Abstract
Introduction
Gardenia blue is currently being considered as a naturally derived food colorant for use in the global marketplace.
Methods
To assess its carcinogenic potential, 100 female and 100 male CByB6F1-Tg (HRAS)2Jic (rasH2) mice were allocated to four dose groups and exposed to gardenia blue in the diet for 26 weeks at dose levels of 0.0% (control), 0.5%, 2.5%, or 5.0% (corresponding to 0.0, 664.8, 3341.0, and 6623.2 mg/kg/day in male mice and 0.0, 1182.7, 5561.1, and 10,440.3 mg/kg/day in female mice, respectively). An additional group of 10 males and 10 females was administered intraperitoneal N-methyl-N-nitrosourea (MNU) as a positive control. Clinical observations, body and organ weights, clinical chemistry, hematology, and hormone analyses were performed in addition to urinalysis and histopathology.
Results
The positive control elicited expected responses specific to rasH2 mice. There were sporadic background non-dose-related findings in clinical pathology parameters and anatomic pathology common to rasH2 mice in the absence of any gardenia blue induced dose-related changes.
Discussion
Under these study conditions, the no-observed-adverse-effect level was 5% gardenia blue (6623.2 mg/kg/day in male mice and 10,440.3 mg/kg/day in female mice).
Conclusions
Based on this study a high dietary level of gardenia blue was negative for carcinogenicity in the rasH2 mouse test system.
Introduction
Gardenia blue, the principal coloring component of gardenia blue powder, is obtained from the fruit of Gardenia jasminoides Ellis. Following aqueous extraction from the fruit, the extraction liquid is passed through a reverse osmotic membrane to yield a high molecular weight fraction, gardenia yellow (i.e., crocin and crocetin), and a low molecular weight fraction consisting of iridoid glycoside (up to 45% geniposide). These iridoid glycosides are converted to their aglycones by treatment with β-glucosidase converting the geniposide to genipin. When genipin is treated with a protein hydrolysate in the presence of oxygen, polymerization occurs and a polymeric blue color, gardenia blue, is formed. The gardenia blue polymer is then heated to denature the enzyme and filtered. The color additive gardenia blue is prepared by mixing the gardenia blue polymer with food grade dextrin or maltodextrin. The amount of dextrin/maltodextrin is added to achieve a specific color value.
Gardenia blue colorant produced by San-Ei Gen, FFI, Inc., conforms to the specifications described in the current ninth edition of “Japan’s Specifications and Standards for Food Additives”. 1 Gardenia blue is permitted for use as a color additive for food and beverage in Japan, China, Taiwan, and Korea and has been used in Japan for over 30 years. Following leads from traditional Chinese medicine, genipin and geniposide extracts of gardenia fruit (Gardenia jasminoides Ellis) have been widely used based on pharmacological and physiological properties including anti-inflammatory properties,2–4 neuroprotective effects5,6 and anti-cancer activity.7–9 Annual production in Japan is about 138 metric tons (as color value 50) with a per capita intake of about 3.0 mg/person/day. 10
An extensive battery of GLP-compliant genotoxicity studies of gardenia blue and its precursor, genipin, including combined micronucleus/comet assays and “reverse” comet assays of gardenia blue, indicate gardenia blue in food presents no significant genotoxic concern for humans. 11 Early toxicity studies at dietary levels as great as 5% have been negative for adverse treatment-related effects.12,13 Recent Good Laboratory Practices (GLP)-compliant studies at dietary doses up to 5% have demonstrated no treatment-related adverse effects for prenatal development, 14 in a 12-months rat toxicity study following in utero exposure, 15 or in a 2-years rat carcinogenicity study. 16
While synthetic color additives still dominate the food coloring marketplace, identification and commercialization of natural source colorants is an ongoing effort to satisfy consumer demand for food to be as natural as possible and to capitalize on potential health benefits of natural pigments.17–20 Identification of new colorants for foods and beverages has prompted the present rasH2 cancer bioassay as part of a safety assessment of the natural food colorant, gardenia blue, conducted under contemporary USFDA GLP. 21
Methods
Identification of test articles
Gardenia blue (lot number 180,910; San-Ei Gen FFI, Inc., Osaka, Japan) consisting primarily of 32.3% gardenia blue color and 62.7% maltodextrin was mixed with Purina Certified 5002 meal diet at dose concentrations of 0.5%, 2.5%, and 5.0% every 2 months and kept at room temperature. N-methyl-N-nitrosourea (CAS 684-93-5) was obtained from Spectrum Chemicals, New Brunswick, NJ and delivered in pH 4.0 citrate buffer from Sigma-Aldrich, St Louis, MO. Dose formulation and analyses were performed at RTI International, Research Triangle Park, NC, and were consistently within acceptable specifications.
