Abstract
Mammary neoplasms are the most common neoplasm in female dogs. Two histologic classification systems for canine mammary tumors and dysplasias have been published: the first in 1974 and a modification in 1999. This article provides a brief overview of the two histologic classification systems. Since the publication of the second system, several new histologic subtypes of canine mammary neoplasms have been described. These have been incorporated into the proposed new classification system. This article also compares the grading systems for canine mammary carcinomas and their use for prognosis, along with the histologic classification.
In 1974, the World Health Organization published the first “International Histological Classification of Tumours of Domestic Animals,” which included tumors and dysplasia of the mammary gland. This classification system was based on three seminal papers on canine malignant mammary tumors published in Veterinary Pathology. 10 –12 These publications included a glossary based on descriptive morphology, which formed the basis for the classification of canine malignant mammary tumors. 11,12 Before this, several classifications of canine mammary tumors had been proposed. 14,15
Classification of Canine Mammary Neoplasms and Dysplasias
Table 1 shows a comparison of the original 7 and more recent 13 classification of canine mammary tumors. Given the previous two histologic classifications, we propose the following histologic classification and nomenclature of neoplasms and dysplasias of the canine mammary gland. Some neoplasms that are incorporated into the new classification system are included as specific entities. These include the cribriform type of simple carcinoma and comedocarcinoma, both of which were described under carcinoma–in situ. 13 Others have been identified as malignant variants of previously described benign neoplasms and include the ductal carcinoma, the malignant counterpart of the ductal adenoma but previously classified as a basaloid adenoma, 13 and the intraductal papillary carcinoma, the malignant counterpart of the intraductal papillary adenoma but previously classified as a duct papilloma. 13 Micropapillary carcinomas, a recently described entity, has been incorporated into the classification. 5 Other new entities, such as the carcinoma and malignant myoepithelioma, have been identified using immunohistochemistry markers for epithelial cells (CK 8, 18, 19, and CK 7) and basal/myoepithelial cells (CK 5, 6, 14, 17, smooth muscle actin, calponin, vimentin, p63). 18
Histologic Classifications: 1974 and 1999

Proposed Histologic Classification: 2010
1: Malignant Epithelial Neoplasms
Carcinoma–in situ
Carcinoma–simple Tubular Tubulopapillary Cystic-papillary Cribriform
Carcinoma–micropapillary invasive
Carcinoma–solid
Comedocarcinoma
Carcinoma–anaplastic
Carcinoma arising in a complex adenoma/mixed tumor
—The benign counterpart is still detectable in the section.
Carcinoma–complex type
—The epithelial component is malignant, and the myoepithelium is benign.
Carcinoma and malignant myoepithelioma
—The epithelial and myoepithelial components are malignant.
Carcinoma–mixed type
—The epithelial component is malignant; the myoepithelial mesenchymal component is benign; and the mesenchymal component is cartilage or bone.
Ductal carcinoma–malignant counterpart of ductal adenoma
Intraductal papillary carcinoma–malignant counterpart of intraductal papillary adenoma
2: Malignant Epithelial Neoplasms—Special Types
Squamous cell carcinoma
Adenosquamous carcinoma
Mucinous carcinoma
Lipid-rich (secretory) carcinoma
Spindle cell carcinomas
Malignant myoepithelioma
Squamous cell carcinoma–spindle cell variant
Carcinoma–spindle cell variant
Inflammatory carcinoma (see Inflammatory Carcinoma section)
3: Malignant Mesenchymal Neoplasms—Sarcomas
Osteosarcoma
Chondrosarcoma
Fibrosarcoma
Hemangiosarcoma
Other sarcomas
4: Carcinosarcoma—Malignant Mixed Mammary Tumor
5: Benign Neoplasms
Adenoma–simple
Intraductal papillary adenoma (duct papilloma 9 )
Ductal adenoma (basaloid adenoma 9 )
With squamous differentiation (keratohyaline granules)
Fibroadenoma
Myoepithelioma
Complex adenoma (adenomyoepithelioma)
Benign mixed tumor
6: Hyperplasia/Dysplasia
Duct ectasia
Lobular hyperplasia (adenosis)
Regular
With secretory activity (lactational)
With fibrosis–interlobular fibrous connective tissue
With atypia
Epitheliosis
Papillomatosis
Fibroadenomatous change
Gynecomastia
7: Neoplasms of the Nipple
Adenoma
Carcinoma
Carcinoma with epidermal infiltration (Paget-like disease)
8: Hyperplasia/Dysplasia of the Nipple
Melanosis of the skin of the nipple
Criteria of Malignancy
A major problem in evaluating canine mammary neoplasms is identifying those neoplasms that are “truly” malignant. The presence of some cells, with enlarged nuclei and prominent nucleoli, often leads to the overdiagnosis of mammary carcinoma. The following are the most significant criteria for the diagnosis of malignant mammary tumors in the dog based on hematoxylin and eosin–stained sections: tumor type, significant nuclear and cellular pleomorphism, mitotic index, presence of randomly distributed areas of necrosis within the neoplasm, peritumoral and lymphatic invasion, and regional lymph node metastasis.
