baseline expression of host defense factors but abrogates the LPS-stimulated expression of tracheal antimicrobial peptide and lactotransferrin. 3 Human influenza virus and herpes simplex virus each suppress baseline expression of human β-defensin-1 in bronchial epithelial cells. 172 Chinchilla airway (nasal) epithelial cells infected in vivo or in vitro with respiratory syncytial virus have reduced β-defensin expression, which permits colonization with H. influenzae; this effect is blocked by prior intranasal administration of the defensin, confirming the specificity of the effect. 129 Thus, these various models provide evidence that impaired defensin expression due to respiratory viral infection predisposes to bacterial pneumonia. In contrast, infection of lambs with BRSV had no effect on expression of sheep β-defensin-1, while infection with parainfluenza virus induced expression of this gene, albeit late in the disease course. 97 The effect on subsequent challenge with bacteria or LPS was not examined.
Th17 cells produce IL-17A, IL-17F, IL-22, and IL-23. In addition to eliciting neutrophil recruitment, these cytokines induce defensin gene expression on mucosal surfaces, 54 and IL-17A has been found to induce tracheal antimicrobial peptide expression in bovine tracheal epithelial cells (unpublished data). Thus, factors that skew the acquired immune response toward a Th1 or Th2 response rather than a Th17 response can interfere with innate immunity on the respiratory mucosa, and indeed the Th2 cytokine IL-13 does suppress TLR2-induced expression of human β-defensin-2 in airway epithelial cells. 219
Micronutrients affect production of host defense factors. Vitamin D, produced locally by airway epithelial cells or activated macrophages, increases expression of cathelicidin and CD14. 79 Thus, vitamin D deficiency is associated with bacterial pneumonia in humans and might partly explain the seasonality of this disease. 79 However, in cattle, vitamin D levels had no effect on response to challenge with BRSV, 173 and injection of vitamin D did not affect the prevalence of BRD. 40
Iodine deficiency might also interfere with production of host defense factors. The Duox/lactoperoxidase system of airway epithelial cells is comparable to the NADPH oxidase/myeloperoxidase system of neutrophils. Tracheobronchial epithelial gland cells secrete lactoperoxidase. In the presence of hydrogen peroxide from the airway surface epithelium, lactoperoxidase catalyzes formation of hypothiocyanite from thiocyanate and hypoiodous acid from iodine. These factors have antibacterial and antiviral activity. The Duox/lactoperoxidase system is defective in cystic fibrosis, where epithelial cells fail to secrete thiocyanate. 136,160 Administration of iodine enhances production of hypoiodous acid. 58 However, observations of diagnostic case material suggest that pneumonia is uncommon in ruminants with iodine deficiency, so the negative effects of iodine deficiency on respiratory defenses are presumably minor. Chromium, copper, selenium, vitamin E, vitamin A, cobalt, and zinc are reported to affect immune function, but dietary levels have not shown a clear association with the development of BRD. 24,40,50,191
Other reported causes for failed regulation of host defense factors may not be relevant to bovine respiratory disease. Vanadium pentoxide and sulfate (air pollutants from diesel exhaust) inhibit LPS-induced expression of tracheal antimicrobial peptide in bovine tracheal epithelial cells. 102 Cigarette smoke reduces β-defensin expression in airway epithelium and may thus play a role in the pathogenesis of COPD. 151 Finally, of possible relevance to the nasal cavity, the normal flora is thought to constantly induce production of innate defense factors, including defensins, thereby protecting the mucosa against colonization by pathogens. There is limited evidence that disruption of this commensal flora may lead to failure of local innate defenses and colonization by pathogens. 204
Together, these findings imply that failure to synthesize host defense factors, including β-defensins, is a likely mechanism by which stress and viral infection predispose to bacterial pneumonia. Nutritional factors, induction of a skewed immune responses, and environmental toxins may have similar effects.
