Abstract
Bovine respiratory syncytial virus (RSV) is a cause of respiratory disease in cattle worldwide. It has an integral role in enzootic pneumonia in young dairy calves and summer pneumonia in nursing beef calves. Furthermore, bovine RSV infection can predispose calves to secondary bacterial infection by organisms such as Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni, resulting in bovine respiratory disease complex, the most prevalent cause of morbidity and mortality among feedlot cattle. Even in cases where animals do not succumb to bovine respiratory disease complex, there can be long-term losses in production performance. This includes reductions in feed efficiency and rate of gain in the feedlot, as well as reproductive performance, milk production, and longevity in the breeding herd. As a result, economic costs to the cattle industry from bovine respiratory disease have been estimated to approach $1 billion annually due to death losses, reduced performance, and costs of vaccinations and treatment modalities. Human and bovine RSV are closely related viruses with similarities in histopathologic lesions and mechanisms of immune modulation induced following infection. Therefore, where appropriate, we provide comparisons between RSV infections in humans and cattle. This review article discusses key aspects of RSV infection of cattle, including epidemiology and strain variability, clinical signs and diagnosis, experimental infection, gross and microscopic lesions, innate and adaptive immune responses, and vaccination strategies.
Keywords
Bovine Respiratory Syncytial Virus Epidemiology and Strain Variability
Epidemiology
The first study to propose an involvement of respiratory syncytial virus (RSV) in respiratory disease in cattle was published several years after the original isolation and characterization of human RSV (hRSV). In 1968, Doggett et al 16 determined that bovine sera contained neutralizing antibody against hRSV, which suggested that a similar virus might exist in cattle. Published reports from respiratory disease outbreaks in several European countries 39,63,91 in the early 1970s identified viruses isolated from cattle that were closely related to hRSV. During this time, Inaba et al 38 isolated what appeared to be a new virus from cases of acute bovine respiratory disease in Japan, initially referred to as Nomi virus but later identified as bovine RSV (bRSV). 37 Subsequently, bRSV was confirmed in bovine respiratory disease outbreaks in the United States in published reports in 1974 67 and 1975. 75
Since these initial reports, bRSV has been detected in cattle herds worldwide, with rates of infection likely affected by management practices. In fact, estimates from several countries suggest that the frequency of bRSV exposure in some dairy and beef herds exceeds 50%. 23 Studies from Sweden have shown that between 40% to 100% of animals in a herd were seropositive, depending on geographic location and animal density. 61 A high frequency of bRSV serum antibodies in cattle (>70%) was reported in a sample of 50 Danish herds. 84 There are fewer published reports on the prevalence of bRSV in Britain, although results from a national survey in England and Wales suggest that most adult cattle have been exposed to bRSV. 64 In South African feedlots experiencing outbreaks of respiratory infections, the seroprevalence to bRSV was found to be 43%. 90 High frequency of bRSV seroconversion during the first month after arrival was found among feedlot calves in Canada. 54 Similarly, in the United States, results from a study of feedlot cattle reported a high rate of seroconversion. 12 Furthermore, the authors found bRSV seropositive rates in range cattle of 28% in calves, 49% in yearlings, and 70% in mature cows. Significant regional differences in the frequency of bRSV have also been reported from studies in Mexico. 70
bRSV-associated disease is most pronounced in calves less than 6 months of age, and infection can occur even in the presence of maternal antibodies. It has been estimated in some geographic areas that more than 70% of calves have been exposed by the age of 12 months. 86 As with hRSV, reinfections in calves are common. Viruses from recurrent bRSV infections varied by up to 11% in sequence, even in closed herds. 47 The authors proposed that based on the high degree of diversity, it is likely that recurrent infections result from reintroduction of bRSV into the herd before each new outbreak. Transmission of bRSV may occur via aerosol droplets from, or direct contact with, infected animals or indirectly via contaminated surfaces. 34 Seasonal periodicity is seen with bRSV, with most common occurrences of infections in the fall and winter months in temperate environments. 89
In addition to temperature fluctuations, additional stress factors that can increase the risk of bRSV infection include but are not limited to weaning, handling, transportation, mixing of cattle from different sources, crowding, and dusty environments. Clinical signs are frequently seen 7 to 10 days following a stressful event (eg, transport to a feedlot) but may be seen as late as 30 days or more after arrival. Respiratory viruses are well recognized as primary pathogens, where even uncomplicated infections cause substantial production losses. However, importantly, these viruses may predispose the calf to secondary bacterial infections, such as Mannheimia haemolyica, Pasteurella multocida, Histophilus somni, or Mycoplasma bovis, resulting in the bovine respiratory disease complex, the leading cause of morbidity and mortality in US feedlots. Its economic impact is considerable due to death losses, reduced performance, and costs of vaccination and treatment.
