Abstract
Juvenile common thresher sharks (Alopias vulpinus) have been recently stranding along the California coastline. Using Illumina sequencing of the bacterial 16S rRNA gene along with necropsy, cytological, bacteriological, and histological techniques, we screened microbial communities and described lesions characterizing affected sharks with the purpose of identifying potential pathogen sources and pathologic processes. Histopathological assessment of moribund sharks revealed severe meningoencephalitis, as previously described in stranded salmon sharks (Lamna ditropis), along with inflammation of the inner ear and subcutaneous tissues surrounding the endolymphatic ducts. Furthermore, inflamed areas were characterized by the prevalence of Carnobacterium maltaromaticum, suggesting this bacterium as a potential pathogen that gains access to the inner ear through the endolymphatic ducts, with subsequent spread into the brain. The absence or low abundance of this bacterium in the spiral valve in both healthy and infected sharks suggests that Carnobacterium is not a commensal member of their digestive communities and the spiral valve is unlikely to be the source of the pathogen. Furthermore, phylogenetic analysis suggests that C. maltaromaticum strains isolated from diseased sharks have minimal genetic variation and differ from other strains originating from food or diseased teleosts. While a C. maltaromaticum–like organism has previously been associated with meningoencephalitis in salmon shark strandings, this is the first study to report common thresher shark strandings associated with C. maltaromaticum, involving the endolymphatic ducts as portals of entry to the brain.
Young of the year salmon sharks (Lamna ditropis) have been observed to strand along West Coast beaches of North America for decades, but this has only recently been attributed to an infection by Carnobacterium maltaromaticum. 27 These stranding events involve disoriented, sick juveniles that swim onto the beach and occur throughout the year, although mostly reported from July to September. 8 More recently (2013–2018), young common thresher sharks (Alopias vulpinus) have also been observed to strand along the California coast, and further examination revealed similar infection with C. maltaromaticum as found in stranded salmon sharks. 23
C. maltaromaticum is a facultative anaerobic, psychrotrophic, lactic acid–fermenting bacterium, of biotechnological interest due to its antimicrobial properties inhibiting Listeria monocytogenes.18,22 It is detected in many natural environments including the gastrointestinal tract of various teleost species,1,15,19,26,29 where it is thought to stimulate the immune system, and thus is sometimes used as a probiotic in aquaculture.12,16 However, C. maltaromaticum is also a pathogen in cold-water teleost fishes exposed to various stressors (eg, spawning, handling events).19,20 In diseased fish, C. maltaromaticum is associated with pseudokidney disease, septicemia, splenomegaly, internal hemorrhages, muscular abscesses, visceral congestion, and thickening of the swim-bladder walls.14,19,20
Common thresher and salmon sharks belong to the order of Lamniformes, are top predators that migrate between coastal and offshore waters of the Northeast Pacific Ocean, and use the continental shelf as nurseries for juveniles. 9 C. maltaromaticum has not been isolated from healthy elasmobranchs; however, this bacterium is known to be a normal component of the intestinal microbiome of many teleost species.1,15,19,26,29 Therefore, if C. maltaromaticum is part of the microbial community of healthy thresher sharks, it is plausible that under certain stressful circumstances, commensal C. maltaromaticum may become pathogenic, initiate disease, and cause sharks to strand.
In this study, we aimed to describe the scope and severity of lesions in stranded sharks to elucidate potential C. maltaromaticum infection pathways. Based on pronounced inflammation in the subcutaneous tissue surrounding the endolymphatic ducts, inner ear, and dorsal meninges of the brain regions closest to the perilymphatic ducts, we hypothesized that endolymphatic ducts serve as routes of entry into the inner ears, and sequentially, perilymphatic ducts as the probable gateway for bacteria to access the cranial vault. We used Illumina 16S rRNA sequencing and conventional bacteriology to characterize microbial communities of the spiral valve, inner ear (mix of perilymph and endolymph fluid), subcutaneous tissues surrounding the endolymphatic ducts, and extradural or cranial fluid surrounding the brain of infected and healthy common thresher sharks. High levels of Carnobacterium in the spiral valves of infected thresher sharks could be an indication that the infection originates by overcolonization of the spiral valve7,21,28,32 and that higher pathogen numbers might affect their microbiome diversity and structure. We also examined the microbial community in the embryonic spiral valve and in the uterine fluid of 2 pregnant females to investigate the uterine environment as another potential source of fetal exposure and infection, as suggested by the young age of stranded salmon sharks.
To elucidate the phylogenetic relationship of the C. maltaromaticum strains isolated from diseased sharks to other strains from different sources, we used the 16S rRNA gene, single-nucleotide polymorphisms (SNPs), and average nucleotide identity (ANI) to construct phylogenetic trees. Despite varying sample origin and collection times, we hypothesized that these shark (SK)-isolated strains would be more phylogenetically closely related to each other than to other C. maltaromaticum isolated from food or disease fish.
Finally, a stranded juvenile shortfin mako (Isurus oxyrinchus) was also diagnosed with C. maltaromaticum infection (CA Department of Fish and Wildlife, unpublished data), constituting the third species of shark affected by this bacterium.
Materials and Methods
Sample Collection and Necropsy
Healthy common thresher sharks were caught via hook-and-line by sport or commercial fishers, and then donated to California State University at Long Beach (CSULB) Shark Lab. Injured and infected sharks that stranded along California beaches (Fig. 1) were collected by members of the general public, volunteers, and a variety of State and local governmental agencies. Stranded and healthy sharks were iced or frozen prior to transport to CSULB. When available, morphometric data (eg, length, body weight, gender) were recorded and age of the shark was estimated based on the Von Bertalanffy equations with corresponding parameters specific for the shark species and sex.11,17,30 Data on sharks used in this study are shown in Supplemental Table S1.
