Abstract
A cutaneous melanocytoma-acanthoma in a 2-year-old female German Shepherd Dog was characterized by the presence of two populations of neoplastic cells: epithelial and melanocytic. The epithelial component consisted of nests of well-differentiated stratified squamous epithelium closely associated with neoplastic melanocytes. The epithelial cells immunoreacted with both monoclonal and polyclonal anti-cytokeratin antibodies, and immunoreaction to S-100 protein and vimentin was observed in the melanocytic cells. This rare pigmented skin neoplasm of the dog apparently has a benign behavior.
Melanocytic tumors represent 6–10% of all skin tumors in dogs. 2 , 12 Benign tumors (melanocytomas) occur more frequently in the eyelids and trunk, and malignant tumors (melanomas) are more frequently found in distal extremities. Macroscopic appearance is not useful in establishing either a diagnosis or a prognosis because basal cell and adnexal tumors may be heavily pigmented. 1 Acanthoma is a squamous cell tumor that may be malignant but well differentiated, benign, or nonneoplastic. In humans, it occurs more frequently in a combined form, i.e., keratoacanthoma and adenoacanthoma. In the dog, two types of tumors with an acanthoma component have been described: infundibular keratinizing acanthoma, which is a common cutaneous tumor similar to keratoacanthoma, and melanocytoma-acanthoma, a rare type of tumor described only once in the veterinary literature. 11
Here, we describe the histologic and immunohistochemical characteristics of a canine skin tumor composed of two different cell populations, epithelial and melanocytic. The lesion is compared with other tumors reported in humans and dogs.
A 2-year-old intact female German Shepherd Dog was presented with a solitary, darkly pigmented, firm 1-cm-diameter nodule of unknown duration. The mass was located in the dermis of the dorsal trunk, was dome shaped, had a smooth hairless surface, and was freely movable. No other clinical signs or evidence of metastasis were noted.
A biopsy specimen was fixed in 10% neutral buffered formalin and embedded in paraffin, and 4-µm-thick sections were stained with hematoxylin and eosin (HE) and periodic acid–Schiff. In addition, an immunohistochemical study was performed to establish the immunophenotype of this tumor. The expression of S-100 protein and vimentin and cytokeratin intermediate filament (IF) proteins was analyzed. Five antibodies were used: a polyclonal antibody (PAB) against human S-100 protein (Dako, Carpinteria, CA, USA), a polyclonal anti-vimentin antibody and three different anti-cytokeratin antibodies, one polyclonal, raised against human cytokeratins from epidermis (Eurodiagnostics B. V., Apeldoorn, The Netherlands), and two monoclonal antibodies (MABs), RCK-102 and NCL-5D3. RCK-102 recognizes cytokeratins 5 and 8 of the Moll catalog of human keratins, 8 and NCL-5D3 recognizes cytokeratins 8, 18, and 19 of the same catalog. The avidin–biotin peroxidase complex (ABC) method (Vector, Burlingame, CA, USA) was used with the different antibodies. Deparaffinized sections were incubated with 1% hydrogen peroxidase in methanol for 30 minutes to block endogenous peroxidase activity. Predigestion of tissue sections for 10 minutes with 0.1% pronase (Sigma Chemical Co., St. Louis, MO, USA) was employed for tissues treated with the two MABs. After proteolytic enzyme treatment, the tissues were covered with 10% normal swine (PABs) and rabbit (MABs) sera in phosphate-buffered saline (PBS) for 30 minutes, followed by incubation with the primary antibodies for 18 hours at 4 C. Slides were incubated with the biotinylated swine anti-rabbit immunoglobulin antibody (Dako) diluted 1:200 in PBS containing 1% normal swine serum (PABs) and with the biotinylated rabbit anti-mouse immunoglobulin antibody (Dako) diluted 1:40 in PBS containing 1% normal rabbit serum (MABs) for 30 minutes and with ABC for 60 minutes, all at room temperature. To develop the immunoreaction, two chromogens were used: 3-3′ diaminobenzidine tetrahydrochloride (DAB) and 3-amino-9-ethylcarbazole (AEC) (Sigma). When DAB was applied, sections were bleached using a 0.25% aqueous solution of potassium permanganate to avoid confusion between the melanin pigment and the brown staining of the substrate. The use of a red chromogen, such as AEC, with the MABs was included in the technique to avoid the application of two pretreatments (pronase digestion and bleaching), which were damaging to the histologic sections. Sections were counterstained with Harris's hematoxylin.
