Abstract
The aim of this study was to investigate the effects of neonatal administration of a relatively high dose of diethylstilbestrol (DES) on the induction of mammary carcinomas (MCs) and benign proliferative lesions (PLs) induced by 7,12-dimethylbenz[a]anthracene (DMBA) in female rats. Three different terms of daily administration of DES (10 μg) were used: 0–14, 0–5, and 6–14 days after birth. Control animals were administered solvent (oil) alone once daily from 0 to 14 days after birth. Rats were given DMBA (10 mg) at 50 days after birth. All rats administered DES showed persistent estrus and anovulatory ovaries. In rats administered DES from 0 to 14 and 0 to 5 days after birth there was protection from development of MCs and PLs, and serum levels of both estrogen and progesterone were significantly lower than in the control animals at 100 days after birth. In rats administered DES from 6 to 14 days after birth, the incidence of MCs was equal to that of the control animals (100%), and the number of PLs was significantly higher than in the control animals. The critical period for exposure to endocrine disruptors, such as DES, affecting the induction of MCs and PLs may be from 0 to 5 days after birth.
Keywords
Introduction
Huggins et al. previously showed that a single administration of 20 mg 7,12-dimethylbenz[a]anthracene (DMBA) at 50 days after birth induced visible mammary cancer in rats by 110 days after birth. 7 Thereafter, this animal has been a useful model for studying the carcinogenesis of the human breast. DMBA is known to be a polycyclic hydrocarbon carcinogen forming DNA adducts. 15 The presence of estrogen and progesterone receptors in DMBA-induced mammary carcinomas (MCs) has been well documented in rats. 19, 20 DMBA-transformed cells (intraductal papillary proliferative lesions) could exist in unpalpable lesions long after the administration of DMBA under conditions in which estrogen was present but progesterone (P) was absent. 16
It is known that the frequency of vaginal clear cell adenocarcinoma was increased in young women whose mothers used diethylstilbestrol (DES) to prevent or lower the risk of abortion. 4 We previously reported that the single administration of 1 µg DES, 10 a synthetic estrogen with strong estrogenic activity and a known endocrine disruptor, or 10 µg 17 β-estradiol (E2), 5 natural estrogen, during the neonatal period in rats promoted mammary carcinogenesis induced by DMBA. We also reported that a single administration of 100 µg DES 10 or 1,000 µg E2 5 during the neonatal period induced persistent estrus (PE) and an absence of corpus lutea (CL) in female rats because of the disturbance of the gonadotropin-secreting system in the hypothalamus, resulting in inhibition of mammary carcinogenesis.
We have reported that rat mammary dysplasia (MD) induced by DMBA is morphologically similar to that in humans and is characterized by gross cysts and solid masses, with microscopic features including fibrotic adenosis and acinar adenosis (AA). 8, 9, 17, 18, 21 MD, FA, and AA are equivalent to benign proliferative lesion (PL), fibroadenoma, and lobular hyperplasia in Fisher rats, respectively. 2
We assumed that the critical period of endocrine disruption in female rats was 0–5 days after birth because a single injection of 1.25 mg testosterone propionate to female rats at 2 or 5 days after birth produced a syndrome characterized by anovulatory ovaries and altered vaginal cyclicity. 1, 3 Recently, we reported that female rats subcutaneously administered a relatively low dose (1 µg) of DES once daily from 0 to 14, 0 to 5, and 6 to 14 days after birth induced PE and an absence of CL because of the disturbance of the gonadotropin-secreting system in the hypothalamus, resulting in effects on the induction of MCs and PLs induced by DMBA. 21 We also showed that mammary epithelial cells in the residual mammary glands of these rats administered 1 µg DES once daily during neonatal periods were displaced by oxyphilic cells, which were similar to the mammary epithelial cells of intact male rats. 21
In the present study, a relatively high dose (10 µg) of DES was administered subcutaneously to neonatal female rats once daily at a critical period of morphogenesis and functional development of the mammary glands to investigate its effect on the induction of mammary tumors induced by DMBA.
Materials and Methods
Animals
The animals were inbred Sprague-Dawley (SD) female rats, maintained in a filtered air laminar flow room at the Institute of Laboratory Animal Sciences, Frontier Science Research Center, Kagoshima University. The animals were given a commercial diet (CE-2, CLEA Inc., Tokyo, Japan) and tap water ad libitum. The room temperature was maintained at 25°C ± 2°C and relative humidity at 55% ± 10%, with a 12-hour light/dark cycle. The use of animals in this research complied with all relevant guidelines set by Kagoshima University.
Study designs
Experiment A (Fig. 1): The rats in group I (n = 10) were subcutaneously administered 0.05 ml sesame oil alone once daily for 0–14 days after birth. The rats in groups II (n = 16), III (n = 8), and IV (n = 9) were subcutaneously administered DES (Sigma Chemical Co., St. Louis, MO, USA) dissolved in 0.05 ml sesame oil at 10 µg/rat once daily for 0–14, 0–5, and 6–14 days after birth, respectively. The rats in groups III and IV were subcutaneously given 0.05 ml sesame oil alone once daily from 6 to 14 and 0 to 5 days after birth, respectively, to provide an overall treatment period comparable to that of group I (control). At 21 days after birth, all rats were weaned. At 50 days after birth, all groups were given 10 mg of DMBA (Wako Pure Chemical Industries Ltd., Osaka, Japan) dissolved in 1 ml sesame oil by gastric intubation. All DMBA-administered animals except those intermediately sacrificed were examined once weekly by palpation to detect mammary tumors from 50 to 363 days after birth. The estrus cycles of all animals were examined once daily from 32 to 363 days after birth by a vaginal smear test after the vaginas of the control animals had opened. At 363 days after birth, all surviving animals underwent necropsy. All palpable mammary masses were weighed, and the longest diameter and shortest diameter of the surface were measured, and the size (cm2) calculated by multiplying their lengths. The body, ovaries, uterus, pituitary glands, and adrenal glands were also weighed.
