Abstract
After initiation with 7,12-dimethylbenz[a]anthracene (DMBA), the promoting potential of 12-O-tetradecanoylphorbol-13-acetate (TPA) on skin tumor development can be detected by an ultra-short-term skin carcinogenicity bioassay using Tg-rasH2 mice. In the present study, 10 chemicals were assessed using this ultra-short-term bioassay as a first step to validate this practical and easy-to-use skin carcinogenicity bioassay. These chemicals belonged to 4 categories: dermal vehicles (acetone, 99.5% ethanol, anhydrous ethanol, and Vaseline), skin noncarcinogens (oleic acid diethanolamine condensate, benzethonium chloride, and diisopropylcarbodiimide), skin tumor promoters (TPA and benzoyl peroxide), and a skin carcinogen (4-vinyl-1-cyclohexene diepoxide). In a first study, DMBA was used as the initiator at a dose of 50 μg according to previous data, but skin tumors were observed in the no-treatment and vehicle groups. Therefore, the dose of DMBA for skin tumor initiation was reevaluated using 12.5 or 25 μg, with 12.5 μg found to be sufficient for initiation activity. In the ultra-short-term assay, the vehicles and skin noncarcinogens were negative while the skin tumor promoters and the skin carcinogen were positive. The detection of skin tumor promotion and carcinogenicity was feasible in only 8 weeks. In conclusion, this carcinogenicity bioassay may represent a useful tool for the assessment of the carcinogenicity potential of topically applied chemicals.
To evaluate the carcinogenicity of pharmaceuticals, the International Conference on Harmonization (ICH) allows 1 of the 2-year carcinogenicity studies conducted in 2 species to be replaced with an alternative short- or medium-term bioassay. 8 The Tg-rasH2 mouse is one of the transgenic mice used in these alternative short-term carcinogenicity bioassays. A 26-week short-term carcinogenicity bioassay using the Tg-rasH2 mouse has been approved by the US, European, and Japanese pharmaceutical regulatory authorities. The Tg-rasH2 mouse is known to be highly sensitive to skin, lung, stomach, and hematopoietic carcinogenesis. 5,13 Another transgenic mouse model, the Tg.AC mouse, is also highly sensitive to skin carcinogenesis; however, this mouse is prone to inflammation, irritation, and hyperplasia, resulting in many false-positive results, 3,21,22 such that the US Food and Drug Administration (FDA) no longer recommends it. 5
Our laboratory has conducted numerous investigations of pharmaceutical products and cosmetic components using a 2-stage skin carcinogenicity model with 7,12-dimethylbenz[a]anthracene (DMBA) as the initiator and CD-1 (ICR) mice as the test animals, and this assay model has recently been scheduled for approval by Japanese pharmaceutical regulatory authorities. 6,7,24 As an alternative to using CD-1 mice, we speculated that using the Tg-rasH2 mouse may enable a more rapid 2-stage skin carcinogenicity bioassay. To test this proposition, we used 12-O-tetradecanoylphorbol-13-acetate (TPA) and found that the skin carcinogenicity promotion effect of TPA was detectable in only 8 weeks. 11
To validate this Tg-rasH2 mouse topical carcinogenicity model, a variety of chemicals of differing carcinogenic potential needs to be tested.
Therefore, in the present study, 10 chemicals belonging to 4 categories—common vehicles of dermally applied products, noncarcinogens when applied dermally, skin tumor promoters, and a skin carcinogen—were tested in the model. These compounds were selected because of their commercial availability and their well-documented carcinogenic or tumor promotion effects on skin after dermal application. 15 –18,20
Materials and Methods
Animals
Six-week-old female mice carrying the human c-Ha-ras gene (Jic: CB6F1-TgrasH2@Jcl.; Tg-rasH2 mice) and their wild-type littermates (non-Tg mice) were provided by Clea Japan, Inc. (Tokyo, Japan). Experiments were started after a 1-week acclimation and quarantine period, when the mice were 7 weeks of age. They were housed in plastic cages (width 143 × depth 293 × height 148 mm) on soft chip bedding (Hara shoten, Aichi, Japan) in an air-conditioned room at a temperature of 22 ± 3°C and a humidity of 55 ± 15% with a 12-hour light, 12-hour dark cycle. The animals were given access to MF basal diet (Oriental Yeast Co., Ltd., Tokyo, Japan) and tap water ad libitum.
Animal care and protocols
The current experiment was approved by the ethics committee of the DIMS Institute of Medical Science, Inc. The animals were maintained in accordance with “Standards Relating to the Care and Management of Experimental Animals” (Notification No. 6, March 27, 1980, Prime Minister’s Office, Japan), Guidelines for Proper Conduct of Animal Experiments (June 1, 2006, Science Council of Japan), and the in-house guideline, Standards for the Care and Use of Laboratory Animals.
