Abstract
Juvenile salmon sharks beach yearly along the California coast, primarily during late summer and early fall. Fresh, frozen, and formalin-fixed tissues from 19 stranded salmon sharks were collected for examination. Histopathology revealed meningitis or meningoencephalitis in 18 of 19 shark brains with intralesional bacteria observed in 6 of the affected brains. Bacterial culture of fresh or frozen brain, liver, and/or heart blood from 13 sharks yielded pure cultures characterized molecularly and/or biochemically as belonging to the genus Carnobacterium. The 16s ribosomal DNA sequence of 7 tissue isolates from 7 separate sharks was 99% homologous to C. maltaromaticum (GenBank FJ656722.1). Sequence of the large ribosomal DNA intergenic spacer region (ISR) was 97% homologous to C. maltaromaticum (AF374295.1). This is the first report of Carnobacterium infection in any shark species, and the authors posit that brain infection caused by Carnobacterium is a significant cause of morbidity and mortality in juvenile salmon sharks found stranded along the Pacific coast of California.
The salmon shark (Lamna ditropis) is an apex predator in the same family as the great white shark. Salmon sharks range from the Gulf of Alaska south to central Baja California and east to Hawaii, with adults reported primarily in the northern part of the range and juveniles in the southern portion. 21 Documentation of juveniles, usually slightly under or over 1 meter in total body length, is composed primarily of reports of stranded individuals found along the coast of California and Oregon. Although these stranding events have been recorded by local biologists for more than 20 years, no specific causes have been identified. Stranded salmon sharks are most commonly found in the summer and fall and are typically either still alive or found as fresh carcasses undamaged by scavengers.
In this study, we examined archived formalin-fixed, frozen, and fresh tissues from stranded juvenile salmon sharks collected from the coast of California between 2002 and 2007. Initially investigating the possible role of marine toxins such as domoic acid in these stranding events, we discovered a severe brain infection that we propose is partially or solely responsible for the disorientation and stranding behavior of these sharks.
Materials and Methods
Specimens
Tissues from 19 stranded salmon sharks were supplied by the California Department of Fish and Game of Monterey, California; the Pacific Shark Research Center of Moss Landing Laboratories; Long Marine Laboratory of the University of California, Santa Cruz; and the Pelagic Shark Research Foundation. Stranded sharks were typically reported to either the Pelagic Shark Research Foundation or the Long Marine Laboratory Marine Mammal Stranding Network. The sharks were then transported to the California Department of Fish and Game facility at Long Marine Laboratory or to the Research Animal Facility at Stanford University for necropsy. Fresh carcasses were evaluated immediately. Archived tissue samples included whole heads and samples of viscera, either frozen (at –20°C from 1 month to 2 years) or immersion fixed in 10% neutral buffered formalin until examination. Frozen brain tissue from 1 healthy adult female salmon shark from a research study in Alaska and fresh brain tissues from 2 healthy leopard sharks and 1 healthy spiny dogfish shark acquired during routine food fish surveys in the San Francisco Bay were used as controls.
Pathology
All available tissues were examined grossly. Fresh and frozen samples were subsequently immersion fixed in 10% neutral buffered formalin. Representative tissue samples were routinely trimmed, processed, sectioned at 4 μm, and stained with hematoxylin and eosin for evaluation.
Microbiology
Routine Gram’s stain was performed on imprints of fresh and frozen brain tissue. Samples of fresh or frozen tissue from 13 individual sharks (12 brains, 9 livers, and 5 samples of heart blood) were plated on Brain Heart Infusion Agar with 5% sheep’s blood (BHIA), Chocolate Agar (CHOC), ROSE (Columbia CNA Agar [nalidixic acid and colistin in Columbia Blood Agar] in Phenylethyl Alcohol Agar Base), and MacConkey Agar and incubated at 25°C in 5% CO2 for 48 to 72 hours or until observable growth was present (all agar media from Hardy Diagnostics, Santa Maria, CA). Brain tissue from 2 leopard sharks, a spiny dog fish and a healthy adult salmon shark was cultured as negative controls.