In addition to these host and viral effects, several bacterial pathogens have evolved mechanisms to evade these antimicrobial factors, although none are directly relevant to BRD. 104,157 Binding of Klebsiella pneumoniae to TLR2 and TLR4 triggers a signaling pathway in host epithelial cells that interferes with NF-κB– and MAP kinase–mediated production of 2 β-defensins. Thus, this provides an example of a bacterial pathogen actively suppressing the antimicrobial peptide response that it would otherwise stimulate. 135 Salmonella typhimurium similarly suppresses α-defensin production by Paneth cells in the duodenum by an unknown mechanism. 175 Finally, procyanin is a metabolite of P. aeruginosa that appears to inhibit Duox and thereby evade the Duox/lactoperoxidase defenses of the airways. 160
Effects of Predisposing Factors on the Function of Innate Defense Molecules
In addition to the aforementioned effects of genetic polymorphisms on the levels of secreted innate defense molecules, genetic variations affecting the function of these molecules may permit bacterial infection of the lung. 113 For example, a nonsynonymous polymorphism has been detected in the coding region of bovine tracheal antimicrobial peptide, which appears to influence the function of the corresponding peptides. 196 Polymorphisms have also been identified in the coding region of genes for cathelicidins, 68 complement proteins, 223 and surfactant protein D. 70
Even when innate defense molecules are secreted appropriately, other substances in the epithelial lining fluid of the lung may interfere with their function. The best studied of these effects is the sensitivity of β-defensin to increased salt concentration, such as that found in the airway fluid of patients with cystic fibrosis. 71 Cathepsins secreted by macrophages cause proteolytic cleavage and inactivation of β-defensins, and neutrophil elastase cleaves lactotransferrin. 195 Thus, the tissue microenvironment may compromise the function of secreted antimicrobial proteins.
Some pathogens are able to evade these defenses directly, either by resisting the effects of antimicrobial factors or by inciting their destruction. 104,157 Group A streptococci such as Streptococcus pyogenes produce the protease SpeB that cleaves and inactivates the human cathelicidin LL-37. 95 Similarly, a protease of Aspergillus fumigatus cleaves several complement proteins and thereby prevents complement activation, 14 while protease IV of P. aeruginosa degrades surfactant proteins. 122 Several Gram-positive pathogens alter the acetylation patterns of peptidoglycan in their cell wall, as a mechanism to resist degradation by lysozyme. 43 The bacterial capsule is important in evading complement 80 but also confers resistance to cathelicidins among some pathogens, including N. meningitidis. 192 Finally, Staphylococcus aureus, Streptococcus agalactiae, and others modify their teichoic acids, phosphatidylglycerol, or lipid A to make the normally anionic bacterial surface more neutral and therefore less attractive to cationic antimicrobial peptides. 157
Despite this diversity of mechanisms by which bacterial pathogens evade antimicrobial peptide defenses, M. haemolytica, H. somni, and P. multocida have uniformly similar sensitivity as Escherichia coli, to the levels of tracheal antimicrobial peptide expected to be induced in vivo. 196 In contrast, Mycoplasma bovis was resistant under the conditions tested.