Table 1 includes a summary of the immunohistochemical procedures and the results obtained with the different primary antibodies. Negative controls included the substitution of the primary specific reagents for either rabbit and mouse nonimmune sera or PBS. Normal routinely processed canine tissues were used as positive controls for all antibodies employed.
Antibodies, procedures, and results of the immunohistochemical study of a canine melanocytoma-acanthoma.
∗AEC = 3-amino-9-ethylcarbazole; DAB = 3-3’ diaminobenzidine tetrahydrochloride.
†Immunoreaction: - = negative; + = scarce; +++ = intense.
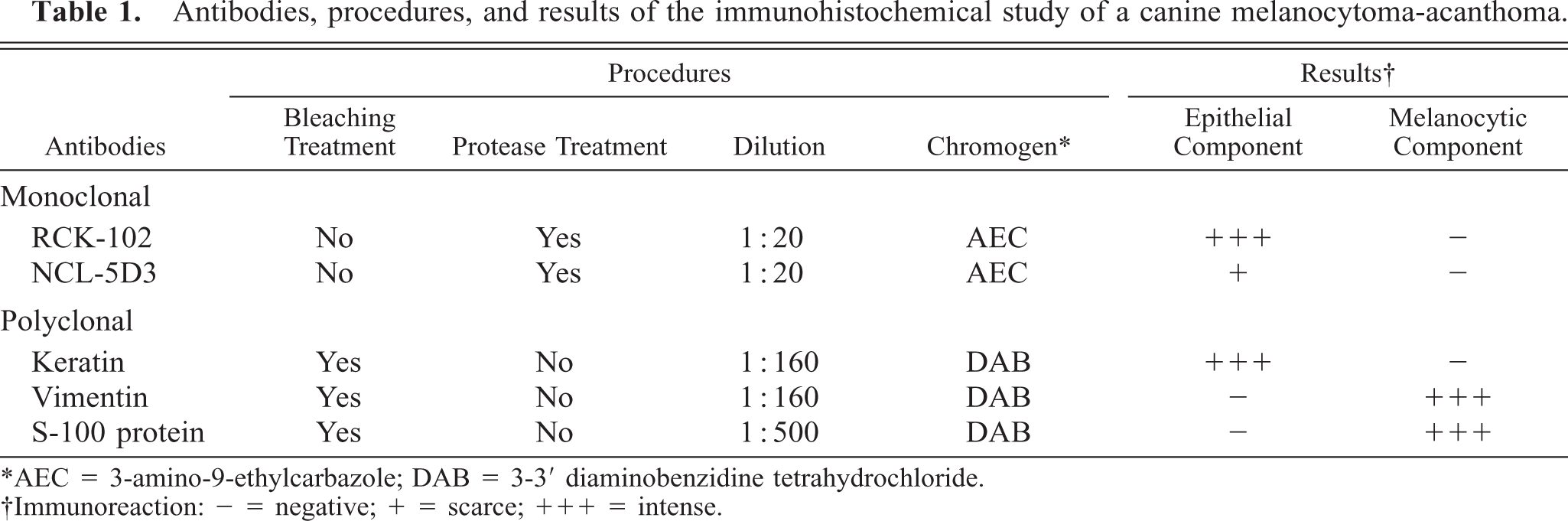
Microscopically, the dermal nodule was focally contiguous with the epidermis (Fig. 1). Two populations of tumor cells were observed: epithelial cells within the dermis and melanocytic cells in both the epidermis and dermis. The epithelial component consisted of nests of well-differentiated stratified squamous epithelium either with gradual keratinization towards the center of the nests or with centers filled with neoplastic epithelioid melanocytes. The basal layer of the epithelial tumor nests showed cordlike expansion into the surrounding compressed dermis but without invasion of tumor cells across the basal lamina. The melanocytic component consisted of intraepithelial nests of epithelioid pigmented cells not only on the basal layer of the overlying normal epidermis but also in the basal layer of the neoplastic epithelial component. In addition, elongated pigmented cells were also observed within the dermis arranged in nervelike whorls surrounded by a delicate fibrovascular stroma or by cords of well-differentiated keratinocytes. These cells were heavily pigmented and uniformly oval. Nuclei were central, oval and uniform, with fine chromatin and very few mitotic figures.