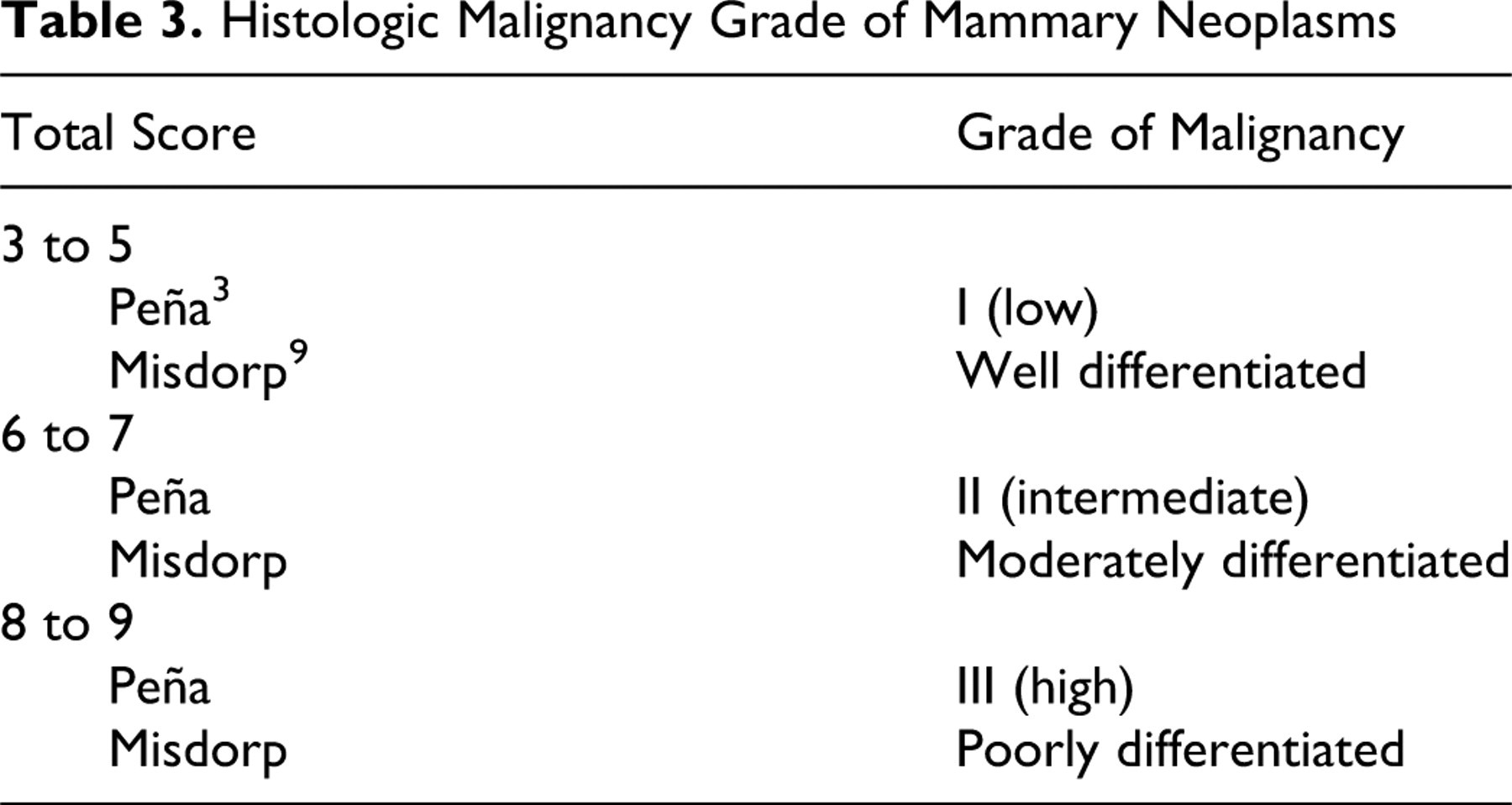
Tables 2 and 3
elaborate on some of these features.
Criteria for Histologic Malignant Grade
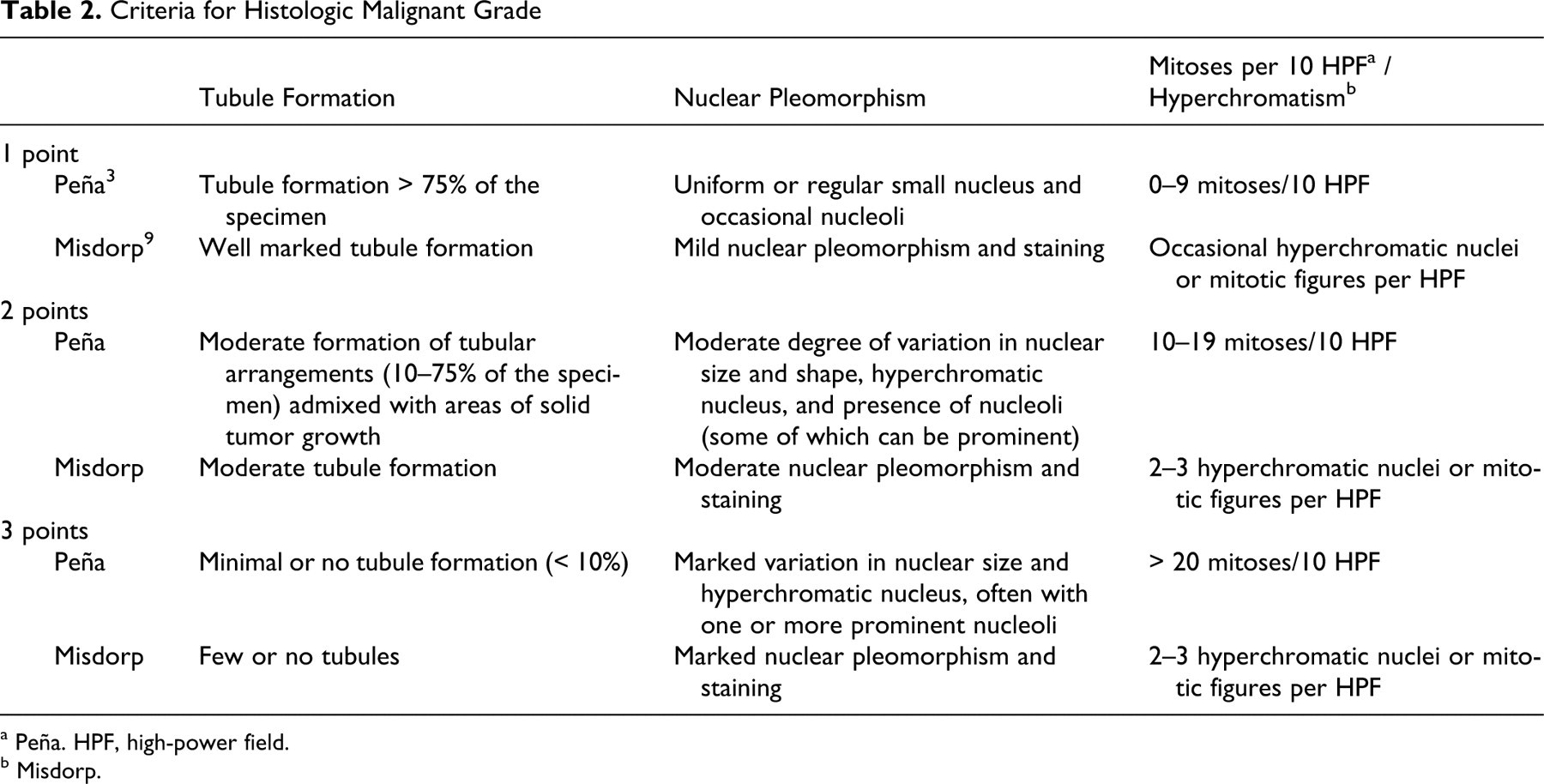
a Peña. HPF, high-power field.
b Misdorp.