Genetic Sequencing
The 16S ribosomal DNA and large intergenic spacer region (ISR) were sequenced from 7 bacterial isolates acquired from 7 separate sharks (sharks Nos. 6, 8–12, 15). Primers and polymerase chain reaction (PCR) cycles are listed in Table 1. Primers were custom designed to amplify regions of the Carnobacterium 16S ribosomal DNA and the large ISR for analysis (Integrated DNA Technologies, Coralville, IA). The ISR primers were designed to amplify a region previously determined to differentiate between Carnobacterium piscicola, Carnobacterium mobile, Carnobacterium divergens, and Carnobacterium gallinarum. 8 PCR reactions were carried out in 25-μl volumes containing 18 μl ddH2O, 2.5 μl PCR buffer, 2.5 μl 0.8 μmol dNTP, 0.5 μl of each primer, 0.06 μl of AmpliTaq (Applied Biosystems, Carlsbad, CA), and 1 μl of bacterial liquid culture. For negative controls, PCR reactions were carried out on 1μl of distilled water and 1 μl of noninoculated broth medium.
Primers.

Sequences of 16S ribosomal DNA and ISR were determined by routine dideoxynucleotide chain termination method using the primers listed in Table 1 (ABI 3100 Genetic Analyzer; Applied Biosystems). Forward and reverse sequences were visually edited and the consensus sequences were compared with known Carnobacterium species 16S and ISR sequences using the GenBank BLAST tool.
Results
Available tissues were from 19 salmon sharks that had stranded between 2002 and 2007 along roughly 300 miles of California coastline. Morphometric data are presented in Table 2. The sharks ranged from 87 to 124.5 cm in total body length (mean = 104.3 cm). There were 9 males, 7 females, and 3 specimens for which the gender was