Abstract
Assessment of the skin tumor–promoting potential of 12-O-tetradecanoylphorbol-13-acetate (TPA) after initiation with 7,12-dimethylbenz[a]anthracene (DMBA) was conducted using rasH2 transgenic (Tg) mice and their nontransgenic (non-Tg) littermates. Mice were treated with DMBA (50 μg/100 μL acetone) on clipped back skin at the commencement of the study, and 1 week thereafter, TPA was applied at 8 μg/200 μL or 4 μg/200 μL acetone, once or twice weekly, for 7 weeks. Skin nodules were observed in the rasH2 Tg mice from week 4, and the incidence reached 100% at weeks 5 and 6. The number of skin nodules (multiplicity) in the 8-μg twice-weekly, 8-μg once-weekly, 4-μg twice-weekly, and 4-μg once-weekly groups was 62.4, 46.2, 62.6, and 36.9, respectively. The non-Tg mice also developed skin nodules, but the sensitivity to induction in the rasH2 Tg mice was higher. No nodules were observed in the acetone groups, but single nodules were apparent in the no-treatment rasH2 Tg and non-Tg groups. In conclusion, skin promotion effects could be detected within only 8 weeks in the rasH2 mice, and the concentration of 4 μg TPA once weekly was sufficient as a positive control. This short-term skin carcinogenesis bioassay using rasH2 mice could represent a useful tool for the assessment of drug and chemical safety with cutaneous treatment.
The standard regulatory requirements for new chemicals include lifetime carcinogenicity testing in 2 rodent species, typically rats and mice, of each sex. This is very time-consuming and expensive in terms of financial and human resources. 15,19 The International Conference on Harmonization (ICH) therefore recommended reducing long-term protocols and using any one species with addition of an alternative bioassay. 6 We have focused on the medium-term carcinogenicity study as one alternative to the long-term carcinogenicity bioassay. This is based on the 2-stage concept of carcinogenesis, and bioassays for skin, liver, urinary bladder, and so on have been established. 3 –5,7,21 We have conducted several investigations of pharmaceutical products or cosmetic components in a 2-stage skin carcinogenesis model with 7,12-dimethylbenz[a]anthracene (DMBA) as the initiator using CD-1 (ICR) mice, and this assay has recently been scheduled for approval by the Japanese pharmaceutical regulatory authorities. 4,5,21
The CB6F1-Tg rasH2 transgenic (rasH2) mouse carries the prototype human c-Ha-ras oncogene and a promoter/enhancer region on the genetic background of a BALB/cByj × C57BL/6 J F1 mouse. A 26-week short-term carcinogenicity study using rasH2 mice, performed with a large number of chemicals, was found to give appropriate results for both genotoxic and nongenotoxic carcinogens. 20,25 –28 On the basis of these results, the US, European, and Japanese pharmaceutical regulatory bodies have approved a short-term carcinogenesis study using the rasH2 mouse to replace a long-term carcinogenicity study. 10
It is known that the rasH2 mouse has high sensitivity to skin, lung, stomach, and hematopoietic carcinogenesis. 2,12 The Tg.AC mouse is also highly sensitive regarding skin carcinogenesis and has been used to evaluate the carcinogenic potential of dermally applied compounds. 1,18 However, the Tg.AC mouse is prone to inflammation, irritation, and hyperplasia and is known to give many false-positive results. 16 In contrast, Urano et al 23 have reported that skin tumors developed in more than 50% of rasH2 mice treated for 26 weeks with 12-O-tetradecanoylphorbol-13-acetate (TPA), a skin tumor promoter, which suggests that an assessment by cutaneous application is possible in a 26-week short-term carcinogenesis study using rasH2 mice. A rapid assay is available to examine lung carcinogenicity in only 9 weeks, 22 and such a short period might also be feasible for a skin 2-stage carcinogenesis model. In addition, it has also been reported that an antitumor activity can be detected in the 2-stage carcinogenesis model using rasH2 mice. 11,14
As a first step for expanding the utility and shortening the experimental period, we evaluated the susceptibility of the rasH2 mouse using a 2-stage skin carcinogenesis bioassay with DMBA as the initiator and TPA as the promoter.