Histologic Malignancy Grade of Mammary Neoplasms
Descriptions
The following are descriptions pertaining to the above classification.
Carcinoma–In Situ
In situ carcinoma is a neoplastic tissue that consists of well-demarcated nodules that have not extended through the basement membrane into the surrounding mammary tissue, that are densely cellular, and that consist of closely packed cells arranged in irregular tubules (Fig. 1 ). 1,16 The cells vary from polygonal to round to cuboidal, and they may have a scant amount of eosinophilic cytoplasm and a high nuclear:cytoplasmic ratio. Nuclei may be hyperchromatic, central, round to oval with clumped chromatin and a single central basophilic nucleolus, or they may be vesicular with a small nucleolus. Anisokaryosis and anisocytosis are moderate, and mitoses are found but vary in number. As is commonly found with carcinoma–in situ in other organs, there is loss of cellular and nuclear polarity and loss of the normal architecture, with layering of cells within the ducts. There are often areas of lobular hyperplasia within the mammary tissue adjacent to the areas of neoplasia.
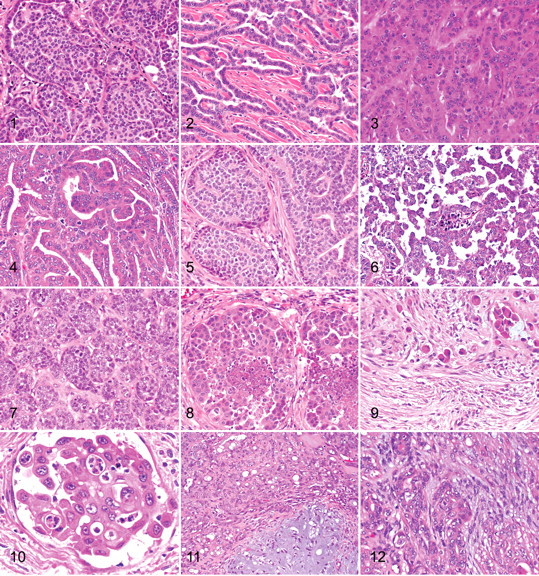
It may be difficult to differentiate among in situ carcinoma, lobular hyperplasia with severe atypia, and atypical epitheliosis in the canine mammary gland, but this is analogous to the situation in breast pathology in women. 13,20 In some cases, carcinoma in situ may represent areas of intraductal neoplastic cell proliferation from a carcinoma elsewhere in the mammary gland. 13
Carcinoma–Simple Types
A simple carcinoma is composed of only one cell type, resembling either luminal epithelial cells or myoepithelial cells. 11,12
Carcinoma–Tubular
Tubular carcinoma is one in which the cells are predominantly arranged in a tubular fashion (Figs. 2, 3). 11,12 Tubular carcinomas are common mammary carcinomas in the dog. The lining of the tubules is usually 1 to 2 cells thick, and cells are variable in their morphology. Nuclei may be hypochromic, normochromic, or hyperchromic. Nucleoli may be single and very large or multiple and small. Cells often have eosinophilic cytoplasm, and cell margins are relatively distinct. Mitotic activity is also variable. Tubule formation, cell morphology, and mitotic rate are the features that form the basis for the grading of mammary carcinomas (Table 2 and 3). The intertubular stroma consists of vessels and fibroblasts, and there may be an infiltrate by plasma cells, lymphocytes, and macrophages. When the neoplastic cells infiltrate into the surrounding mammary tissue, they can evoke a stromal response, including extensive myofibroblast proliferation. The infiltrative nature of the neoplasm in conjunction with anisokaryosis and increased mitotic activity is a feature that differentiates adenomas from carcinoma–in situ and carcinomas.
Carcinoma–Tubulopapillary
Tubulopapillary carcinoma is one in which the neoplastic tubules are predominantly arranged in a sessile or pedunculated papillary fashion (Fig. 4). 11,12 The tubulopapillary type of mammary carcinoma differs from the tubular type by having papillae that extend into tubular lumina. The papillae are supported by a fine fibrovascular connective tissue stroma. The remaining features are as described under carcinoma–tubular.
Carcinoma–Cystic-Papillary
The cystic-papillary type of mammary carcinoma differs from the tubular type in that there are papillae extending into marked dilated and cystic tubular lumina. The papillae are supported by a fine fibrovascular connective tissue stroma, and the ectatic tubular lumina may contain an eosinophilic, often finely flocculent material admixed with degenerating granulocytes and foamy macrophages. The remaining features are as described under carcinoma–tubular.
Carcinoma–Cribriform
Cribriform carcinoma, which is uncommon, is characterized by the proliferation of a population of neoplastic epithelial cells forming a sievelike arrangement (Fig. 5). The lumina formed are very small and often round, and they are surrounded by bridges of neoplastic cell. The surrounding interstitial connective tissue is sparse. Neoplastic cells vary from columnar to polygonal and often have scant homogeneous eosinophilic cytoplasm. Anisokaryosis and anisocytosis are moderate, and the number of mitoses is variable.