Strain Variability
bRSV belongs to the family Paramyxoviridae, subfamily Pneumovirinae, and genus Pneumovirus. Pneumoviruses are single-stranded, negative-sense RNA viruses with a genome of approximately 15.2 kb. The RSV viral RNA is transcribed into 10 major subgenomic mRNAs encoding 11 proteins, because the M2 gene encodes 2 proteins. Associated with the genomic RNA are nucleocapsid (N), phosphoprotein, large polymerase, and associated proteins, as well as transcriptional antitermination factor M2-1 and RNA regulatory protein M2-2. There are 3 transmembrane surface glycoproteins: attachment (G), fusion (F), and small hydrophobic. A nonglycosylated matrix or membrane protein (M) is associated with the inner face of the envelope. Finally, there are 2 nonstructural (NS) proteins that accumulate in infected cells: NS1, NS2.
RSV has been classified into 2 subgroups, A and B, based on antigenic and genetic differences. 1,13 bRSV isolates can be classified into subgroups based on reactivity of mAb to the G protein, although these may represent variants of a single major antigenic group. 15,19 The F protein is a type I viral fusion protein synthesized as a precursor that is proteolytically cleaved by a furin into disulfide-linked fragments. 57 Although bRSV isolates possess antigenically heterogeneous G proteins, a type II integral membrane protein, the nucleotide sequences have been shown to be less variable than for hRSV. 19,87 The F and G glycoproteins contain the main neutralization and protective epitopes. For RSV, the attachment protein is a major target of the host anti-RSV antibody response, 22 and certain regions may be under immune selection. 95 However, immunologic pressures may differ between bRSV and hRSV, as suggested by differences in the variability of the central hydrophobic region of the G protein of these viruses. 65
bRSV Diagnosis, Experimental Infection, and Pathology
Clinical Signs and Diagnosis of Field Cases
The incubation time for bRSV is estimated to be 2 to 5 days. Infection can be subclinical and involve the upper respiratory tract or both the upper and lower regions of the respiratory tree. 87 Clinical signs of bRSV infections can range from minimal to severe with dyspnea and death. Affected calves can have tachypnea, ocular serous secretions, dry muzzle, reduced activity, anorexia, and fevers up to 40°C. With severe infections, dyspnea can be pronounced and marked. These signs coincide with bRSV infection, during which viral replication is detectable beginning at 2 to 3 days postinfection and continues until 7 to 10 days postinfection. There has been a biphasic pattern of clinical signs noted following some BRSV infections. 10 However, the mechanistic basis underlying this phenomenon is not fully understood. In grouped calves, individual animals can become infected, but there can be spread involving multiple calves. With subclinical infections, animals may have some loss of feed intake and, thereby, a subtle reduction in weight gain and activity. With severe infections, however, anorexia and reduced water consumption can result in weight loss and dehydration in just a few days. Those animals that do survive severe infection lag to varying degrees in weight gain and growth. They are also susceptible to secondary bacterial infections with pathogens such as M. haemolytica, P. multocida, H. somni, and M. bovis that further alter growth.
