Abstract
Cyclooxygenase (COX) enzymes catalyze the synthesis of prostaglandins and exist as two isoforms, COX-1 and COX-2. COX-2 is a potent inducible mediator of inflammation. COX-2 is also upregulated in several human tumors and in canine squamous cell, renal cell, and transitional cell carcinomas, prostatic adenocarcinoma, and intestinal neoplasia. The purpose of this study was to determine whether COX-2 is expressed in various feline tumors. Results of this study may help determine whether COX-2 is a potential target for therapeutic and preventive strategies in cats. Immunohistochemical studies were performed on paraffin-embedded tissues using the amplified streptavidin-biotin-horseradish peroxidase system. COX-2 was found in 7 of 19 (37%) feline transitional cell carcinomas and in 2 of 21 (9%) feline oral squamous cell carcinomas. No COX-2 immunoreactivity was detected in cutaneous squamous cell carcinomas (6), adenocarcinomas (nine mammary, eight pulmonary, seven intestinal), lymphomas (six nasal, six intestinal), or 10 vaccine-associated sarcomas. The widespread absence of COX-2 expression in most feline neoplasms might suggest that COX-2 inhibitors would have a low potential as anticancer agents.
In the past two decades, numerous experimental, clinical, and epidemiologic studies have linked tumor development and progression with the presence of cyclooxygenase (COX) in tumor cells in humans. COX enzymes exist as two different isoforms and catalyze conversion of arachadonic acid to an array of prostaglandins. COX-1 is expressed constitutively in many cells and is involved in normal physiologic activities such as cytoprotection of the gastric mucosa, regulation of renal blood flow, and platelet aggregation. COX-2 is an inducible enzyme that is involved in the production of prostaglandins, which modulate pathologic events such as inflammation, wound healing, and neoplasia. A variety of agonists including cytokines, growth factors, and oncogenes can induce COX-2 expression. 7 COX-2 immunoreactivity is found in a number of human tumors including colon, lung, breast, gastric, prostatic, head and neck, and bladder carcinomas. 7,20 More recently, COX-2 has been demonstrated in canine squamous cell, renal cell, and transitional cell carcinomas, prostatic adenocarcinomas, and intestinal neoplasias. 4,8,9,12,15,21
Recent studies also demonstrate that nonsteroidal anti-inflammatory drugs (NSAIDs), which inhibit COX enzymes, can reduce the incidence of cancer in humans and experimental animal models and may be potential targets for therapeutic and preventive strategies. 7,11,13,18,20 Importantly, the relative risk of developing epithelial cancer, including colonic carcinoma, is lower in persons receiving aspirin therapy. 7,18,20 In veterinary medicine, dogs with transitional cell carcinoma of the urinary bladder may have complete or partial reductions in tumor size when treated with piroxicam, a potent NSAID. 10,13 Recently, antitumor activity was reported in dogs with oral squamous cell carcinoma treated with piroxicam. 17
The objective of this study was to characterize the expression of COX-2 in a variety of feline neoplasms. Results of this study may help determine whether COX-2 is a potential target for therapeutic and preventive strategies in cats with tumors.
Materials and Methods
Biopsy samples selected for inclusion in the study were obtained from the surgical pathology and necropsy services of the Department of Biomedical Sciences, Cornell University. Biopsy samples from cats with squamous cell carcinomas (
Immunoperoxidase (biotin-streptavidin) staining was performed on all sections using a rabbit polyclonal antibody to COX-2 (PG27B, Oxford Biomedical Research, Oxford, MI). Four-micron-thick tissue sections mounted onto poly-
Immunoreactivity was evaluated in a blinded fashion (SLB), which assessed grade (percentage of positive cells) and staining intensity. Five 10×; fields from each slide were evaluated. The grade was evaluated by the following scoring system: 15 0 = negative; 1 = <10% of cells staining positive; 2 = 10–30%; 3 = 31–60%; 4 = >60%. Intensity was evaluated by the following scoring system: 0 = negative; 1+ = weak staining; 2+ = moderately intense staining; and 3+ = intense staining.