Carcinoma–Micropapillary Invasive
Micropapillary invasive carcinoma is a type of mammary neoplasm that has been recently described in dogs (Fig. 6). 5 Within the mammary tissue are one or more nodules characterized by an intraductal neoplastic population forming small intraluminal irregular aggregates and small papillae that do not have a supporting fibrovascular stalk and are surrounded by empty lacunar spaces. The neoplastic cells grow along ductal walls and infiltrate the periductal collagenous stroma. Cells are pleomorphic and cuboidal to polygonal and have scant eosinophilic cytoplasm. The nuclear:cytoplasmic ratio is high, and nuclei are central and oval, with a large central nucleolus and hyperchromatic granular chromatin. Anisokaryosis and anisocytosis are moderate to severe, and the mitotic index is high. The infiltrating micropapillary areas may be seen in association with tubulopapillary or solid areas. 5 This tumor frequently shows vascular invasion and has a high metastatic potential.
Carcinoma–Solid
Solid carcinoma is one in which the cells are predominantly arranged in solid sheets, cords, or masses, without lumina (Fig. 7). 11,12 This neoplasm is seen less frequently than the tubular type of mammary carcinoma, and it is composed of closely packed cells to form dense, irregularly sized lobules supported by a fine fibrovascular stroma. The cells are polygonal to oval, and they often have poorly demarcated cell margins and scant cytoplasm that may be lightly eosinophilic to basophilic. Nuclei are oval and often hyperchromatic with coarsely stippled chromatin and a single central basophilic nucleolus. Anisokaryosis and anisocytosis are moderate to severe, and the number of mitoses is variable. Infiltration of neoplastic cells into lymphatic vessels can be found at the periphery of the neoplasm, with metastasis to regional lymph nodes.
Comedocarcinoma
Comedocarcinoma is a neoplasm characterized by the presence of necrotic areas within the center of the neoplastic cell aggregates (Fig. 8). In areas of necrosis, there is abundant amorphous eosinophilic material admixed with cell debris, necrotic neutrophils, and macrophages. The necrosis is thought to be a combination of apoptosis and karyorrhexis. The peripheral tissue of the affected area and any foci not showing necrosis consist of aggregates of closely packed cells arranged in solid foci, nests, cords, or tubules, supported by a fine fibrovascular connective tissue stroma. The neoplastic cells and their arrangement and morphology have characteristics as described above, although the majority of cases show no tubule formation (ie, they are solid carcinomas). Infiltration of neoplastic cells into lymphatic vessels can be found at the periphery of the neoplasm, with metastasis to regional lymph nodes.
Carcinoma–Anaplastic
Anaplastic carcinoma is the most malignant of the mammary carcinomas, often showing diffuse invasion of interlobular connective tissue and lymphatic vessels by the neoplastic cells (see inflammatory carcinoma) (Figs. 9, 10). The origin of the neoplasm within the mammary tissue is often difficult to find because no neoplastic nodules may be identified within the tissue submitted for histopathology.
Neoplastic cells are often individualized or grouped in small nests; they are also round, oval, or polygonal and 15 to 70 μm in diameter, with moderate to abundant eosinophilic cytoplasm. Nuclei are round to oval, occasionally indented or convoluted with coarsely stippled chromatin. Multiple variably sized nucleoli are frequently present (usually, 2 or 3). Anisokaryosis and anisocytosis are severe, and mitoses are common. Multinucleated cells containing several irregularly sized nuclei are found in some cases. The invading carcinoma cells often evoke a marked desmoplastic host response with myofibroblast proliferation, which may be accompanied by an infiltrate of lymphocytes, plasma cells, mast cells, occasional neutrophils or eosinophils, and macrophages. The interstitial tissue is often edematous, with numerous ectatic lymphatics. Neoplastic cells, either as single cells or in aggregates, are often present within lymphatic vessels, with metastasis to regional lymph nodes and, subsequently, the lung. Radiographic examination of the lungs shows an interstitial pattern rather than a nodular pattern.
Carcinoma Arising in a Complex Adenoma or Benign Mixed Tumor
The benign counterpart of this neoplasm, either a complex adenoma or a mixed tumor, is still detectable in the section (Fig. 11). However, multifocally with the section there are areas of much more pleomorphic epithelial cells with increased numbers of mitoses (foci of carcinoma cells). This marked increase in nuclear and cellular pleomorphism, when compared to the preexisting benign epithelial component, should be a feature required to make this diagnosis.
Carcinoma–Complex Type
Complex-type carcinoma has a malignant epithelial component and a benign myoepithelial component (Fig. 12). 11,12 The neoplasm is characterized by the presence of two cell populations supported by a fibrovascular stroma. The first population of epithelial cells is arranged in irregular tubules lined by a single layer to several layers of cuboidal to columnar cells with a scant to moderate amount of eosinophilic cytoplasm. These cells exhibit moderate to marked anisokaryosis and anisocytosis, and variable numbers of mitoses are present. Necrosis of these epithelial cells may be focal or multifocal, and occasional foci of squamous differentiation may be present. The second population is composed of spindle-shaped cells (myoepithelial cells) within the interstitium, arranged in irregular bundles within a fibrillar basophilic (myxoid) matrix. These cells have poorly demarcated cell borders, scant to moderate homogeneous slightly eosinophilic cytoplasm, and round to ovoid central nuclei with finely stippled chromatin and a small central nucleolus. Anisokaryosis and anisocytosis are minimal. At the periphery of the neoplasm, there may be inflammatory foci primarily consisting of lymphocytes and plasma cells. Complex carcinomas must be differentiated from complex adenomas by their greater cellularity and the significant pleomorphism of the epithelial component, an increased mitotic index, foci of necrosis, and an infiltrative growth pattern.