Test chemicals
Dermal application of oleic acid diethanolamine condensate (OADC) and benzethonium chloride (BC) has no carcinogenic activity in F344 rats or B6C3F1 mice and no genotoxic activity. 15,17 Dermal application of diisopropylcarbodiimide (DC) also has no carcinogenic activity in F344 rats or B6C3F1 mice, but there are reports of equivocal genotoxicity. 16 Benzoyl peroxide (BP) is a tumor promoter using a 2-stage skin mouse carcinogenicity model, 20 but the reports of genotoxicity are equivocal. 12,14 Dermal application of 4-vinyl-1-cyclohexene diepoxide (4-VCD) caused tumors in a 2-year carcinogenicity study using F344/N rats and B6C3F1 mice. 18 DMBA (CAS No. 57-97-6), TPA (CAS No. 16561-29-8), and 4-VCD (CAS No. 106-87-6) were purchased from Sigma Aldrich (St. Louis, MO), and 99.5% ethanol and anhydrous ethanol (CAS No. 64-17-5), acetone (CAS No. 67-64 -1), and OADC (CAS No.93-83-4) were purchased from Wako Pure Chemical Industries Ltd. (Osaka, Japan). BC (CAS No. 121-54-0), DC (CAS No. 693-13-0), and BP (CAS No. 94-36-0) were purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan).
Preparation of the dermal application area
Three days prior to DMBA initiation, the back skin of each mouse was shaved with electric hair clippers (Thrive model 6000 AD; Daito Electric Machine Industry Co., Osaka, Japan) (area approximately 2 × 4 cm). The telogen phase is the resting phase of the hair follicle, and the skin is more sensitive to carcinogens during the telogen phase than the anagen phase (hair follicular growth). 1,2,4 Mice of 7 weeks of age are in the telogen phase, 10,19 and this was confirmed in the present study by examination of hair growth after shaving. After dermal application of DMBA, clipping was performed weekly.
Experimental Procedure
Experiment 1 (initial investigation with various chemicals)
Tg-rasH2 mice were divided into 15 groups of 5 or 10 mice/group 1 day before initiator treatment. Five groups (5 mice/group) were not treated with DMBA and 10 groups (10 mice/group) were given a single topical application of DMBA at a concentration of 50 μg/100 μL dissolved in acetone using a micropipette with disposable tips (100 μL/mouse). One week after the initiator treatment (week 1), the mice in the non-DMBA treatment groups were given topical applications of OADC (30 mg/kg/d), BC (1.5 mg/kg/d), DC (40 mg/kg/d), BP (20 mg/mouse), or 4-VCD (10 mg/mouse), and the mice in the plus-DMBA treatment groups either were not treated or given topical applications of 99.5% ethanol, anhydrous ethanol, Vaseline, OADC (30 mg/kg/d), BC (1.5 mg/kg/d), DC (40 mg/kg/d), BP (1 mg or 20 mg/mouse), or 4-VCD (10 mg/mouse). DC, BC, and 4-VCD were applied 5 times per week (Monday to Friday), and the other chemicals were applied every day using a micropipette with disposable tips until the termination of the experiment at week 8. DC and BC were dissolved in anhydrous ethanol, OADC was dissolved in 99.5% ethanol, and BP and 4-VCD were dissolved in acetone. Dose levels were set based on data from previous carcinogenicity studies. 15 –18,20
Nodules on the clipped back skin were carefully evaluated macroscopically once weekly. At the termination of the experiment, all animals were placed under deep isoflurane anesthesia and euthanized by exsanguination from the abdominal aorta. Administration sites and main organs in the thoracic and abdominal cavities were examined macroscopically. All macroscopically visible skin nodules at the administration site were counted, and the average number of nodules per mouse was calculated. Back skins were removed, pressed between paper filters, and fixed in 10% buffered formalin. For all animals, back skin and all skin nodules were trimmed, embedded in paraffin wax, sectioned (2–3 μm), and stained with hematoxylin and eosin for histopathological examination.
Experiment 2 (appropriate dose of the initiator)
Since several skin nodules were observed in the no-treatment, vehicle (anhydrous ethanol), and skin noncarcinogen (OADC, BC, and DC) groups in experiment 1, we concluded that the dose of the initiator (DMBA) was too high. Therefore, in experiment 2, lower doses of DMBA were used to determine a dose that would not result in false positives for noncarcinogenic compounds.
Ninety Tg-rasH2 mice were divided into 10 groups of 5 or 10 mice/group 1 day before initiator treatment. Animals were given a single topical application of DMBA at concentrations of 12.5 μg/100 μL acetone or 25 μg/100 μL acetone (100 μL/mouse). Starting 1 week after treatment, mice were treated for 7 weeks with 99.5% ethanol or anhydrous ethanol as vehicles, OADC (30 mg/kg/d) or BC (1.5 mg/kg/d) as skin non-carcinogens, and BP (20 mg/mouse) as a skin tumor promoter. The other experimental methods were the same as in experiment 1.
Experiment 3 (appropriate dose of TPA as a positive control)
From the results of experiment 2, 12.5 μg was found to be an appropriate dose for DMBA in this 2-stage skin carcinogenicity model. The objective of experiment 3 was to determine an appropriate dose of TPA for use as a positive control when using 12.5 μg DMBA as the initiator.