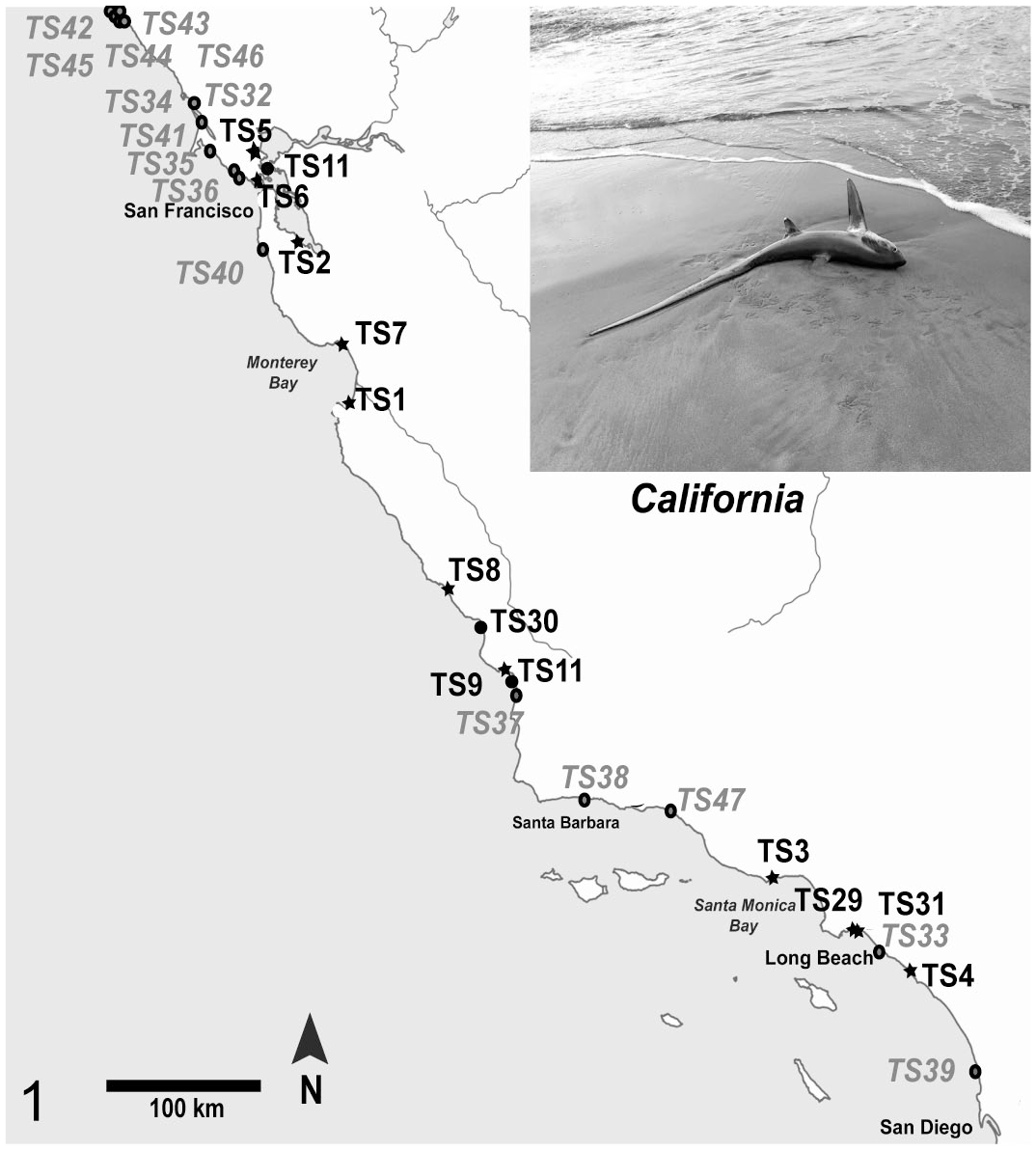
Locations of shark strandings along the California coast. Common thresher sharks (TS) in bold indicate they were infected with Carnobacterium maltaromaticum, and the location is marked with a star. Stranded sharks that did not show any evidence of infection are depicted with a black circle. Gray circles and letters are stranded sharks not collected in this study but reported at the iNaturalist website (www.inaturalist.org). Information about the sharks is shown in Supplemental Table S1.
Necropsies were performed on adult, juvenile, and fetal common thresher sharks obtained from 2 pregnant females (Thresher 24 and Thresher 25; Supplemental Table S1) caught by fishers. Dissection was focused on the spiral valves, uterine horns, endolymphatic ducts, brain, and inner ears to obtain baseline data on normal anatomy, cytology, microbiology, and histology and to provide insights into pathology and pathogenesis in infected sharks. Detailed description of the dissection procedure, cytology, bacteriology, and histology is provided in the Supplemental Material.
Cytology
Cytology samples were taken using sterile plastic pipettes from: (1) periductal subcutaneous tissues surrounding the endolymphatic ducts; (2) inner ear fluid (a mix of perilymph and endolymph); and (3) extradural fluid overlying the brain. Fluids were evaluated for the presence and type of inflammatory cells and bacteria. Bacteria were assessed based on size, shape, and motility.
Bacteriology
Duplicate samples of extradural fluid, inner ear fluid, and subcutaneous fluid surrounding endolymphatic ducts were taken for bacteriologic assessment using sterile cotton-tipped swabs and inoculated onto tryptic soy agar (TSA) plates or blood agar plates (TSA with 5% sheep blood). A conventional isolation streak pattern was used on TSA and blood agar plates to identify major bacterial pathogens and to separate them from contaminant colonies. Agar plates were incubated aerobically at room temperature (24°C–27°C). Bacterial colony growth was assessed daily and characterized by shape, color, and size. Next, DNA from subcultured bacterial colonies was extracted and confirmed as C. maltaromaticum using Sanger sequencing as previously described. 23
Histology
To assess the degree and extent of bacterial infection and damage to tissues in stranded thresher sharks, brain, inner ear, calvarium, and tissues surrounding the endolymphatic ducts were histologically examined. In a few sharks, olfactory bulbs and tracts were also histologically examined. Samples for histopathology were fixed in 10% formalin solution for at least 48 hours prior to trimming. Trimmed samples were washed with phosphate-buffered saline, dehydrated in an ethanol series, and embedded in paraffin wax. Tissues were serially sectioned at 6 µm using a microtome, mounted onto Superfrost Plus slides (Fisher Scientific, Hampton, New Hampshire), and stained with hematoxylin and eosin for histological analysis.
To establish baseline data on normal anatomy and histology, common thresher sharks caught by fishers via hook-and-line were used as putatively healthy controls. Necropsy, histological sampling, processing, and assessments were identical to those used with stranded sharks. Six areas of the brain were evaluated histologically to determine the presence or absence of bacteria, as well as the type and degree of inflammation and necrosis in the meninges and parenchyma of the brain. The degree of congestion of meningeal blood vessels and the thickening of meninges due to fibroplasia (ie, “scarring”) and neovascularization were also assessed. Regions of the brain that were specifically examined included coronal sections of the rostral, medial, and caudal telencephalon (ie, olfactory lobes), optic tectum, cerebellum, and medulla oblongata (Fig. 2). For each region, the degree of inflammation was determined semi-quantitatively by counting layers of inflammatory cells (ie, mononuclear and polymorphonuclear cells) associated with the meninges. Inflammation scores were used to determine which specific regions of the brain were targeted and to define whether inflammation was distributed equally within an area of the brain. Due to their irregular 2-dimensional nature, the meninges did not lend itself well to conventional morphometric analysis (eg, inflammation within a circular objective field); thus, counting layers of inflammatory cells was deemed a more suitable method for the evaluation of inflammation in this organ (Supplemental Methods).