Materials and Methods
Animals and Maintenance
Fifty-five female mice with introduction of the human prototype c-Ha-ras gene (Jic: CB6F1-TgrasH2@Jcl; rasH2 or Tg mice hereafter) and 60 rasH2 mice littermates without the transgene (non-Tg mice) were provided by Clea Japan, Inc. (Tokyo, Japan) at 6 weeks of age. They were housed in plastic cages (143 [width] × 293 [depth] × 148 [height] mm) on soft chip bedding (Hara syoten, Aichi, Japan) in an air-conditioned room at a temperature of 22 ± 2°C and a humidity of 55% ± 15% with a 12-hour light, 12-hour dark cycle. The animals were given ad libitum access to NIH-07 basal diet (Clea Japan, Inc.) in feeders and tap water in water bottles.
Experimental Procedure
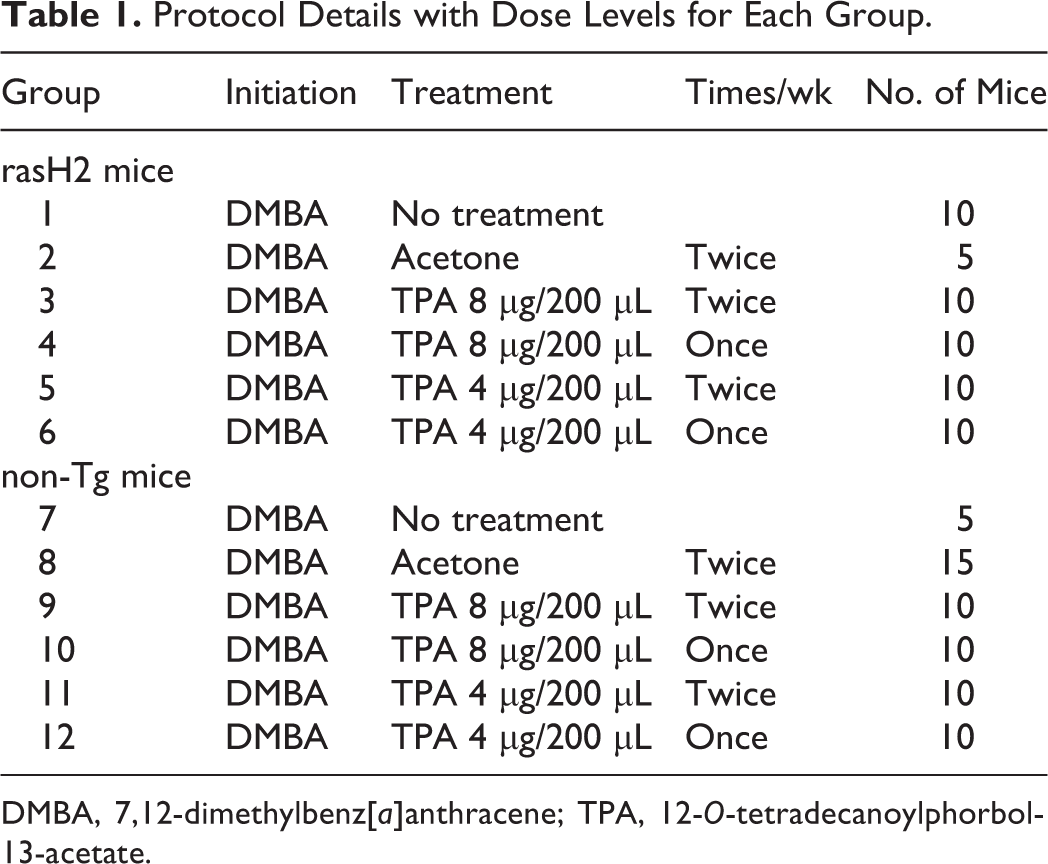
The experimental design is shown in Table 1. Three days prior to treatment, the back skin of each mouse (area; ∼2 × 4 cm) was shaved with electric hair clippers (Thrive model 6000 AD; Daito Electric Machine Industry Co., Ltd., Osaka, Japan), and the hair cycle was confirmed to be in the telogen stage by checking the skin condition after shaving. After the commencement of the study, clipping was conducted weekly. After confirmation of normal status, the animals were entered into the study at 7 weeks of age.
Protocol Details with Dose Levels for Each Group.
DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate.