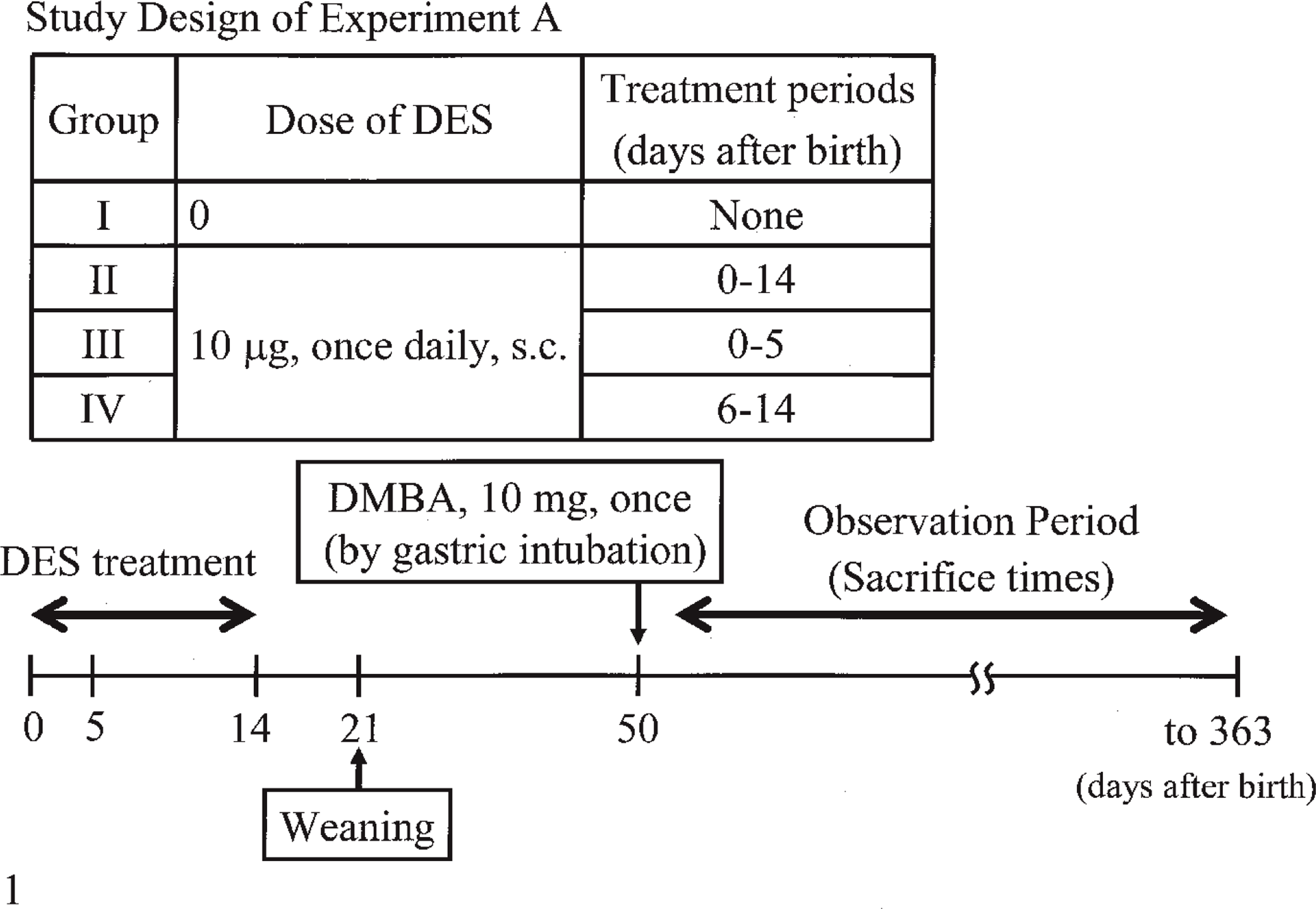
Summarized protocol of Experiment A.
Experiment B (Fig. 2): At 0–14 days after birth, 10, 9, 8, and 10 rats underwent the same treatment as groups I, II, III, and IV, respectively, in Experiment A. At 100 days after birth (without administration of DMBA at 50 days after birth), all rats underwent necropsy. The body, ovaries, uterus, pituitary glands, and adrenal glands were weighed.
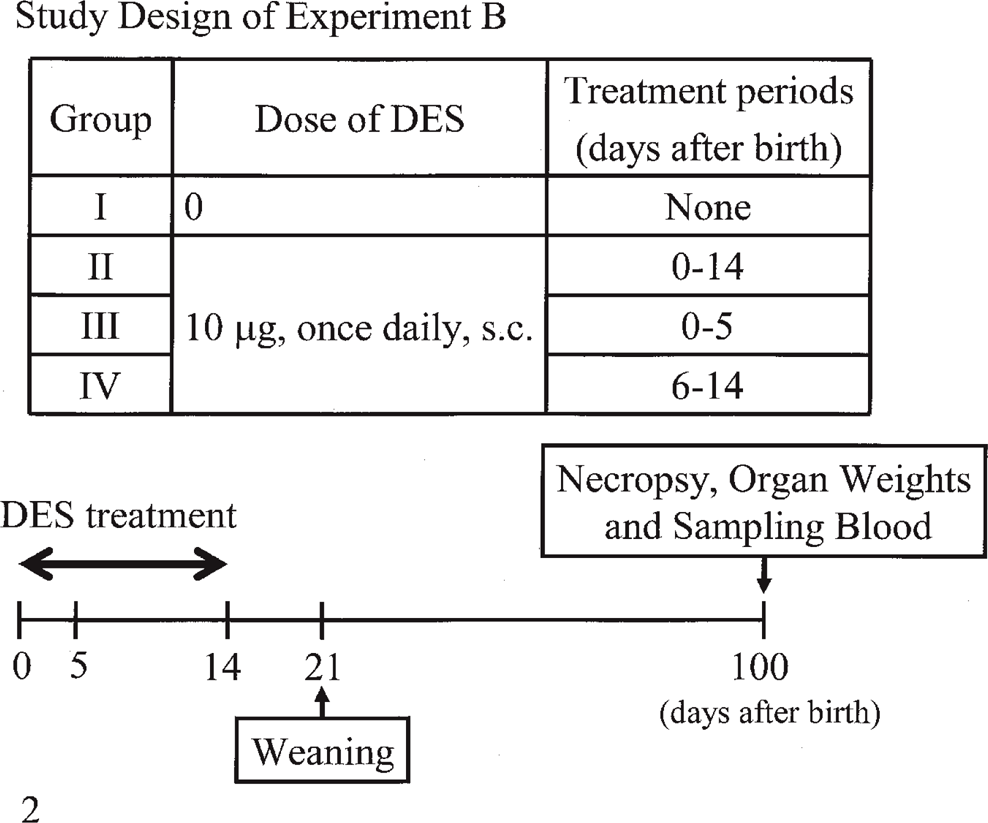
Summarized protocol of Experiment B.
Histopathologic examination (Experiment A)
All palpable mammary masses, residual mammary glands, ovaries, and pituitary were fixed in 10% phosphate-buffered formalin, dehydrated, and embedded in paraffin. The widest cut surfaces of the mammary masses, ovaries and pituitary glands, and all areas (L1-6 and R1-6) of the residual mammary glands were sectioned to 5 µm, stained routinely with HE, and then examined histopathologically.
Serum hormone levels (Experiment B)
At 100 days after birth, 16, 4, 4, and 4 rats in groups I, II, III, and IV, respectively, had blood taken from the abdominal aorta under anesthesia for determination of serum E2 and P levels. Serum hormone levels of E2 and P were determined by Shin Nippon Biomedical Laboratories, Ltd. using radioimmunoassay (RIA), with an estradiol RIA Kit (Coat-A-Count Estradiol, Diagnostic Products Corp., Los Angeles, CA) and progesterone RIA Kit (Coat-A-Count Progesterone, Diagnostic Products Corp.), respectively.
Statistics
The mean differences were evaluated by the Student's t-test. Data are shown as the mean ± standard deviation (SD). The incidences (percentages) of rats with MCs, PLs, and PE and without CL were tested using a fourfold contingency table (chi-square test).
Results
Pathology of mammary masses induced by DMBA (Experiment A)
Single or multiple mammary masses, diagnosed as MC (Figs. 3, 4) or PL, were seen in rats, while no metastasis was recognized. These masses were localized in all areas (L1-6 and R1-6) of the mammary glands. MCs were briefly subclassified into papillary type, solid type, or secretory type in this study as described previously. 6, 11 PLs consisted of solid masses, which included fibroadenoma and lobular hyperplasia, and gross cysts in this study. Fibroadenoma, by far the most common benign neoplasm of the rat mammary gland, 2 consisted of rich hyalinized stroma and developed alveoli (Fig. 5). Lobular hyperplasia consisted of large lobular structures composed of alveoli and ductules. The lumen of the ductules were dilated and filled with acidophilic secretions (Fig. 6). Gross cysts were grossly soft, yellow, oval, and filled with milky fluid. Microscopically, gross cysts were lined with a single layer of flattened or cuboidal epithelium (Fig. 7). In 4 rats in group IV, 8 masses, whose mean latent period was 290.6 days after birth, consisted of both MC and PL.