Results
Seven of 19 (37%) cases of transitional cell carcinoma of the urinary bladder displayed positive immunoreactivity for COX-2. All transitional cell carcinomas were well differentiated with varied patterns including papillary and infiltrative (3); nonpapillary and infiltrative (3); and papillary with occasional glandular differentiation and infiltrative (1). The overall pattern of cellular staining was random. Often, the positive epithelial cells were arranged individually or in small clusters. In one sample, numerous smooth muscle cells within the tunica media of a few blood vessels stained with moderate intensity (Fig. 1). In another sample, there were a few scattered positive-staining fibroblastic cells in the schirrous stroma associated with the neoplastic epithelium. The accumulation of COX-2 within neoplastic transitional cell epithelium was located diffusely within the cytoplasm (Fig. 1). In several weakly stained cells, the area around the nuclear envelope was stained, whereas the cytoplasm showed negative or very poor staining. COX-2–positive staining within neoplastic epithelial cells was grade 1 (<10%) in six cases and grade 2 (10–30%) in one case. Staining intensity varied between mild and intense with four of six being moderately intense (2+). Two of 21 samples (9%) of oral squamous cell carcinoma stained positive for COX-2, with less than 10% (grade 1) of neoplastic epithelial cells staining positively. Staining intensity was 1+ and 2+, respectively. The remaining feline tumors evaluated were all negative for COX-2 immunoreactivity (Table 1). Numerous inflammatory cells within granulation tissue at ulcerated surfaces in two oral squamous cell carcinoma, two intestinal lymphoma, and one intestinal adenocarcinoma specimens stained positively for COX-2. Although no special stains were used to identify the cells, the morphology of the inflammatory cells was consistent with that of macrophages or myofibroblasts (Fig. 2). The staining pattern for these cells was similar to the predominantly diffuse cytoplasmic staining of the positive transitional cell carcinoma cells. Staining was absent in negative controls. The six canine tumors, used as positive