not recorded. Ten sharks stranded in the region of Monterey Bay and Santa Cruz, 7 along the western coast of the San Francisco peninsula, 1 shark at Morro Bay, and 1 at Bodega Bay. Sharks most commonly stranded between July and September (8 sharks). Whole fresh or frozen carcasses or subsets of tissues including fresh, frozen, or formalin-fixed head, brain, and/or viscera were available for evaluation.
Morphometric Data, Histologic Features, Culture Results, and PCR Results From 19 Stranded Salmon Sharks Collected From the California Coast.
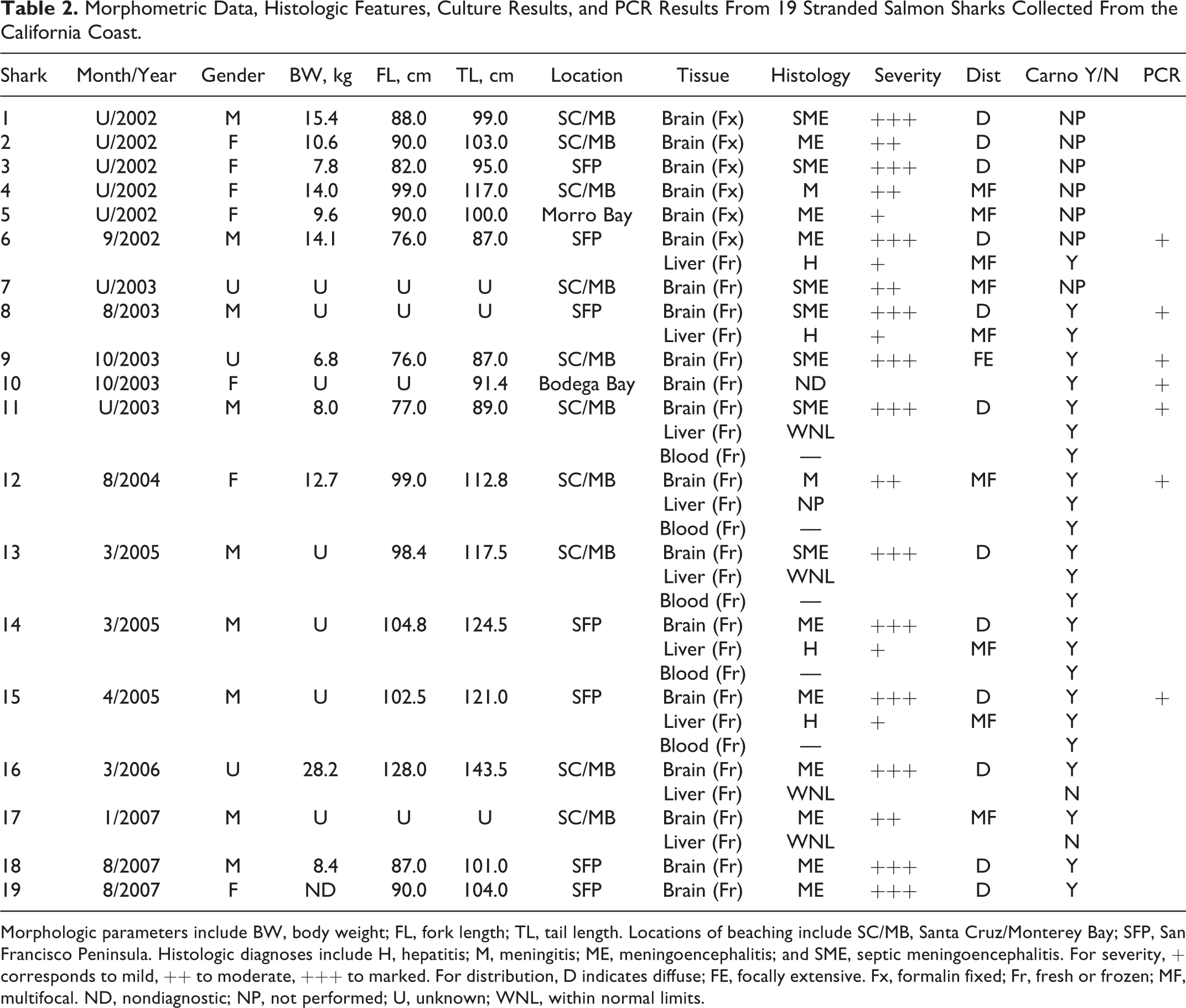
Morphologic parameters include BW, body weight; FL, fork length; TL, tail length. Locations of beaching include SC/MB, Santa Cruz/Monterey Bay; SFP, San Francisco Peninsula. Histologic diagnoses include H, hepatitis; M, meningitis; ME, meningoencephalitis; and SME, septic meningoencephalitis. For severity, + corresponds to mild, ++ to moderate, +++ to marked. For distribution, D indicates diffuse; FE, focally extensive. Fx, formalin fixed; Fr, fresh or frozen; MF, multifocal. ND, nondiagnostic; NP, not performed; U, unknown; WNL, within normal limits.
Grossly, the meningeal surfaces of the brain were reddened and variably thickened in 18 of 19 brains (Fig. 1). Multiple pinpoint (<1 mm diameter), white to yellow foci were scattered throughout the hepatic parenchyma of 2 of 5 livers available for examination (Fig. 2). The remaining livers were too autolyzed for gross interpretation. Incidental gross findings included superficial dermal abrasions and ocular perforations (postmortem damage), copepod parasites attached to gill and skin surfaces, and cestodes in the spiral valve and rectal gland.