Animal husbandry and maintenance
CByB6F1-Tg (HRAS)2Jic (rasH2) mice of approximately 5 weeks of age were obtained from Taconic Biosciences, Inc. (Germantown, NJ), individually identified by transponder chip (BioMedic Data Systems, Inc., Seaford, DE), and housed individually in polycarbonate caging with microinsulator tops and absorbent heat-treated hardwood bedding (Northeastern Products Corp., Warrensburg, NY) that was changed weekly. Prior to assignment to treatment groups, animals were given Purina Certified 5002 meal diet (Ralston Purina Co, St Louis, MO) ad libitum as the carrier diet and reverse osmosis-treated tap water. The mice were allowed at least 7 days acclimation prior to inclusion in the study and were approximately 7–9 weeks of age at the start of dosing. Animal weights at start of dosing were 18.2–21.6 grams for females and 22.8–28 grams for males. Following approval from the Integrated Laboratory Systems (ILS), Inc. (Research Triangle Park, NC, USA) Animal Care and Use Committee, all procedures during the study were carried out in compliance with the Animal Welfare Act Regulations (9 CFR 1–4), and animals were handled and treated according to the Guide for the Care and Use of Laboratory Animals. 22
Experimental design
Group designation, animal identification, and dose levels.
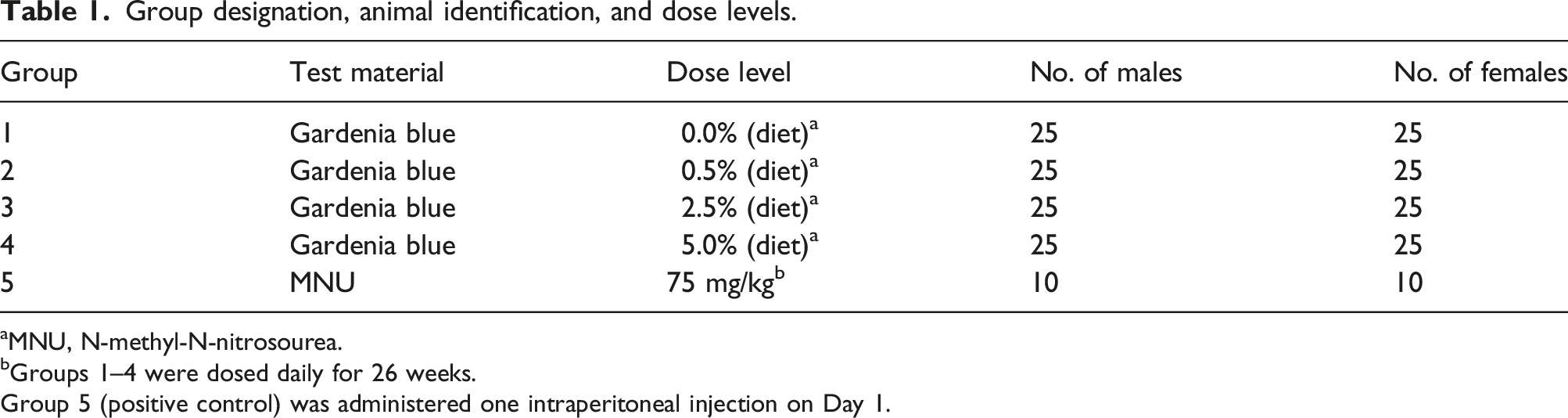
aMNU, N-methyl-N-nitrosourea.
bGroups 1–4 were dosed daily for 26 weeks.
Group 5 (positive control) was administered one intraperitoneal injection on Day 1.
Viability, clinical signs, body weight and food consumption
Cage-side observations for individual animals were performed daily following the initiation of dosing on day 1. Observations for morbidity and mortality were performed twice weekly on weekdays and once daily on weekends and holidays. Observations for clinical signs and body weight measurements were performed within 2 days of arrival, during allocation to an exposure group, prior to exposure on day 1, weekly thereafter, and at termination. Food consumption was measured at least once weekly with the exception of mice administered the positive control, MNU. Urine samples from 10 males and 10 females per dose group were collected prior to termination.
