Abstract
Scuticociliatosis is an economically important, frequently fatal disease of marine fish in aquaculture, caused by histophagous ciliated protozoa in the subclass Scuticociliatida of the phylum Ciliophora. A rapidly lethal systemic scuticociliate infection is described that affected aquarium-captive zebra sharks (Stegostoma fasciatum), Port Jackson sharks (Heterodontus portusjacksoni), and a Japanese horn shark (Heterodontus japonicus). Animals died unexpectedly or after a brief period of lethargy or behavioral abnormality. Gross findings included necrohemorrhagic hepatitis and increased volumes of celomic fluid. Histologically, 1 or more of a triad of necrotizing hepatitis, necrotizing meningoencephalitis, and thrombosing branchitis were seen in all cases, with necrotizing vasculitis or intravascular fibrinocellular thrombi. Lesions contained variably abundant invading ciliated protozoa. Molecular identification by polymerase chain reaction from formalin-fixed tissues identified these as the scuticociliate Philasterides dicentrarchi (syn. Miamiensis avidus), a novel and potentially emergent pathogen in sharks.
Sharks are some of the most iconic and charismatic species displayed in public aquaria. Reports of protozoal disease in this group are sparse, 4,5 mostly individual cases reported by personal communication with limited description or further investigation.
Scuticociliatosis is an economically important and frequently fatal disease of fish, caused by histophagous ciliated protozoa in the subclass Scuticociliatida of the phylum Ciliophora. One parasite, Philasterides dicentrarchi (syn. Miamiensis avidus), has been identified as the cause of disease outbreaks in a range of marine teleost fish in aquaculture and aquarium settings. 2,6,8,9 Previous reports of scuticociliate infection in elasmobranchs are very limited. 4,5
The present report describes a series of rapidly fatal systemic scuticociliate infections in sharks, frequently characterized by 1 or more lesions from a triad of necrotizing hepatitis, meningoencephalitis, and branchitis. This highlights a significant, novel, and potentially emerging cause of mortality in sharks in captivity.