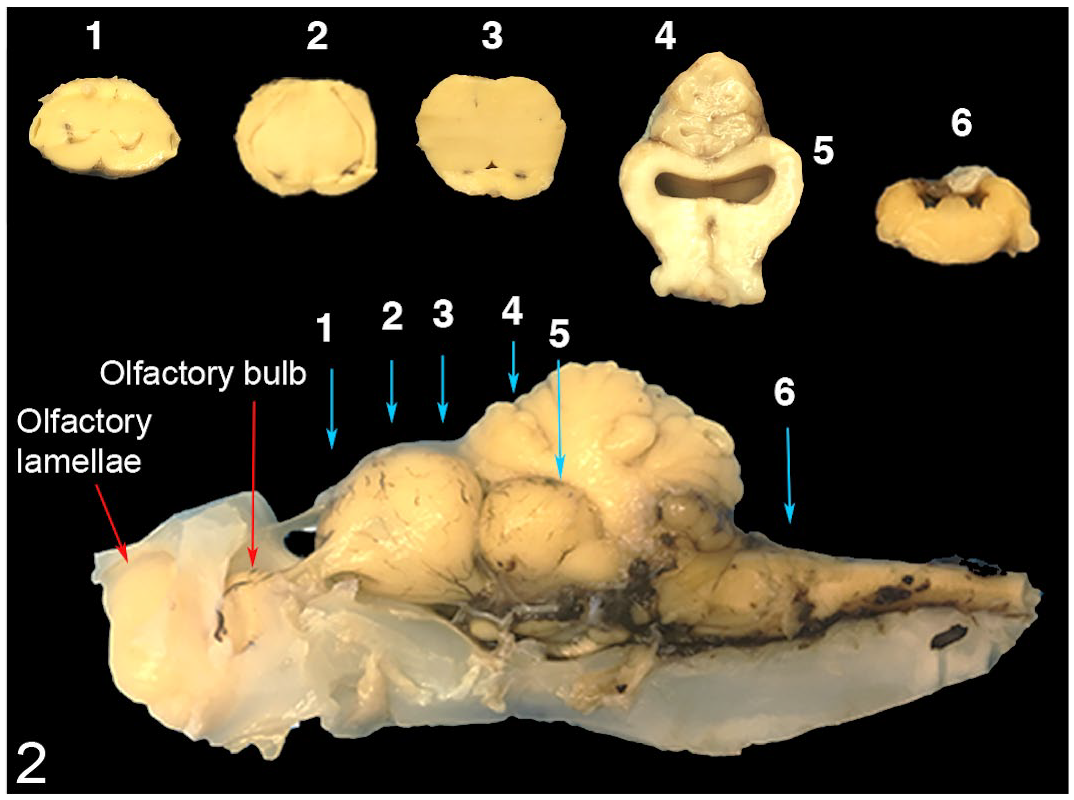
Regions of the common thresher shark brain used for histological assessment were rostral telencephalon (1), middle telencephalon (2), caudal telencephalon (3), cerebellum (4), optic tectum (5), and medulla oblongata (6). Blue arrows indicate the location of coronal sections. Red arrows indicate adjacent regions. Cartilage from the base of the skull, including around the olfactory bulb and lamellae, was left to preserve the shape and integrity of the brain.
Microbial Communities and C. maltaromaticum Phylogeny
The methods for DNA bacterial sequence retrieval, phylogeny of C. maltaromaticum isolated from diseased sharks, and corresponding bioinformatics analysis are described in the Supplemental Material. Briefly, for microbiome analysis, DNA extracted from the extradural fluid, inner ear endolymph and perilymph, uterine fluid (from pregnant females), and spiral valve contents (from the distal segment closest to the cloaca) was used to amplify the V4 variable region common to all bacterial archaeal 16S rRNA genes, and samples were paired-end sequenced on an Illumina MiSeq (v2, 2 × 150 bp) instrument. Retrieved sequences were analyzed using QIIME (Quantitative Insights Into Microbial Ecology—V. 1.9.1), 4 and the final OTU (Operational Taxonomic Unit) table was imported to R studio (V. 1.0.153) 33 for further analysis. For the characterization of C. maltaromaticum strains isolated from diseased sharks, the draft genomes of 9 C. maltaromaticum strains (SK) isolated from the brain and inner ear of 3 stranded thresher sharks and 2 stranded salmon sharks 23 (Supplemental Table S2) necropsied and sampled in this study (with the exception of salmon shark 0 sampled by the CA Department of Fish and Wildlife) were compared with 8 other strains isolated from various environments and 37 other publicly accessible genomes from the NCBI databases covering different species from the genus Carnobacterium and 1 outgroup (Supplemental Table S3). First, the 9 SK strains were phylogenetically identified by comparing the complete 16S rRNA gene sequences. Next, tetranucleotide correlation search (TCS) 25 and ANI 34 were performed and trees based on identified core genome SNPs and ANIb values were built and evaluated.
Results
Thirty-six common thresher sharks were sampled (Supplemental Table S1). Fourteen were healthy asymptomatic sharks caught via hook-and-line, 8 were healthy embryos from 2 pregnant females, and 11 thresher sharks were found stranded on beaches in Central and Southern California (Fig. 1). In addition, 3 nonlethal cloacal swabs were collected from live sharks caught during an annual National Oceanic and Atmospheric Administration (NOAA) shark tagging cruise off the coast of Southern California during August 2016.
Of the 11 thresher sharks that stranded, 8 had gross, cytological, and histological lesions in the subcutaneous tissues surrounding the endolymphatic ducts, inner ears, meninges, and brain. One stranded thresher shark (Thresher 4) likely had lesions associated with Carnobacterium infection (ie, inflammatory exudate surrounding endolymphatic ducts and thickened meninges), but was severely autolyzed and could not be adequately evaluated at necropsy. These 9 stranded thresher sharks (including Thresher 4) were also positive for C. maltaromaticum infection, based on aerobic culture on agar plates, polymerase chain reaction (PCR), Illumina sequencing, and cytology.