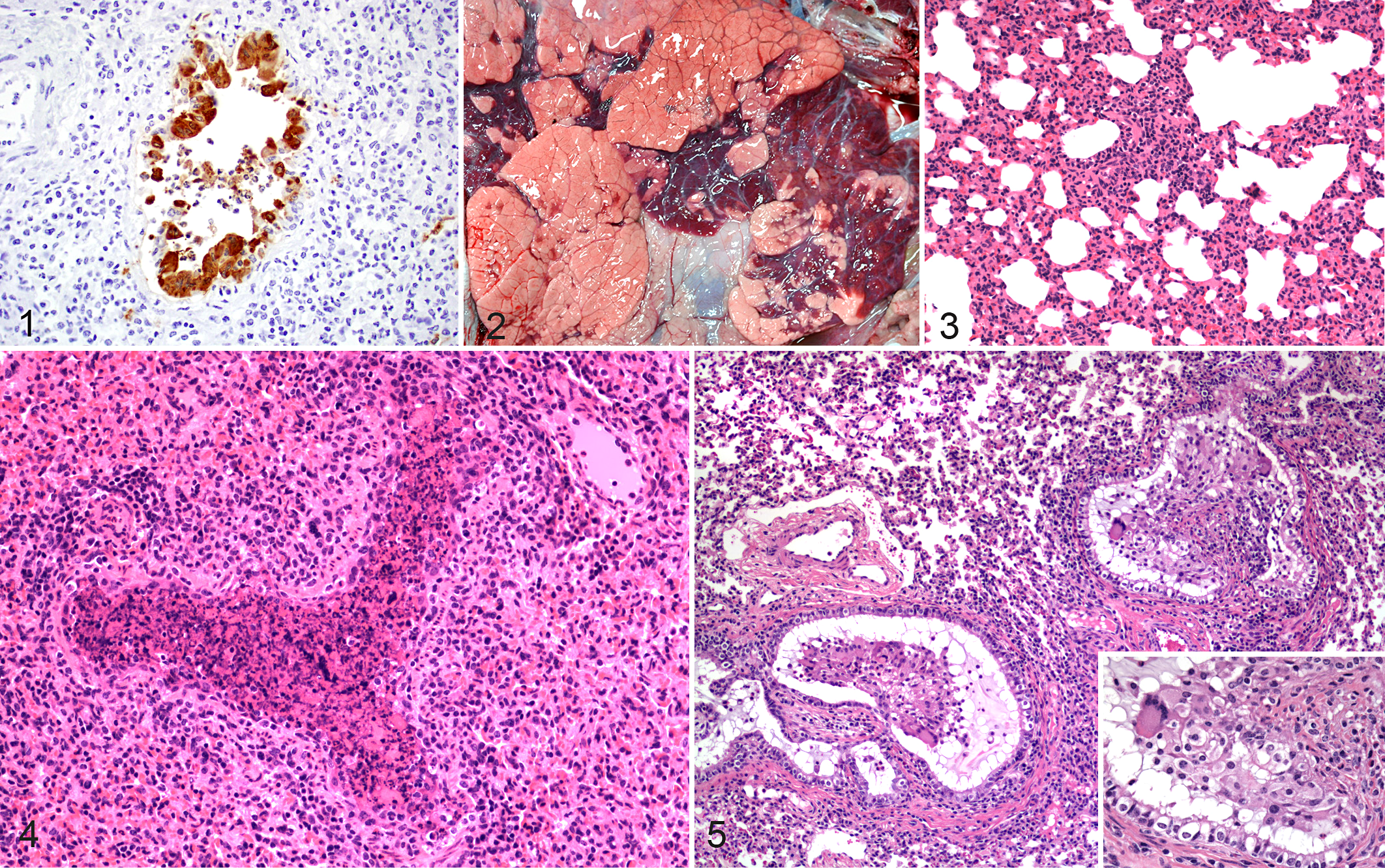
Immunofluorescence stains have been used to identify bRSV antigens in frozen lung samples; however, cellular and structural resolution is difficult with fluorescent images, and tissues can have endogenous (background) fluorescence. In many laboratories, the virus is detected with immunohistochemistry (IHC) of lung tissue fixed in formalin and processed (Fig. 1). IHC sections allow excellent assessment of cellular structures and lesions compared to immunofluorescence, although in practical terms, many samples submitted to diagnostic laboratories have some degree of autolysis due to the time that it takes for samples to be collected from cattle in the field or in feedlots and for immersion into formalin. Autolysis can affect fluorescence and IHC staining and assessment of histopathologic lesions.