During 2 months in 2010, 6 deaths occurred in a single group of wild-caught juvenile and adult sharks. These comprised 2 zebra sharks (Stegostoma fasciatum), 3 Port Jackson sharks (Heterodontus portusjacksoni), and 1 Japanese horn shark (Heterodontus japonicus) in a European aquarium. These sharks were maintained in an established mixed species (teleost and elasmobranch) artificial seawater exhibit and died unexpectedly or following nonspecific clinical signs, including abnormal behavior, lethargy, and anorexia. Prior to the outbreak, there had been no recent introductions or changes in husbandry. No other species in the exhibit were affected at this time.
In addition, sudden unexpected deaths were recorded in 2 juvenile zebra sharks in a separate North American display aquarium. As with the first animals, these sharks were held in a mixed-species marine exhibit.
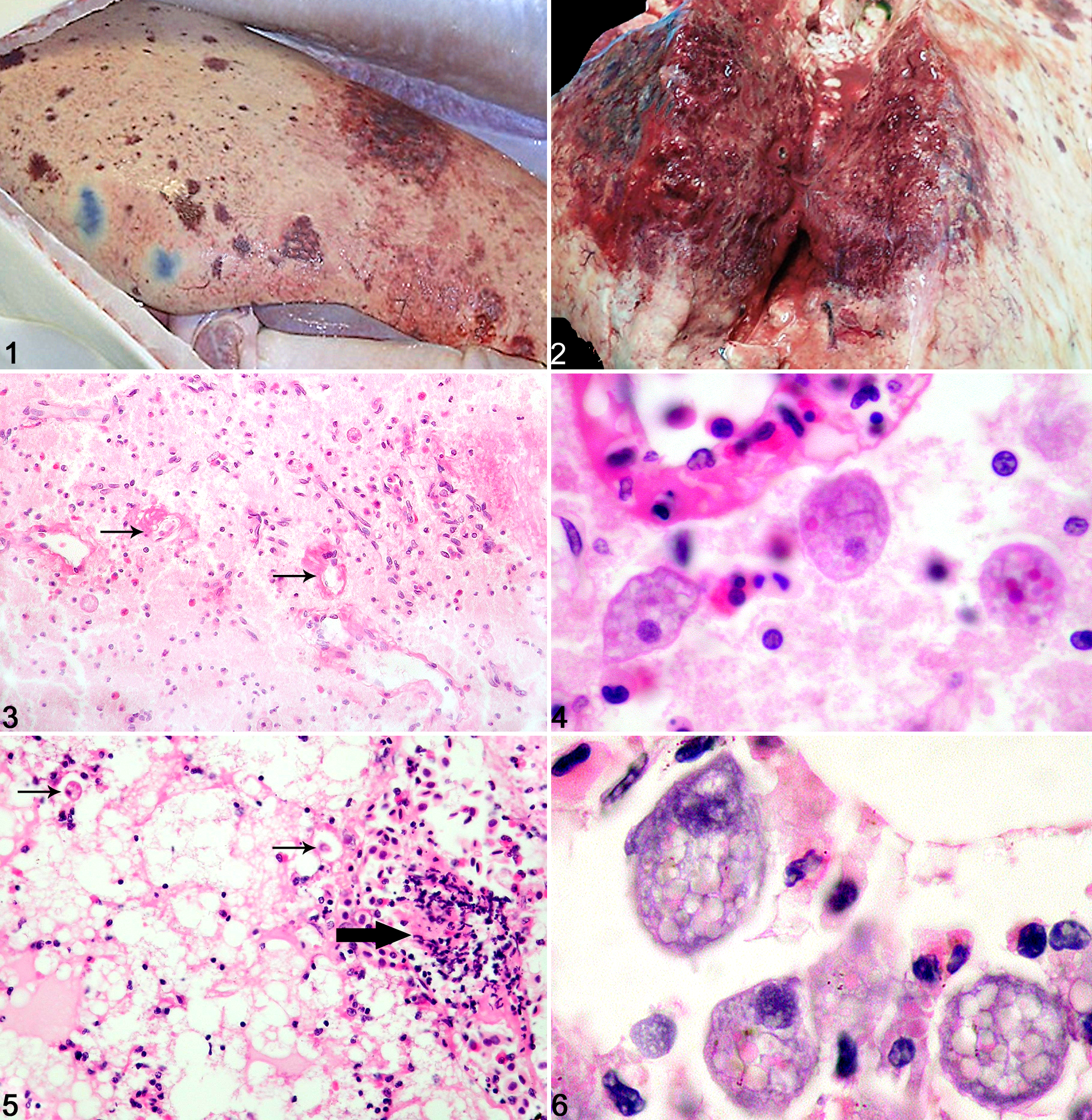
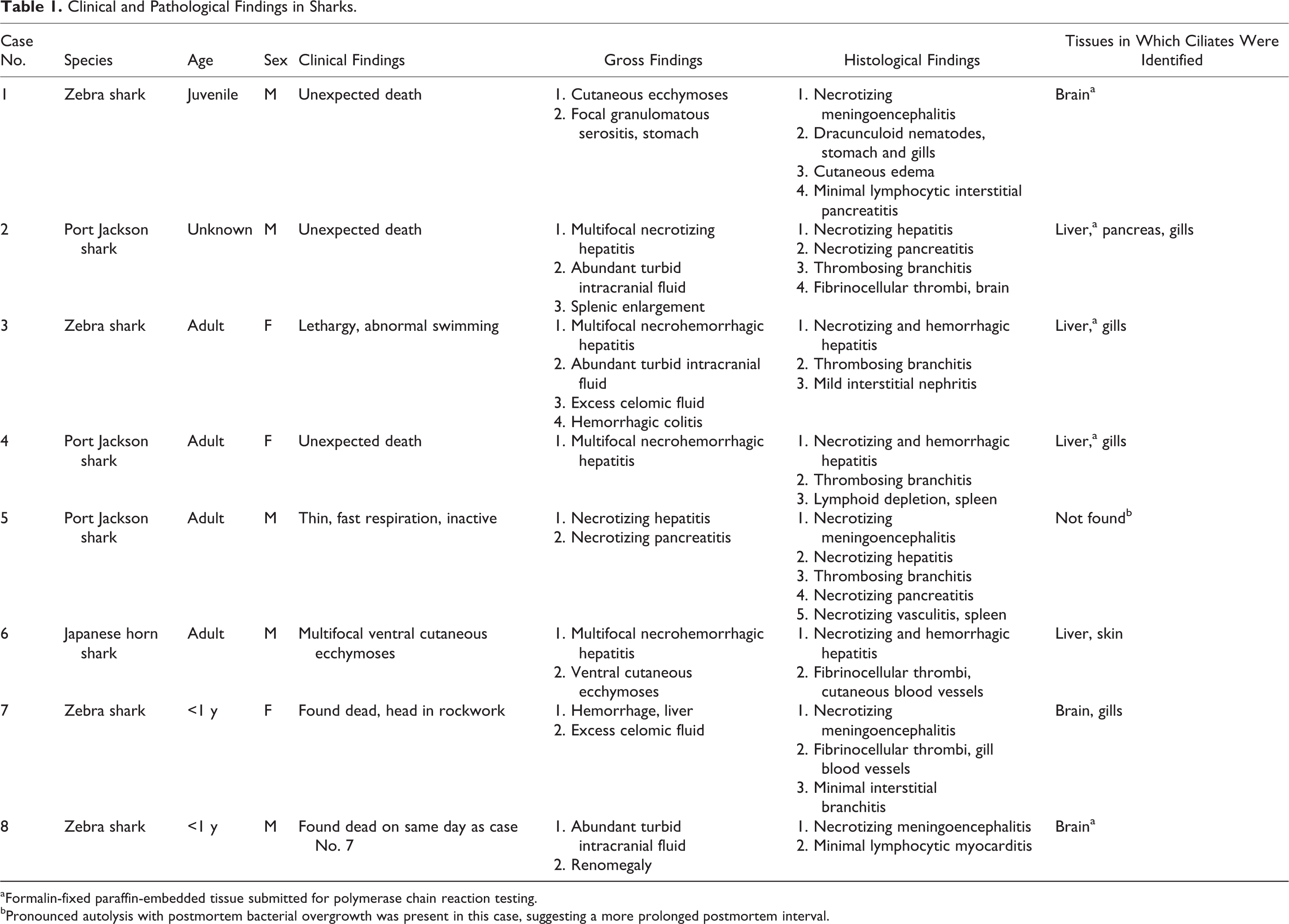
Gross postmortem examinations were performed routinely, and samples were submitted for histopathology. Clinical, gross, and histological findings in each case are summarized in Table 1. Gross abnormalities included severe multifocal necrotizing and hemorrhagic lesions of the liver (Figs. 1, 2), accompanied in 2 cases by increased volumes of celomic fluid (approximately 100 ml), which was clear and watery or brown-tinged. In 3 cases, slight turbidity of abundant intracranial fluid was noted. Cutaneous ecchymoses, necrotizing pancreatitis, and enlargements of spleen and kidney were occasionally recorded. Typically, no abnormalities were recognized grossly in the remaining organs.
Clinical and Pathological Findings in Sharks.
aFormalin-fixed paraffin-embedded tissue submitted for polymerase chain reaction testing.
bPronounced autolysis with postmortem bacterial overgrowth was present in this case, suggesting a more prolonged postmortem interval.