Forty Tg-rasH2 mice and 40 non-Tg mice were divided into 4 groups of 10 mice/group. Twenty Tg-rasH2 mice and 20 non-Tg mice were given a single topical application of DMBA (12.5 μg/100 μL acetone), and the remaining 20 Tg-rasH2 and non-Tg mice were treated with 100 μL acetone as non-DMBA treatment controls. Starting 1 week after the initiator treatment, mice were treated once per week for 7 weeks with 4 μg or 8 μg/200 μL TPA dissolved in acetone (200 μL/mouse). The other experimental methods were the same as in experiment 1.
Experiment 4 (reconfirmation of the carcinogenicity of 4-VCD)
In experiment 1, the incidence of skin nodules in mice treated with 4-VCD (10 mg/mouse, 5 times a week), a skin carcinogen, and DC (40 mg/kg/d), a noncarcinogen, were similar to the anhydrous ethanol and no-treatment groups, with skin nodules developing in 1 to 3 of 10 mice. Therefore, we reexamined the carcinogenicity of 4-VCD and DC using 12.5 μg DMBA (12.5 μg/100 μL acetone) as the initiator. Fifty Tg-rasH2 mice were divided into 5 groups of 10 mice/group. Animals were given a single topical application of 12.5 μg DMBA as the initiator. Starting 1 week after initiator treatment, mice were treated daily for 7 weeks with acetone, DC (40 mg/kg/d), or 4-VCD (10, 15, or 20 mg/mouse/d). The other experimental methods were the same as in experiment 1.
Statistical Analyses
Statistical analysis was not performed in experiments 1 to 3. In experiment 4, the significance of the differences between the acetone and DC groups was assessed using the F test. If homogeneous, the data were analyzed with the Student' t test (one-sided), and if not homogeneous, they were analyzed with the Welch test (one-sided). Analysis of incidence was performed with Fisher’s exact probability test (one-sided). The significance of the differences between the acetone and 4-VCD groups was analyzed with Bartlett’s test. If homogeneous, the data were analyzed using Dunnett’s multiple comparison test (one-sided), and if not homogeneous, they were analyzed with Steel’s test. Analysis of incidence was performed using Fisher’s exact probability test (one-sided). P values <.05 were considered significant.
Results
Experiment 1
