Abstract
Squamous cell carcinoma represents 47.4% of all malignant canine digital lesions, but despite its frequency, there are few published studies available. Pathology submission records of 154 cases and follow-up of 49 animals were analyzed. On the 49 cases, histological evaluation was performed of the differentiation degree, mitotic index, presence of emboli, and immunohistochemical expression of vimentin and E-cadherin. The mean (SD) age of affected animals was 10.2 (2.3) years; no sex predisposition was recorded. Beauceron and Briard were 2 new overrepresented breeds. Dark-haired animals comprised 97 of 105 (92%); 94 dogs of 125 (75.2%) belonged to large and giant breeds. The forelimb was affected twice more than the hind limb. Probable metastases were observed in 4 dogs; new tumor development was recorded in 11 of 49 (22.4%). Epidemiologic factors, histological grade, mitotic index, and expression of immunohistochemical markers seemed not to be related to the clinical outcome.
Keywords
Malignant neoplastic processes account for 53.5% of all digital lesions in dogs, 15 and squamous cell carcinoma (SCC) is the most commonly diagnosed tumor. Affected animals have a mean age range between 7 and 9.8 years, depending on the study. 7,15 No sex predisposition has been described, and some breeds have been reported as overrepresented, particularly Rottweiler, Giant Schnauzer, Standard Poodle, and Dachshund. 4,11,12 Multiple digits can be affected, 3,4,12 and metastases have been recorded, with a prevalence ranging from 5% to 25% to local lymph nodes and lungs. 8,9,11,15
Although canine digital squamous cell carcinoma (CDSCC) is a frequent tumor, with an incidence of 47.4% of all digital malignant lesions, 15 clinical information is limited. The veterinary literature of digital SCC consists of 4 articles and 3 case reports, 5,8 –12,15 and thus epidemiological data may be considered incomplete, and no analyses of prognostic factors have been reported, to the best of our knowledge. CDSCC is histologically similar to SCC arising in other parts of the body, 3 but the application of histological grade criteria for cutaneous SCC does not predict the development of metastasis or multicentric tumors. 10
The aims of our study were (1) to provide a better characterization of the epidemiology of this tumor, (2) to better characterize the clinical outcome, and (3) to identify clinically relevant prognostic factors.
The pathology submission records of 154 biopsy specimens with histologically confirmed CDSCC were collected from the archives of the Laboratoire d’Anatomie Pathologique et d’Histologie of Vetagro Sup between January 1, 2000, and June 30, 2010, and from the archives of IDEXX Laboratory, Alfort, between January 1, 2009, and June 30, 2010. Age at surgical excision, sex, breed, hair color, dog size, and tumor location were analyzed.
A questionnaire about the follow-up and the development of multiple tumors was sent to all submitting veterinarians, and 49 replies were received. The answers to the questionnaire were obtained in May 2012, and thus the follow-up ranged from 23 to 28 months. Data concerning survival, the eventual cause of death, the presence or absence of metastasis, and new tumor development (occurrence, number of developed tumors, disease-free period) were analyzed.
For the 49 cases in which follow-up data were available, 1 or more paraffin blocks derived from the same mass or multiple masses (5 cases) were obtained. For each block, 4 serial sections of 4 μm each were cut; the first one was routinely stained with hematoxylin and eosin (HE), and the other 3 were used for immunohistochemical analysis. For each HE slide, the percentage of well, moderately, and poorly differentiated areas of the tumor was evaluated; a global differentiation score for each tumor was calculated applying this formula: %W*3 + %M*2 + %P*1, where %W, %M, and %P are respectively the percentage of well, moderately, and poorly differentiated areas for each tumor (the obtained score ranges from 100 for the completely poorly differentiated tumors to 300 for the completely well-differentiated neoplasms). Mitotic index was calculated by counting the number of mitoses in ten 400× high-magnification fields. The presence or absence of emboli was recorded. In 2 cases, the local lymph node was available and was examined for tumoral emboli or metastasis.