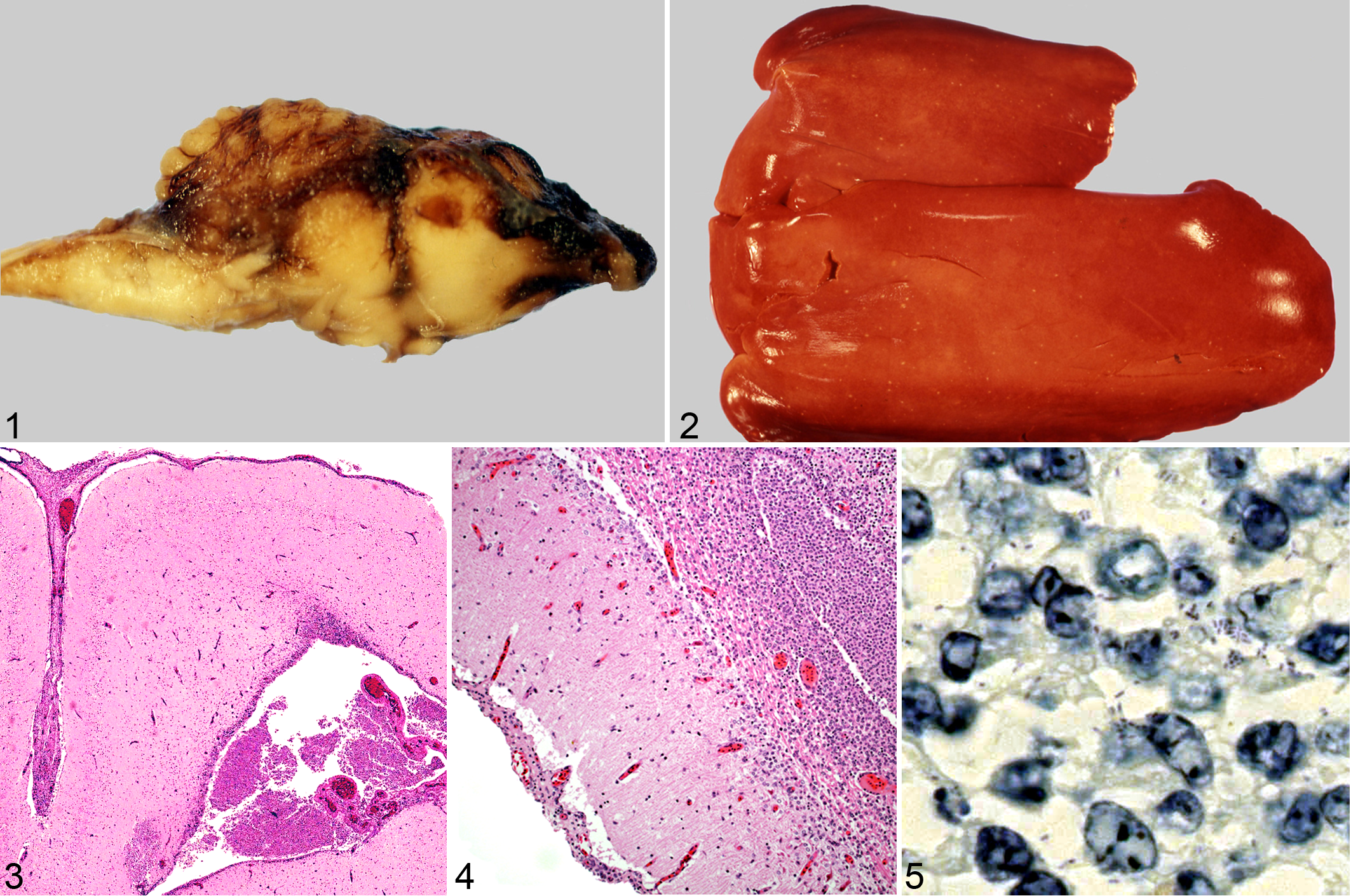
Eighteen of 19 brains were characterized histologically by inflammatory infiltrates in one or more histologic sections of the telencephalon, diencephalon, mesencephalon, and/or metencephalon. Brain inflammation ranged from mild to marked and tended to be multifocal in more mildly affected brains and diffuse in those more severely affected (Table 2). Typical histological findings in the brain included mild to marked expansion of the leptomeninges with hemorrhage, eosinophilic protein-rich fluid, mononuclear inflammatory cells, necrotic cell debris, and congested meningeal vessels (Fig. 3). Ventricles were filled with inflammatory cells in more severely affected animals, and inflammation frequently extended into the neuropil beneath inflamed meninges and surrounding severely affected ventricles. The ventricular ependymal lining was frequently disrupted and expanded by mononuclear cells mixed with necrotic cell debris (Fig. 4). Microabscesses with gram-positive, non-acid-fast, extracellular and/or intracellular bacilli were occasionally observed in the ventricles. Bacteria within areas of inflammation were best identified in Giemsa-stained sections (Fig. 5). Small vessels in the neuropil adjacent to inflamed meninges and ventricles were cuffed by inflammatory cells. Liver foci observed grossly were correlated microscopically with multifocal irregular foci ranging from a few microns to 2 mm in diameter, characterized by mononuclear cell infiltrates that displaced and replaced hepatocytes. All tissues displayed some degree of freeze-thaw artifact and/or autolysis. However, although tissues were frequently disrupted by ice crystal formation and rare tissues displayed physical disruption and/or autolysis that precluded making a histological diagnosis, most tissues were of adequate quality for microscopic evaluation of inflammation. Brain tissues were not compromised by overgrowth of postmortem bacteria.