During the study, no mortality was observed in any of the groups. No tumors were observed in any of the non-DMBA treatment groups. The incidence and average number of skin nodules (multiplicity) in the DMBA-initiated groups are shown in Supplemental Table S1. The incidence of skin nodules in the 1- and 20-mg/mouse BP groups was 100%, and the multiplicity was 1.3 and 52.5, respectively. The incidence of skin nodules in the 99.5% ethanol and Vaseline groups was 0%, and in the other groups, the incidences of skin nodules were 10% to 30% with a multiplicity of 0.1 to 0.5. Histopathological findings are shown in Supplemental Table S2. The skin nodules were diagnosed as squamous cell hyperplasia, squamous cell papilloma, squamous cell carcinoma, or keratoacanthoma. The incidence of squamous cell hyperplasia and squamous cell papilloma in the BP (20 mg/mouse/d) groups was 100%. In contrast, the incidence of skin lesions in the 4-VCD group was very low. No lesions in the thoracic or abdominal cavities were observed in any of the groups.
Experiment 2
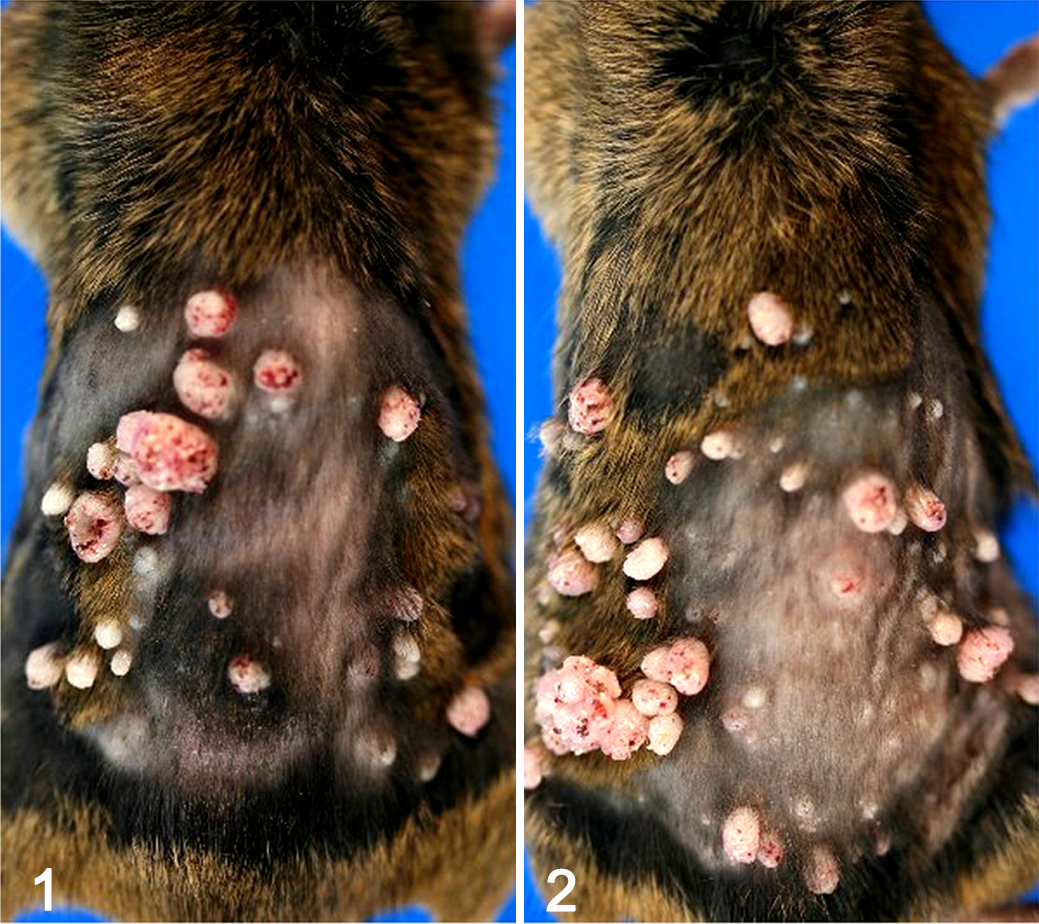
Since several skin nodules were observed in the no-treatment, vehicle, and skin noncarcinogen groups in experiment 1, we concluded that application of 50 μg DMBA to initiate skin tumors was too high. In experiment 2, a dose range finding study was conducted for DMBA with dose of 12.5 or 25 μg. No skin nodules were observed in the 99.5% ethanol, anhydrous ethanol, OADC, or BC groups using 12.5 or 25 μg DMBA (Table 1). In contrast, mice treated with BP, a skin tumor promoter, did develop skin nodules: the incidence and multiplicity of skin nodules were 100% and 29.2 for 12.5 μg DMBA and 100% and 35.6 for 25 μg DMBA (Table 1 and Figures 1, 2). The skin nodules observed in the BP groups were diagnosed as squamous cell hyperplasia or squamous cell papilloma (Table 2). No lesions in the thoracic or abdominal cavities were observed in any of the groups.
Incidences and Multiplicity of Skin Nodules in Tg-rasH2 Mice Treated with Low-Dose DMBA Followed by Test Chemicals.
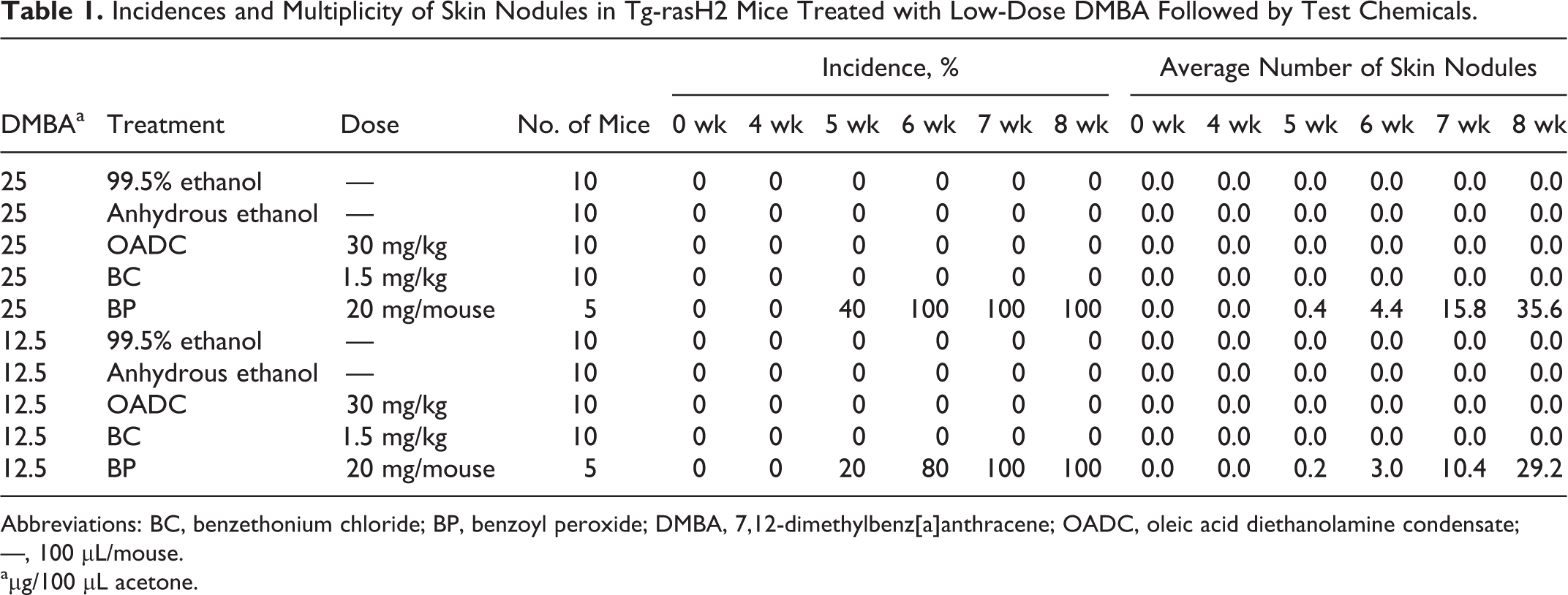
Abbreviations: BC, benzethonium chloride; BP, benzoyl peroxide; DMBA, 7,12-dimethylbenz[a]anthracene; OADC, oleic acid diethanolamine condensate; —, 100 μL/mouse.
aμg/100 μL acetone.