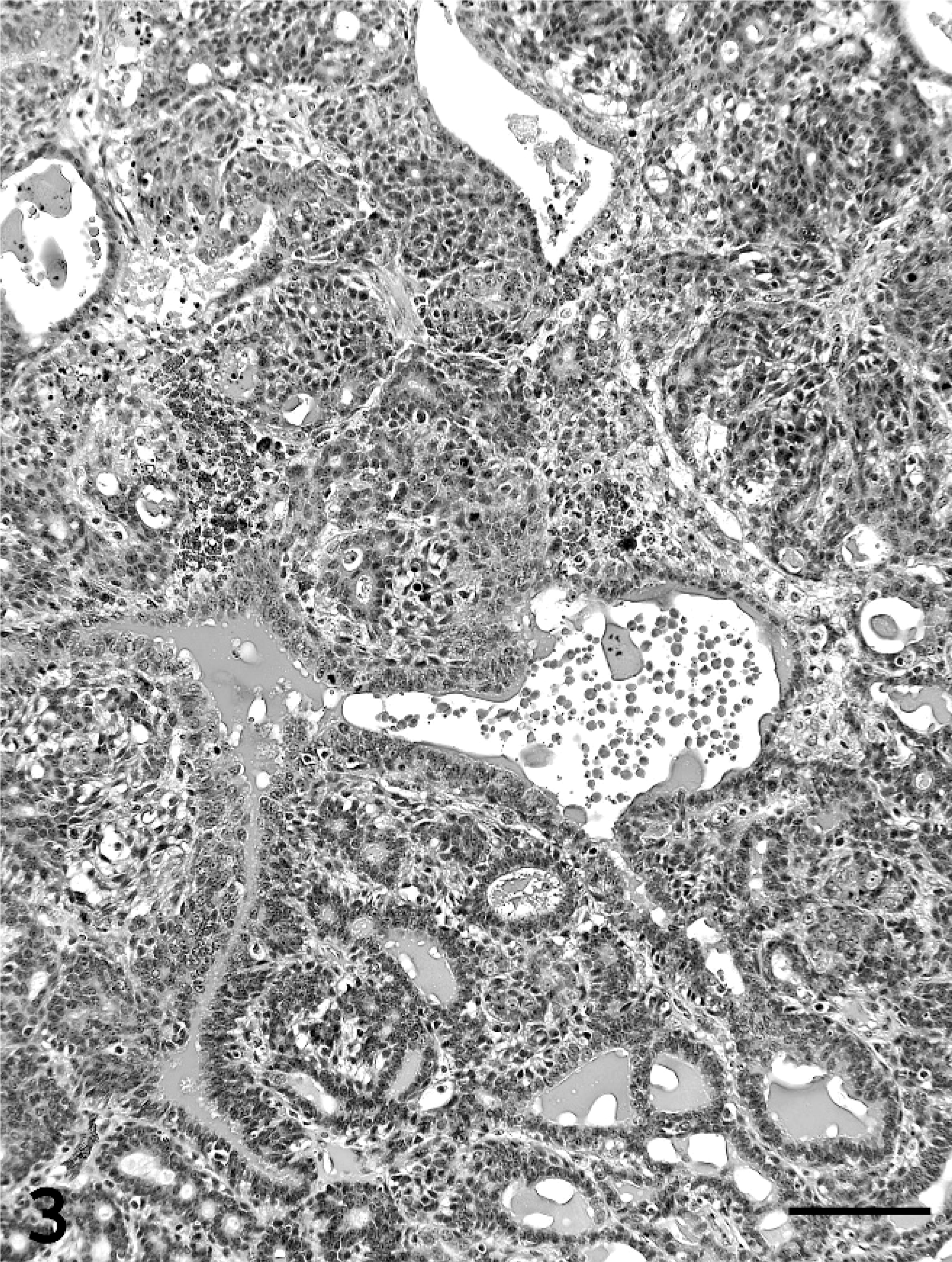
Mammary carcinoma in group IV. Tubular proliferation containing acidophilic secretions and solid pattern and multilayered epithelium are seen. Bar = 100 µm. HE stain.
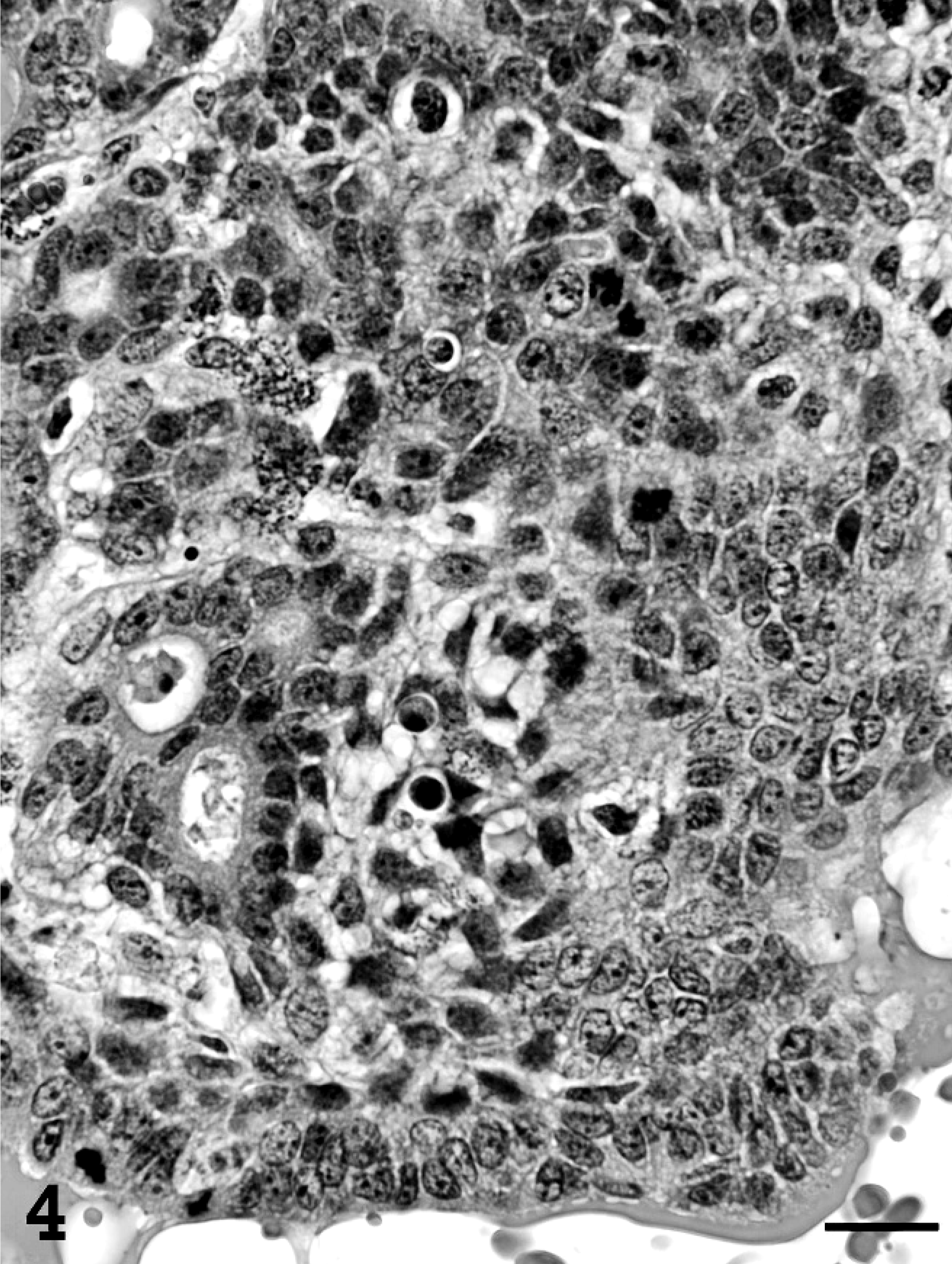
Mammary carcinoma in group IV. Mitoses, apoptosis, and prominent nuclei are seen. Bar = 20 µm. HE stain.