Carcinoma and Malignant Myoepithelioma
Both the epithelial and the myoepithelial components are malignant (Fig. 13 ). This malignant neoplasm is characterized by the presence of two cell populations supported by moderate fibrous stroma. The first population is composed of cuboidal to columnar cells arranged in irregular tubules and nests (see above). Expanding the interstitium, peripherally to the tubules and nests, is the second population of cells. These cells may be spindle shaped and have poorly demarcated cell borders and a moderate homogeneous eosinophilic or basophilic cytoplasm with round central nuclei, finely stippled chromatin, and a single nucleolus. Around this population of cells is a scant basophilic fibrillar material that may occasionally be found within the cytoplasm of the cells. Cells exhibit anisokaryosis and anisocytosis, and variable numbers of mitoses are found; or, the second population of cells may have oval to fusiform vesicular nuclei with an extensive amount of eosinophilic cytoplasm and distinct cell margins. Considerable anisokaryosis and anisocytosis are found with variable numbers of mitotic figures. Immunohistochemistry is required to confirm the presence of the neoplastic myoepithelial component (smooth muscle actin, calponin, vimentin, and p63) and epithelial component (CK 8,18,19). 18
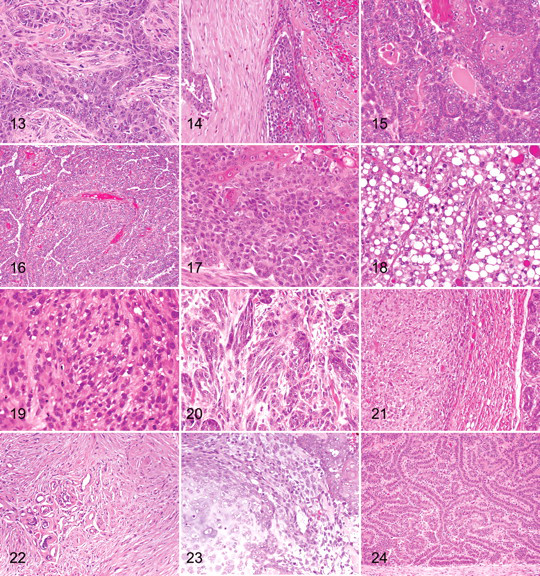
Carcinoma–Mixed Type
Mixed-type carcinoma, which is uncommon, has a malignant epithelial component and benign mesenchymal component, which are cartilage and/or bone and/or adipose tissue (Fig. 14). The neoplasm is characterized by the presence of three or more cell populations supported by a fibrovascular stroma. The first population is composed of cells arranged in irregular tubules (see above). The second population is composed of spindle-shaped cells (myoepithelial cells; see above). The third component consists of foci of cartilage and/or bone and/or adipose tissue, which exhibit no atypia. Bone that is being formed by intramembranous ossification may be lined by plump osteoblasts that are reactive but not neoplastic.
Carcinomas that are mixed type must be differentiated from benign mixed tumors by their greater cellularity and the significant pleomorphism of the epithelial component, an increased mitotic index, foci of necrosis, and an infiltrative growth pattern.
Ductal Carcinoma
Ductal carcinoma is a neoplasm that shows differentiation to interlobular ducts and is the malignant counterpart of the ductal adenoma (Fig. 15). The neoplastic cell population is arranged in cords and tubules that surround slitlike lumina that are often lined by a double layer of epithelial cells that exhibit significant anisokaryosis and anisocytosis; there are also numerous mitotic figures. Focal or multifocal areas of squamous differentiation and keratinization are present, with intracytoplasmic keratohyaline granules within some cells. The morphology of this neoplasm is identical to the apocrine ductal carcinoma of the skin. 6
Intraductal Papillary Carcinoma
With intraductal papillary carcinoma, there is proliferation of a multilayered (stratified) population of epithelial cells with characteristics of malignancy not found in duct papillary adenomas, such as a higher nuclear:cytoplasmic ratio, nuclear and cellular pleomorphism, and increased numbers of mitotic figures (Fig. 16). The fibrous connective tissue and myoepithelial cells are still retained as the supporting stroma for the papillae. In many cases, the intraductal papillary carcinoma may be found in close association with an intraductal papillary adenoma. The papillary intraductal growth pattern distinguishes this neoplasm from a complex carcinoma, even though there are myoepithelial cells within the supporting connective tissue stroma.
Malignant Epithelial Neoplasms: Special Types
Squamous Cell Carcinoma
Squamous cell carcinoma is composed solely of squamous epithelium (to differentiate from adenosquamous carcinoma). The neoplasm has its origin either from squamous cells of the teat duct or from ductal epithelial cells that had undergone squamous metaplasia and neoplastic transformation. It is often difficult to differentiate squamous cell carcinomas that have their origin in the mammary gland from those that have their origin from the overlying epidermis and invaded into the underlying mammary gland. Histologically, the neoplasm is identical to those that occur in the skin. Islands and cords of epithelial cells are seen with the formation of keratin pearls. Cells and nuclei are large; the cells produce intracytoplasmic keratin tonofilaments; and intercellular bridges may be found. A marked secondary acute and chronic inflammatory cell infiltrate often occurs in these neoplasms.
Adenosquamous Carcinoma
Adenosquamous carcinoma is composed of areas of carcinoma of any type (see above) admixed with foci where the neoplastic cells exhibit squamous differentiation and features of malignancy as described above (squamous cell carcinoma) (Fig. 17).
Mucinous Carcinoma
Mucinous carcinoma is a rare carcinoma characterized by abundant mucin production. The neoplastic epithelial mucus-producing cells can be individual cells or form tubules and nests. They are periodic acid–Schiff–diastase positive and are mucicarmine positive. The mucus-secreting cells stain cytokeratin positive on immunohistochemistry. This neoplasm must be differentiated from complex carcinomas with an abundant extracellular mucinlike matrix.
Lipid-Rich Carcinoma
Lipid-rich carcinoma may be found in younger intact bitches (Fig. 18). The neoplasm consists of sheets of cells with inconspicuous fibrovascular trabeculae and foci of necrosis. The cells are round to oval, with distinct cell borders and moderate to abundant cytoplasm containing numerous small vacuoles or a single large vacuole that occasionally peripheralizes the nucleus. Nuclei are round to oval and vesicular, with clumped chromatin and 1 or 2 nucleoli. Anisokaryosis and anisocytosis are moderate to severe, with variable numbers of mitoses. Some cases show lymphatic invasion and lymph node metastasis.
Spindle-Cell Neoplasms
Within spindle-cell neoplasms, the morphology of the neoplastic cells may be insufficient to arrive at a definitive diagnosis, and immunohistochemistry will be required in these cases and to differentiate them from mesenchymal spindle cell sarcomas.