To evaluate the acquisition of a mesenchymal phenotype, which is correlated with an increased ability to infiltrate and metastasize, we examined tumoral cells for immunohistochemical expression of vimentin (dilution 1:50, clone V9; DakoCytomation, Carpinteria, CA) and E-cadherin (dilution 1:100, clone NCH-38; DakoCytomation). Negative controls used diluent in place of the primary antibody, and positive controls were internal in each slide (normal epidermis and dermis). The percentage of vimentin and E-cadherin–positive tumoral cells was semi-quantitatively calculated, and a score was given for each area of differentiation as follows: for vimentin, the applied score was 0 for 0% to 10%, 1 for 11% to 40%, 2 for 41% to 70%, and 3 for 71% to 100%. For E-cadherin, it was 0 for 0% to 5%, 1 for 6% to 25%, 2 for 26% to 50%, 3 for 51% to 75%, and 4 for 76% to 100%. 2,13
All statistical analyses were carried out with R (R Development Core Team, Vienna, Austria). A P value <.05 was considered statistically significant. Clinical outcome was also compared for the group of animals that developed only 1 tumor with the group of animals that developed more than 1 tumor. To test the effects of age, sex (male or female), breed, hair color (light, dark, tricolor, or black and white), and size (small, middle, or large) on the multiplicity of tumors, Pearson χ 2 tests were used. The localization of tumors (forelimb or hind limb, left or right, and the specific digit affected) was also analyzed by Pearson χ 2 . To test the effects of the global differentiation score, mitotic index, and vimentin and E-cadherin expression on the multiplicity of tumors, analyses of variance were used. Means (SD) were expressed.
Results
At the time of the first tumor submission, the age was available for 133 of 154 animals. The mean (SD) age was 10.2 (2.3) years, ranging from 4 to 16 years. The sex was known for 149 of 154 animals. Sixty-nine were females (33 spayed, 35 intact, and 1 unknown) and 80 were males (6 castrated, 71 intact, and 3 unknown). On average, females were 10.2 years old at first surgical submission, whereas males were 10.0 years old.
Breed information was available for 152 of 154 animals. We registered 30 different breeds, with the most represented breeds being Rottweiler (29 animals + 3 crossbred Beauceron), Schnauzer (24), Beauceron (19), Briard (11 + 2 crossbred), and Miniature Poodle (9 + 1 crossbred). Ninety-four percent (93.8%) of known cases (121 animals) had dark coats, and 3.9% had light coats (5 dogs) (2 animals were tricolors, 1 was black and white, and 41 were of unknown coat color). Considering the size, on the basis of the breed, 75.2% (94 of 125) of affected animals belonged to large and giant breeds, whereas 16.8% (21 of 125) were small breeds.
Forelimbs presented an incidence of affected digits of 66.9% (97 of 145), which was double the rate compared with the hindlimb (33.1%) (48 of 145). Nevertheless, there was no difference between right vs left limbs or among the different digits.
Survival information was available for 49 cases. Among them, 61.2% (30) dogs were dead when the questionnaire was answered, 36.7% (18) were alive, and 2% (1) were alive and lost to follow-up 13 months after the surgery. Among the dead animals, 20% (6) died of a cause directly related to CDSCC: in 2 cases, the owner decided for euthanasia because of new tumor development and in 4 cases because of probable metastasis (no necropsy was performed to confirm the metastatic disease). The euthanasia in the probable metastatic disease occurred within 1 year of the surgical excision. Thirteen of 154 animals had multiple tumors. Considering the 49 cases for which follow-up was available, 22.4% (11 cases) developed new tumors on other digits: 8 animals developed 2 neoplasms in total, 2 animals presented 3 tumors, and only 1 animal had 4 new CDSCC. For the disease-free period, 6 new tumors appeared after 1 month and 4 after 12 months. In the other 5 cases, new tumor development was recorded respectively at 4, 7, 9, 10, and 24 months after the previous excision.