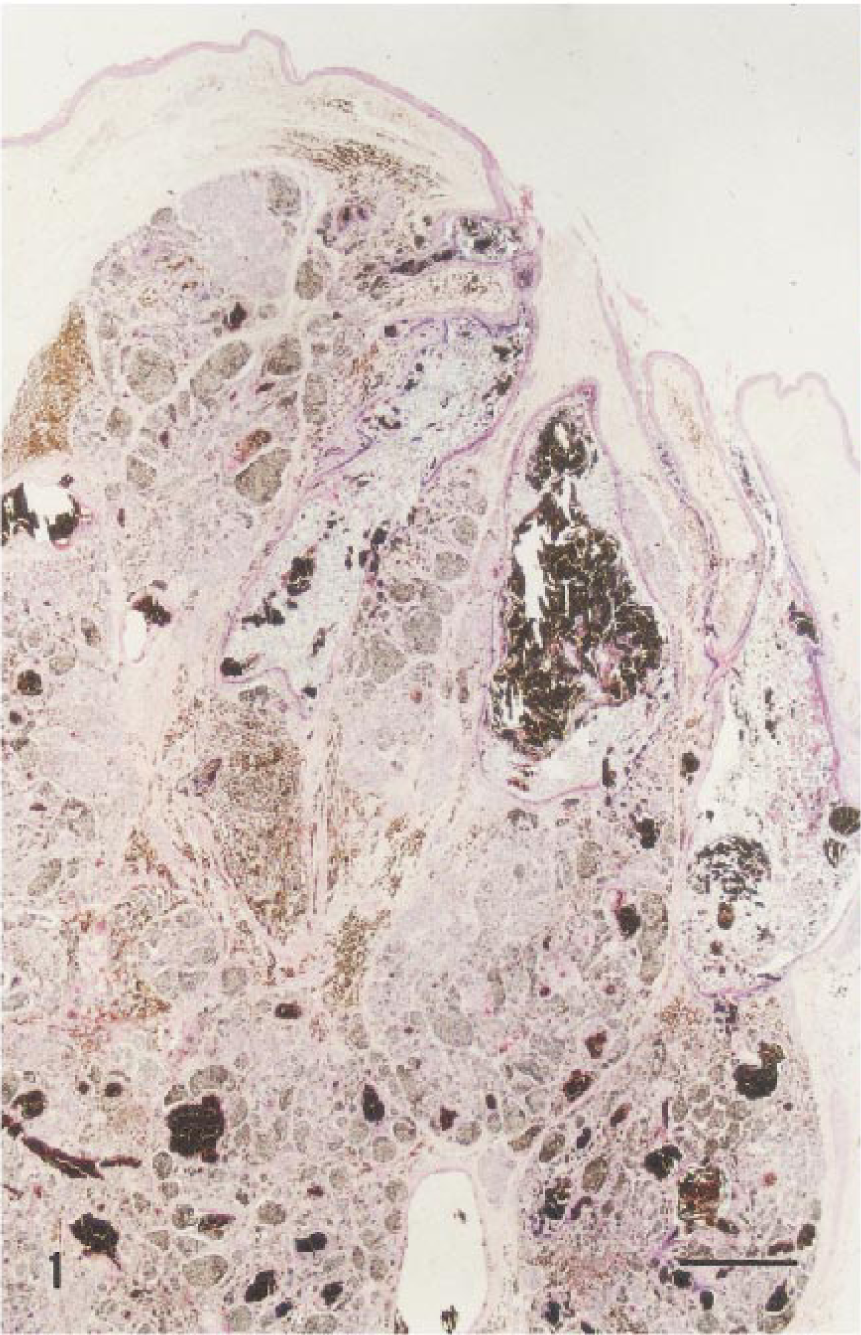
Melanocytoma-acanthoma; German Shepherd Dog. Neoplasm consists of nests of well-differentiated squamous epithelium and nests of epithelioid pigmented cells. HE. Bar = 240 µm.
All three anti-keratin antibodies reacted with the epithelial component of the neoplasm, and the anti-S-100 protein and anti-vimentin antibodies stained the melanocytic component at the epidermal and dermal levels. The staining pattern with anti-cytokeratin antibodies varied according to the degree of differentiation of neoplastic epithelial cells. The Keratin PAB reacted with the more differentiated cells either grouped at the center of the nests or distributed randomly throughout the tumor, whereas MAB RCK-102 reacted with the more basal cells of the epithelial nests and with those surrounding the melanocytic component (Fig. 2). Scattered positive epithelial cells were observed with MAB NCL-5D3. A homogeneous and strong vimentin immunoreactivity was found in the cytoplasm of melanocytic cells and in the stroma of the neoplastic growth and normal tissues. Immunoreactivity to S-100 protein was observed homogeneously in the nucleus and cytoplasm of the melanocytic cells (Fig. 3).