Effects of Predisposing Factors on Antibody Defenses
In addition to the above innate defense factors, IgM, IgG—particularly IgG2—and IgA are effective in defense of the bovine respiratory tract against bacterial pathogens by activating complement, opsonizing for enhanced recognition by macrophages and neutrophils, neutralizing bacterial toxins, and blocking sites of colonization. Lack of prior exposure is 1 reason for failure of this defense mechanism. At the time of arrival to feedlots, the prevalences of serum antibody titers to M. haemolytica and H. somni vary considerably but are generally high, presumably because of prior exposure or vaccination. In several studies, high on-arrival antibody titers to bacterial pathogens were associated with lower incidence of BRD, although that finding was not demonstrated in other studies. 123 –125,198 In contrast, generally low titers to Mycoplasma bovis prior to mixing of calf groups suggests that prior exposure is infrequent, and titers have not been correlated with later frequency of disease. 8,25
Maternal antibody is considered important in defense against BRD in the first 3 months of life. Failure to acquire colostral immunoglobulin is associated with an increased prevalence of respiratory disease in this age group and may result from inadequate levels of protective antibodies in colostrum, poor quality or inadequate storage of colostrum, or failure of the neonatal calf to ingest colostrum. 209
Viral infections may interfere with an adequate immune response after exposure to a bacterial pathogen. Even if the acquired immune response develops too slowly to influence the course of acute disease in an immunologically naive calf, development of an antibody response may influence the longer term course of disease. The evidence that BHV-1 and BVDV negatively affect humoral immune responses has been previously reviewed. 22,33,193
The bacterial pathogens causing BRD have several mechanisms to limit antibody-mediated immunity, although it should be emphasized that induction of humoral immunity does confer protection in challenge models of the disease. The capsule of M. haemolytica confers partial resistance to antibody, whereas an acapsular mutant had similar sensitivity to complement as the parent strain. 131 Formation of biofilm is described for H. somni and several other bovine respiratory pathogens, 148,176 although the functional role in resisting the host immune response during development of BRD has not been tested. M. haemolytica secretes a protease that cleaves IgG1, and this pathogen also encodes genes homologous to IgA protease of other Pasteurellaceae bacteria even though IgA proteolytic activity has not been detected. 69,109 H. somni secretes immunoglobulin-binding proteins, including 3 IgG2-binding proteins, which are associated with serum resistance. 182 Thus, degradation or binding of antibody and formation of a capsule are mechanisms by which these pathogens may evade the host antibody response.
Leukocyte Responses
Effects of Predisposing Factors on Pulmonary Alveolar Macrophages
Alveolar macrophages—considered distinct from other histiocytes in the lung, including intravascular and interstitial macrophages as well as dendritic cells—have considerable heterogeneity in function. Research in mice has revealed plasticity of macrophage responses and diversity of macrophage subsets; evidence for the validity of these concepts in cattle remains limited. Resident alveolar macrophages form a self-renewing cell population supplemented at a low level by immigration of blood monocytes. These resident cells in mice are quiescent, with low major histocompatibility complex (MHC) II expression, weak antigen-presenting function, and low production of inflammatory cytokines. In contrast, an inflammatory stimulus elicits a more substantial influx of monocytes to form so-called exudative macrophages and monocyte-derived dendritic cells in the lung tissue; these cells present antigen effectively and activate T cells, produce nitric oxide and TNF-α, and stimulate injury to lung tissue. 199
Macrophages of mice and other species have functional differences depending on the stimuli they encounter during differentiation. Classically activated (M1) macrophages develop in response to IFN-γ and TNF-α; stimulate inflammation by secreting TNF-α, IL-12, and chemokines; and have substantial bactericidal activity by undergoing an oxidative burst as well as generating nitric oxide from arginine. In contrast, alternately activated (M2) macrophages develop in response to IL-13 and IL-4, express arginase I, and promote tissue repair and restoration by secreting IL-10, TGF-β, and IL-6. 2,206 Comparable subsets have been suggested in cattle, although they have not been extensively characterized. 52 Thus, macrophages in the lung maintain alveolar homeostasis by recycling surfactant, phagocytose particulates including bacteria, generate reactive oxygen and nitrogen species, and secrete cytokines and proteases, yet the spectrum of these functions depends on the monocyte subpopulation from which they were derived, the method of recruitment, and the stimuli encountered in the lung microenvironment. 2,206
Most research on alveolar macrophages of cattle, particularly the effects of viral infection and corticosteroids, predated these concepts of macrophage heterogeneity, which are of considerable relevance to understanding the findings. For example, if viral infections elicit exudative macrophages, then macrophages isolated from infected calves would be expected to respond differently than normal resident macrophages infected in vitro, yet this difference would not represent a direct effect of viral infection on macrophage function. Furthermore, the diversity of methods used to probe macrophage function over the years has probably contributed to the conflicting data in the published literature.