Histopathological Findings of Skin Nodules in Tg-rasH2 Mice Treated with Low-dose DMBA Followed by Test Chemicals.
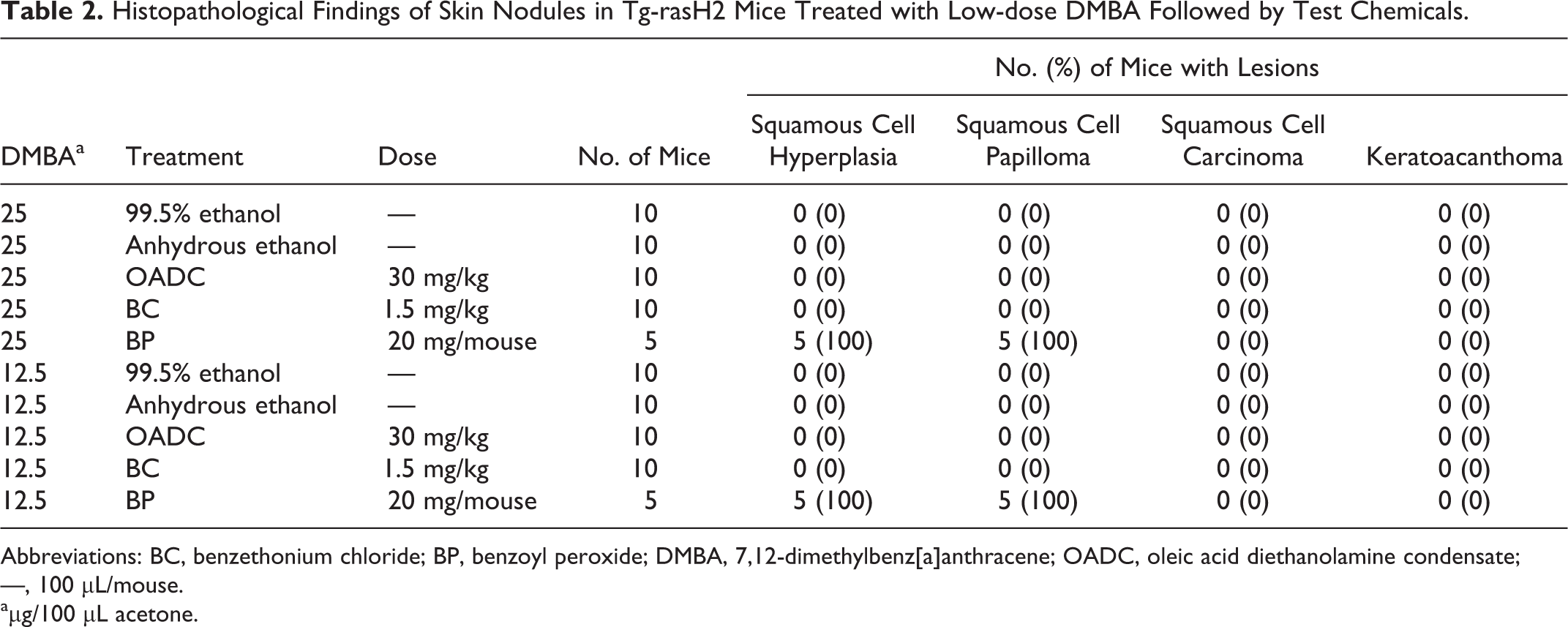
Abbreviations: BC, benzethonium chloride; BP, benzoyl peroxide; DMBA, 7,12-dimethylbenz[a]anthracene; OADC, oleic acid diethanolamine condensate; —, 100 μL/mouse.
aμg/100 μL acetone.
Experiment 3
Experiment 3 was conducted to determine the concentration of TPA, a positive control substance that can be used when skin tumors are initiated with 12.5 μg DMBA. The incidence and multiplicity of skin nodules in Tg-rasH2 and non-Tg mice treated with TPA are shown in Supplemental Table S3. In the Tg-rasH2 mice initiated with 12.5 μg DMBA and then treated with 4 μg TPA, skin nodules developed by week 5, and the incidence reached 100% on week 7 with a multiplicity of 10.9. In the mice treated with 8 μg TPA, skin nodules developed by week 4, and the incidence reached 100% on week 6 with a multiplicity of 20.5. The multiplicity showed dose dependency. All skin nodules were diagnosed as squamous cell hyperplasia or squamous cell papilloma (Suppl. Table S4).
In the Tg-rasH2 mice treated with 4 or 8 μg TPA but without DMBA initiation and in all non-Tg mice groups, no skin nodules were observed throughout the treatment period. No lesions in the thoracic or abdominal cavities were observed in any of the groups.