Histologically, the observed tumors were located in the superficial, mid, and deep dermis, often invading the adjacent bone. Nail bed origin was evident in some samples (14), but serial sections were not performed in the other cases. The tumor was multilobular, not encapsulated, and moderately cellular. Three degrees of differentiation could be observed. 7 In the well-differentiated areas, cells arranged in a lobular pattern showed an evident squamous differentiation with formation of abundant keratin in the lobule center (Fig. 1). In the moderately differentiated areas, lobules were smaller and scant keratin was present in the middle. The poorly differentiated areas were characterized by an anaplastic morphology: cells were arranged in cords or were isolated; they were polygonal and smaller in size, with an amphophilic cytoplasm. Neither keratin formation nor dyskeratosis was present (Fig. 2). Neither cellular nor architectural morphology reminiscent of a spindle cell tumor was observed. The stroma was moderate in amount and infiltrated by lymphocytes and plasma cells. When keratin debris was free in the dermis (due to the rupture of tumoral lobules containing keratin), an intense pyogranulomatous reaction was present as well as new vessel formation. Tumors often showed more than one differentiation type. Considering all the examined tumors, the mean tumor proportions comprising well, moderately, and poorly differentiated areas were respectively 61.2%, 27%, and 11.8%. The poorly differentiated tissue, when present, was located mainly at the invasive margin of the tumor (Fig. 2). It represented 100% of the section in only 2 cases. For 4 categories of the global differentiation score (100–150, 151–200, 201–250, and 251–300), 2, 10, 11, and 30 cases were respectively recorded. Mitotic index ranged from 0 to 46. In 4 cases, we observed neoplastic emboli in the superficial and mid lymphatic vessels (Fig. 1), and in 1 case, neoplastic cells were present in the capsular sinus of the submitted lymph node. Emboli were very different in morphology: they could be small or large, composed of few or several (up to 50) cohesive cells characterized by a polygonal shape and severe anisocytosis and anisokaryosis; dyskeratosis was occasionally present. Some were completely anaplastic, whereas others contained some cells with a squamous epithelial phenotype. In 1 case, neoplastic emboli were found at cytological examination of fine-needle aspirates from the draining lymph node; the histology was done on a biopsy specimen where a scant amount of tumoral cells was present and no lymphatic vessels were visible. The global differentiation score of cases with emboli were 300 in 2 cases; 270, 240, and 190 in 3 cases; and 100 for 1 case.
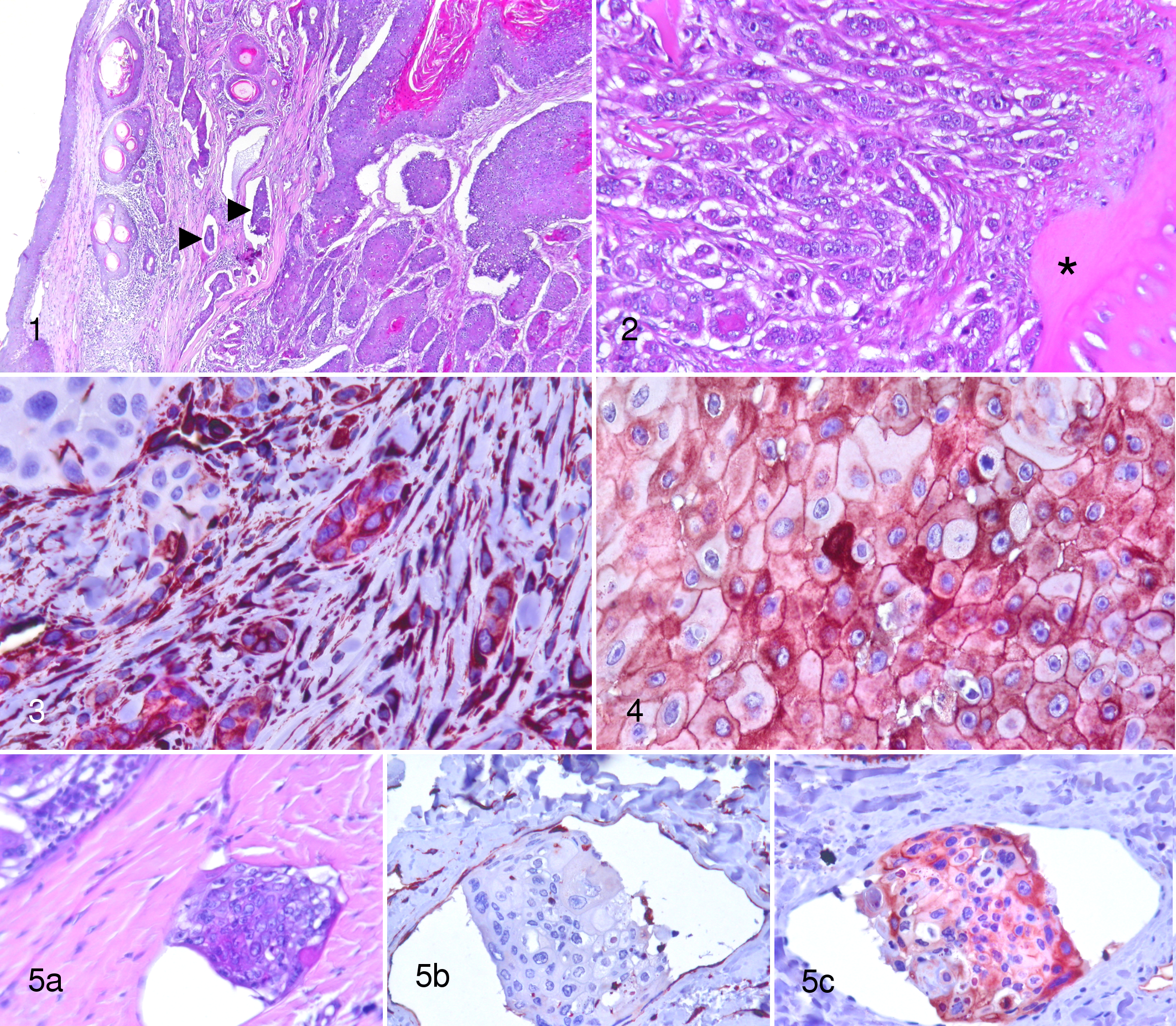