Fifty-five rasH2 mice and 60 non-Tg mice were divided into 12 groups (5–15 animals each), respectively, the day before initiator treatment. All animals were given a single topical application of DMBA (CAS No. 57-97-6; Sigma-Aldrich, St Louis, MO) at the concentration of 50 μg/100 μL dissolved in acetone (Wako Pure Chemical, Inc., Ltd., Osaka, Japan) using a micropipette with disposable tips as an initiator. Starting 1 week after the initiation treatment, mice were treated with TPA (8 μg/200 μL acetone, twice [groups 3 and 9] or once [groups 4 and 10] weekly; 4 μg/200 μL acetone, twice [groups 5 and 11] or once [groups 6 and 12] weekly) using a micropipette with disposable tips until the termination at week 8. The dose levels in this study were selected based on medium-term skin carcinogenesis studies using CD-1 mice. 4,21 Animals were carefully checked once weekly for back nodules grossly and data were recorded. Body weights were measured once weekly. At the termination, all animals underwent euthanasia by whole-blood collection via the postcava under deep anesthesia, and skin nodules on the clipped back skin, as well as the thoracic and abdominal cavities, were examined macroscopically and data were recorded. Back skins were removed, pressed between paper filters, and fixed in 10% buffered formalin. For all animals, back skin and skin nodules were trimmed, embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin for histopathological examination.
Statistical Analyses
The significance of differences between the control and TPA groups was analyzed and evaluated at P < .05 or P < .01. The significance of differences between the no treatment and acetone groups, as well as the acetone and TPA treatment groups, for numerical data was assessed using the F test. If homogeneous, the data were analyzed with the Student’s t test (1-sided), and if not homogeneous, they were analyzed with the Welch test (1-sided). Analysis of incidences was performed with Fisher’s exact probability test (1-sided).
Animal Care and Protocols
The present study was performed in accordance with Standards Relating to the Care and Management of Laboratory Animals and Relief of Pain (notice No. 88 of the Ministry of Environment, dated April 28, 2006) and Standards for Care in Use of Laboratory Animals of the DIMS Institute of Medical Science, Inc. (September 1, 2010). The protocol was approved by the animal experimental committee of the DIMS Institute of Medical Science, Inc.
Results
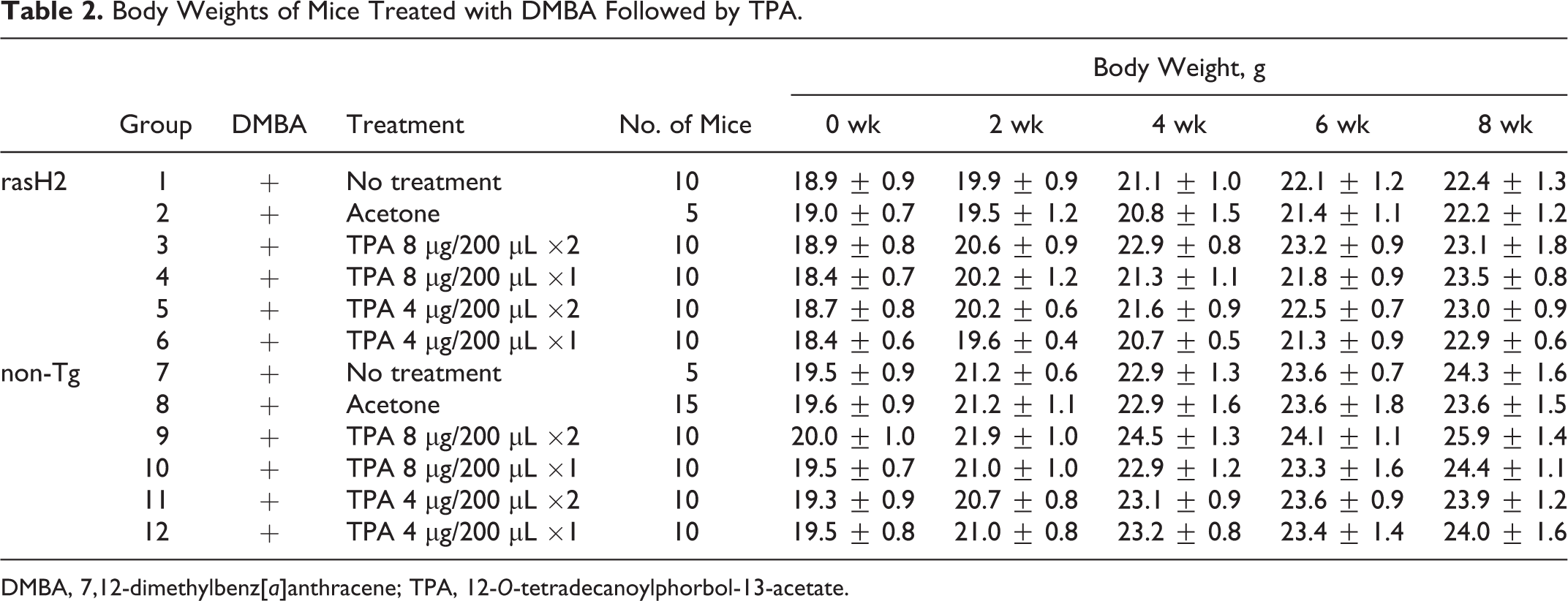
During the study, no mortality was observed in any of the groups (data not shown). Body weight data during the study are shown in Table 2. No statistically significant differences in body weights were observed throughout the study in the Tg and non-Tg groups.