Experiment 4
Experiment 4 was conducted to examine the carcinogenicity of 4-VCD and DC in Tg-rasH2 mice initiated with 12.5 μg DMBA; vehicle control mice were treated with acetone. The incidence and multiplicity of skin nodules are shown in Table 3. Skin nodules developed by week 5 in the 20-mg/mouse 4-VCD group, and the incidence and multiplicity at week 8 were 80% and 1.8, respectively. In the 15-mg and 10-mg/mouse 4-VCD groups, skin nodules were observed at week 6, and the final incidence and multiplicity were 50% and 0.9 for 15 mg 4-VCD and 50% and 0.6 for 10 mg 4-VCD. Skin nodules did not develop in the acetone-treated (vehicle control) or DC-treated (skin noncarcinogen) mice.
Incidences and Multiplicity of Skin Nodules in Tg-rasH2 Mice Treated with 12.5 μg DMBA Followed by Acetone, DC, or 4-VCD.
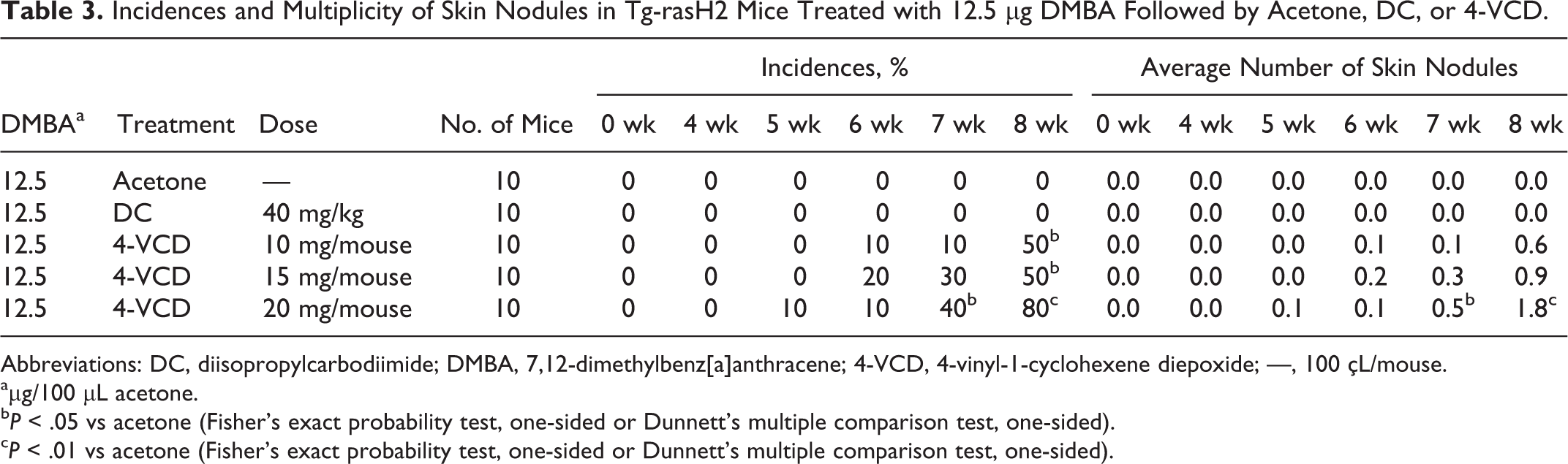
Abbreviations: DC, diisopropylcarbodiimide; DMBA, 7,12-dimethylbenz[a]anthracene; 4-VCD, 4-vinyl-1-cyclohexene diepoxide; —, 100 çL/mouse.
aμg/100 μL acetone.
bP < .05 vs acetone (Fisher’s exact probability test, one-sided or Dunnett’s multiple comparison test, one-sided).
cP < .01 vs acetone (Fisher’s exact probability test, one-sided or Dunnett’s multiple comparison test, one-sided).