For immunohistochemical analysis, all tumors were stained for pancytokeratin AE1/AE3 to confirm the epithelial origin of the neoplasia, and all tumoral cells evaluated for vimentin and E-cadherin expression were AE1/AE3 positive (data not shown). In 4 cases, vimentin expression was not evaluable because of a technical problem or the presence of too many inflammatory cells. In the other cases, vimentin expression was more frequent in the peripheral, invasive front of the tumor (Fig. 3). The intensity was mostly moderate, but some strongly positive cells were observed. All emboli were negative (Fig. 5b). For the E-cadherin immunostain, 6 cases were excluded because the epidermis (internal positive control) was unevenly stained or not stained at all. In the remaining cases, the intensity of the membranous staining was lower than in the epidermis. Occasionally, cytoplasmic staining was observed in all 3 degrees of differentiation (Fig. 4). We could evaluate only 3 cases with emboli, because in the others they were not present on the available sections. The E-cadherin antigen was detected in more than 70% of the cells within the emboli, with some presenting a cytoplasmic location (Fig. 5c).
Cases with multiple tumors compared with the other animals were not statistically different for age, sex, breed, hair color, size, location, differentiation, mitotic index, and E-cadherin expression. Vimentin expression was statistically increased in cases with multiple tumors compared with the others (P < .05).
Discussion
CDSCC is the most common tumor on the dog’s digit. 15 It has some peculiar characteristics, most notably its multicentric development 12,15 and a potential for metastatic disease, with a prevalence, recorded to local lymph nodes and lungs, that varies among different studies from 5% to 25%. 8,9,11,15 Owners confronted by multiple consecutive tumors or distant metastasis often choose euthanasia of their dogs. To the best of our knowledge, few data are available on multicentric tumors, and the only factor reported to predict metastatic disease is histological grade, which does not seem to be reliable. 10
In our study, affected animals had a mean age of 10.2 years without a sex predisposition, as reported in the literature. 7,9,11,15 A breed predilection for large breed, black-coated dogs has been reported. 7,11,12,15 In our study, the most commonly represented breeds were Rottweiler and Schnauzer. Interestingly, we found a high percentage of Beauceron and Briard, and this has not been described before: the present study was conducted in France, where these breeds are quite common compared with other nations. Unfortunately, we did not have access to the proportion of these breeds among the entire canine population in France for the studied years to estimate a real breed predisposition.
Seventy-five percent of affected animals in our study were large or giant. This has already been reported in literature, 4 but it is the first time that a prevalence in the studied population has been provided. In previous reports, neither digit nor limb predisposition was found. Wobeser and colleagues 15 first described a higher incidence of digital lesions in the forelimb but without specifying the type of tumor. In our study, digits of the pectoral limb were 2 times more affected than digits of the pelvic limb; new studies would be needed to explain this predisposition.