Two stranded thresher sharks were Carnobacterium-negative. The first Carnobacterium-negative stranded shark (Thresher 10) was a moribund shark collected live with severe eye and head wounds, and was likely hit by a motorized watercraft. The second Carnobacterium-negative stranded shark (Thresher 11) was found deceased with external wounds suspected to be the cause of death and related to injury associated with fishing capture. The 2 sharks that were Carnobacterium-negative did not have gross cytological or histological lesions associated with otitis and meningoencephalitis.
None of the 14 putatively healthy thresher sharks caught via hook-and-line had evidence of otitis, meningitis, or encephalitis. C. maltaromaticum was not cultured from the subcutaneous interstitial fluid surrounding the endolymphatic ducts, nor from inner ear perilymph or extradural fluid. Furthermore, no bacteria were detected from DNA extracted from these samples using conventional PCR.
Gross Pathology
Stranded thresher sharks infected with C. maltaromaticum were pristine externally, with no major lesions or wounds, and internal lesions were confined to the head. Infected sharks had lesions in the subcutaneous tissues surrounding the endolymphatic ducts. Peri-ductal subcutaneous lesions were centered over the endolymphatic fossa of the chondrocranium and were typically characterized by large amounts of a thick, yellow-white, viscous exudate. In some sharks, the inflammatory exudate was primarily hemorrhagic or serosanguinous (Fig. 3). Comparatively healthy sharks collected via hook-and-line had no gross lesions in the subcutaneous tissues surrounding the endolymphatic ducts (Fig. 4) above the endolymphatic fossa (Fig. 5). In a thresher shark (Thresher 1), inflammation was marked by discrete abscess formation over the endolymphatic fossa.
Meningitis was a consistent finding in all infected sharks that stranded. In some thresher sharks, meningitis was characterized by congestion of meningeal blood vessels and only mild thickening of the meninges (Fig. 6). In the majority of sharks, however, meningitis was characterized by marked thickening, combined with a thick, yellow-white exudate that obscured the dorsal surface of the brain. The inner perichondrial lining of the calvarium was similarly thickened and inflamed. Blood vessels over the surface of the brain were consistently congested and prominent. Extradural fluid and inner ear perilymph in infected sharks were cloudy and opaque. There were no gross lesions in the gills, hearts, kidneys, or coelomic organs of infected sharks that stranded. Overall, sharks with meningitis had empty stomachs but were well-muscled and had large livers (Supplemental Fig. S1), with no gross signs of emaciation or liver atrophy, suggesting they died quickly (without depletion of hepatic lipid stores or due to muscle atrophy).
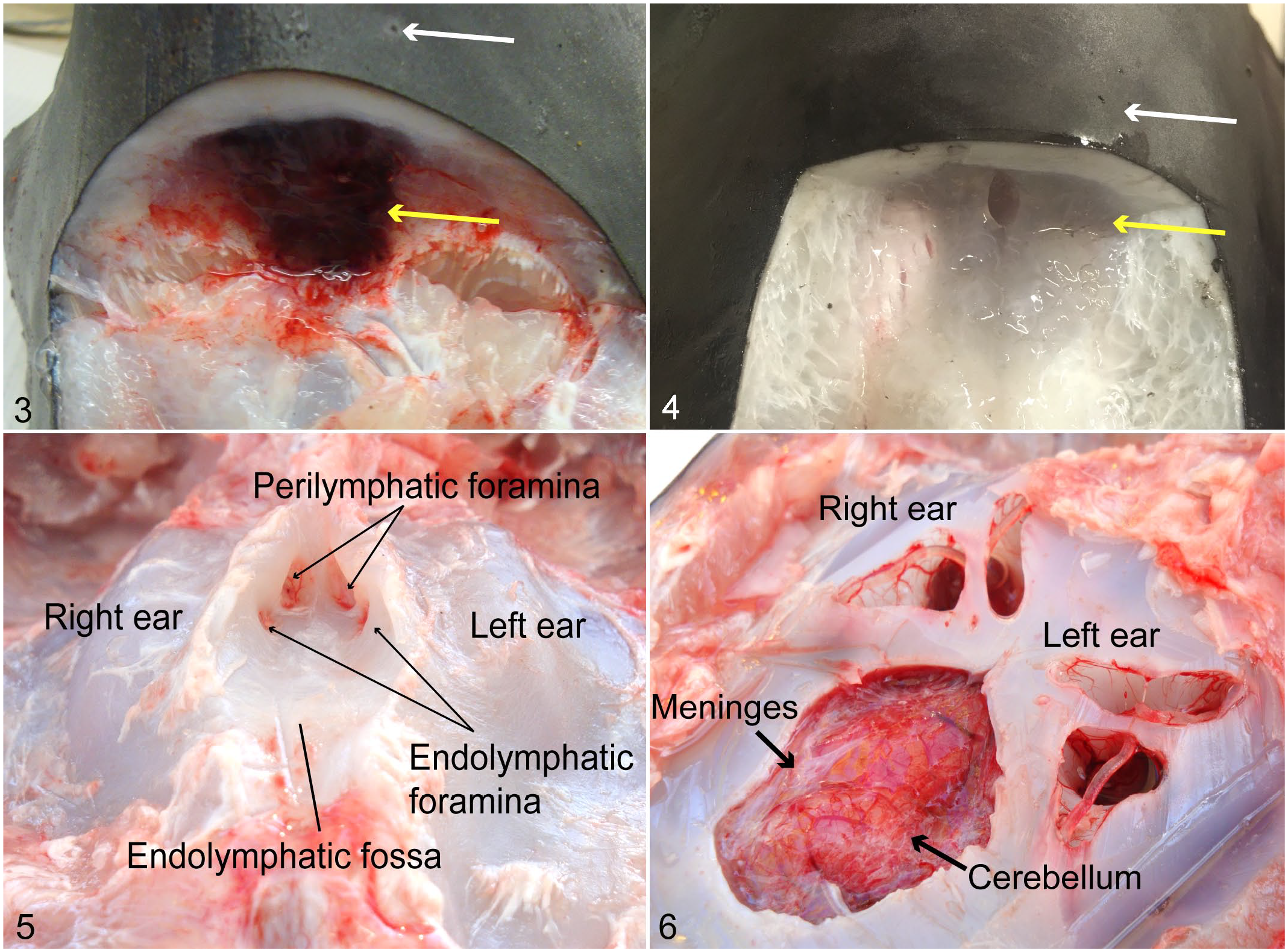
Dissection of the endolymphatic ducts and chondrocranium, common thresher sharks. Dorsocaudal view of the head, incised transversely at the level of the endolymphatic pores overlying the endolymphatic fossa. Figure 3. Infected shark. The subcutaneous tissue surrounding the endolymphatic duct (yellow arrow) is hemorrhagic. Endolymphatic pores (white arrow). Figure 4. Uninfected healthy shark. Endolymphatic ducts and subcutaneous connective tissue. Figures 5–6. Infected shark. Figure 5. Endolymphatic fossa ventral to the endolymphatic ducts and dorsal to the brain. Figure 6. Removal of the calvarium exposes the brain and both inner ears. The meninges are thickened.