Noncytopathic BVDV has a suppressive effect on alveolar macrophage function. Infection reduces expression of receptors for immunoglobulin and complement; interferes with phagocytosis and fusion of phagosomes to lysosomes; reduces bactericidal activity; reduces the secretion of TNF-α, IL-1β, IL-6, and neutrophil chemoattractants; and enhances secretion of prostaglandin E2. However, it has no effect or a stimulatory effect on production of IL-12 and type I interferon. 110,115,207,215 Similarly, BPIV-3 and BHV-1 have shown mainly suppressive effects on alveolar macrophage function, including reductions in phagocytosis and oxidative burst, 53,112,185 although TNF-α production in response to LPS was enhanced by BPIV-3 infection, 17 and BHV-1 infection enhanced TLR2 and TLR4 gene expression in blood mononuclear cells. 83
In contrast to the situation with BVDV and BPIV-3, BRSV infection has shown more variable effects on alveolar macrophages. BRSV infection recruits macrophages into the lung, resulting in increased levels of nitrate in lung tissue (of postnatal lambs). 189 Alveolar macrophages infected in vitro show early induction of IL-1β, IL-6, and IL-8 gene expression, as well as more minor expression of TNF-α, IL-12, IL-4, and IL-10. 56,161 The level of induction was less than for other TLR agonists, consistent with the mild but significant neutrophil infiltrates within the resulting lesions. 29,56 Phagocytosis of nonopsonized and of opsonized particles is reported to either increase or decrease depending on the study. 27,98,145,202 BRSV infection has generally had no effect on the oxidative burst of alveolar macrophages. 145 Thus, BRSV induces production of inflammatory mediators by alveolar macrophages, but the effects of this infection on macrophage function are not clear.
Studies in mouse models of influenza inform our understanding of how viruses affect pulmonary alveolar macrophage function. Nasal infection with influenza virus triggered type I interferon production that interfered with production of the chemokine CCL2, thus reducing recruitment of macrophages to the lung and predisposing to colonization by S. pneumoniae. 138 Pulmonary infection with influenza virus also induced IFN-γ production by T lymphocytes, which reduced expression of the scavenger receptor MARCO on pulmonary alveolar macrophages, thereby impairing phagocytosis and elimination of S. pneumoniae. 194 Finally, influenza infection in mice reduces recruitment, activation, and TNF-α production by NK cells, and this deficiency in TNF-α–mediated activation of alveolar macrophages increases the susceptibility to infection with S. aureus. 188 These studies in mice thus provide detailed examples of how the host antiviral response abrogates defenses against bacteria.
The effects of stress on alveolar macrophage function have been difficult to study. There is a widespread assumption that stress impairs the function of these innate immune cells, but much evidence suggests that acute stressors—which are of most relevance to recently arrived feedlot cattle—enhance many aspects of the innate immune response. 1,59 For example, in a rat model, stress induces release of heat shock protein 72 from cells, which acts similarly to a damage-associated molecular pattern (DAMP) to enhance production of nitric oxide from macrophages. 59 This idea of stress-associated enhancement of innate immunity is clearly sensible in an evolutionary context, yet contradicts the observed occurrence of respiratory disease in stressed cattle. These observations suggest that the vague concept of “immunosuppression” must be abandoned and more appropriately focused on specific elements of innate immune responses (Table 1). 48,73,149
Summary of Some Positive and Negative Effects of Acute Stress and Glucocorticoids on Measures of Innate Immunity Relevant to Bovine Respiratory Disease. It is erroneous to think of stress as immunosuppressive: glucocorticoids and the body s response to acute stress have stimulatory and suppressive effects on various elements of the innate immune response, with differing outcomes in different models of disease.

BRD, bovine respiratory disease; IL-8, interleukin 8; LPS, lipopolysaccharide; TLR, Toll-like receptor; TNF-α, tumor necrosis factor–α.
Effects of Predisposing Factors on Neutrophils
Adverse effects of stress and viral infections on neutrophil responses include neutropenia, altered neutrophil recruitment to the lung, effects on sensitivity to leukotoxin, and effector responses to bacterial infection. Neutropenia is the most readily measured effect on neutrophil responses in the lung and predisposes to pneumonia, such as that caused by P. aeruginosa in humans. Noncytopathic BVDV infection induces a sustained neutropenia, in part due to reduced production in the bone marrow. 99 The effect of other respiratory viral infections, including BHV-1, on blood neutrophil counts is less consistent. Conversely, abrupt weaning stress results in a slight increase in the number of blood neutrophils. 119
Stress and viral infections may interfere with neutrophil recruitment to the lung. This is observed in mouse models of influenza, where the virus-triggered type I interferon response limits the production of CXC chemokines, thus increasing the susceptibility to pulmonary S. pneumoniae challenge. 178 Similar findings have not been reported with viral infections of cattle.