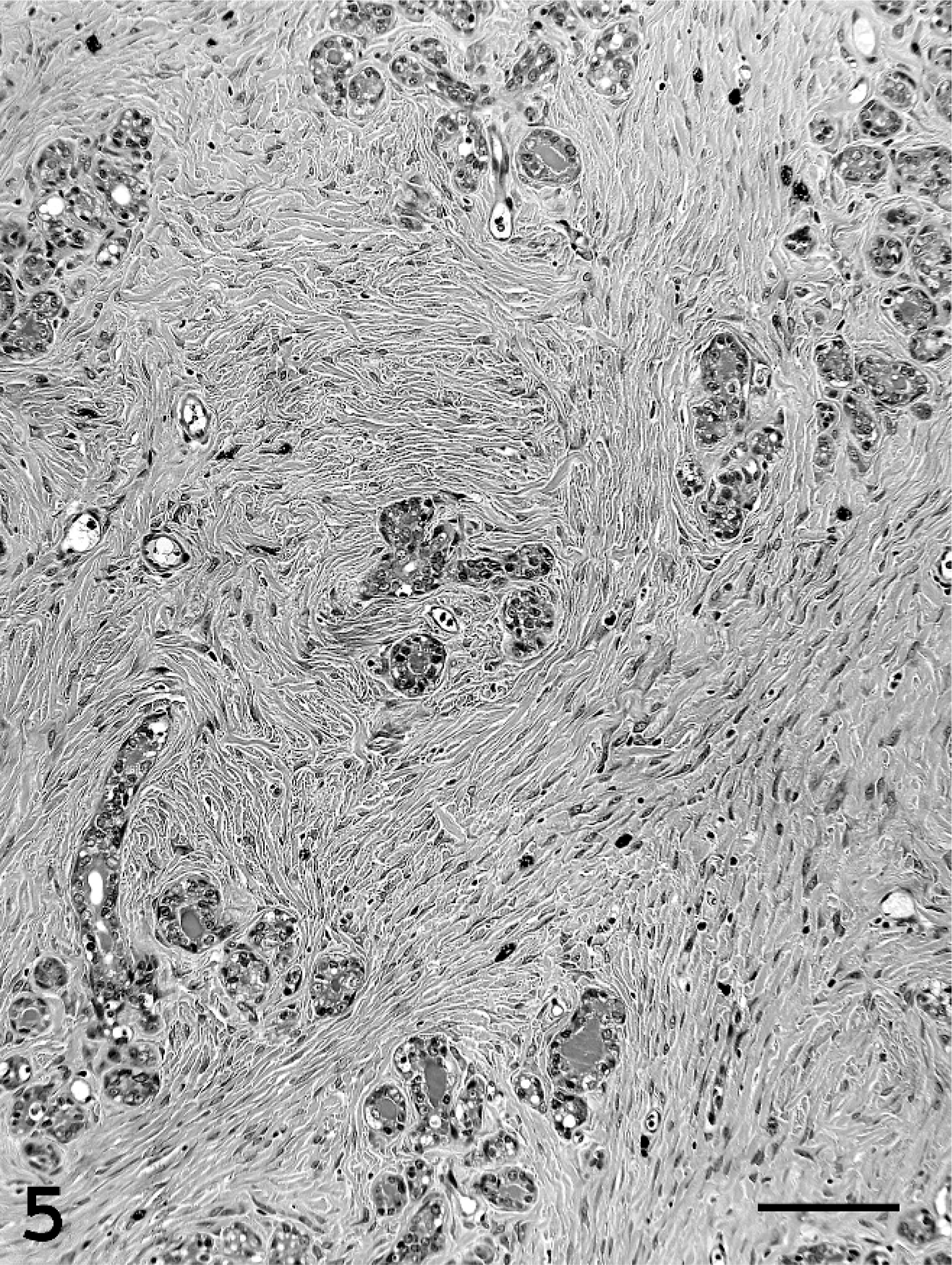
Fibroadenoma in group IV. Fibroadenoma consists of rich hyalinized stroma and developed alveoli. Bar = 100 µm. HE stain.
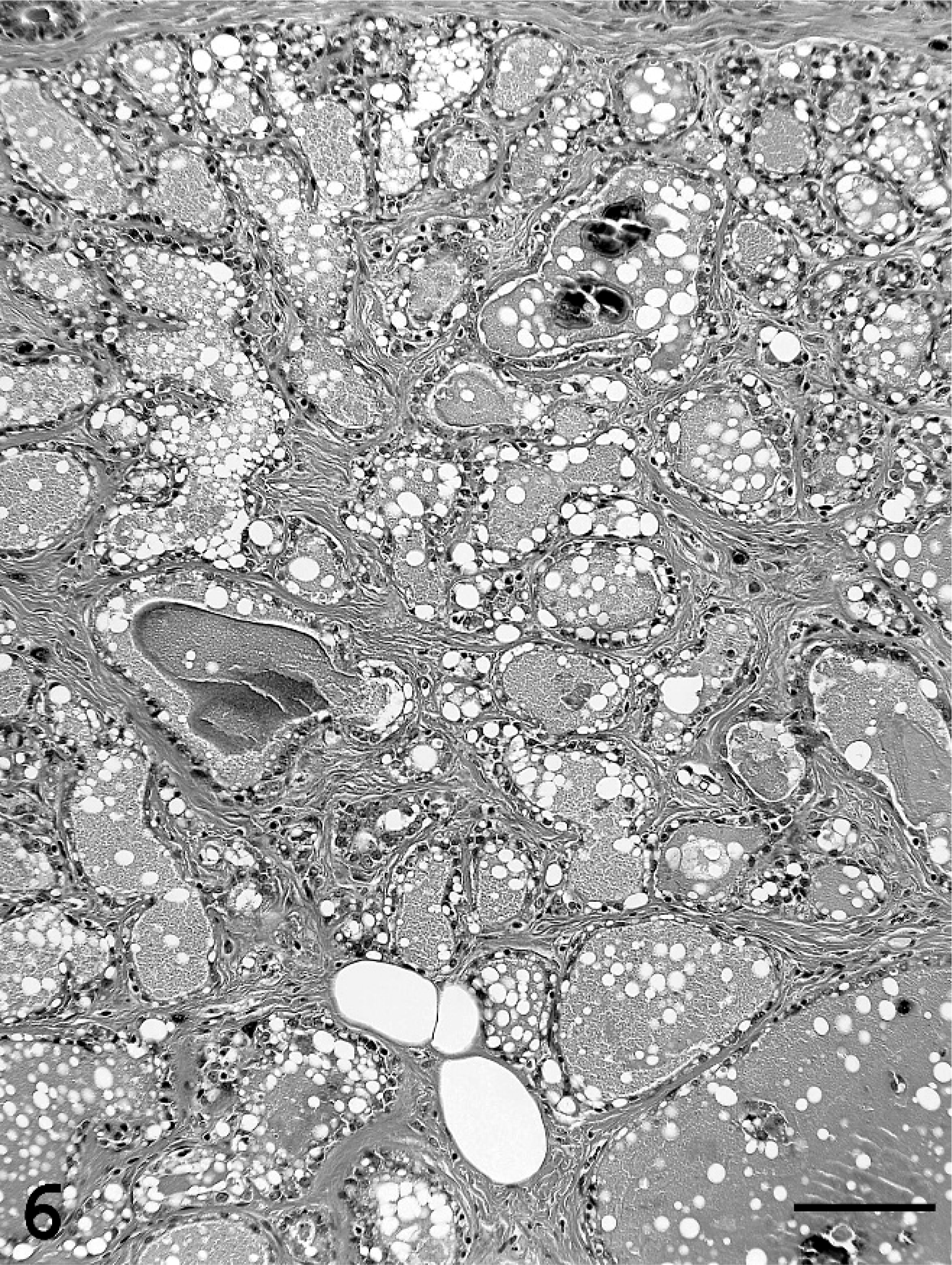
Lobular hyperplasia in group IV. Lobular hyperplasia consists of large lobular structures composed of alveoli and ductules. The lumen of the ductules were dilated and filled with acidophilic secretions. Bar = 100 µm. HE stain.