Samples were examined histologically following routine processing, sectioning, and staining (hematoxylin and eosin [HE] and special stains, including Gram, periodic acid–Schiff [PAS], and Ziehl-Neelsen as appropriate). At least 1 lesion from a characteristic triad of histological changes was present in each case (Table 1). The triad consisted of acute multifocal necrohemorrhagic hepatitis with thrombosing vasculitis, acute multifocal necrotizing meningoencephalitis with vasculitis, and acute multifocal thrombosing branchitis. In the brain, focally extensive areas of edema, necrosis, malacia, and neutrophilic inflammation were associated with intravascular fibrinocellular thrombi and necrotizing vasculitis (Figs. 3, 4). Hepatic lesions comprised multiple large foci of coagulative to liquefactive necrosis and cavitation, severe hemorrhage, and typically sparse infiltrates of inflammatory cells, mainly neutrophils (Fig. 5). Some of these lesions were clearly centered on or adjacent to blood vessels with necrotizing vasculitis and intravascular fibrinocellular thrombi. In the gills, multiple lamellar capillaries were dilated and occluded by similar fibrinocellular thrombi.
Elliptical ciliated protozoa were found in 1 or more of these lesions (Figs. 3–6) in each animal, except in 1 case (which had significant postmortem autolytic changes; case No. 5). Organisms were most commonly identified either within blood vessels or in the immediate perivascular space associated with vasculitis, or in foci of necrosis. Organisms were occasionally present on the lamellar epithelial surface of the gills, with minimal epithelial hyperplasia. In 2 cases, severe necrotizing pancreatitis with intralesional ciliates was present, and 1 case had scuticociliates in cutaneous blood vessels. In 1 case, marked lymphoid depletion was noted in the spleen, and occasional fibrinocellular thrombi were noted in the heart and splenic blood vessels. Special stains ruled out concurrent bacterial, mycobacterial, or fungal involvement. Additional changes identified in these cases were limited to occasional aggregates of dracunculoid nematodes in gills and gastric serosa, as well as mild chronic interstitial infiltrates of lymphocytes into gills, kidneys, and pancreas. All of these were interpreted as incidental.