Healthy sharks had no lesions in the endolymphatic ducts, inner ears, meninges, or brain. Extradural fluid and inner ear perilymph from healthy sharks were clear.
Cytology and Bacteriology
No bacteria or inflammatory cells were found in wet mount cytology samples of periductal interstitial fluid (from subcutaneous tissues surrounding the endolymphatic ducts), extradural fluid, or inner ear perilymph taken from healthy thresher sharks caught via hook-and-line. Similarly, no bacteria or inflammatory cells were observed in cytology fluid samples taken from the 2 stranded thresher sharks that were Carnobacterium-negative. In contrast, all stranded sharks that were Carnobacterium-positive had cytological lesions in periductal subcutaneous tissues overlying the endolymphatic fossa, in extradural fluid, and in inner ear perilymph. Cytological assessment consistently revealed myriad small nonmotile coccobacilli (short bacterial rods; morphology consistent with C. maltaromaticum) mixed with moderate to severe inflammation (Supplemental Fig. S2). Similarly, agar plates streaked with swabs from infected sharks usually showed typical C. maltaromaticum white opaque colonies (Supplemental Fig. S3), while those caught by fishers had no growth.
Histopathology
A total of 8 infected and 7 uninfected common thresher sharks were used for the histopathology assessment (Supplemental Table S1). The uninfected sharks included 2 thresher sharks that stranded but had no infection (Threshers 10 and 11) and 5 putatively healthy sharks caught by hook-and-line (Threshers 18, 20, 23, 24, and 25). In some sharks, the tissue was damaged or missing; thus, it was not used for histological assessment (Supplemental Table S4). No histological lesions were observed in the periductal subcutaneous tissues, inner ears, meninges, or brains of presumed healthy sharks caught via hook-and-line. Contrarily, among infected sharks, histological findings consisted of severe diffuse meningoencephalitis, bilateral otitis interna, and severe, focally extensive cellulitis in the subcutaneous tissues surrounding the endolymphatic ducts.
While there was severe mixed inflammation in periductal subcutaneous tissues of infected sharks (Figs. 7 and 8), the endolymphatic ducts were intact and pseudostratified, sometimes with mild infiltration of the duct epithelium by few inflammatory cells (Fig. 9). Inflammation consisted of large numbers of mononuclear and polymorphonuclear cells (Fig. 8). In some sharks, inflammatory cells were mixed with large numbers of small basophilic coccobacilli (presumed to be C. maltaromaticum). Subcutaneous blood vessels were often congested and, in some areas, were markedly inflamed, with thickening of vessel walls, mural necrosis, and infiltration by inflammatory cells. Subcutaneous inflammation extended from the superficial subcutis, adjacent to dermal collagen, down to the endolymphatic fossa of the skull. There was often significant hemorrhage in subcutaneous tissues as well. Periductal inflammation in 7 stranded and infected sharks was uniformly severe. By comparison, none of the 4 control sharks (3 caught by hook-and-line and 1 a stranded trauma victim) had any inflammation in subcutaneous tissues surrounding the endolymphatic duct connective tissue. The tissue was characterized by attenuated fibroblasts and thin strands of collagen supporting an acellular gelatinous matrix (Fig. 10).
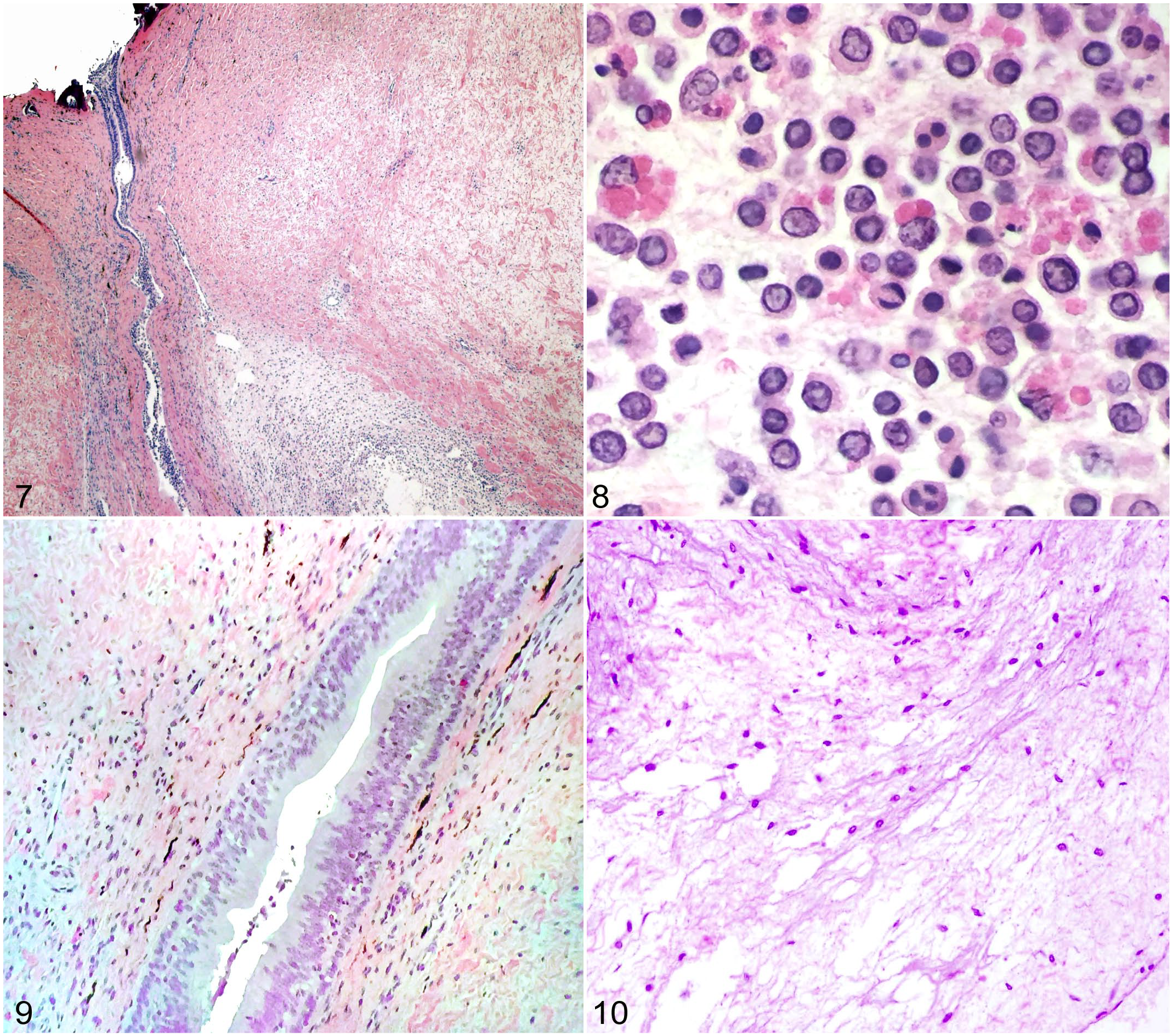
Endolymphatic duct area, common thresher sharks. Hematoxylin and eosin. Figure 7. Infected shark. The endolymphatic duct is surrounded by subcutaneous tissue with myriad inflammatory cells. Figure 8. Infected shark. The subcutaneous tissue surrounding the duct contains numerous mononuclear and polymorphonuclear inflammatory cells. Figure 9. Infected shark. Intact endolymphatic duct with mild infiltration of the duct epithelium by few inflammatory cells. Figure 10. Healthy shark. Loose connective tissues surrounding the endolymphatic ducts do not contain inflammatory cells.