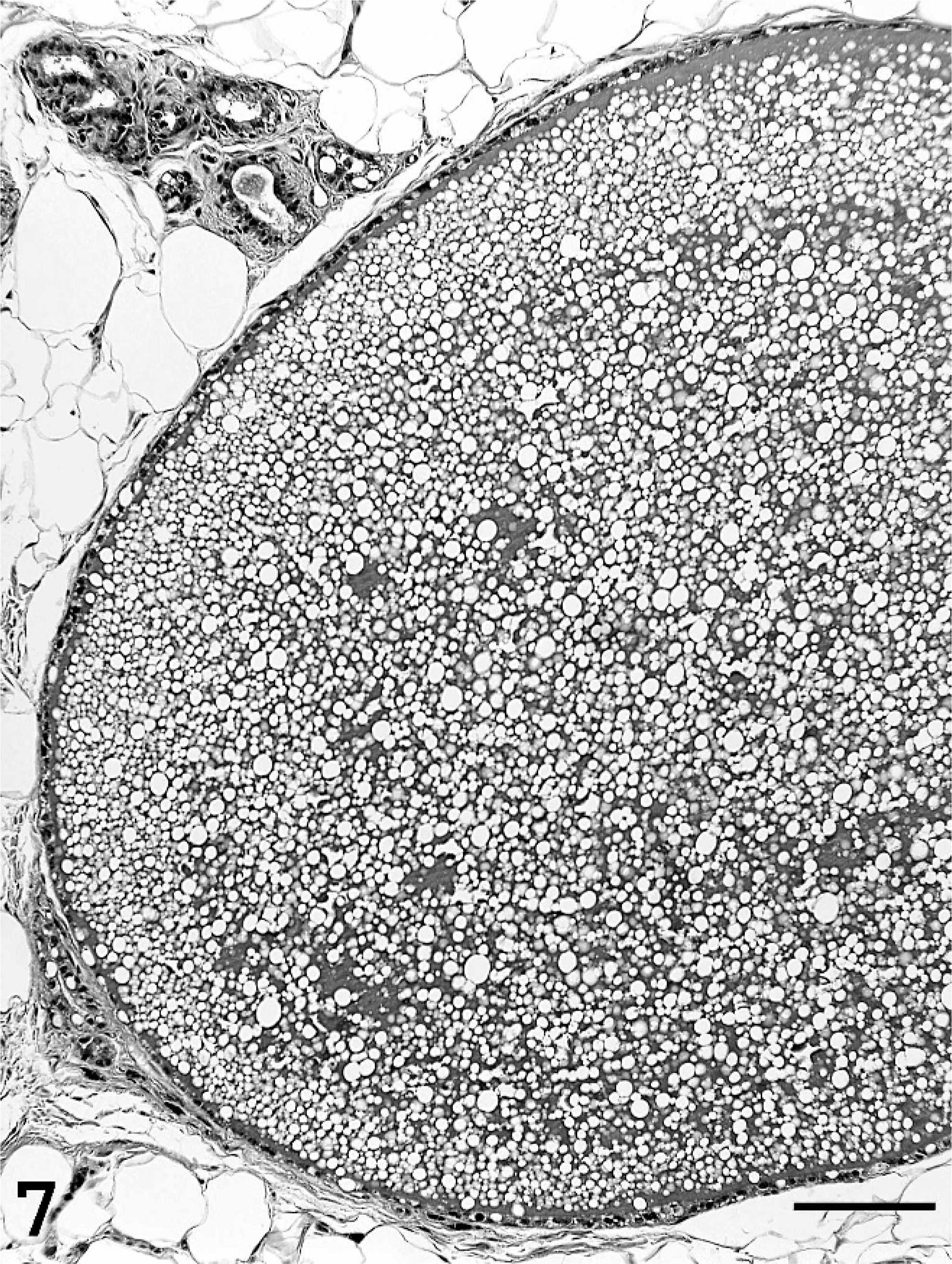
Gross cyst in group IV. Gross cyst is filled with milky fluid and lined with a single layer of flattened or cuboidal epithelium. Bar = 100 µm. HE stain.
Experiment A
The vagina opened immediately after administration of DES in groups II and III, and the day of opening in groups II, III, and IV was significantly earlier than in the control animals (all at P < .01) (Table 1). The incidence of rats with PE and an absence of CL in groups II, III, and IV was higher than that of the control animals at necropsy (all P < .01).
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the day of vaginal opening and the number (incidence) of rats with persistent estrus (PE) and without corpus lutea (CL) in ovaries (Experiment A).
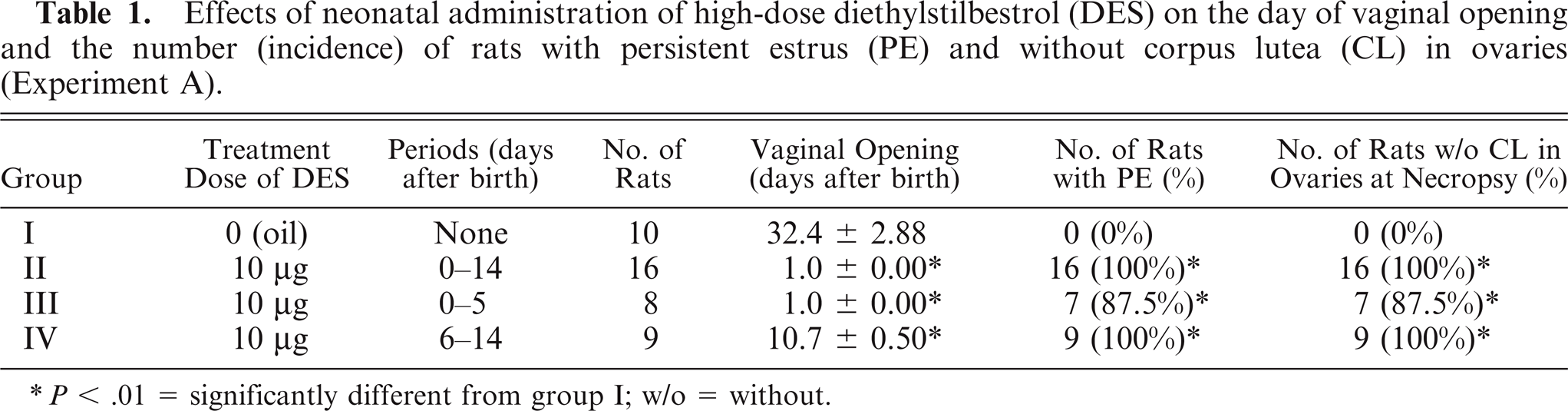
P < .01 = significantly different from group I; w/o = without.