Malignant Myoepithelioma
For malignant myoepithelioma, cells may be oval to spindle shaped (Fig. 19). Oval cells often have poorly demarcated cell borders and a moderate amount of eosinophilic or basophilic cytoplasm, with occasional intracytoplasmic clear vacuoles. Nuclei are round and central, with finely stippled chromatin and a single nucleolus. Around this population of cells is a scant, variably basophilic fibrillar material (myxoid matrix) that can be seen within vacuoles in the cytoplasm of the cells. Cells exhibit anisokaryosis and anisocytosis, and variable numbers of mitoses are found. These cells often express markers for basal cytokeratins (CK 5, 6, 14, 17) and markers for contractile proteins (smooth muscle actin, calponin, vimentin). 18
Spindle cells usually have fusiform vesicular nuclei with an extensive amount of brightly eosinophilic cytoplasm with distinct cell margins. Considerable anisokaryosis and anisocytosis are found with variable numbers of mitotic figures. Immunohistochemistry (smooth muscle actin, calponin, vimentin, p63) is required to confirm the presence of the neoplastic myoepithelial cells. 18
Squamous Cell Carcinoma–Spindle Cell Variant
The spindle cell variant of squamous cell carcinoma consists of islands and cords of epithelial cells supported by a fine fibrous stroma. Cells and nuclei are large and fusiform, and the cells produce intracytoplasmic keratin tonofilaments and intercellular bridges, with the formation of occasional irregular foci of squamous epithelial differentiation.
Carcinoma–Spindle Cell Variant
The spindle cell variant of carcinoma is one in which the cells are predominantly spindle shaped (Fig. 20). 11,12 The neoplasm consists of islands and cords of epithelial cells, often with a direct association with areas of tubular carcinoma. Surrounding the neoplastic elements is a fibrovascular stroma. Cells and nuclei are large and fusiform. Nuclei often have a prominent nucleolus; the cytoplasm is eosinophilic but does not show accumulation of intracytoplasmic keratin tonofilaments; and desmosomes are not seen. There is moderate anisokaryosis and anisocytosis and variable numbers of mitoses. Immunohistochemistry (AE1/AE3), a pancytokeratin marker, will confirm the epithelial origin of the neoplasm.
Inflammatory Carcinoma
The term inflammatory carcinoma denotes a clinical entity characterized by a fulminant clinical course, sudden presentation, edema, erythema, firmness, and warmth of the mammary glands, with or without mammary nodules. 3 Histologically, several types of highly malignant mammary carcinomas have been described. The hallmark for the histologic confirmation of inflammatory mammary carcinoma is the invasion of dermal lymphatic vessels by neoplastic emboli. The blockage of the superficial lymphatics by tumor cells is responsible for the severe edema found in the region.
Malignant Mesenchymal Neoplasms (Sarcomas)
Osteosarcoma
Osteosarcoma is the most common mesenchymal neoplasm of the canine mammary gland, and there is often a history of recent rapid growth of a mammary mass that had been present for some time (years) (Fig. 21). Within the neoplasm, there is proliferation of cells that vary from fusiform to stellate to ovoid, as associated with islands of tumor osteoid and/or bone formation. Mitoses are frequently found. The biological behavior of this neoplasm is similar to that of osteosarcomas at other sites, with metastasis via the hematogenous route, mainly to the lungs.
Chondrosarcoma
Chondrosarcoma is an uncommon mammary neoplasm that is often multilobulated. The neoplastic cells at the periphery of the lobules are small with round hyperchromatic nuclei and occasional binucleate or multinucleate neoplastic cells. The nuclear outline is often irregular, and nucleoli are prominent. Mitoses are variable in number and more common in less well-differentiated neoplasms. 21 The basophilic chondroid matrix that is associated with the neoplastic nuclei is variable in amount, but the amount and quality of the matrix is not of value as a prognostic indicator. 21
Fibrosarcoma
Fibrosarcoma is an uncommon mammary gland neoplasm that can arise within a preexisting mammary gland neoplasm or from the interstitial stroma of the mammary gland (Fig. 22). There is proliferation of fusiform cells with a distinctly interwoven pattern. Neoplastic cells have indistinct cell borders, a small amount of eosinophilic fibrillar cytoplasm, and oval to elongate nuclei that contain finely stippled chromatin and variably distinct nucleoli. Anisokaryosis and anisocytosis are moderate to marked, and mitoses are common. Fibrosarcomas must be differentiated from malignant myoepitheliomas, spindle cell carcinomas (by immunohistochemistry), and canine hemangiopericytomas, which are common on the ventral thorax and abdomen and which are composed of fusiform cells with a typical storiform and perivascular whorled pattern.
Hemangiosarcoma
Primary hemangiosarcomas may be found arising within the mammary tissue (not dermal or subcutaneous). Their histopathologic features are identical to these neoplasms in the spleen and subcutis.
Malignant Mixed Mammary Tumor (Carcinosarcoma)
The malignant mixed mammary tumor is composed partly of cells morphologically resembling the epithelial component and partly of cells morphologically resembling connective tissue elements, both types of which are malignant (Fig. 23). 11,12 It is an uncommon mammary gland neoplasm, but it most often presents as a carcinoma and osteosarcoma (see above). The epithelial component metastasizes via lymphatic vessels to regional lymph nodes and the lungs, and the mesenchymal component, via the hematogenous route to the lungs.
Benign Mammary Neoplasms
Adenoma–Simple
Simple adenomas are well-demarcated noninfiltrative nodular lesions composed of cells arranged in tubules that occasionally contain an amorphous amphophilic secretion (Fig. 24). The supporting fibrovascular stroma is scant to moderate. The tubules are lined by a single layer of cuboidal to columnar cells with a moderate amount of eosinophilic cytoplasm. Nuclei are central and round to oval, with finely stippled chromatin and a small central nucleolus. Anisokaryosis and anisocytosis are minimal with few mitotic figures.