The inner ears of infected sharks were similarly inflamed, with large numbers of mixed inflammatory cells trapped in the perilymphatic space located between the cartilaginous and membranous labyrinths. In some infected sharks, moderate to large numbers of inflammatory cells were present within the endolymphatic space of the sacculus as well (Fig. 11). Seven of the 8 infected sharks had severe inner ear inflammation. In comparison, none of the 6 uninfected control sharks (ie, C. maltaromaticum-negative sharks) evaluated histologically had inflammation in the inner ears, including the semicircular canals, sacs, and sacculus in either endolymphatic or perilymphatic fluid (Fig. 12).
The brains of 8 stranded common thresher sharks were completely (n = 4) or partially (n = 4) examined histologically (Supplemental Table S4). Only well-preserved tissue samples were used in this study; thus, histological assessment was incomplete in 4 sharks because of damage to the brain from autolysis (shark 1) or damage to the area during sampling or processing. Meningeal inflammation was widespread among infected sharks, with diffuse involvement of all regions of the brain. Inflammation was most severe over the telencephalon and cerebellum and was worse over dorsal and lateral aspects of the brain. Inflamed meninges were markedly expanded with congested blood vessels and dense sheets of mixed inflammatory cells. Inflammatory infiltrates were largely mononuclear (primarily macrophages), mixed with fewer polymorphonuclear cells (Fig. 13). Many macrophages were enlarged with phagocytized cellular debris. In some sharks, inflammatory cells were mixed with moderate to large numbers of small coccobacilli (presumed to be C. maltaromaticum). There was mild inflammation of adjacent cerebral or cerebellar parenchyma in a few areas.
The brains of uninfected healthy thresher sharks were covered by a simple meningeal lining composed of a highly vascularized membrane of loose connective tissues (Fig. 14). Among 5 healthy sharks caught via hook-and-line and Carnobacterium-negative shark No. 11, there was no meningeal inflammation. Carnobacterium-negative shark No. 10 had evidence of intracranial hemorrhage (ie, red blood cells overlying the cerebellum) and mild meningeal inflammation composed of mononuclear cells over the optic tectum and medulla oblongata.
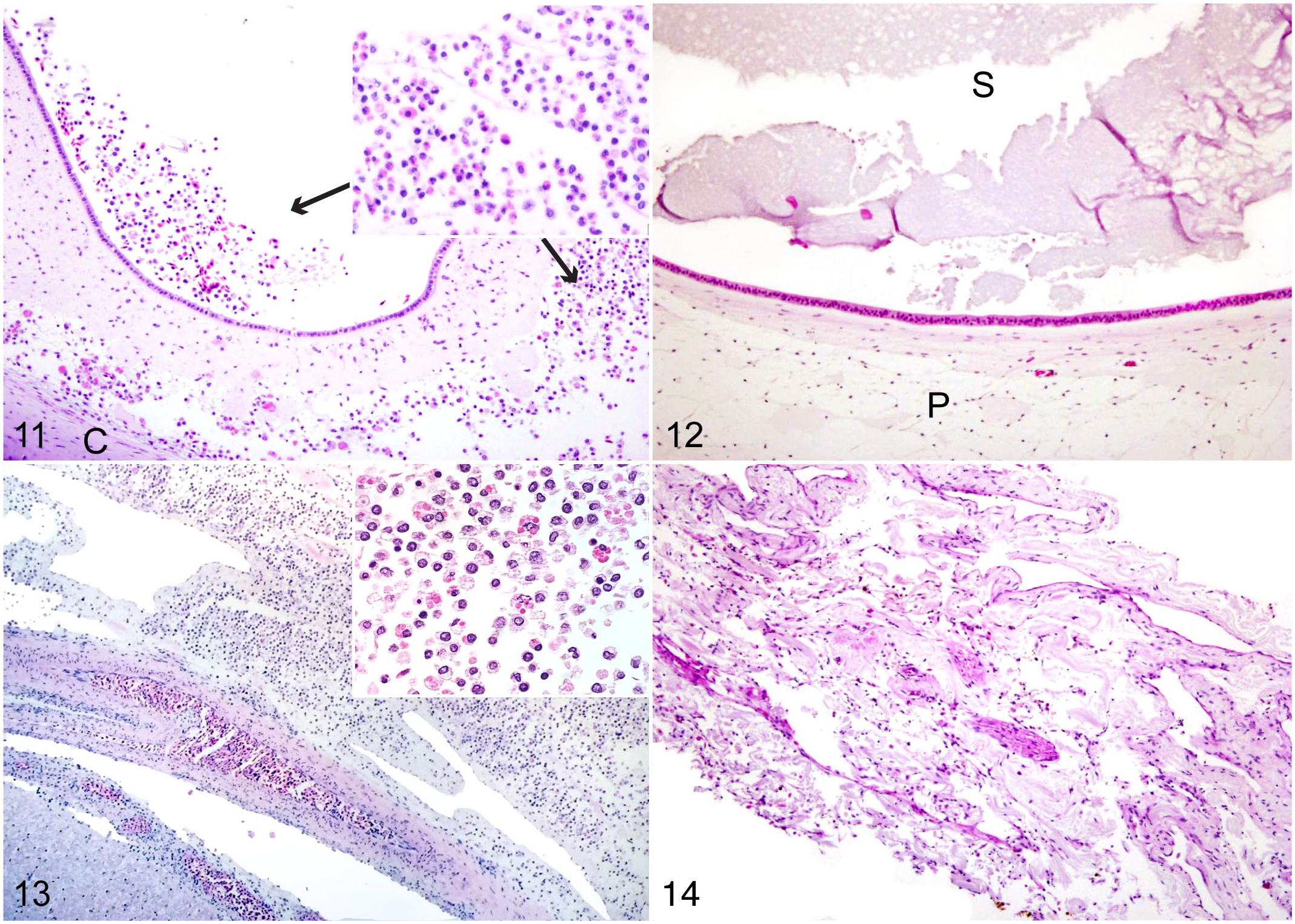
Inner ear and meninges, common thresher sharks. Hematoxylin and eosin. Figure 11. Infected shark. The inner ear contains mononuclear and polymorphonuclear inflammatory cells (inset) in the sacculus and perilymph space. Figure 12. Healthy shark. Inner ear with no inflammatory cells. Figure 13. Infected shark. Severely thickened meninges overlying the cerebellum, largely composed of mononuclear inflammatory cells. Some larger cells have phagocytosed debris (inset). Figure 14. Healthy shark. The meninges are normally thick with no inflammatory cells. C, cartilage; P, perilymphatic space; S, sacculus.