Stress or glucocorticoid treatment reduces expression of L-selectin—and to a lesser degree CD18—on bovine neutrophils, 23,119,214 reduces TLR2 and TLR4 messenger RNA (mRNA) expression, 55 and delays apoptosis. 121 Despite these effects, dexamethasone-treated cattle had greater numbers of neutrophils after administration of IL-8 into the lung or uterus, 103,134 with similar findings in the mammary gland of cows experiencing transport stress. 222 These findings surprisingly suggest that either recruitment or survival of neutrophils is increased by glucocorticoid.
Glucocorticoids induce synthesis of the anti-inflammatory protein annexin A1 and stimulate release of this protein from the secondary granules of neutrophils. 16,156 Calves with lower levels of annexin A1 at the time of arrival to the feedlot are more likely to later develop pneumonia; 177 this finding, along with the observation that weaning stress induces expression of proinflammatory cytokines, 146 may suggest that calves that are better able to downregulate these neutrophil inflammatory responses are less likely to develop disease.
Viral infections sensitize neutrophils to the damaging effects of M. haemolytica leukotoxin. Bovine mononuclear cells infected with BHV-1 produce IL-1β and other cytokines that upregulate expression of CD18 (LFA-1) on neutrophils. Since CD18 is the receptor for M. haemolytica leukotoxin, this increases the binding of this toxin and exacerbates the cytotoxic effect on bovine neutrophils. 111
Laboratory studies of the phagocytic and bactericidal functions of neutrophils are complex and often contradictory, perhaps because of the diversity of analytical methods and the considerable variability among individual animals. Abrupt weaning has relatively minor adverse effects on phagocytic activity of neutrophils and no effect on oxidative burst. 119 Weaning stress induces gene expression of IL-1β, TNF-α, IFN-γ, IL-8, and other chemokines in whole-blood leukocytes of calves. 146,147 Castration of calves (by the Burdizzo method) results in higher numbers of blood neutrophils and a mild increase in respiratory burst. 153 Neutrophils from temperamental bulls (which have elevated serum levels of cortisol) have slightly lower levels of oxidative burst and phagocytosis than do neutrophils from calm bulls. 88 Glucocorticoid treatment has been shown to inhibit phagocytosis, oxidative burst, and antibody-dependent cell-mediated cytotoxicity in bovine neutrophils. 103,150,162,169 Although these studies show significant influences of stress and glucocorticoids on neutrophil function, the effects are generally not large, usually affect only one of several time points, and differ between studies.
BRSV infection induces neutrophil and macrophage infiltration of airways and alveoli, 189 associated with increased levels of IL-8. 29 However, these BRSV-elicited neutrophils have lower myeloperoxidase levels as assessed by immunohistochemistry (IHC), suggesting dysfunction or immaturity. 189 Similarly, cattle infected with BPIV-3 or BHV-1, or mice infected with influenza, have evidence of reduced neutrophil phagocytosis or bactericidal activity. 13,18,224
Of relevance to dairy cows experiencing negative energy balance, β-hydroxybutyrate has adverse effects on neutrophil function, including reduced phagocytosis and formation of neutrophil extracellular traps. 75 Thus, many factors affect neutrophil recruitment and function, including stress, glucocorticoids, viral infection, and negative energy balance. However, these effects are neither uniformly negative nor positive (Table 1), and their contribution to development of pneumonia is uncertain.