Intraductal Papillary Adenoma (Duct Papilloma)
With the intraductal papillary adenoma, the neoplasm has a papillary, arborescent growth pattern supported by a fibrovascular stalk (Fig. 25). 13 The papillae may develop at a single focus or at multiple foci in multiple ducts. Adjacent ducts are often ectatic and lined by attenuated epithelial cells. The surface epithelial cells, which form a single layer, have an oval normochromic nucleus and a small amount of eosinophilic cytoplasm. Below the epithelium, a layer of myoepithelial cells can be readily identified. The supporting stroma consists of fibroblast, collagen, and blood vessels. Mitoses are rare.
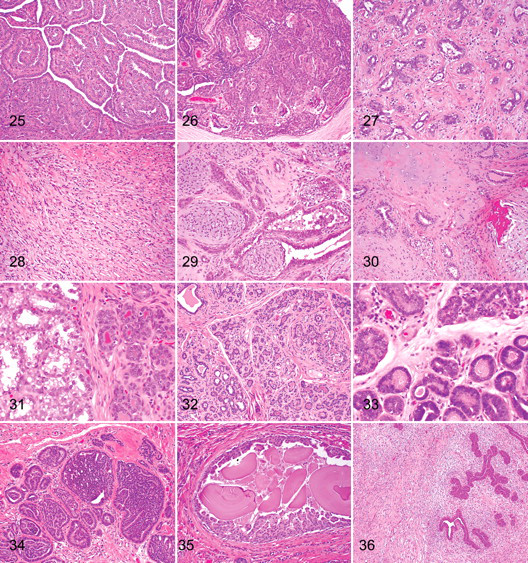
Ducts may become distended (papillary cystadenoma). Sclerosis may occur, but an underlying cause is not known. Areas of hyalinization are occasionally present with entrapment of epithelial elements, which may be misinterpreted as carcinoma.
Ductal Adenoma (Basaloid Adenoma)
With ductal adenoma, the neoplastic mass is often found within the lumen (or lumens) of an ectatic duct (or ducts) (Fig. 26). 13 It is composed of cells arranged in uniform bilayered cords with two distinct rows (luminal and basal). There is often the formation of slitlike lumina. The luminal cells are cuboidal to columnar with relatively indistinct cell margins and scant to moderate eosinophilic cytoplasm. Nuclei are round to oval, central to basal, and occasionally hyperchromatic, with a prominent basophilic nucleolus. Anisokaryosis and anisocytosis are minimal with few mitoses. Multifocally, areas of squamous differentiation (ductal differentiation) are present, with some accumulation of keratinous debris within the ductal lumina, mimicking the change normally seen at the junction of the proximal ducts and the teat sinus. The morphology of this neoplasm is identical to the apocrine ductal adenoma of the skin. 6
Fibroadenoma
The fibroadenoma is a neoplastic mass that consists of tubules lined by cuboidal or columnar cells, with nuclei that are round and uniform (Fig. 27). An extensive stroma of loose connective tissue, rich in mucopolysaccharides, surrounds the tubules. The fibroblastic stromal cells have elongated nuclei, with little cytoplasm and indistinct cell margins. More long-standing neoplasms have a denser fibrous connective tissue and hyalinization of the stroma.
Myoepithelioma
Myoepithelioma is a rare neoplasm composed of spindle-shaped cells arranged in short bundles admixed with an extracellular fibrillar basophilic material (myxoid matrix) (Fig. 28). The cells have poorly demarcated cell borders, a small amount of fibrillar cytoplasm, and a round hypochromatic nucleus with stippled chromatin. Anisokaryosis and anisocytosis are minimal, and the mitotic index is low. Myoepithelial cells can be positively identified on immunohistochemistry (smooth muscle actin, calponin, vimentin, p63). 18
Complex Adenoma (Adenomyoepithelioma)
Complex adenoma has both epithelial (tubular) and myoepithelial proliferation (Fig. 29). This neoplasm consists of two cell populations and variable amounts of fibrous stroma. The first population is composed of tubules lined by cells that are cuboidal to columnar and that have a moderate amount of eosinophilic cytoplasm. Nuclei are round to oval with finely stippled or marginated chromatin and a single central basophilic nucleolus. Anisokaryosis and anisocytosis are usually minimal. The second population is composed of spindle to stellate cells with poorly demarcated cell borders and a moderate amount of cytoplasm. Nuclei are round to fusiform with finely stippled chromatin and a single nucleolus. Intimately associated with these cells is an abundant basophilic fibrillar myxoid matrix.
Benign Mixed Tumor
Benign mixed tumor has both epithelial (tubular) and myoepithelial proliferation with foci of cartilage and/or bone (Fig. 30). This neoplasm consists of more than two cell populations and variable amounts of fibrous stroma. The first population is composed of tubules and cords lined by cells that are cuboidal to columnar and that have a moderate amount of eosinophilic cytoplasm. Nuclei are round to oval with finely stippled or marginated chromatin and a single central basophilic nucleolus. Anisokaryosis and anisocytosis are minimal. The second population is composed of spindle to stellate cells with poorly demarcated cell borders and a moderate amount of cytoplasm. Nuclei are round to fusiform with finely stippled chromatin and a single nucleolus. Intimately associated with these cells is an abundant basophilic fibrillar myxoid matrix. Multifocally, areas of cartilage and/or bone and/or adipose tissue are present.
There is some debate on the origin of the cartilage and bone within these benign mixed tumors. 13 Bone and cartilage could result from metaplastic change of the epithelial cells, 14 myoepithelial cells, 4,17,19 and interstitial stromal cells. 22 Recent studies appear to reinforce the putative role of myoepithelial/basal cells in mesenchymal differentiation. The bone in mixed tumors occurs by endochondral ossification of cartilage formed by the myoepithelial cells or by intramembranous ossification of stroma connective tissue.
Mammary Hyperplasia and Dysplasia
Duct Ectasia
With duct ectasia, there is cystic dilation of large ducts with accumulation of necrotic debris, variable numbers of foamy macrophages mixed with lipid material, and cholesterol clefts in the lumina of the ducts. Duct ectasia may be secondary to occlusion of duct lumina by intraductal neoplasms.
Lobular Hyperplasia–Regular (Adenosis)
Regular lobular hyperplasia is a nonneoplastic proliferation of the intralobular duct/ductules resulting in increased numbers of ducts/ductules and acini per lobule. 13 The epithelial cells exhibit no atypical changes. Lobular hyperplasia may be found in mammary tissue adjacent to benign or malignant mammary neoplasms.