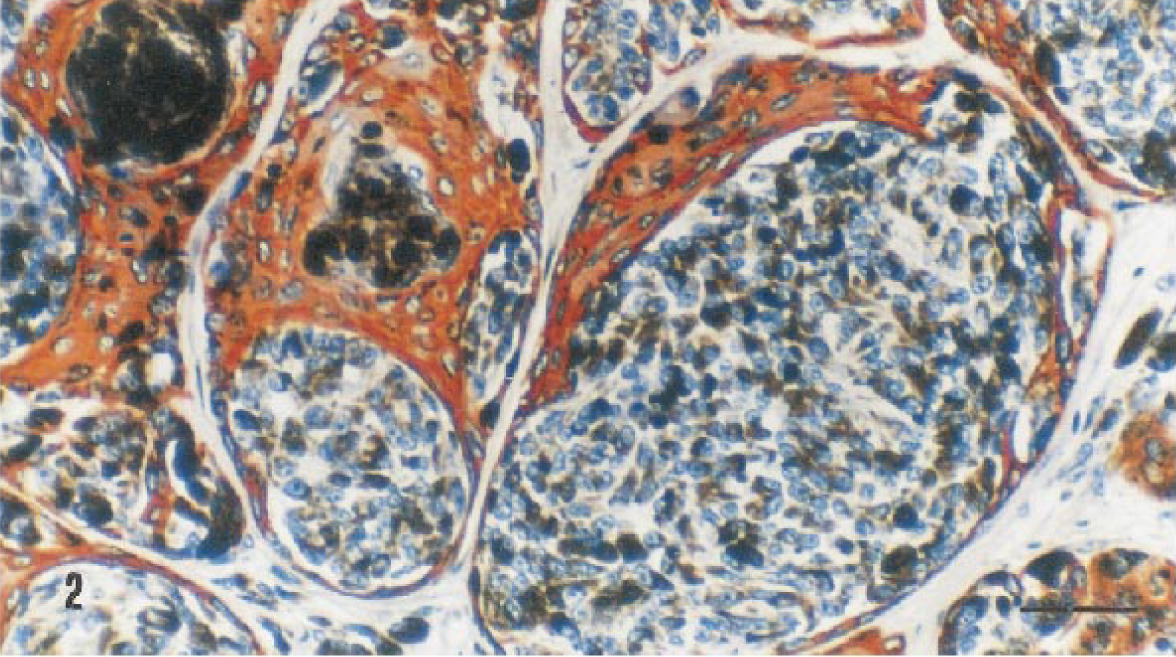
Melanocytoma-acanthoma; German Shepherd Dog. RCK-102 antibody staining reaction in the epithelial nests and in those surrounding the melanocytic cells. ABC method, AEC chromogen, Harris's hematoxylin counterstain. Bar = 25 µm.
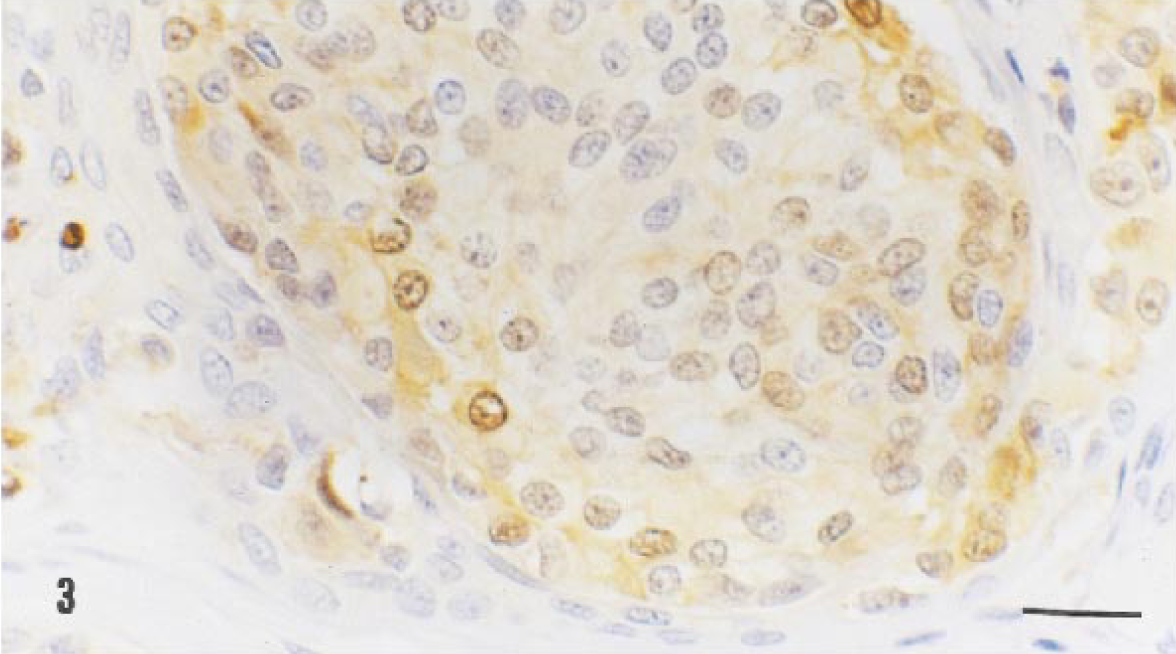
Melanocytoma-acanthoma; German Shepherd Dog. S-100 protein antibody staining reaction in a nest of melanocytic cells. Immunoreactivity is found in both nuclei and cytoplasms of the neoplastic cells. ABC method, DAB chromogen, Harris's hematoxylin counterstain. Bar = 15 µm.