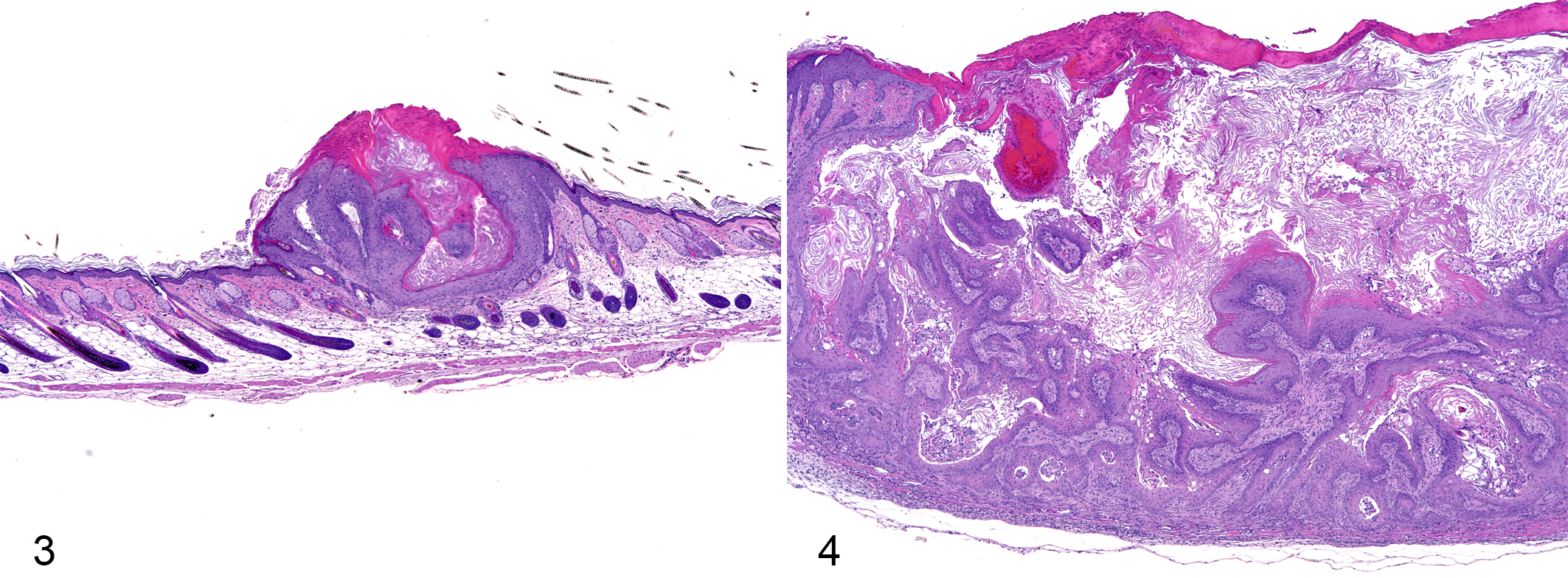
Histopathological findings are summarized in Table 4 and illustrated in Figs. 3 and 4. Squamous cell hyperplasia and squamous cell papillomas were observed in all three 4-VCD groups. The incidence of squamous cell hyperplasia and squamous cell papilloma in all groups and the average number of squamous cell papillomas in the 20-mg/mouse group were significantly increased compared to the acetone group. Squamous cell carcinoma developed in 1 mouse in the 4-VCD 15-mg/mouse group. No lesions in the thoracic or abdominal cavities were observed in any of the groups.
Histopathological Findings of Skin Nodules in Tg-rasH2 Mice Treated with 12.5 μg DMBA Followed by Acetone, DC, or 4-VCD.

Abbreviations: DC, diisopropylcarbodiimide; DMBA, 7,12-dimethylbenz[a]anthracene; 4-VCD, 4-vinyl-1-cyclohexene diepoxide; —, 100 �L/mouse.
aμg/100 μL acetone.
bP < .01 vs acetone (Fisher’s exact probability test, one-sided).
Discussion
The present study was conducted in 4 steps: first, Tg-rasH2 mice were treated with 100 μL DMBA at a concentration of 50 μg/100 μL, followed by topical application of commonly used vehicles (99.5% ethanol, anhydrous ethanol, or Vaseline), skin noncarcinogens (OADC, BC, or DC), a skin tumor promoter (BP), or a skin carcinogen (4-VCD). While there was an increase in the development of skin nodules in mice treated with the skin tumor promoter BP, there was no increase in the development of skin nodules in mice treated with the skin carcinogen 4-VCD. Furthermore, untreated mice and mice treated with anhydrous ethanol, OADC, BC, and DC also developed skin nodules. These data indicated that the dose of 50 μg DMBA was too high for the initiator in this model. Therefore, a second experiment was performed to determine an appropriate dose of DMBA for the model. In this study, mice were treated with lower doses of DMBA (12.5 and 25 μg/mouse) followed by topical application of 95% ethanol, anhydrous ethanol, OADC, BC, or BP treatment. Only BP, a skin tumor promoter, induced skin nodules. Since the incidence of skin nodules in BP-treated mice with 12.5 and 25 μg DMBA was 100%, the dose of 12.5 μg DMBA was considered suitable. In the third experiment, the appropriate dose of TPA for use as a positive control was determined to be 4 μg/200 μL applied topically once per week. The fourth experiment reexamined the carcinogenicity of 4-VCD (a skin carcinogen) and DC (a skin noncarcinogen) using a single application of 12.5 μg DMBA as the initiator with acetone as the vehicle control. In this experiment, only 4-VCD caused development of skin nodules. Overall, commonly used vehicles (99.5% ethanol, anhydrous ethanol, and Vaseline) and skin noncarcinogens (OADC, BC, and DC) were negative, while skin tumor promoters (TPA and BP) and a skin carcinogen (4-VCD) were positive (Table 5) in this ultra-short-term skin carcinogenicity assay (Figure. 5). Therefore, these results were consistent with those of traditional 2-year carcinogenicity studies 15 –18,20 and a 26-week carcinogenicity study using Tg-rasH2 mice treated with ethanol, Vaseline, and acetone, administered cutaneously. 25
Summary of Results in the Ultra-Short-Term Skin Carcinogenesis Study.