Presence of Carnobacterium in the Microbiome
The presence of Carnobacterium in the spiral valve’s bacterial communities was examined in 5 groups: (1) 11 presumed healthy sharks; (2) 5 stranded sharks infected with C. maltaromaticum; (3) 2 stranded sharks with no signs of infection (stranded noninfected); (4) 3 nonlethal cloacal swabs from live sharks (collected on the annual NOAA thresher shark tagging cruise); and (5) 8 fetal sharks. In addition, the uterine fluid from the 2 pregnant females was also screened for Carnobacterium.
Carnobacterium was present in the spiral valves of 2 of 11 healthy thresher sharks, 3 of 5 infected sharks, 0 of 2 stranded noninfected sharks, and 1 of 3 cloacal swabs from live thresher sharks (Supplemental Fig. S4). There was a significant difference in relative abundances of Carnobacterium between healthy and infected sharks (Kruskal-Wallis, chi-square = 4.006, P = .045); however, the overall relative OTU abundance was very low (less than 0.1%). Similarly, Carnobacterium was identified in 3 of the 4 uterine horns sampled from the 2 pregnant females and in the spiral valve of 6 of 8 embryos; however, when present, Carnobacterium relative abundance was quite low (less than 0.05% of the reads) (Supplemental Fig. S4). The predominant bacteria in the uterine fluid and in the spiral valve of the embryos were Mycoplasma sp (99%).
While no bacteria were detected in the periductal subcutaneous tissues surrounding the endolymphatic ducts, extradural fluid, or inner ear of healthy and stranded noninfected sharks, these regions in infected sharks were heavily colonized. Carnobacterium dominated the microbial communities in stranded sharks with otitis and meningitis, accounting for more than 50% of relative abundance in the extradural fluid and in subcutaneous tissues surrounding the endolymphatic ducts. Carnobacterium was especially prominent in the inner ears of infected sharks that stranded, comprising more than 90% of the relative abundance at that location. Two other commonly detected genera were Photobacterium and Mycoplasma (Supplemental Fig. S5). Similarly, the affected areas in stranded Mako and salmon sharks had high prevalence of Carnobacterium, except the adult salmon shark (Supplemental Fig. S6).
Strain Identification
According to complete 16S rRNA gene analysis (less than 1% of 16S rRNA variation13,19) (Supplemental Fig. S7), the tetranucleotide correlation analysis (TCS >0.99), and AIN (ANIb >98%), all 9 SK strains were identified as C. maltaromaticum. Across SK genomes only, ANIb were >99.9%. Furthermore, 16S rRNA sequences from all publicly accessible genomes of Carnobacterium (n = 37) revealed little phylogenetic divergence among sequenced genomes of C. maltaromaticum with the exception of C. maltaromaticum 757 CMAL, derived from human skin (ANIb = 67.7%) (Supplemental Fig. S7). SK strains belonged to the same more distant C. maltaromaticum cluster in the SNP (Supplemental Fig. S8) and ANIb (Supplemental Fig. S9) trees. However, both the identification of 91,236 identified SNPs distributed throughout the core genomes of the 17 C. maltaromaticum genomes (Supplemental Fig. S8) and the ANIb tree (Supplemental Fig. S9) produced a diverged branched cluster with the 9 SK strains.
Discussion
Reports of elasmobranch stranding events have recently (2011–2018) become more common, and while the underlying cause is not always the same, stranded sharks frequently show signs of central nervous system infection.24,27,31 For example, the protozoan Miamiensis avidus has been associated with massive leopard shark (Triakis semifasciata) stranding events in San Francisco Bay, 24 and a C. maltaromaticum–like organism has been attributed as the cause of salmon shark strandings along the West Coast of North America. 27 Thus, it seems that elasmobranch stranding events are commonly associated with central nervous system infections, possibly resulting in disorientation and inadvertent swimming onto shore. In this study, we analyzed healthy and stranded common thresher sharks using necropsy, bacteriology, cytology, histology, and Illumina sequencing. Furthermore, we reported a third lamniform species—the shortfin mako—with a potential C. maltaromaticum infection.
Through genomic sequencing, this study identified C. maltaromaticum both in stranded common thresher and in salmon sharks with meningoencephalitis and otitis. First, the systematic comparison of sequenced Carnobacterium genomes highlighted phylogenetic cohesion of all C. maltaromaticum isolates. Furthermore, all SK isolates of C. maltaromaticum clustered as a monophyletic clade distantly related to other lineages of C. maltaromaticum. This suggests that C. maltaromaticum isolated from infected thresher and salmon sharks has few genetic variations and thus may share the same evolutionary history, which could indicate a common source of this strain of C. maltaromaticum.