Melanocytoma-acanthoma is a benign mixed tumor of the skin composed of neoplastic melanocytes and squamous epithelial cells and has been previously described in the dog. No data concerning breed, age, or sex were available in the cases of melanocytoma-acanthoma previously reported. 11 The growth pattern and cell morphology of this tumor have some features in common with trichoepithelioma, tricholemmoma, and canine infundibular keratinizing acanthoma. 3 , 11 However, pigmented trichoepitheliomas and tricholemmomas lack the intradermal melanocytic components; pigmented cells observed in these lesions are macrophages or melanized epithelial cells. 12 The most common type of melanocytoma in the dog is the compound (intraepidermal and dermal) melanocytoma. 9 The tumor described here has the histologic features of compound melanocytomas, and the immunophenotype of the tumor cells is characteristic of melanocytes. 11 The blue nevus and the pilar neurocristic melanoma, described in humans and dogs, respectively, 1 result from the proliferation of melanocytic neoplastic cells with a marked perifollicular orientation but without the epithelial neoplastic component seen in the present tumor. The epithelial component of the tumor described here is a well-differentiated squamous epithelial growth with a distribution pattern of cytokeratins similar to those described in humans, dogs, and cats in the normal epidermis and epidermis-derived tumors; 4 , 5 this cytokeratin pattern is not inconsistent with hair follicles and/or follicular neoplasms. 4 The lesion described in this dog closely resembles the acanthotic variant of seborrhoeic keratosis observed in human. 6 This skin lesion is a benign tumor with a typical shape of an epidermal plaque, 6 whereas the melanocytoma-acanthoma described previously and in this dog are intradermal nodules. 12 Histologically, seborrohoeic keratosis is characterized by proliferation of basaloid cells with squamoid differentiation accompanied by a wide infiltration of melanocytes and is occasionally referred to as a melanoacanthoma. 10 In contrast to the lesion in this dog case, in which the epithelial and melanocytic components were equally present in the tumor, the epithelial cells of the lesion in seborrhoeic keratosis are the predominant component. Malignant changes with seborrhoeic keratosis, represented by basal cell carcinomas and squamous cell carcinomas, have been documented in humans. 7
Because this tumor occurred in only one dog and there is little clinical data in the previous report, no conclusions concerning age, breed, sex, or site predilections can be made. The dog was in good health 2.5 years after tumor excision, and there was no evidence of local regrowth or metastasis. There appear to be no clinically distinctive features of canine melanocytoma-acanthoma. The diagnosis of this neoplasm can be based on the histologic appearance of the tumor, but the characteristic immunohistopathologic findings can be used to differentiate the two different neoplastic cell populations occurring at the same anatomical site.
This is the first time that the two cell populations of this recently recognized canine neoplasm have been described immunohistochemically. In addition, it is the first report of dermal melanocytoma-acanthoma with long-term clinical follow-up (2.5 years without evidence of recurrence). This neoplasm thus far appears to have benign behavior but, as described in the seborrhoeic keratosis of humans, it could later develop malignant changes represented by cutaneous carcinomas or melanomas.
Footnotes
Acknowledgements
We thank P. Castro (Department of Comparative Pathology) for technical assistance.