Body Weights of Mice Treated with DMBA Followed by TPA.
DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate.
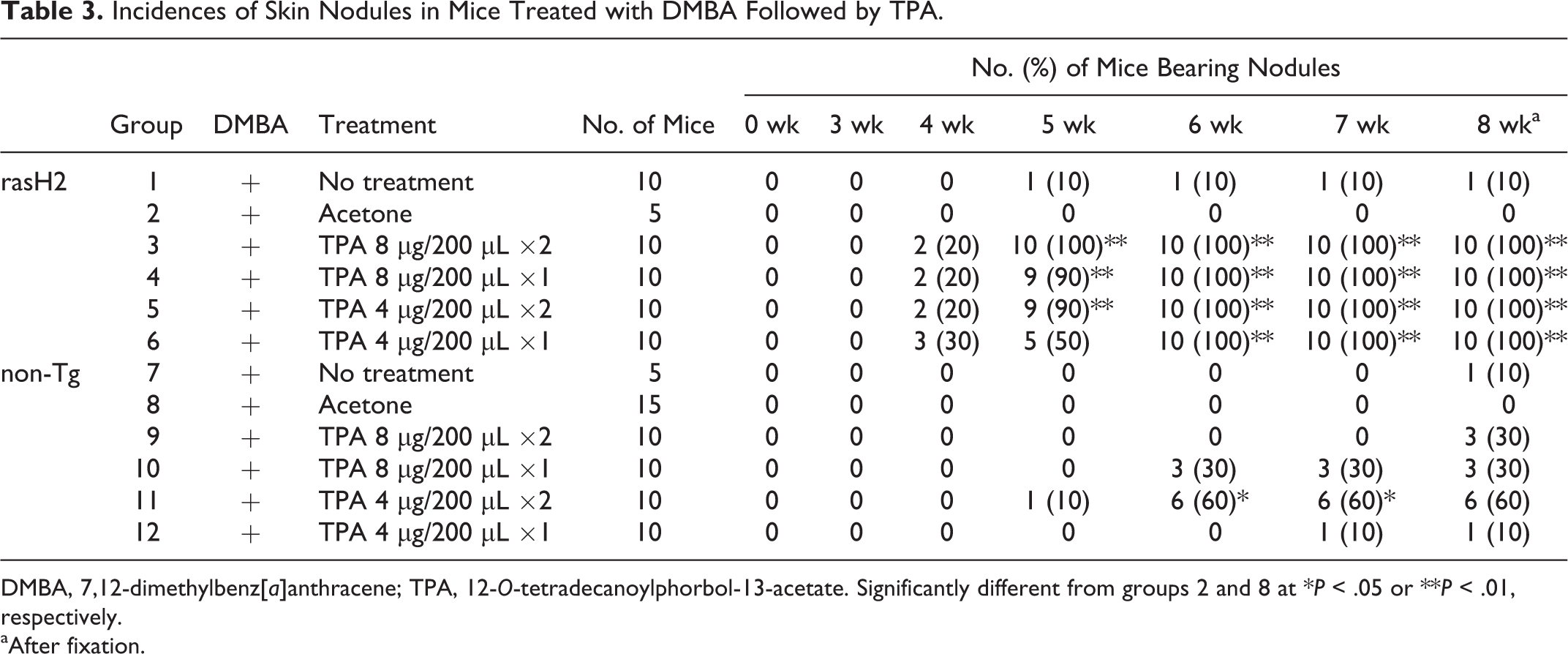
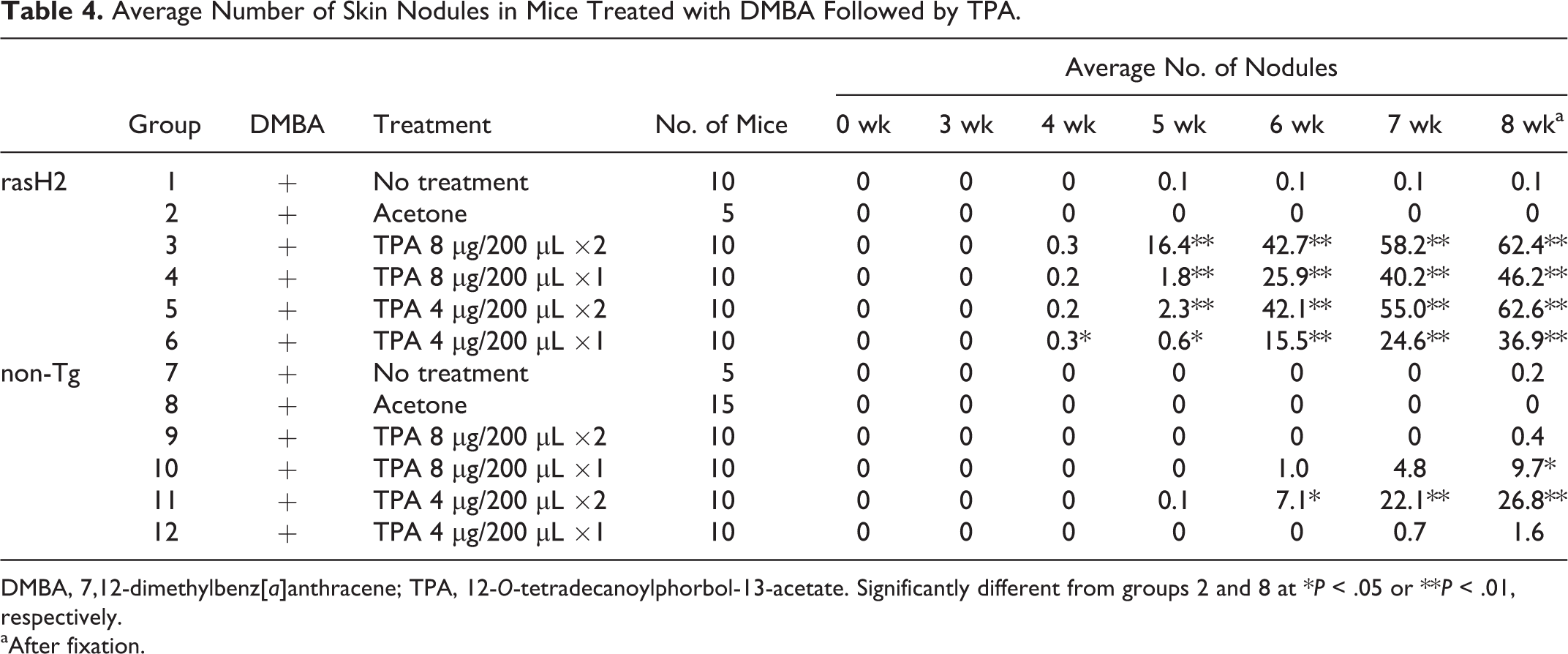
Sequential observation data for incidence of skin nodules in clipped back skin are shown in Table 3, and average numbers of nodules (multiplicity) are given in Table 4. In rasH2 mice treated with TPA, skin nodules developed from week 4, and the incidence reached 100% in week 5 (group 3) or 6 (groups 4, 5, and 6). The multiplicities in the 8-μg twice-weekly (group 3), 8-μg once-weekly (group 4), 4-μg twice-weekly (group 5), and 4-μg once-weekly (group 6) groups at week 5 were 16.4, 1.8, 2.3, and 0.6, respectively. Large numbers of nodules on the back skin were observed at week 8, and the multiplicities in the 8-μg twice-weekly (group 3), 8-μg once-weekly (group 4), 4-μg twice-weekly (group 5), and 4-μg once-weekly (group 6) groups (after fixation) were 62.4, 46.2, 62.6, and 36.9, respectively (Figs. 1–4). Statistical significance was found in all TPA-treated groups as compared with the acetone group.
Incidences of Skin Nodules in Mice Treated with DMBA Followed by TPA.
DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate. Significantly different from groups 2 and 8 at *P < .05 or **P < .01, respectively.
aAfter fixation.
Average Number of Skin Nodules in Mice Treated with DMBA Followed by TPA.
DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate. Significantly different from groups 2 and 8 at *P < .05 or **P < .01, respectively.
aAfter fixation.