Lobular Hyperplasia With Secretory (Lactational) Activity
Lobular hyperplasia with secretory (lactational) activity is a nonneoplastic proliferation of intralobular ducts/ductules and acini resulting in increased numbers of ducts/ductules and acini per lobule (Fig. 31). The acini are lined by actively secreting cuboidal cells (lactational changes) with accumulation of secretion within acini and ductules. The acinar cells have intracytoplasmic vacuoles and blebbing on their luminal surfaces.
Lobular Hyperplasia With Fibrosis (Interlobular Fibrous Connective Tissue)
Lobular hyperplasia with fibrosis is a nonneoplastic proliferation of intralobular ducts/ductules and acini resulting in increased numbers of ducts/ductules per lobule and with increased amount of interlobular fibrous connective tissue (Fig. 32).
Lobular Hyperplasia With Atypia
Lobular hyperplasia with atypia is a nonneoplastic proliferation of the intralobular duct/ductules and acini per lobule with increased numbers of ducts/ductules and acini per lobule (Fig. 33). The epithelial cells exhibit atypical changes, including epithelial hyperplasia, nuclear hyperchromasia, anisokaryosis, and anisocytosis with variable numbers of mitotic figures. Differentiating lobular hyperplasia with severe atypia from carcinoma in situ is very difficult. 13 When this change is seen diffusely within large areas of the mammary tissue, it is more likely to be hyperplastic.
Epitheliosis–Intraductal Hyperplasia Without Atypia
Epitheliosis (intraductal hyperplasia without atypia) is a lesion that shows the regular proliferation of epithelial cells within the lumen of ducts (Fig. 34). The aggregates of cells often fill the duct lumina. The cells have hyperchromatic nuclei with little nuclear or cellular pleomorphism and scant cytoplasm. Larger lesions can be difficult to differentiate from in situ carcinoma.
Papillomatosis–Intraductal
With intraductal papillomatosis, the lesion shows focal to multifocal intraductal papillary epithelial proliferations that are not supported by a fibrovascular stroma (Fig. 35).
Fibroadenomatous Change
Fibroadenomatous change is an uncommon lesion in the dog that shows proliferation of interlobular ducts and periductal stromal cells (Fig. 36). The stroma is often edematous or myxomatous. The fibroblast nuclei and ductal cell nuclei exhibit some pleomorphism and increased numbers of mitoses.
Gynecomastia
Gynecomastia refers to enlargement of the mammary glands in a male with duct ectasia and lobular hyperplasia found on histopathology. One or more glands may be affected. This change is most often seen with testicular Sertoli cell tumors.
Pathology of the Nipple
Ductal Adenoma and Carcinoma
Ductal adenoma and carcinoma are rare neoplasms that involve only the tissue of the nipple with no neoplastic tissue in the underlying mammary gland. The nipple is enlarged and firm. Within the neoplastic tissue, the ducts are lined by one or more layers of epithelial cells, and the lumina are open and often appear slitlike. The epithelial cells have little pleomorphism (adenomas), or the cells have increased anisocytosis and anisokaryosis and aggregate within the ductal lumina (carcinoma).
Carcinoma With Epidermal Infiltration
Carcinoma with epidermal infiltration is a neoplasm occasionally seen in the dog. A carcinoma is present within the underlying mammary gland, and carcinoma cells (either as individual cells or small aggregates) are present within the epidermis of the nipple.
Nipple Hyperplasia/Dysplasia: Melanosis of the Skin of the Nipple
Nipple hyperplasia/dysplasia is a lentigo-like proliferation of melanocytes within the epidermis. The lesion is focal and surrounds the teat ducts. The epidermis is mildly hyperplastic with rete peg formation. In the basal cell layer are increased numbers of melanocytes that often occur as small groups, or the cells may have a clear cytoplasm. There is an increased amount of melanin within the cytoplasm of keratinocytes in the basal and spinous layer, and occasional melanophages are present in the superficial dermis.
Conclusion
The proposed classification system and grading system, when applied to retrospective and prospective studies, will make it easier to compare studies and the significance of other parameters in the prognosis of canine mammary neoplasms.
Histologic Grading of Canine Mammary Carcinomas and Prognosis
A review of the prognostic significance of tumor and host characteristics was published as part of the Histological Classification of Mammary Tumors of the Dog and the Cat. 13 Since that time, there have been several publications that correlate tumor type (histologic classification) with grade and prognosis. Most grading systems of mammary carcinomas in dogs are a modification of the numeric method of Ellston and Ellis. 3,8 There are slight differences between the criteria used by Peña 3 and Misdorp. 9
There are subtle differences between these grading systems, and it is not known how this would affect the outcome of cases of mammary neoplasia. This would be most significant in cases of grade II (ie, intermediate or moderately differentiated neoplasm), and a prospective study comparing these two systems would be indicated.
The following features have also been shown to be of prognostic significance in the evaluation of mammary carcinomas and their survival 2 years after surgery. Karayannopoulou et al found significant differences in survival between cases with different tumor grades. 8 Survival was worse in dogs with grade III carcinomas than in those with grade I or grade II. Dogs with simple carcinomas had a worse prognosis than with other carcinomas; there was no significant difference in survival between grade II and grade III cases, with both having a very poor prognosis. Undifferentiated carcinomas (grade III) had an increased risk of death when compared with differentiated carcinomas (grade I and II). As noted in many of the studies, lymphatic/vascular invasion and lymph node metastasis are, as expected, associated with a poor prognosis. 2
Our knowledge of mammary neoplasms and dysplasia in the dog has advanced significantly over the last 35 years. Separating malignant mammary neoplasms into categories with a meaningful clinical outcome will require additional retrospective and prospective studies, in conjunction with the uniform use of the proposed classification and grading system in this publication.
Footnotes
Acknowledgement
This study represents an initiative of the American College of Veterinary Pathologists' Oncology Committee. The manuscript has also been reviewed and endorsed by the World Small Animal Veterinary Association. We would like to thank both organizations for their support and guidance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