At necropsy, the body weight (BW) of rats in group III was significantly higher than in the control animals (P < .01) (Table 2). For the ovaries and uterus, the absolute weight (AW) and the AW/BW in groups II, III, and IV were significantly lower than in the control animals (all P < .01). For the pituitary glands, the AW in group II and the AW/BW in groups II and III were significantly lower than in the control animals (all P < .01). For the adrenal glands, the AW in group II (P < .01) and the AW/BW in groups II (P < .01), III (P < .05), and IV (P < .05) were significantly lower than in the control animals.
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the body, genital, and endocrine organ weight at necropsy (Experiment A).
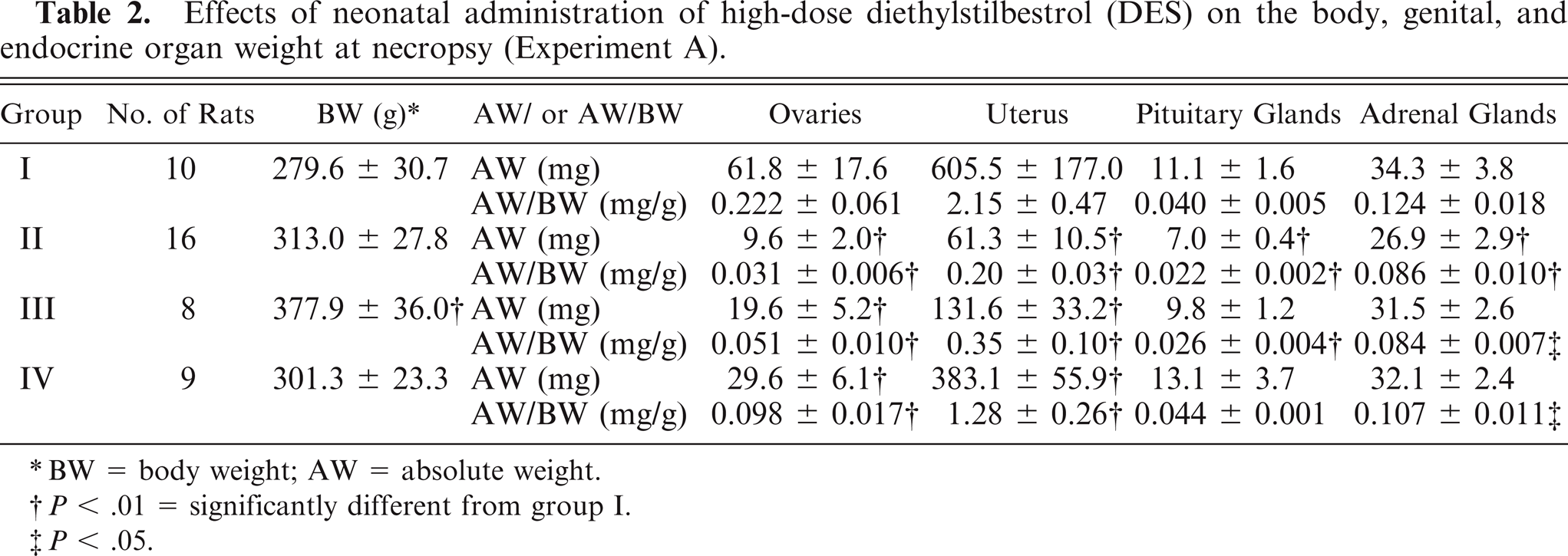
BW = body weight; AW = absolute weight.
P < .01 = significantly different from group I.
P < .05.
Single or multiple MCs or PLs were seen in rats in groups I and IV, while neither MCs nor PLs were seen in groups II and III (Tables 3, 4). The incidence of MCs and the number of MCs per rat in group IV were equivalent to that of the control animals, while the mean day of detection (latent period) in group IV (P < .01) was significantly longer than that of the control animals (Table 3). The number of PLs per rat in group IV (P < .05) was significantly higher than that of the control animals (Table 4). The number of fibroadenomas and gross cysts per rat in group IV (P < .05) was significantly higher than that of the control animals (both P < .05) (Table 5).
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the number (incidence) of mammary carcinomas (MCs) (Experiment A).
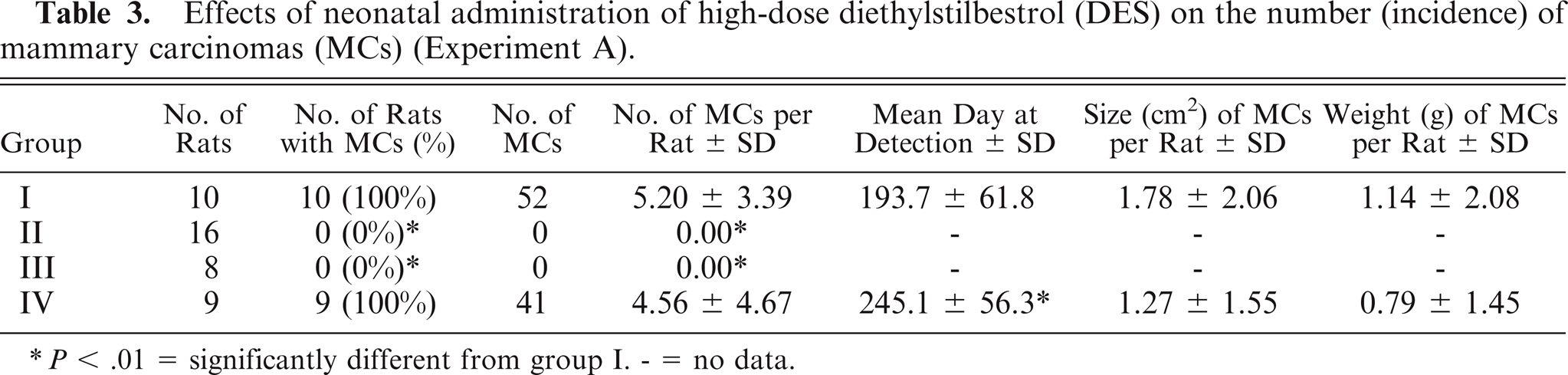
P < .01 = significantly different from group I. - = no data.