Furthermore, the prevalence of Carnobacterium in the extradural fluid, inner ear, and endolymphatic ducts further supports C. maltaromaticum as the cause of inflammation in infected sharks and consequent disorientation resulting in stranding. The presence of other bacterial strains in infected sharks such as Photobacterium and Aliivbrio, also found in the spiral valve, was likely a result of postmortem contamination. Postmortem contamination of inner ear and brain may have followed the same route as for C. maltaromaticum (ie, via open endolymphatic pores and ducts) or could have resulted from vascular invasion and hematogenous migration following bacterial overgrowth in the spiral valve after the shark stranded and died. It is also possible, although less likely, that bacteria from the spiral valve were antemortem invaders, associated with bacteremia in moribund sharks.
Consistent gross cytological, bacteriological, and histological findings in infected common thresher sharks revealed that the pathogenesis of C. maltaromaticum infection may involve bacterial entry through the endolymphatic skin pores and then passage through the endolymphatic ducts to the inner ears. This is supported by the presence of severe inflammation combined with pathogen isolation and identification in these areas. Short perilymphatic ducts, linking the inner ear with the cranial cavity, could subsequently allow bacteria to spread to the meninges and brain. Pathogenesis in infected thresher sharks differed from that in leopard sharks that stranded in San Francisco Bay in 2017, where the protozoan pathogen M. avidus accessed the brain through the nares. Inflammatory and necrotizing lesions in leopard sharks were prominent in the olfactory lamellae and bulbs, while no inflammation was observed in the inner ears or tissues surrounding the endolymphatic ducts. 24 While a nasal route of entry cannot be totally ruled out (ie, some stranded thresher sharks did have olfactory bulb inflammation), prominent lesions found in the periductal subcutis, inner ears, and dorsal meninges of the brain all support the hypothesis that C. maltaromaticum enters into stranded thresher sharks through endolymphatic pores and ducts that remain open throughout the shark’s life.
Alternatively, meningitis-causing bacteria could originate from the spiral valve. Unfortunately, limitations associated with “short-reads” of the 16S rRNA gene (200–300 bp) prevented the identification of the OTUs at species level in the microbiomes. Therefore, although small traces of Carnobacterium were found in the spiral valves and uterine environment, we cannot confirm that these were actually C. maltaromaticum. Regardless, we can conclude that no species of Carnobacterium appear to be a large part of the microbial community of normal or infected shark spiral valves. Thus, meningitis is unlikely to have resulted from intestinal overgrowth, bacteremia, and breaching of the blood-brain barrier.
Only 1 adult salmon shark and 1 adult thresher shark were found stranded during the course of this study, and neither showed any evidence of Carnobacterium in the extradural fluid or inner ears. In contrast, young salmon, thresher, and mako sharks that stranded consistently had abundant Carnobacterium in the brain, inner ears, and periductal tissues surrounding the endolymphatic ducts. We could speculate that, although at low levels, the presence of Carnobacterium in the uterine environment and the young age of stranded sharks might indicate that the pathogen accesses the embryos through the endolymphatic ducts during gestation or during parturition, and thus sharks may be born infected. Infection with C. maltaromaticum could occur when a female adult shark has sufficient numbers of pathogenic Carnobacterium in the spiral valve, or cloaca, to expose vulnerable fetal sharks either in utero or during parturition. Another possibility is that embryonic sharks are exposed because of Carnobacterium contamination of the uterine environment that occurs when sharks are mating (ie, male sharks transfer pathogenic bacteria to females via contaminated claspers).
All lamniform sharks are regional endotherms2,3,5,6,10 because young lamniform sharks are less capable of maintaining a stable body temperature; abrupt exposure to cold water upwelling events has been cited as a possible contributing factor in salmon shark strandings. 8 It is thought that the combination of the sudden exposure to cold water and the lower thermal inertia of the young sharks potentially results in sufficient physiological stress to suppress the immune system, thereby making them more vulnerable to the invasion of opportunistic pathogens. 8 We could additionally speculate that not only does thermal shock predispose young lamnoid sharks to C. maltaromaticum infection, but also that colder seawater may harbor more of the pathogenic bacteria, as C. maltaromaticum is known to inhabit colder and deep oceanic waters. 18
When compared with relative high frequency of annual strandings of juvenile salmon sharks (eg, as many as 50 in 2018—CA Department of Fish and Wildlife personal communication), strandings of common thresher sharks are more irregular (eg, only one confirmed CA stranding in 2018). Furthermore, infected thresher sharks strand at an older age (1–3 years), compared with salmon sharks that typically strand when less than a year old. The cause for the difference in age at stranding for these 2 species is unknown.
This study details the likely pathogenesis of C. maltaromaticum in stranded common thresher sharks, with a focus on the endolymphatic ducts as probable conduits of pathogen entry into the inner ear and brain. We hope that it will serve as a guide for the investigation of future studies of shark diseases by using cytology, histology, bacteriology, and molecular biology techniques, as the cause of elasmobranch stranding events often remains elusive. 31 It is difficult to determine the impact that this disease has on wild shark populations because comparatively little is known about the cause of C. maltaromaticum otitis and meningoencephalitis in sharks, the source of infections, progression of the disease, or the proportion of individuals that strand (ie, lacking a swim bladder, many infected sharks may simply sink and never reach the coast). Further studies should focus on determining where reservoirs of pathogenic strains of C. maltaromaticum reside (eg, baitfish species, intestinal tracts of healthy sharks, or cold marine waters at depth) and tracking environmental sources of this specific C. maltaromaticum strain by screening metagenomic databases.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221102600 – Supplemental material for Carnobacterium maltaromaticum associated with meningoencephalitis and otitis in stranded common thresher sharks (Alopias vulpinus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221102600 for Carnobacterium maltaromaticum associated with meningoencephalitis and otitis in stranded common thresher sharks (Alopias vulpinus) by Laura Martinez Steele, Mark S. Okihiro, Renaud Berlemont, Jesse G. Dillon, Kelly A. Young, Shohreh Hesami, Sean Van Sommeran and Christopher G. Lowe in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the recreational fish community and the Pfleger Institute of Environmental Research (Oceanside, CA) who donated a lot of their thresher shark catches. Thanks also to the staff at the National Oceanic and Atmospheric Administration (NOAA) for taking cloacal swabs during their annual thresher shark cruise. We are equally grateful to those people who took the time to collect and send us stranded sharks such as Mike Harris and Dayv Lowry from the California and Washington Department of Fish and Wildlife, respectively, and to the staff at the Marine Mammal Center in Sausalito (San Francisco, CA).
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received financial support through several small grants/awards. The sources for the funds originated from the following groups: Los Angeles Rod and Reel Club, The Southern California Academy of Sciences, The Southern California Tuna Club, and The Council on Ocean Affairs of Science and Technology.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