controls, consistently stained positively. Three canine controls were of grade 2, and three controls were of grade 3. Staining intensity was 3+ in all canine tissues.
Cyclooxygenase-2 (COX-2) Immunoreactivity in feline tumors by location and diagnosis.

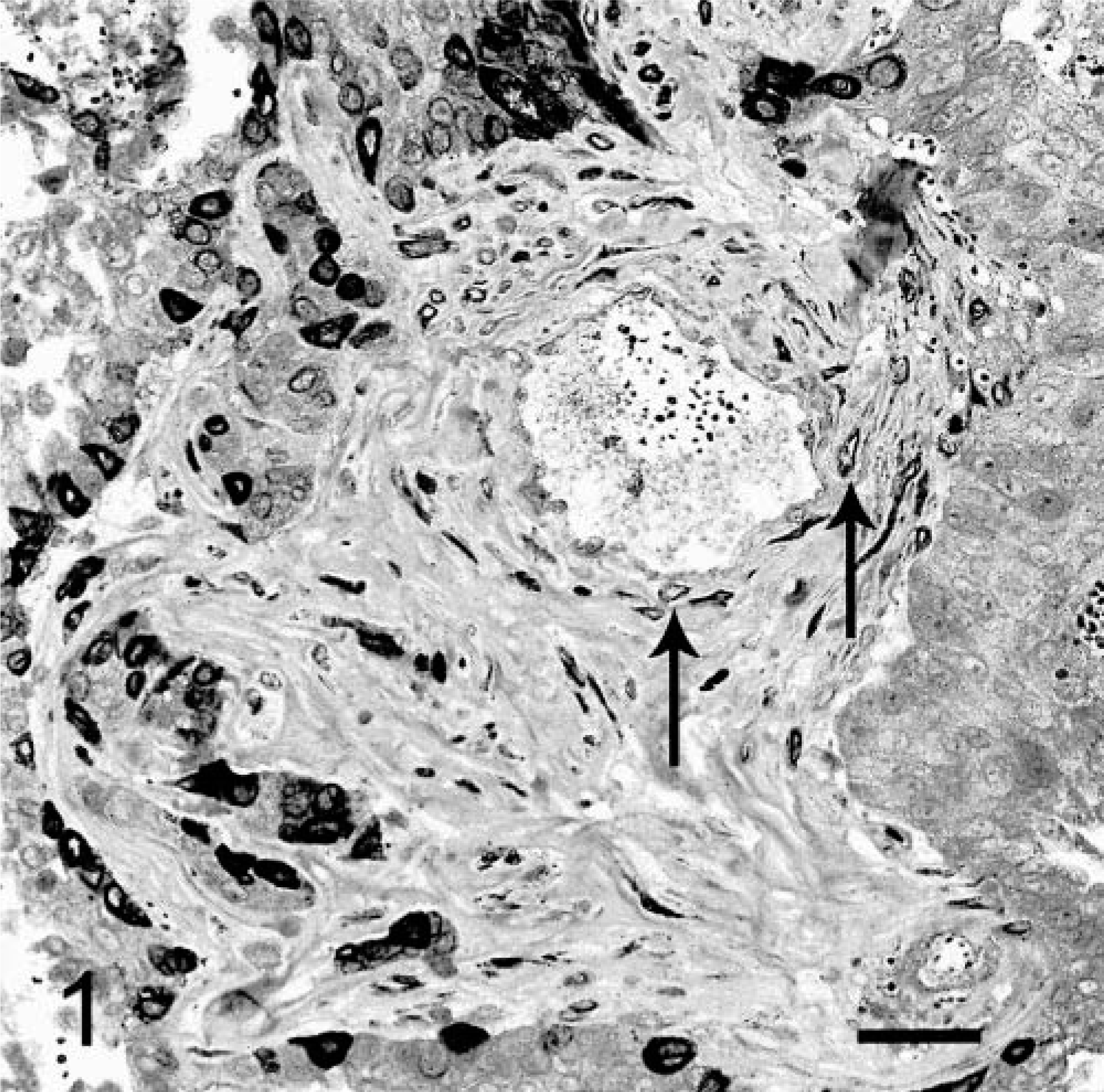
Urinary bladder. Nests of neoplastic transitional epithelial cells are intensely positive for COX-2 immunoreactivity, which is localized diffusely within cytoplasm. Numerous positive-staining smooth muscle cells in the tunica media (arrows) of many intratumoral vessels. DAB chromagen, Gill's hematoxylin counterstain. Bar = 40 µm.
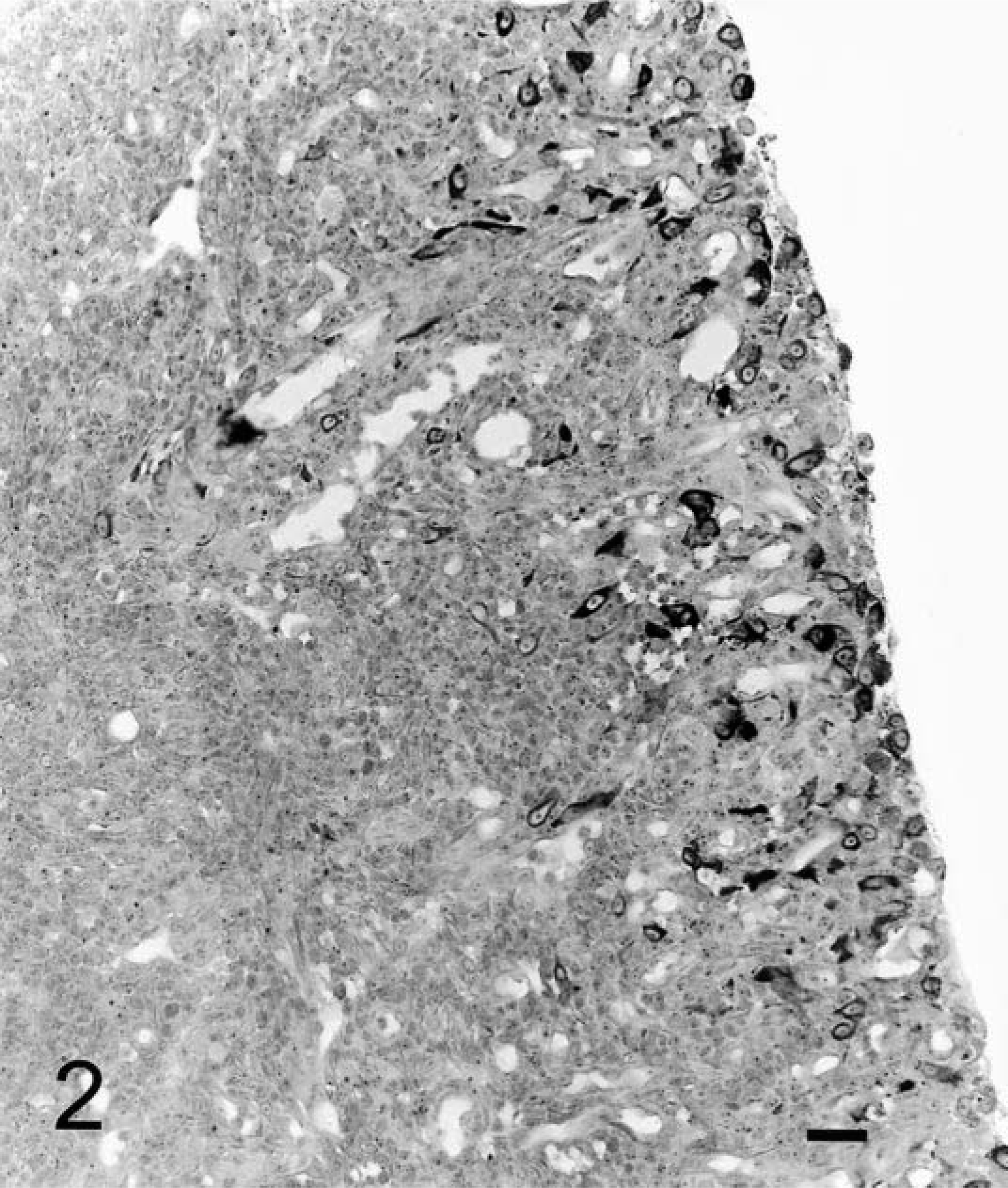
Jejunum. Nonneoplastic COX-2–positive staining cells, macrophages, and myofibroblasts based on HE morphology, located exclusively at ulcerated surfaces of intestinal lymphoma in areas of developing granulation tissue. COX-2 immunoreactivity in the granulation tissue cells was diffusely cytoplasmic, similar to the neoplastic transitional carcinoma cells in Fig. 1. DAB chromagen, Gill's hematoxylin counterstain. Bar = 228 µm.