Ninety-four percent of the dogs in our study had a dark coat (black, gray, or black and tan), even in affected small-size animals, confirming that there is a connection between the development of this kind of tumor and hair color. However, we did not know the nail color, which could have a major role in the development of SCC of subungual origin.
Among the 6 cases with emboli, 4 died because of problems related to the tumor within 1 year of the diagnosis, 1 animal died 2 years later because of poor body condition, and 1 was still alive 3 years later. Marino and colleagues 9 reported that the survival rate of non-subungual digit SCC was less than that of subungual SCC. We were only able to confirm nail bed origin in 14 cases. Nevertheless, subungual origin was not confirmed in any cases with intravascular emboli. Although our survival rate could be biased by this fact, our data suggest that CDSCC has a scant risk of emboli and a lower risk of metastasis. We can speculate that although some tumoral cells can have a malignant phenotype, expressed by the differentiation degree, the mitotic index, and mesenchymal features, they may not have acquired yet all the characteristics that render them prone to develop metastases. Thus, an early surgical treatment is curative in most cases.
Multicentric tumor development, from our study, seems to be more frequent than metastasis-related death. We found a multicentric pattern in 22.4% of animals, and most developed within 1 month of the initial surgical excision, although new tumor development was recorded up to 2 years afterward. In our study, no epidemiological factors were linked to an increased predisposition to develop multiple tumors.
Taking into account the animal breed and size, location predisposition of the tumor, and the multicentric feature of the CDSCC, we can speculate that there is an important genetic component related to the breed and/or hair color, on which repeated micro-traumatic injuries can play an important role to allow, on several epithelial cells of different digits, the genetic modifications necessary to acquire a tumoral phenotype. New investigations on the genetics and the tumoral microenvironment may lead to an improved understanding of carcinogenesis in these patients.
The grading system for SCC is based on histological differentiation of the tumor. 3 We observed that in a single sample, different grades of differentiation were present, with well-differentiated tissue being most represented. Nevertheless, neither the differentiation nor the mitotic index was statistically significantly different for multicentric tumors compared with the others. Cases with metastasis and emboli were too few for statistical analysis, but our data indicate that they cannot be predicted by histology. Thus, the current grading system seems not to be a useful marker to define CDSCC prognosis, as suggested by Meleo. 10
Epithelial-mesenchymal transition (EMT) is defined as the loss of epithelial characteristics and the acquisition of a mesenchymal phenotype in epithelial cells. It is reversible and occurs during embryogenesis (EMT type 1), in wound healing (EMT type 2), and in carcinogenesis (EMT type 3). 1 In neoplasia, it is considered essential for tumoral cells for the acquisition of invasive properties. Cells lose expression of epithelial markers, such as cytokeratins and E-cadherin, and acquire a mesenchymal phenotype, expressing vimentin, N-cadherin, and other markers. 6,14 We evaluated the percentage of tumoral cells positive for vimentin and E-cadherin, to identify a malignant phenotype and link it to prognosis. In some CDSCC, we found a mesenchymal phenotype, which was linked to the differentiation degree of tumoral cells. The less differentiated cells, mostly at the periphery of the neoplasia, on the invading edge, were vimentin positive and E-cadherin negative. Statistical analysis demonstrated that multiple tumors, in our study, had an increased amount of tumoral cells positive for vimentin but not for E-cadherin. Although their prognosis seems to be no different from solitary tumors, this study was not sufficiently statistically powered to identify differences between groups when one considers that all tumors were completely excised.
In conclusion, this study provides new data about the epidemiology of this tumor and the clinical outcome. CDSCC seems to have a benign behavior, despite the histological and the immunohistochemical results. Currently, no prognostic factors have been identified that can help us to predict metastatic disease or new tumor development, but future studies focused on cellular genetic mutations may provide new insight.
Footnotes
Acknowledgements
We thank IDEXX Laboratory Alfort for having provided the case material and all the veterinarians who participated to the study. We also thank Sabine Ballaydier for technical assistance and Dr Gerry Polton for manuscript revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