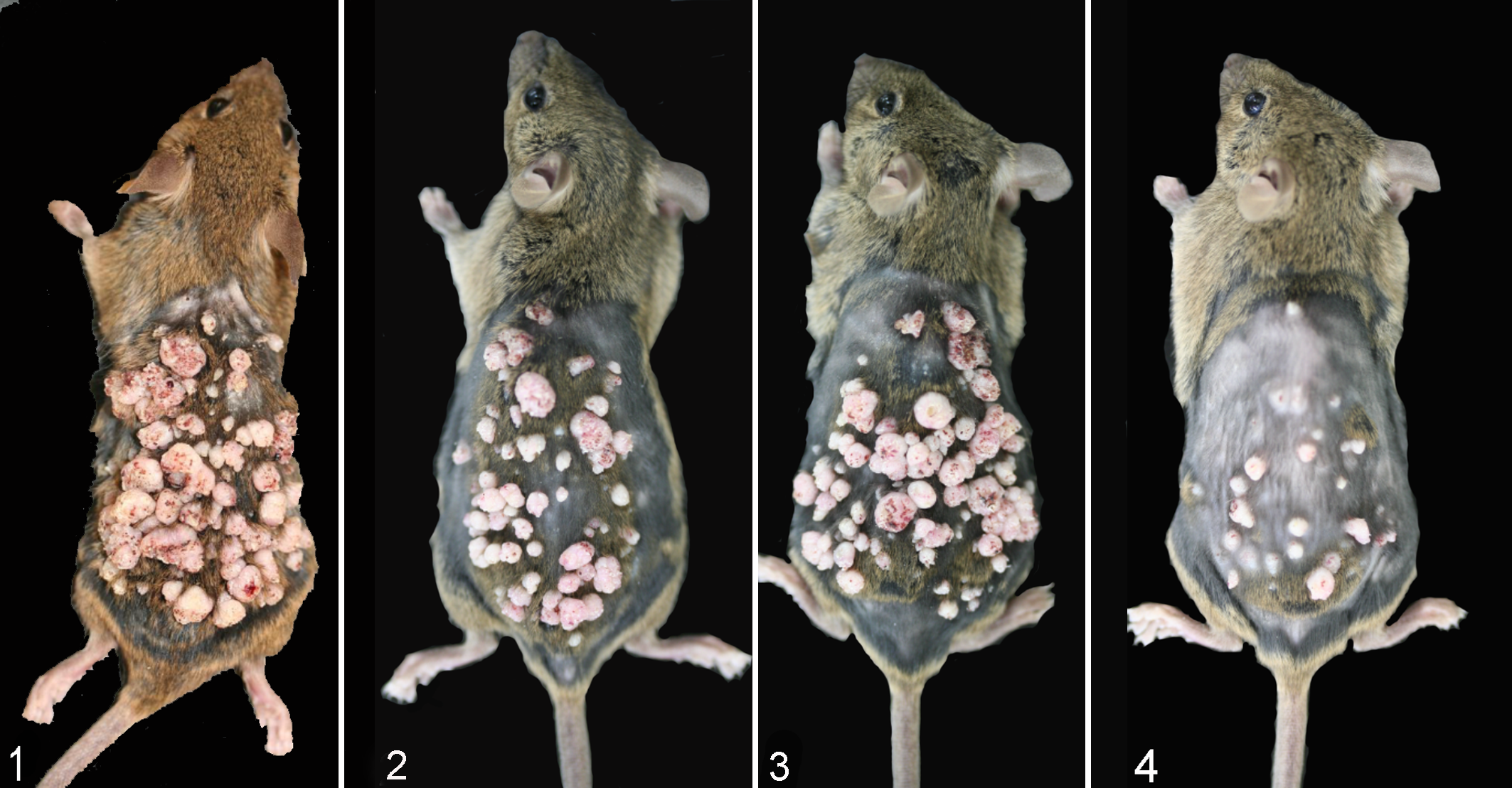
No nodules were observed in the acetone group, but a single nodule was found in the no treatment group of rasH2 mice (group 1). In the non-Tg groups, 1 nodule was found in 1 mouse of the 4-μg twice-weekly group (group 11) at week 5. The multiplicity at week 8 was 26.8, with statistical significance compared with the acetone group (group 8). In the 8-μg twice-weekly (group 9), 8-μg once-weekly (group 10), and 4-μg once-weekly (group 12) groups, skin nodules had developed by weeks 8, 6, and 7, respectively. The multiplicities at week 8 were 0.4, 9.7, and 1.6, respectively, and statistical significance was found only for the 8-μg once-weekly group (group 10) as compared with the acetone group (group 8). No nodules were observed in the acetone group (group 8), but 1 nodule was evident in the no treatment group (group 7) after fixation.