Additional diagnostic tests include polymerase chain reaction (PCR), to identify the simple presence of viral genome, or real-time reverse transcription PCR (RT-PCR), to determine the relative level of bRSV mRNA of either F protein or nucleoprotein. A commercial kit detects all bRSV strains assessed with 99.3% efficiency and a detection limit of 0.1 TCID50 (50% tissue culture infective dose). 83 This kit had much higher sensitivity than that of fluorescent antibody assays. A 1-step multiplex real-time RT-PCR assay for bRSV, bovine herpesvirus 1, and bovine parainfluenza virus 3 was also found to be more sensitive than immunofluorescence (and virus isolation). 81 Intranasal vaccination (Rispoval RS with PI3 IntraNasal, Pfizer) can result in bRSV detection by an RT-PCR kit (Taqvet BRSV, Laboratoire Service International) for beyond 14 days in some calves. 82
Viral isolation and titers can be determined from lung samples as well. Unfortunately, bRSV is somewhat labile and can be difficult to isolate and titrate from tissue samples. This is especially true for samples from the field or tissues that are packaged and shipped to diagnostic laboratories. A 1-step enzyme-linked immunosorbent assay has been developed recently and appears to have advantages in terms of sensitivity in comparison to an antigen-capture enzyme immunoassay. A strip test designed for detection of human strains of RSV can detect bRSV in experimentally infected calves. 76
At the diagnostic and pathology laboratories at Iowa State University, bRSV cases are routinely assessed by postmortem examination, histopathology, IHC, RT-PCR, and, less frequently, viral isolation. In 2012, the diagnostic laboratory had 45 bovine respiratory disease cases diagnosed with bRSV. Cases originated from not only Iowa but also Nebraska, Illinois, Minnesota, Ohio, and Virginia, and the average age of affected calves was 6 to 7 months. Peak numbers of cases were seen in the fall, primarily in November and December. Concurrent infections with infectious bovine rhinotracheitis, bovine viral diarrhea virus, P. multocida, M. haemolytica, M. bovis, H. somni, Arcanobacterium pyogenes and Bibersteinia trehalosi were diagnosed in many of these cases. Although bacterial pathogens were not always isolated, the majority of cases had microscopic evidence of concurrent bacterial bronchopneumonia. The routine postmortem tissue work up in the diagnostic laboratory includes gross and histopathologic evaluations, routine bacterial culture of lung, IHC staining of lung sections, and a multiplex RT-PCR assay which detects multiple bovine viral respiratory pathogens and was developed at Iowa State University. 35 In the early stages of clinical disease, tissue from lesions typical of bRSV in the cranioventral lung fields is frequently the best sample to use for virus detection. 10,48
Experimental Infection
Clinical signs of respiratory disease observed following experimental infection of calves with bRSV, while similar to those seen in field cases, have been of varying degrees of severity. Plausible reasons for the variable results between studies include differing routes of inoculation, dose of inoculum, number of times that inoculum was administered, age of calves, or complicating factors such as secondary bacterial infection. In comparing a sample of experimental infection studies, routes of inoculation have included intratracheal, intranasal, aerosol, or a combination of challenge routes, given once or multiple times on consecutive days. 30,49,69,94,96 In addition, doses of inocula utilized in these studies have ranged from 102 to 105 tissue culture infective doses and, in others, up to 105 plaque-forming units. Experimentally infected calves have been shown to have an elevated temperature beginning at day 2 that persists until approximately day 7 postinfection. Additionally, increased respiration rates, nasal discharge, and coughing have been observed for several days following infection.
Gross and Microscopic Lesions
Lesions observed in field cases or following experimental infection with bRSV are of a similar nature. In the lungs of affected animals, gross lesions of bRSV infection have a cranioventral distribution and are characterized by atelectatic, collapsed, deep red-purple lesions that are rubbery upon palpation. Consolidated lesions can be present throughout the cranial, middle, and accessory lobes or lobular in distribution with areas of consolidation distributed multifocally and surrounded by lobules of pink, overinflated lung (Fig. 2). 68 The nasal meatus, trachea, bronchi, and bronchioles can contain foamy to mucopurulent discharge. In contrast, caudodorsal lobes are often distended with interlobular, lobular, or subpleural emphysematous and edematous lesions. Caudodorsal lung regions may fail to collapse and, due to emphysema, may be pale. The demarcation between cranioventral consolidation and caudodorsal edematous and emphysematous changes can be well delineated. Variations in gross lesions may occur with a generalized red discoloration to all lung lobes and rubbery texture in both cranioventral and caudodorsal regions. Animals that exhibit severe respiratory distress may present with emphysematous lesions in interlobular or subpleural locations, resulting in pneumothorax, pneumomediastinum, or pneumopericardium. 8,44 In some cases, subcutaneous emphysematous bullae may develop along the shoulders, back, neck, or perineum. 8,44 Pulmonary lymph nodes (ie, mediastinal and tracheobronchial) may be enlarged, edematous, or even hemorrhagic.