The histological appearance of the intralesional protozoa was identical in all cases. Organisms were typically circular, oval, or pyriform and approximately 20 μm wide and 20 to 50 μm long, with a single variably positioned hyperchromatic circular or oval macronucleus (approximately 10 μm in diameter). Cytoplasm was pale basophilic and variably vacuolated, and it frequently contained globules of homogeneous eosinophilic debris, presumptive erythrophagocytosis. The best preserved were clearly covered by rows of cilia. The appearance of the organisms closely resembled that previously described in cases of scuticociliatosis in teleost fish species. 1
To confirm that the parasites were scuticociliates and further identify them to species level, we undertook molecular investigations using formalin-fixed paraffin-embedded tissues from several of the cases (case Nos. 1–4, 8). Paraffin blocks were selected that contained tissues with definite lesions, including histologically apparent ciliates. Briefly, DNA was extracted from five 10-μm sections of a fixed paraffin-embedded sample using a commercial kit (Qiagen DNeasy; Qiagen, Valencia, CA) according to the manufacturer’s instructions, with an additional step of removing paraffin from the top while warmed during the initial DNA lysis. A portion of the 18S ribosomal DNA (rDNA) was amplified by polymerase chain reaction (PCR) using universal Ciliophora primers as previously described. 7 Nucleotide sequencing of amplicons was carried out by a commercial company (GENEWIZ, South Plainfield, NJ). Sequences were confirmed by sequencing both strands. Forward and reverse sequences were aligned by a sequence alignment program, 10 and each sequence was compared by use of the GenBank nucleotide sequence database for similarity with sequences in an online genomic database (Nucleotide Basic Local Alignment Search Tool; National Center for Biotechnology Biotechnology Information, National Institutes of Health, Bethesda, MD [www.ncbi.nlm.nih.gov/blast/blast]).
PCR testing yielded a positive result from case No. 8. Based on the nucleotide sequence, P. dicentrarchi (syn. M. avidus) was identified. The small subunit (SSU) rDNA sequence (designated GenBank accession # KC285109) completely matched that of M. avidus (100% sequence identity [602/602 bp] with GenBank accession EU831196 and others). See online version for Supplementary Figs. S1 and S2. The next closest species match was to Homalogastra setosa, with only 96% sequence identity. Samples from cases Nos. 1 to 4 failed to yield a PCR product. This was likely due to DNA fragmentation associated with prolonged formalin fixation, since a control PCR for β-actin, which is highly conserved between species, was also negative.
Previous descriptions of ciliate infection in elasmobranchs are very limited, and reports implicating scuticociliatosis are limited to Uronema-like gill and renal infections in Southern stingrays (Dasyatis americana). 4,5 Other ciliates that have been reported include Brooklynella sp in 2 Heterodontus species, and Cryptocaryon sp infection was generated experimentally in dusky smoothhounds (Mustelus canis) and spiny dogfish (Squalus acanthias). 5 Molecular identification by PCR is not reported for these cases. Interestingly, presumptive amebic infections of brain (in Stegostoma fasciatum) and liver (in sandbar shark, Carcharhinus plumbeus) have also been reported as personal communications. 5 The availability of molecular testing for scuticociliates by PCR facilitates the reevaluation of such cases with presumptive etiologic diagnoses based on morphology alone. This is desirable since the definitive identification of unicellular protozoa based on morphological characteristics in histological sections is difficult, 1 particularly when complicated by necrosis or postmortem degeneration (which occurs rapidly in fish samples). Review of the archive of one of the authors (M.F.S.) has found 6 additional histologically similar cases, each in different aquaria. Species affected have included zebra, Port Jackson, bowmouth (Rhina ancylostoma), and nurse (Ginglymostoma cirratum) sharks. This suggests that this disease may be a problem that is either emerging or has gone unrecognized previously in captive sharks.
In conclusion, although reports of protozoal disease are less common in captive elasmobranchs than in teleosts, the cases described above indicate that sharks are susceptible to spontaneous infection by scuticociliates under captive conditions and that the infection rapidly follows a systemic and lethal course. In teleost fish in aquaculture, simultaneous infections by genotypically and serologically distinct strains of P. dicentrarchi appear to contribute to outbreaks of disease 2 and may be differentially susceptible to potential treatments. 3 Therefore, work remains to be done to clarify the epizootiology in elasmobranchs and to establish whether teleost or elasmobranch coinhabitants of these exhibits represent an important reservoir of infection. In the future, such investigations may yield information with implications for scuticociliate control in mixed exhibits containing sharks.
Footnotes
Acknowledgements
The assistance of aquarists and curators within the Merlin Entertainments group, particularly Robin James, is gratefully acknowledged, as is that provided by Daniela Denk and Penny Cusdin (IZVG) and Liz Post and Christie Buie (Northwest ZooPath).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