Standard protocol of the ultra-short-term skin carcinogenicity bioassay using Tg-rasH2 mice as described in this report. ▾, 7,12-dimethylbenz[a]anthracene (DMBA) (12.5 μg/100 μL acetone, dermal application). 12-O-tetradecanoylphorbol-13-acetate (TPA) (positive control): 4 μg/200 μL acetone, once weekly, dermal application.
The selection of the dose levels of the compounds used in experiment 1 was based on previous reports. 15 –18,20 The 4-VCD dose was based on the results of 14-day and 13-week dermal toxicity studies and a 2-year carcinogenicity study using B6C3F1 mice; 18 a dose of 10 mg/mouse applied dermally 5 times/wk was not lethal and did not cause skin ulceration at 13 weeks. In the 2-year study, long-term survival was lower than in the vehicle controls, primarily because of neoplasms: all 50 male mice and 40 of 50 female mice treated with 10 mg/mouse died by week 83. However, in our study (experiment 1), the incidence and the number of skin nodules were very low, suggesting that the 7-week treatment period may have been too short to allow skin nodules to develop when 4-VCD is applied at 10 mg/mouse 5 times/wk. Therefore, increased doses of 4-VCD were used in experiment 4: 10 mg, 15 mg, and 20 mg per mouse applied daily for 7 weeks. These increased doses resulted in a dose-dependent increase in the incidence and multiplicity of skin nodules. It is noteworthy that in experiment 1, 1 of 10 mice developed skin nodules, whereas in experiment 4, 5 of 10 mice treated with 10 mg/mouse 4-VCD developed skin nodules, even though the amount of DMBA used for initiation was 50 μg in experiment 1 and 12.5 μg in experiment 4. While the difference in skin nodule development is not statistically significant, these results suggest that the 2-day interval in the 5 times/wk treatment may have affected the carcinogenicity of 4-VCD, although the total doses administered (350 mg and 490 mg for the 5 times/wk and the daily administration, respectively) may account for the difference in skin nodule incidence. In an earlier study, we treated mice with 4 μg TPA (4 μg/200 μL) 2 times/wk for 7 weeks or with 8 μg TPA (8 μg/200 μL) 1 time/wk for 7 weeks. 11 Although the total dose of TPA applied per mouse was the same in that study, the dosing regimen of 4 μg TPA 2 times/wk caused the development of 62.6 nodules/mouse compared to 46.2 nodules/mouse in the other dosing regimen, demonstrating that application interval as well as total dose can affect the results of a carcinogenicity bioassay.
In the current study, the incidence and multiplicity of skin nodules induced by the tumor promoter, BP, were higher than those of the skin carcinogen, 4-VCD. This apparent discrepancy is most likely due to the difference in the promoting activities of BP and 4-VCD. Initiators have genotoxic effects on cells, enabling initiated cells to respond to promoting agents, and promoters in turn induce initiated cells to eventually develop into neoplasms. Complete carcinogens have both initiation and promoting activities; however, the strength of the respective initiation and promoting activities of different carcinogens can vary widely. Since a complete carcinogen can have weaker promoting activity than a promoting agent, the combination of an initiator plus a complete carcinogen may have stronger initiation activity but weaker promoting activity than the combination of an initiator plus a promoter, which would lead to differences in overall tumor formation.
Experimental models are increasingly being modified depending on the use and purpose of the drugs being tested. Recently, we have conducted a 26-week short-term carcinogenicity study of 1,2-dichloroethane, a known lung carcinogen, using dermal application to Tg-rasH2 mice. 23 This study confirmed that the evaluation of the lung carcinogenicity of at least some drugs is possible using a short-term dermal application assay. A 26-week short-term internal organ carcinogenicity study using dermal application could also represent an alternative assay for carcinogenicity testing.
Since the ICH guidance on testing for carcinogenicity of pharmaceuticals was issued, medium-term carcinogenicity bioassays and 26-week short-term carcinogenicity studies using transgenic mice have been conducted as an alternative to a second traditional long-term (ie, 2-year) carcinogenicity study. In Japan, the 2-stage skin carcinogenicity bioassay with CD-1 (ICR) mice is recognized as a tool for the assessment of topical carcinogenicity in the development of dermal liniments by the Pharmaceuticals and Medical Devices Agency (PMDA) of Japan, and in the United States, more than 75% of mouse carcinogenicity studies are now conducted using Tg-rasH2 mice. 9 The ultra-short-term bioassay described in this report may represent a useful screening tool to predict skin carcinogenesis during the early stages of drug development, prior to more laborious and time-consuming regulatory studies.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819854440 - Establishment and Validation of an Ultra-Short-Term Skin Carcinogenicity Bioassay Using Tg-rasH2 Mice
Supplemental Material, DS1_VET_10.1177_0300985819854440 for Establishment and Validation of an Ultra-Short-Term Skin Carcinogenicity Bioassay Using Tg-rasH2 Mice by Mayumi Kawabe, Koji Urano, Mayuko Suguro, Tomomi Hara, Yasushi Kageyama, Yukinori Mera and Hideki Tsutsumi in Veterinary Pathology
Footnotes
Acknowledgement
We thank Dr. David B. Alexander from Nagoya City University Graduate School of Medical Sciences for critical reading of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