In cranioventral lobes, microscopic lesions are those of a bronchointerstitial pneumonia, necrotizing bronchiolitis, syncytia formation, type II pneumocyte hyperplasia, and exudative or proliferative alveolitis (Figs. 3, 4). 10 Alveolar lumens often contain seroproteinaceous fluid, fibrin casts, cell debris, few alveolar macrophages, and occasional neutrophils. Nearby alveoli can contain air and be modestly dilated due to hyperinflation. The most consistent feature, necrotizing bronchiolitis, is characterized by degenerative or necrotic ciliated and nonciliated bronchiolar epithelium. Epithelial cell necrosis leads to ulcerated foci covered by necrotic cell debris with nearby degenerate cells exhibiting rounded cell borders. Erosion and ulceration of the epithelial layer may also lead to attenuation of epithelial cells as they extend over exposed basement membrane. This results in bronchioles lined by flattened cells that sometimes have a squamous morphology. IHC may reveal viral antigen in bronchiolar epithelial cells. 23 Multinucleated syncytial cells can often be found projecting from the bronchiolar wall or free in the lumen. Syncytial cells may also be found in alveoli. Uncommonly, eosinophilic intracytoplasmic viral inclusions may be seen in syncytial cells as well as mononuclear cells or epithelia. Neutrophilic exudates may be present within bronchi, bronchioles, or alveoli. Lobular to diffuse interstitial thickening of alveolar septae is seen and is the result of alveolar capillary congestion, infiltration of inflammatory cells (ie, lymphocytes, macrophages), and type II pneumocyte hyperplasia. Interlobular septae are often widened or expanded by edema and dilation of lymphatic vessels. Lesion severity does not often correlate with viral load or viral distribution, suggesting that much of the tissue damage is the result of host response rather than primary viral infection. 23 Tracheobronchial lymph nodes of affected cattle are often characterized by enlarged cortical areas with prominent follicles and expanded parafollicular areas and medullary cords due to lymphocytic hyperplasia. Medullary sinuses often contain macrophages, with variable numbers of lymphocytes, plasma cells, and hemosiderin-laden macrophages and occasional neutrophils and eosinophils. With secondary bacterial infections or bovine respiratory disease complex, tracheobronchial lymph nodes can have a variety of additional histologic features, ranging from suppurative to lymphocytic and granulomatous.
As the infection progresses, an attempt to repair necrotic airways may result in epithelial hyperplasia and bronchiolitis obliterans, also known as bronchiolitis fibrosa obliterans, obliterative bronchiolitis, or organizing bronchiolitis (Fig. 5). 10 Bronchiolitis obliterans can result after damage to the bronchiolar epithelium and has been characterized as “wound healing gone awry.” 10 Persistent inflammation with fibrinous exudates results in attempts to heal through fibroblast infiltration and neovascularization. 10 In as little as 10 days after infection, the result may be fibrous polyps, covered by respiratory epithelium, extending into the airway lumen, and resulting in permanent decreases in airflow and alveolar ventilation. Bronchiolitis obliterans can be induced by various agents (eg, viral, bacterial, parasitic, toxic) and is not specific for bRSV. Ultrastructurally, changes in ciliated and nonciliated epithelial cells, as well as type I and type II pneumocytes, can range from acute cell swelling to necrosis. 44 Intercellular areas can be expanded, and epithelial cells may lose intercellular junctions.