Discussion
On the basis of the immunohistochemical results of this study, it was found that COX-2 was expressed in over a third of feline transitional cell carcinomas and in only a small number of squamous cell carcinomas of the oral cavity. COX-2 was not detected in feline mammary carcinoma, pulmonary adenocarcinoma, intestinal adenocarcinoma, cutaneous squamous cell carcinoma, nasal and enteric lymphomas, and vaccine-associated sarcomas. These results are in striking contrast to those in dogs, where COX-2 immunostaining has been demonstrated in the majority (47–100%) of canine squamous cell and transitional cell carcinomas, prostatic adenocarcinomas, and intestinal neoplasias. 4,8,12,15,21
Lack of detectable COX-2 expression in the majority of feline epithelial neoplasms in this study may have been caused by sample size, failure of the antigen retrieval method used, or other confounding variables such as the type of fixative or prolonged fixation. Formalin fixation or tissue processing (or both) destroys or denatures several proteins, thereby altering antigenic recognition by immunohistochemistry. 6,16,19 However, positive staining of seven of 19 transitional cell carcinomas and two of 27 oral squamous cell carcinomas indicates that the COX-2 antigenic epitopes are expressed in cats and are detectable with the polyclonal antibody used. In addition, inflammatory cells (presumptive macrophages and myofibroblasts) at ulcerated sites in five of the feline tumors displayed COX-2 immunoreactivity, which served as positive internal controls. The polyclonal antibody used in this study has detected COX-2 in canine tissues in previous studies 9,13 as well as in control canine tissues used in this study. These results suggest a basic species difference between feline and dog neoplasms in COX-2 expression. It is possible that the polyclonal antibody used may not be a sensitive indicator of COX-2 antigenic epitopes present in the cat. Potentially, other polyclonal COX-2 antibodies or a feline monoclonal antibody could be used to more fully characterize the presence of COX-2 in feline tumors. An alternative explanation is that COX-2 concentrations in feline tumors are below the level of immunohistochemical detection. Although not attempted in the present study, the use of frozen sections for these studies may yield more reliable results. Determination of prostaglandin E2 (PGE2) concentration is commonly used as a marker for COX-2 activity. 5,14 Future studies to evaluate a larger number of feline tumors with these techniques would be useful to confirm the findings of the present study.
The mechanism of action of COX-2 and COX metabolites such as PGE2 in tumors is beginning to be elucidated. Prostaglandins, produced by COX-2 activity, increase cell proliferation, inhibit apoptosis, promote angiogenesis, alter cellular adhesion that allows for metastasis, inhibit immune surveillance, and may activate xenobiotics to reactive substances that are carcinogenic. 7,11–13,18,20,22 Mechanisms responsible for initial upregulation of COX-2 in neoplastic tissues are still unclear.
COX and PGE2 are being evaluated as targets for cancer prevention and treatment. In numerous studies on humans, the chronic use of NSAIDs is linked to a lower incidence of several types of epithelial neoplasms including colon, breast, gastric, and esophageal carcinomas. 7,18,20 COX-2 inhibitors cause reductions in neoplastic cell numbers in vitro and tumor regression in mouse tumor models. 7,18,20 In phase I and II clinical trials, COX-2 inhibitors reduced tumor size in naturally occurring canine transitional cell and squamous cell carcinoma. 10,13,17
This study represents, to our knowledge, the first attempt to evaluate COX-2 expression in feline tumors. The widespread absence of COX-2 expression in most feline neoplasms might suggest that COX-2 inhibitors would have a low potential as anticancer agents for cat. However, COX inhibitors have demonstrated antiproliferative effects in human cell lines that are independent of COX-2 expression. 22 Preliminary studies indicate that NSAIDs may exert anticancer effects by both COX-dependent and COX-independent mechanisms. 11,18 Although the molecular targets of these COX-independent effects are not known, recent studies have identified several signaling pathways, including peroxisome proliferator–activated receptor-γ (PPAR-γ), 15-lipoxygenase-1, and Akt/protein kinase B (PKB), which may potentially play roles in NSAID-mediated anticancer cellular actions. 1 PPAR-γ is a member of a nuclear hormone receptor family that plays a key role in inducing terminal cell differentiation or inducing apoptosis. 2,3 PPAR-γ is also antiangiogenic by mechanisms that involve apoptosis or decreased expression of a vascular endothelial growth factor receptor (or both). 3 15-Lipoxygenase-1 is upregulated by COX inhibition. 15-Lipoxygenase-1 catalyzes the production of 13-S-hydroxyoctadecadienoic acid, which has growth-inhibitory and proapoptotic properties. 22 Finally, the Akt/PKB pathway is a major antiapoptotic pathway. 1 In vitro and in vivo studies have demonstrated that NSAIDs reduce phosphorylation (thus activation) of Akt/PKB pathways by inhibiting 3-phosphoinositide–dependent kinase 1. Further studies of these molecular targets in feline tumors or clinical studies with COX-2 inhibitors are needed to fully evaluate the potential role of COX-2 inhibitors as therapeutic adjuncts in cats with tumors.