Gram’s stain of fresh tissue imprints revealed small, bipolar, lancet-shaped, Gram-positive rods, mostly single with some in pairs and short chains. Large capsules were evident on many of the organisms. The smears also demonstrated few gram-negative organisms with the same morphology, indicating that they are easily destained.
Fresh or frozen tissue from 13 sharks (12/13 brains, 7/9 livers, and 5/5 blood samples) yielded pure bacterial cultures on BHIA, ROSE, and CHOC. The cultured organisms grew as small, <1-mm diameter, pearly white colonies and were gram positive with the same morphology as the tissue organism. Gram-positive Biochemical tests were performed with the BIOLOG GP microbial identification system, which identified the bacterium as a Carnobacterium spp (Biolog, Inc, Hayward, CA). When multiple tissues were available from the same animal (brain, liver, blood), heaviest growth came from brain. Carnobacterium was not isolated from any control shark tissue samples.
Seven separate isolates were subjected to PCR analysis using primers for the 16S ribosomal DNA gene to amplify a region totaling 1022 base pairs in length (GenBank accession number JN314890). The 16S ribosomal DNA sequences from the separate isolates were 100% homologous to each other and 99% homologous to multiple C. maltaromaticum isolates listed in GenBank. Many other as-yet uncharacterized Carnobacterium species shared 97% to 99% homology with the 16S ribosomal DNA of our isolate. PCR with primers for the ISR yielded a region totaling 578 base pairs (GenBank accession JN314891). All of the ISR sequences obtained from our bacterial isolates were 100% homologous to each other. The consensus sequence was 100% homologous to Carnobacterium sp 17–4 (CP002563.1), an isolate from Norwegian seawater, and 90% to 97% homologous to strains of C. piscicola (now reclassified as C. maltaromaticum).
Discussion
Meningoencephalitis has been documented as causing disorientation and death in elasmobranch species, although reports are rare. 19 Parasitic meningoencephalitis has been described in a group of nurse sharks infected with dracunculoid-type nematode larvae. 3 Pathogenic Vibrio sp was associated with encephalitis in a naturally infected brown shark, and encephalitis was subsequently induced in spiny dogfish and lemon sharks exposed to the isolated organism by intraperitoneal injection. 5 Pyogranulomatous meningitis of unknown etiology was identified in a stranded basking shark. 3
Species of Carnobacterium are frequent inhabitants of the intestine, urogenital tracts, and gills of both fresh and salt water teleosts, in both aquaculture and in wild populations. 1,7,9,15 –17 These organisms can also be cultured from temperate and polar ocean waters, as well as certain terrestrial environments. 11,20 Some species of Carnobacterium are known to produce bacteriocins, proteins that kill or inhibit the growth of other bacteria, and this property has led to continuing interest and use of Carnobacterium species as probiotics in aquaculture and food preservation. 4,9,11,13,14,18
Despite their role as normal flora and the protective effects of some bacteriocin-producing strains, some Carnobacterium species, particularly C. maltaromaticum, have been associated with morbidity and mortality in a variety of fish species. 1,5 Rainbow trout (Oncorhynchus mykiss) challenged with C. maltaromaticum (previously C. piscicola) died within 5 to 9 days of exposure with histological evidence of hepatic necrosis and encephalitis. 1 A C. maltaromaticum–like bacterium was isolated in association with histological abnormalities in lake whitefish (Coregonus clupeaformis) from Lakes Michigan and Huron. 12 In support of the role of C. maltaromaticum as a fish pathogen, Leisner et al 10 identified specific genes in the C. maltaromaticum genome encoding virulence factors. Although members of the genus Carnobacterium are not considered human pathogens, they have been recovered from wounds and blood cultures in human patients. 2,6