Immune Responses to RSV
Innate Immunity
The initiation of an innate immune response is dependent on the recognition and binding of evolutionarily conserved pathogen-associated molecular patterns (PAMPs) to pattern recognition receptors (PRRs). Recognition of viral PAMPs involves at least 3 distinct classes of PRRs: toll-like receptors (TLRs), retinoic acid–inducible gene I–like receptors, and nucleotide-binding oligomerization domain–like receptors. Recognition of RSV infection involves several TLRs, including TLR2, 3, 4, 7, and 8. 4,98 TLR3, which recognizes double-stranded RNA, and TLR7, which recognizes single-stranded RNA, are endosomal receptors that are key for the innate recognition of RNA viruses. It has been shown that RSV stimulates airway epithelial cells via signaling through TLR3 and retinoic acid–inducible gene I–like receptors, which have been linked to distinct pathways controlling NF-κB activation. 50 The F protein of bRSV has been reported to bind to TLR4 and, in concert with MD2 and CD14, also mediate NF-κB activation. 51
Recognition of viral infection through PRRs results in production of type I IFN and induction of IFN-stimulated genes, including MxA protein, protein kinase R, ISG15, and 2′-5′ oligoadenylate synthetase. Paramyxoviruses are known to utilize different mechanisms to affect IFN signaling. hRSV and bRSV have evolved strategies to inhibit the IFN-induced cellular response that are dependent on NS proteins. As is characteristic of pneumoviruses, these viruses have 2 genes that encode for NS proteins. It has been shown that NS1 and NS2 proteins cooperatively mediate resistance of bRSV and hRSV to IFN-stimulated responses in a species-specific manner. 6,71 In fact, the precise mechanism whereby type I IFN responses are altered varies between these viruses. In the case of bRSV, NS proteins block phosphorylation and activation of IRF3. 7 By comparison, hRSV NS1 and NS2 modulation of type I IFN responsiveness involves inhibition of Stat2 expression. 52
In addition to induction of the IFN response, ligation of PRRs by viral PAMPs stimulates the early release of inflammatory chemokines and cytokines that are important in the initiation of adaptive immunity. In particular, activation of antigen-presenting cells such as dendritic cells and macrophages is critical for this function. Stimulation of bovine dendritic cells with bRSV in vitro results in increased expression of transcripts for the chemokines RANTES, MIP-1α, MIP2α, MIP3α, and MCP-2 and the cytokine receptor CCR3. 92 Our laboratory has demonstrated that bRSV infection also induces inflammatory cytokine production by alveolar macrophages. Interestingly, the kinetics of induction following bRSV stimulation differs between cytokines. For example, we found peak induction of IL-1β and IL-12p40 mRNA in alveolar macrophages occurs on day 3 postinfection in the neonatal ruminant RSV model, whereas IL-6 mRNA was higher at day 5 than day 3 of infection. 18
In addition to antigen-presenting cells, another cell type thought to be important in bridging the innate and adaptive immune response is the γδ T cell. Recently, our laboratory demonstrated that stimulation of bovine γδ T cells with TLR3 or TLR7 agonists or with in vitro or in vivo bRSV infection results in robust production of message and protein for the chemokines MIP-1α, GM-CSF, and MCP-1. 55 Bovine γδ T cells can express the workshop cluster 1 (WC1) receptor and can be further divided serologically into WC1.1+ and WC1.2+ subsets. Production of proinflammatory mediators following bRSV stimulation was primarily restricted to WC1.1 and WC1neg γδ T-cell subsets, with the WC1.2+ γδ T-cell subset producing regulatory cytokines, including IL-10 and TGF-β, suggesting a unique functional difference amongst bovine γδ T-cell populations following bRSV infection.
Recently, our laboratory has shown a significant upregulation of the innate chemokine IL-8 in areas of lungs with lesions of RSV-infected calves on day 7 postinfection. 69 While the source of IL-8 in our model is not clear, Gershwin has suggested that bRSV-infected bovine respiratory/turbinate epithelial cells upregulate transcripts for the proinflammatory cytokines IL-6 and IL-8. 24 Interestingly, our results fit well with data showing that IL-8 is elevated in the respiratory tract of children with RSV bronchiolitis. Rodents lack a bona fide homologue of IL-8, although mice have what are considered to be functional homologues, CXCL1 (GRO/KC), CXCL2 (MIP-2), and CXCL5-6 (LIX), which belong to the same major cluster of chemokines. Thus, it is important to note that the calf can prove useful as an in vivo model for future exploration into pathways that specifically regulate innate immune responses during RSV infection.