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the number (incidence) of benign proliferative lesions (PLs) (Experiment A). ∗
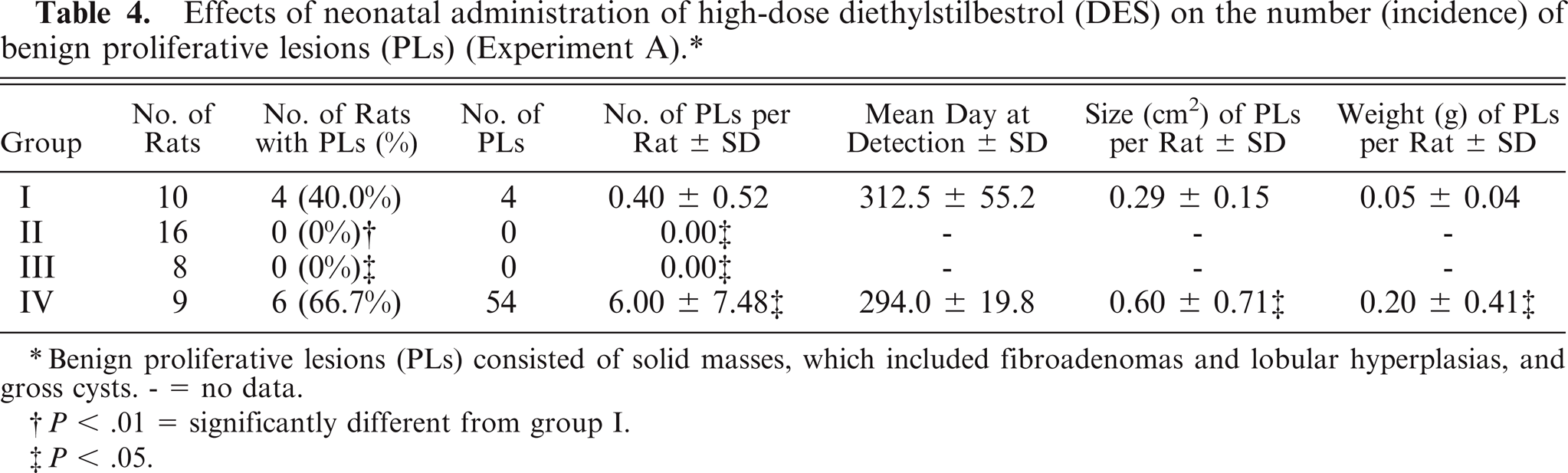
Benign proliferative lesions (PLs) consisted of solid masses, which included fibroadenomas and lobular hyperplasias, and gross cysts. - = no data.
P < .01 = significantly different from group I.
P < .05.
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the number (incidence) of solid masses and gross cysts in benign proliferative lesions (Experiment A). ∗
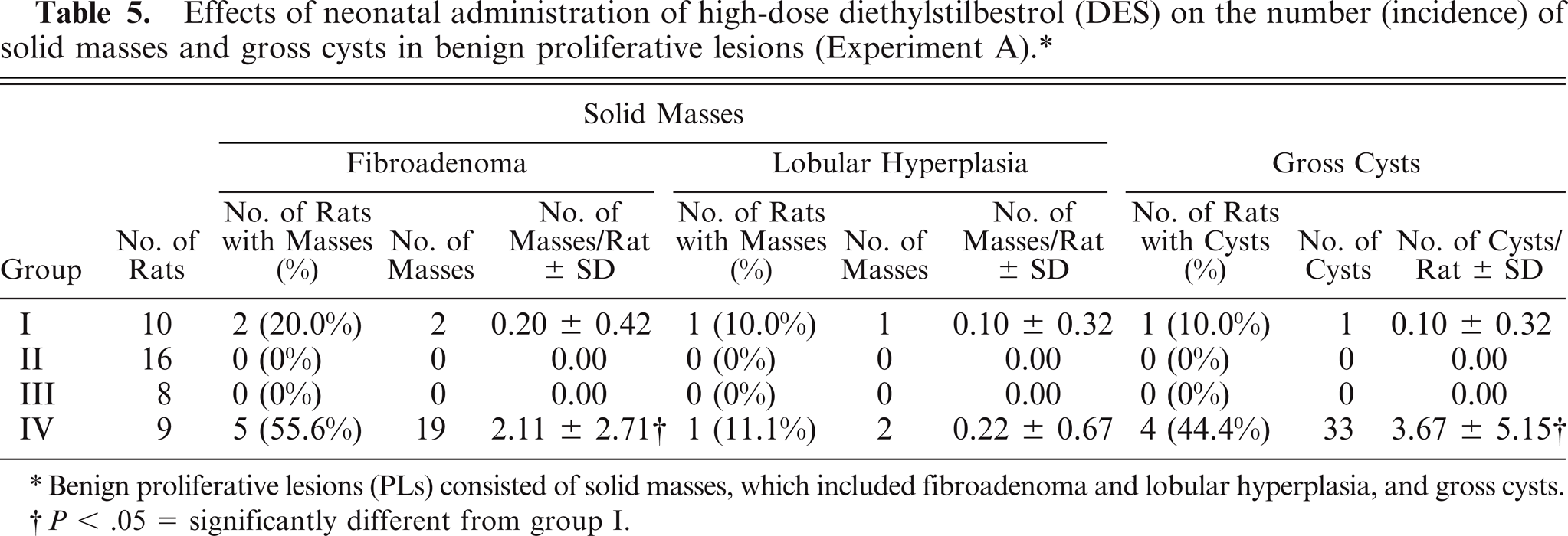
Benign proliferative lesions (PLs) consisted of solid masses, which included fibroadenoma and lobular hyperplasia, and gross cysts.
P < .05 = significantly different from group I.
The incidence of rats with active lactation in the residual mammary glands in group IV (P < .01) was significantly higher than that in the control animals at necropsy (Table 6). The incidence of rats with oxyphilic cells in groups II and III was significantly higher than that in the control animals at necropsy (both P < .01). The oxyphilic cells had rich oxyphilic cytoplasm, displaced the residual mammary glands, and were similar to the mammary glands of intact male adult SD rats as in the previous study. 21
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the number (incidence) of rats with active lactation and oxyphilic cells in residual mammary glands (Experiment A).

P < .01 = significantly different from group I.
Experiment B
At 100 days after birth, the BW of rats in groups II and III was significantly higher than in the control animals (both P < .05) (Table 7). AW and the AW/BW of the ovaries in groups II, III, and IV (all P < .01), AW of the uterus in groups II (P < .01), III (P < .01), and IV (P < .05), AW/BW of the uterus in groups II (P < .01), III (P < .01), and IV (P < .05), AW of the pituitary glands in groups II (P < .01) and III (P < .05), AW/BW of the pituitary glands in groups II (P < .01) and III (P < .05), AW of the adrenal glands in group II (P < .01), and AW/BW of the adrenal glands in groups II and III (both P < .01) were significantly lower than in the control animals. No mammary masses were grossly seen in any animal.
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on the body, genital, and endocrine organ weights at 100 days after birth (Experiment B).