Histopathological findings are summarized in Table 5. The skin nodules, observed in back skin of rasH2 mice, were diagnosed as squamous cell hyperplasia, squamous cell papilloma, squamous cell carcinoma, or keratoacanthoma. Squamous cell carcinoma was observed in 1 to 3 mice in the no-treatment (group 1), 8-μg twice-weekly (group 3), 8-μg once-weekly (group 4), and 4-μg once-weekly (group 6) groups. In the non-Tg group, squamous cell hyperplasia and squamous cell papillomas developed, and squamous cell carcinomas and keratoacanthomas were not observed. The histopathological findings for rasH2 and non-Tg mice were similar to those observed in the 2-stage skin carcinogenesis bioassay using CD-1 (ICR) mice. The susceptibility to skin tumor development of the rasH2 mice was higher than that of non-Tg mice or CD-1 (ICR) mice.
Histopathological Findings in Skin in Mice Treated with DMBA Followed by TPA.
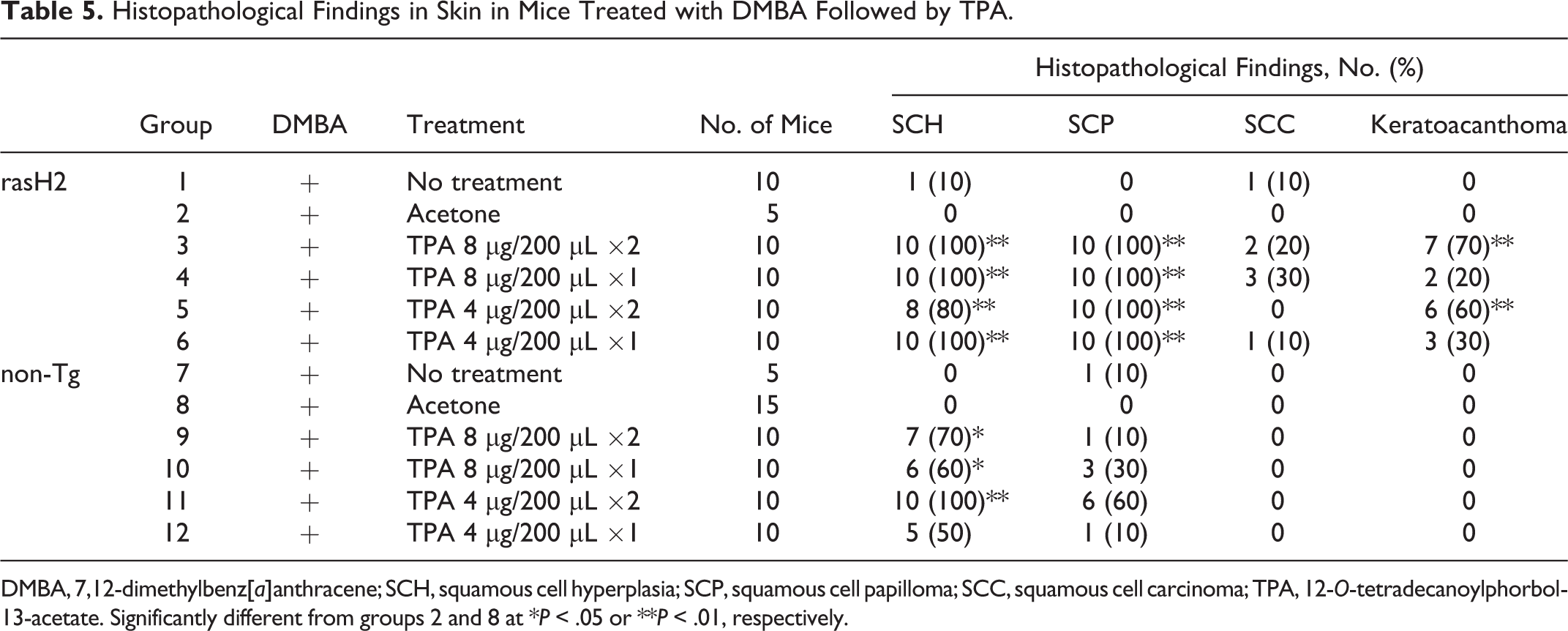
DMBA, 7,12-dimethylbenz[a]anthracene; SCH, squamous cell hyperplasia; SCP, squamous cell papilloma; SCC, squamous cell carcinoma; TPA, 12-O-tetradecanoylphorbol-13-acetate. Significantly different from groups 2 and 8 at *P < .05 or **P < .01, respectively.