Adaptive Immune Responses
The development of an adaptive immune response is required for the control and clearance of established RSV infections. Following infection, cattle mount virus-specific antibody and T-cell responses; however, these responses are weak and transient, as animals can be continuously reinfected throughout life. It is clear from work in mice and humans that CD4 and CD8 αβ T cells mediate elimination of a primary RSV infection. T-cell responses are directed at epitopes within several RSV proteins, including the N, M, NS2, M2-1, F, and G. 58 The F and G proteins are the major histocompatibility antigen class II–restricted targets in humans and cattle, 9,58 with the F protein of hRSV being the most thoroughly studied and described to contain multiple antigenic regions. 88 To our knowledge, there are currently no specific bovine leukocyte antigen class II epitopes defined for bRSV in the bovine.
RSV infection in humans induces a mixed Th1- and Th2-type cytokine response. 62 Production of IL-12 by dendritic cells and early IFNγ are required for the priming of an effective Th1-type cytokine response; however, hRSV has been shown to interfere with dendritic cell cytokine production and its ability to initiate the development of a Th1 response. 3,9,31 The ensuing Th2 polarized response leads to increased disease severity and lung injury and is thought to block the development of an effective CD8 T-cell response during primary and secondary challenge. 5,9,17,36
Like humans, calves infected with bRSV develop a mixed cytokine response but favor the development of a Th2-type immune response following infection. Studies of the cells and lymph fluid from bRSV-infected calves reveal enhanced IL-4 and IL-13 production in the serum and tissues as early as day 4 postinfection and increased serum levels of virus-specific IgE, indicating the establishment of a Th2-type response. 59,77,78 Calves develop IFNγ-producing cells and levels of the cytokine increase in the serum, but neither cell numbers nor IFNγ levels correlate with positive disease outcome. 26 Evidence from humans has suggested that due to the Th2 nature of the antiviral immune response, hRSV infection may predispose children to the development of allergies and asthma later in life. 73,74 Interestingly, bRSV infection in calves has also been shown to predispose to allergic sensitization. 25,27 Gershwin et al demonstrated that exposure to the model allergen ovalbumin during bRSV infection resulted in significantly increased levels of IL-4, IL-13, and ovalbumin-specific IgE compared to uninfected control calves. 25
Cytotoxic CD8 T cells play a critical role in the control and clearance of RSV infection. Bovine CD8 T cells target the M2, F, N, and G proteins of bRSV. 20,79 Calves infected with bRSV exhibit increased CD8 T-cell infiltration in the lungs, trachea, and nasopharynx, 56 with cytotoxic T-cell activity peaking between 7 and 10 days postinfection. 97 As evidence of their importance during infection, depletion of CD8 T cells from bRSV-infected calves results in more severe disease and increased and sustained viral shedding compared to nondepleted control animals. 80 Although bRSV elicits a cytotoxic T-cell response, it is weak and transient. 21,97 Anecdotally, this is evidenced by the recurring infections that occur commonly in humans and calves. 29,89 Calves infected with bRSV exhibit limited bRSV-specific cytotoxicity during primary infections and impaired memory responses following challenge or vaccination. 21,97 This may be due to the strong Th2 skewing that occurs during RSV infection, which acts to inhibit the development of an efficient CD8 T-cell response and prevent the establishment of long-lived memory. 9
Humoral immunity plays an important role in defending the host from RSV infection. While not fully effective, maternal antibodies may provide some level of protection from severe bRSV infection; 46 however, their presence has also been described to suppress the development of antibody and T-cell responses during acute infection. 14,45 Calves mount antibody responses to several bRSV antigens, but the primary targets for protective humoral responses are the F, G, and NP proteins. 79 bRSV-specific IgM and IgA can be detected in the nasal secretions and serum of bRSV-infected calves as early as 8 days postinfection. 45 BRSV-specific IgG2 is not detected in the serum until 1 to 3 months postinfection, while virus-specific IgG1 is detectable starting at 13 days postinfection. 45 In a separate study, virus-specific IgE, another antibody associated with Th2 skewing and airway hyperresponsiveness, was detectable in the serum concurrent with the development of clinical signs. 77,78