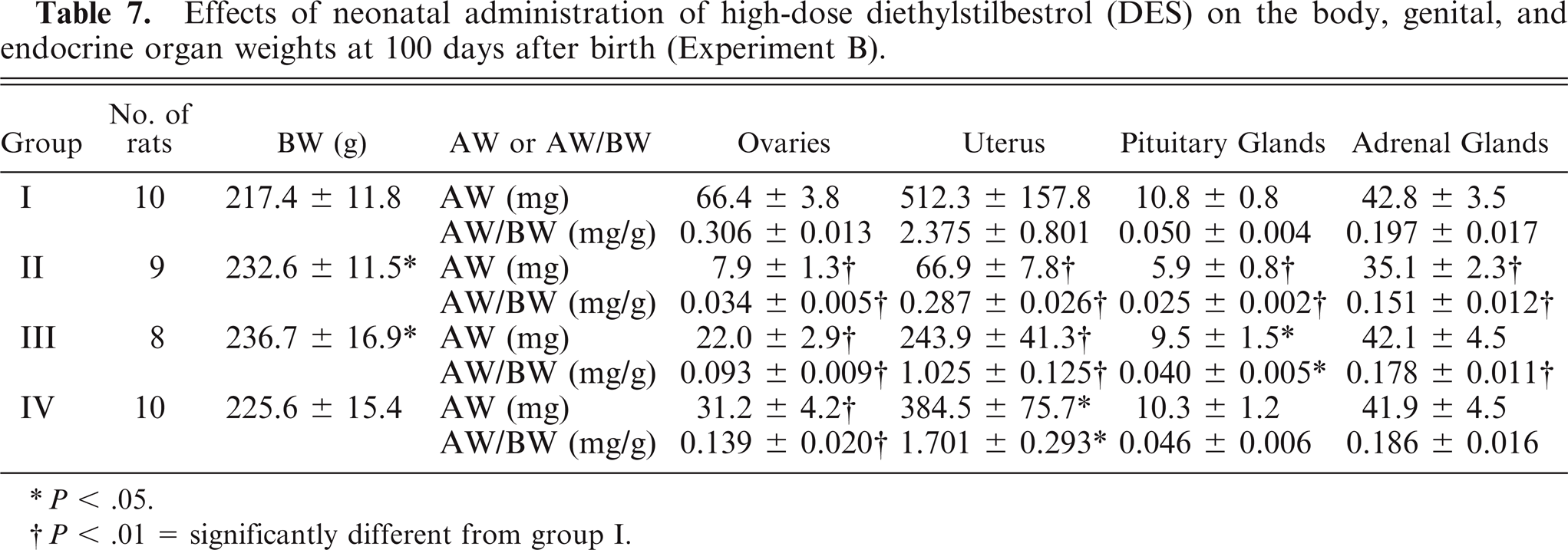
P < .05.
P < .01 = significantly different from group I.
Serum hormone levels (Experiment B)
Serum E2 and P levels in groups II, III, and IV were significantly lower than in the control animals at 100 days after birth (all P < .01) (Table 8).
Effects of neonatal administration of high-dose diethylstilbestrol (DES) on serum hormone levels at 100 days after birth (Experiment B). ∗
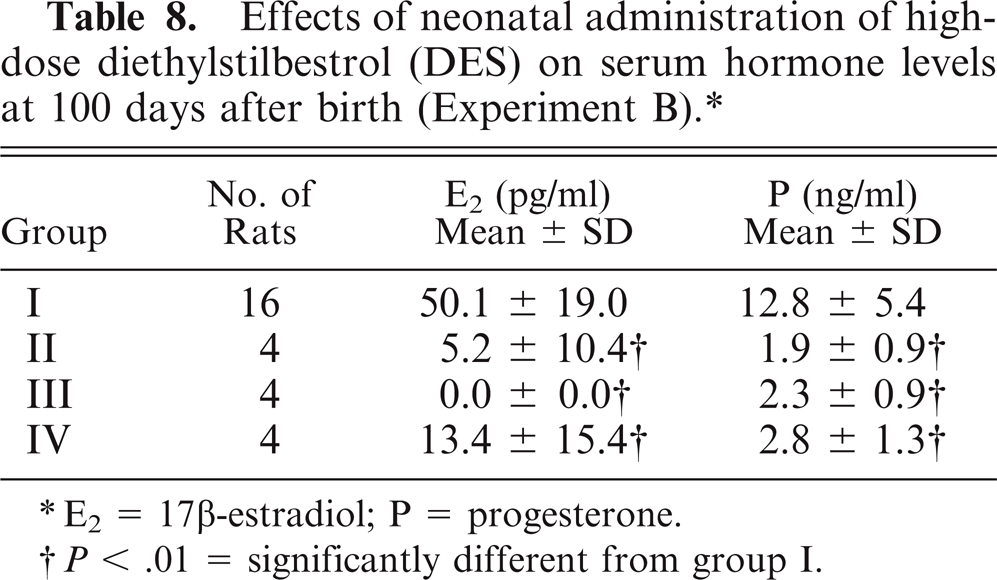
E2 = 17β-estradiol; P = progesterone.
< .01 = significantly different from group I.
Discussion
Experiment A revealed that the daily administration of a relatively high dose (10 µg) of DES from 0 to 14, 0 to 5, and 6 to 14 days after birth induced disturbance of the gonadotropin-secreting system, resulting in rats with early opening of the vagina, PE, and animals lacking CL, as with the daily administration of a relatively low dose (1 µg) of DES. 21 It was also shown that daily administration of high-dose DES decreased the weights of the ovaries, uterus, pituitary glands, and adrenal glands, possibly because of the disturbance of the gonadotropin-secreting system.
We conducted Experiment B after Experiment A to briefly investigate the effects of daily administration of a high dose of DES on serum hormone levels at 100 days after birth, at which point the rats were sexually mature. Experiment B demonstrated that daily administration of high-dose DES decreased the weights of the ovaries, uterus, pituitary glands, and adrenal glands at 100 days after birth as in Experiment A. Experiment B also showed that daily administration of a high dose of DES during the neonatal period did not induce mammary masses.
With the daily administration of low-dose DES from 0 to 14 days after birth, both MCs and PLs were inhibited because of the depletion of serum E2 and P; however, the daily administration of low-dose DES from 0 to 5 days after birth inhibited MCs, while it promoted PLs, possibly a result of hormonal conditions with a relative excess of estrogen. 21 Experiment A demonstrated that the daily administration of high-dose DES from not only 0 to 14, but also from 0 to 5 days after birth could strongly inhibit both MCs and PLs. This phenomenon is thought to be caused by greater depletion of serum E2 and P (Experiment B) than with the daily administration of low-dose DES from 0 to 5 days after birth.
In the daily administration of low-dose DES from 6 to 14 days after birth, no inhibition of MCs was seen, possibly caused by the same serum E2 and P levels as the control animals, and promotion of PLs was seen, possibly caused by hormonal conditions with a relative excess of estrogen and active lactation in the residual mammary glands. 21 The daily administration of high-dose DES from 6 to 14 days after birth resulted in a significant decrease of serum E2 and P levels in Experiment B but no inhibition of MCs in Experiment A. The precise mechanisms of these results are unknown, but it was considered that the critical period affected by endocrine disruptors may be 0–5 days after birth. Experiment A also showed that control animals had more, larger, and earlier MCs than treated animals. These results are thought to be caused by greater concentrations of E2 and P (Experiment B) in control animals than in treated animals. Alternatively, terminal end buds (TEBs) differentiate to more mature structures, namely, alveolar buds (ABs) and lobules, which are less susceptible to carcinogens in rats. 13, 14 TEBs that had already differentiated into ABs before DMBA administration did not develop carcinomas but remained unmodified or underwent dilatation, giving rise to hyperplastic lobules. 12 However, it is possible that dysplastic lobules may develop carcinomas because both MCs and dysplastic lobules were found in the same rat or in the mass following daily administration of a high dose of DES from 6 to 14 days after birth, and the masses consisting of both MC and PL had long latent periods in Experiment A.
Experiment A also showed that the daily administration of high-dose DES from 6 to 14 days after birth promoted PLs. This phenomenon is thought to have been caused by a relative excess of estrogen (Experiment B) and active lactation (Experiment A) in the residual mammary glands as described previously. 21
These results demonstrated that administration of high-dose DES from 6 to 14 days after birth did not inhibit MCs or PLs induced by DMBA. It is suggested that the critical period for exposure to endocrine disruptors, such as DES, affecting the induction of MCs and PLs may be from 0 to 5 days after birth.
Footnotes
Acknowledgements
This work was supported in part by the Kodama Memorial Fund Medical Research. We are grateful to Mr. T. Kodama for his valuable technical assistance.