Bacterial pathogens use diverse mechanisms to evade killing by neutrophils and macrophages. The capsule of M. haemolytica and P. multocida confers resistance to phagocytosis. 32,80 H. somni is capable of inhibiting the oxidative burst in neutrophils and alveolar macrophages, and the immunoglobulin-binding protein A inhibits phagocytosis and induces cytotoxicity of bovine monocytes. 72,85 The M. haemolytica leukotoxin induces apoptotic or oncotic death of ruminant neutrophils and macrophages. 183 Thus, the pathogens causing BRD are well equipped to interfere with the bactericidal effector functions of neutrophils and macrophages.
Conclusions
The mechanisms by which the risk factors for BRD affect lung defenses are complex. Earlier reviews of this topic focused mainly on the impact on mucociliary clearance and phagocytes, as well as considered which of these effects were more important. 91,224 Since that time, the recognized mechanisms of lung defense have flourished, leading to the following conceptual model of how such a multifarious system could fail.
Respiratory defenses can be considered a layered system, albeit one with considerable functional interaction among the layers. Specifically, for a bacterial pathogen to infect the lung, it must either evade or take advantage of the failure of these sequential defenses, including those of the nasal cavity, antimicrobial molecules in the airway lining fluid, mucociliary clearance, alveolar resident and exudate macrophages, and neutrophils (Fig. 1). In cattle that develop aspiration pneumonia, these barriers are still functioning but are overwhelmed by multitudinous bacteria. In contrast, for “shipping fever” or “community-acquired” bronchopneumonia in cattle and humans, it is likely that many of these defenses must be overcome before opportunistic pathogens are able to colonize the lung and cause bronchopneumonia. But pneumonia in cattle is not caused by common environmental bacteria or mucosal commensals—the example of aspiration pneumonia notwithstanding—implying that simultaneous failure of all respiratory defenses is uncommon. Instead, the risk factors for BRD cause failure of some respiratory defenses, while the bacterial pathogens that take advantage of this opportunity to colonize the lung are those that have virulence attributes allowing them to overcome the remaining defenses.
There are 2 major implications of this model. First, it raises the opportunity for collaboration between infecting pathogens. Most cases of bacterial pneumonia in cattle are polymicrobial, including various Pasteurellaceae, Trueperella pyogenes, and a diversity of Mycoplasma spp. This may reflect the importance of the predisposing factors that weaken the lung defenses, as infection with the causative bacteria is so common. It is, in theory, possible that each pathogen could interfere with different respiratory defenses, allowing 2 or more bacteria to colonize the lung together when either pathogen alone would be unable. However, mechanisms of polymicrobial pneumonia are little studied in humans and less in cattle.
Second, this model implies that disease prevention does not require that all of the failed lung defenses be cured. Instead, restoration of one or a few of these defenses—particularly those that the pathogens are ill-equipped to evade—may be adequate to eliminate bacterial infection and prevent disease. This concept is important for developing alternatives to antibiotics for prevention of BRD in feedlot, veal, and dairy calves.
Bacterial bronchopneumonia in cattle results from complex interactions among the stresses of weaning, transport and disruption of social groups, adverse climatic conditions, poor air quality, and viral and mycoplasmal infections, as well as infection with the bacteria that ultimately elicit the disease-causing host response. This complexity leads to challenges in investigating the mechanisms by which the respiratory defense system fails, yet it is this complexity of interacting causes that is lacking in mouse models of bacterial pneumonia. Nonetheless, a limitation of in vivo studies of BRD is the difficulty in measuring the inducible responses. A major criticism of past research is the focus on static parameters: blood neutrophil counts, baseline levels of cytokines and antimicrobial factors in body fluids, and ex vivo studies of leukocyte function. Most of the innate and acquired defenses of the lung are dynamic, inducible, and dependent on the microenvironment in the lung; such studies fail to capture the true nature of these immune responses. Thus, advancing this understanding will require synthesis of knowledge acquired from cell culture systems, mouse models, experiments in cattle, and observations of natural disease in cattle and other species, including humans. Understanding how host factors and pathogens interact to cause bovine respiratory disease clarifies the pathogenesis and may lead to improved strategies for disease prevention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Natural Sciences and Engineering Research Council (NSERC) of Canada, the Ontario Cattlemen’s Association, and the Ontario Ministry of Agriculture and Food.