BRSV Vaccines
There are multiple vaccines currently marketed for bRSV (killed and modified live) that are generally provided as part of multivalent products. In the 1960s, a formalin-inactivated RSV vaccine was developed that not only failed to protect against hRSV disease but also induced an exaggerated clinical response to RSV infection in infants who were RSV näive before vaccination, and 2 infants died. 11,41,42 Similar vaccine-enhanced disease has been reported from 2 cases of natural bRSV infections in calves. 43,72 In the former case, an outbreak of respiratory tract disease among 5- to 7-month-old calves on a beef-fattening farm in the Netherlands started 2 days after administration of a modified-live bRSV vaccine. 43 The disease was severe among vaccinates but absent in nonvaccinated calves 8 months of age or older. In the latter case, 30% of 8-month-old Belgian blue calves vaccinated with a β-propiolactone inactivated bRSV vaccine died during a naturally occurring bRSV outbreak. Interestingly, no deaths were recorded among younger calves not vaccinated. 72 Experimentally, vaccine-enhanced disease has been reproduced in some studies of calves vaccinated with formalin-inactivated bRSV preparations 2,28 but not in others. 60,93 Where bRSV vaccine-enhanced disease has been observed, there is a bias toward a Th2-like response, characterized by a reduced CD8 T-cell response and decreased production of IFNγ compared to unvaccinated bRSV-infected animals. 97 Others also observed increases in serum IgE 2,40 and eosinophils in lavage fluid, 2 similar to that observed in children with vaccine-enhanced disease.
Although there is widespread use of bRSV vaccines in calves, their efficacy is controversial, and given the significant disease burden associated with bRSV infection, there is a definite need for improved technologies. BRSV and its target population pose several obstacles with respect to vaccine development, particularly the need to vaccinate calves with immature immune systems, induce a response in the face of maternal antibodies, and effect an appropriate, robust, and long-lasting immune response.
Researchers are currently studying the use of subunit-based vaccines and live attenuated bRSV. 58 One promising example reported by Valarcher et al was the success of 2 bRSV strains, one devoid of NS1 and the other lacking NS2. 85 Calves vaccinated with either deletion mutant exhibited a robust virus-specific antibody and CD4 T-cell response and were protected against virus challenge. 85 Interestingly, the NS2 mutant was more effective than its counterpart. Another group used a subunit approach by immunizing calves with nanoparticles encapsulating the N protein of hRSV. In this study, investigators observed N-specific antibodies and cellular responses, as well as reduced viral shedding and lung pathology in vaccinated calves. Furthermore, the calves did not exhibit vaccine-enhanced disease. 66
An additional area being extensively pursued against bRSV is the use of new adjuvants coupled with inactivated bRSV or subunit vaccines. Among those showing promise is the use of CpG containing oligodeoxynucleotides 53 and immunostimulating complexes, 32,33 both of which induce a robust Th1 skewing. Immunostimulating complexes are multimers composed of cholesterol, phospholipids, proteins, and Quillaja saponins. Recent studies by Hagglund et al have described the ability bRSV immunostimulating complexes to successfully induce bRSV-specific cellular and humoral responses and protect from virulent bRSV challenge in neonatal calves aged 3 to 8 weeks. Interestingly, this protection was robust despite the presence of significant levels of maternally derived antibodies. 32,33 Further information on novel approaches to bRSV vaccine development have been recently reviewed. 58
Summary
RSV infection in cattle continues to be a problematic disease resulting in significant economic losses to the industry due to decreased productivity coupled with increased costs for treatment. RSV is well recognized as a primary pathogen causing serious respiratory disease that predisposes cattle to secondary infection. hRSV and bRSV are closely related viruses with similarities in histopathologic lesions and mechanisms of immune modulation induced following infection. The innate and adaptive responses to RSV in calves are becoming increasingly understood; however, additional mechanistic insight is needed to better identify prophylactic and therapeutic compounds and vaccine targets.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