Discussion
The present study indicated that TPA applied after DMBA treatment at doses of 4 or 8 μg, once or twice weekly to rasH2 and non-Tg mice, caused significantly increased skin nodule development. Dose and application times did not exert a major influence. These results suggest that rasH2 mice have higher sensitivity with regard to skin tumor development as compared with their non-Tg littermates.
It has been previously reported that skin nodules developed in CD-1 (ICR) mice treated with DMBA. 4,5 In that bioassay, TPA was applied at 4 μg/200 μL twice a week for 19 weeks, and the multiplicity ranged from 12 to 28. 4,5,21 In the present study, DMBA used as initiator was applied to back skin at a dose of 50 μg/100 μL acetone, and this dose was half that used in the 2-stage skin carcinogenesis bioassay with CD-1 (ICR) mice. 4,5,21 When we performed a 26-week short-term carcinogenicity study in rasH2 mice given DMBA by cutaneous application at the doses of 10, 25, and 50 μg/100 μL acetone, the development of skin nodules was limited, with no dose dependence (data not shown). In an investigation to examine if the difference between the codons of the c-Ha-ras gene in human and mouse contributed to the tissue-specific sensitivity to DMBA, the dose of the DMBA was 25 μg/100 μL acetone. 13 In the present study, skin nodules also developed in the no treatment groups of the rasH2 and non-Tg mice, and large numbers of nodules were observed in the groups treated with TPA. Considering the development of skin nodules, these results suggested that 50 μg DMBA in 100 μL acetone may effectively initiate the skin of rasH2 mice but may be an excessive and not optimal dose as an initiator.
Tg.AC transgenic mice have been approved by the US Food and Drug Administration (FDA) and Committee for Proprietary Medicinal Products for use in short-term carcinogenicity studies with cutaneous treatment. 10 Spalding et al 17 reported that the Tg.AC transgenic mouse model can discriminate between carcinogens and noncarcinogens, and both mutagenic and nonmutagenic carcinogens can be detected. Moreover, the Tg.AC transgenic mouse has been used to screen tumor promoters and for assessing antitumor and antiproliferative agents. 8 However, in these reports, skin nodules also developed upon treatment with vehicles such as acetone or ethanol. 1,8,9,17 Lynch et al 9 considered that skin nodules observed in acetone or untreated groups developed possibly as a consequence of skin shaving, fighting, or abrasion. At present, the US FDA no longer recommends the Tg.AC model based on the problems observed with this model, and the number of Tg.AC studies has been decreasing. 2
In the present study, tumor development in the no-treatment group of rasH2 mice was limited, and no nodules were observed in mice treated with acetone. Similarly, in a 26-week short-term carcinogenecity study using rasH2 mice treated with ethanol, Vaseline, and acetone topically, skin nodules were not evident in any mice. 23 Moreover, when IC tags were worn by rasH2 mice for 26 weeks, tumorigenic changes were not observed at the sites of contact. 24 We, therefore, considered that an evaluation of skin promotion by TPA was possible with an exposure period of only 8 weeks when using rasH2 mice, although a more appropriate dose of DMBA as the initiator should be determined in future experiments.
The Tg.AC alternative model for topical medium-term carcinogenicity assessment produced false-positive results related to vehicle effects and/or local irritation, and this model is no longer recommended by the US FDA. As described above, an 8-week initiation-promotion model in rasH2 mice is proposed as an alternative model for carcinogenicity assessment of dermally applied products. However, a large amount of validation data still need to be collected with a variety of chemicals to confirm that this model is both sensitive and specific. Therefore, although our proposed ultra short-term carcinogenesis bioassay methodology still needs more validation studies before it could be used for routine regulatory use, we anticipate that this bioassay may be useful as a screening tool to predict skin carcinogenesis in early stages of drug development.
In conclusion, the detection of skin tumor promotion was possible after only 8 weeks of treatment in the current short-term skin carcinogenicity bioassay using rasH2 mice. As a positive control, the 4-μg dose of TPA once weekly was sufficient for effective promotion.
Footnotes
Acknowledgement
We thank Dr Malcolm A. Moore for critical reading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