Carnobacterium has not been reported as normal flora in sharks, and this is the first report of Carnobacterium-associated pathology in any shark species. In the present study, a C. maltaromaticum–like bacterium was isolated in pure culture from all sharks for which fresh or frozen tissue was available. Histologic evidence of intracellular and extracellular bacteria within areas of brain inflammation strongly supports a causative role for this organism. The severity of the meningoencephalitis identified in the present study strongly suggests that this process contributed to altered mentation, stranding behavior, and mortality. Isolation of the organism from blood suggests that affected sharks were bacteremic with involvement of liver and brain coming via blood circulation. Interestingly, the 16S and ISR sequence data demonstrate that the isolates are genetically identical to each other at these 2 loci, although the sharks were collected from geographically and temporally separate stranding events. This suggests an as yet unidentified common source of exposure to the organism.
Specific identification of this Carnobacterium isolate was hampered by the small number of reports of the same or similar organisms in microbiological and genetic databases. Standardized identification schema of Carnobacterium spp are still being established, resulting in a recent reclassification of the type strain C. piscicola to C. maltaromaticum. 11 BIOLOG, the only phenotypic identification system, to our knowledge, listing Carnobacterium species, is limited to 6 type strains out of the 10 ATCC (American Type Culture Collection)–listed type strains and does not reflect a comprehensive identification to species level. Genetic comparisons are also based on very small numbers of submissions from naturally occurring (nonengineered) strains to the GenBank database and may similarly fail to provide accurate identification to species level. Additional verified sequence and phenotypic data are needed from culture collection databases to determine relatedness of this organism to previously described Carnobacterium species.
Future studies are needed to address the source of exposure for juvenile salmon sharks and to further determine and evaluate any predisposing factors such as age or environment. The infections may be peculiar to juvenile salmon sharks because of unknown aspects of their life history such as coastal migration, behavior, or diet, as well as physiologic stressors and aspects of immunologic function not shared by adults. It is possible that adult salmon sharks are exposed and infected, but not reported because they inhabit the northern Pacific Ocean, where coastal communities are less frequent, making identification, reporting, and retrieval of stranded adults less likely. In addition, the adults are pelagic, and thus ill or dead sharks are more likely to be preyed upon or scavenged in deep water and less likely to observed on beaches.
We report a C. maltaromaticum–like organism isolated in pure culture from the brain, liver, and blood of stranded salmon sharks suffering from meningoencephalitis. Bacterial isolates from 7 separate sharks spanning multiple years and extensive geographic distances shared 100% homology of DNA base pairs at both the 16S and large IRS loci. The epidemiology of this disease will be understood with further investigation into the natural habitat of the organism, as well as the lifestyle and ecology of juvenile and adult salmon sharks. Continued monitoring of stranding events and evaluation of additional samples from diseased and healthy juveniles as well as adult salmon sharks will aid in elucidating the pathogenesis of this disease.
Footnotes
Acknowledgements
The authors greatly appreciate Robert Lea of the California Department of Fish and Game, Monterey, California, and Dr David Ebert at the Pacific Shark Research Center, Moss Landing Marine Laboratories. We also thank Dr Stephen Palumbi, Stanford University’s Hopkins Marine Station, for guidance and laboratory support of molecular studies; Dr Sal Frasca Jr, University of Connecticut, for initial review of neurohistopathology; and Dr Linda Cork, Department of Comparative Medicine, for her support of the project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Department of Comparative Medicine, Stanford University.