Survival, necropsy, blood collection, and tissue handling
All animals in the control and test article-treated groups (Groups 1–4) survived to scheduled sacrifice except for the following: one found dead male in the control group; 2 found dead or moribund females in the 0.5% group; 3 moribund males and 1 found dead female in the 2.5% group; and two moribund or found dead males and 4 moribund or found dead females in the 5.0% group. Lymphoma was determined to be the cause of death for 2 of the early mortality mice while cause of death for the remaining early deaths was not determined. The animals that did not survive to the terminal necropsy did not exhibit any toxicity or carcinogenicity effects attributable to the test article. Animals of both sexes in the MNU positive control group were euthanized at study termination or found dead prior to the terminal necropsy.
Tissues preserved for histopathology. a

aTissues were paraffin-embedded, sectioned, and stained with hematoxylin and eosin for histopathology from all control and high dose mice, except for spleen, harderian glands, and lungs, which were processed and examined from all gardenia blue-dosed groups. 15
bTissues were weighed at necropsy. Thyroids/parathyroids were weighed after fixation.
Statistical analysis
Group means and standard deviations were calculated and reported. All data were analyzed using Statistical Analysis System version 9.2 (SAS Institute, Cary, NC), including final body weight, body weight gain, food consumption (g/kg/day), urinalysis endpoints, clinical pathology endpoints, absolute and relative organ weights, and histopathological endpoints. First, studentized residual plots were used to detect possible outliers in the data. Homogeneity of variance was then analyzed using Levene’s test. 29 If the data were heterogeneous, then appropriate transformation of the data was performed (logarithm, square root, and/or multiplicative inverse), and the data were re-analyzed for homogeneity of variance. Data were then analyzed using a one-way analysis of variance (ANOVA) and treated groups were compared to the appropriate control group using Dunnett’s test. 30 Finally, dose-dependent changes were evaluated using a linear regression model. If data could not be transformed to be homogeneous, data were evaluated using the non-parametric Dunn’s test 31 and dose-dependent changes analyzed via the non-parametric Jonckheere’s trend test. 32
Results
Survival, clinical observations, body weights, and test article consumption
Male body weight changes, food consumption, and gardenia blue consumption.
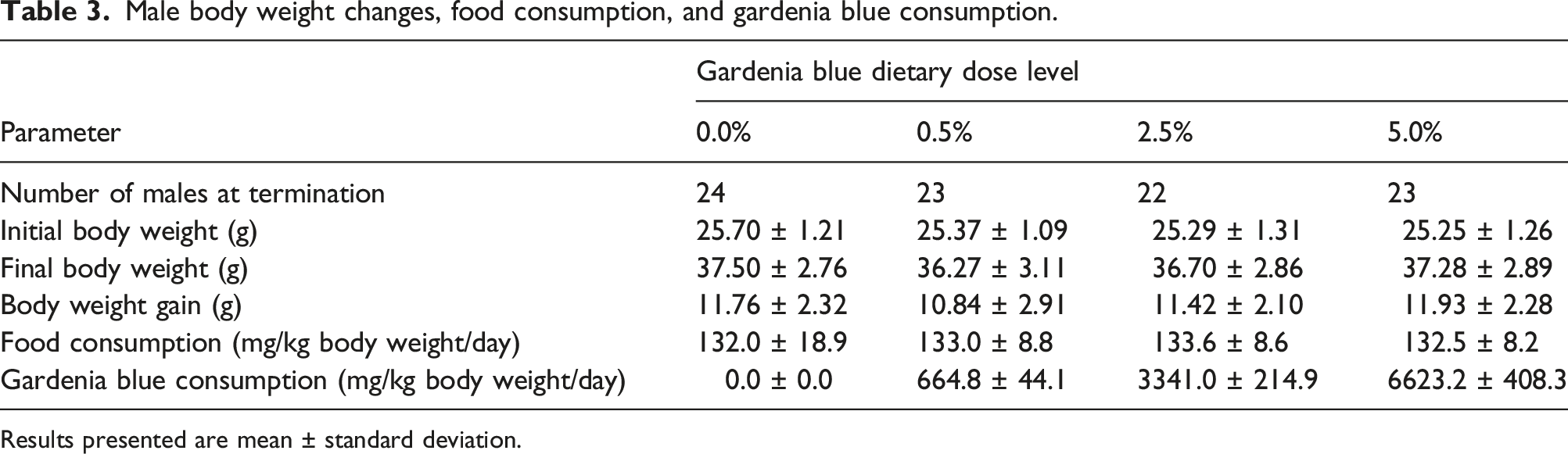
Results presented are mean ± standard deviation.
Female body weight changes, food consumption, and gardenia blue consumption.
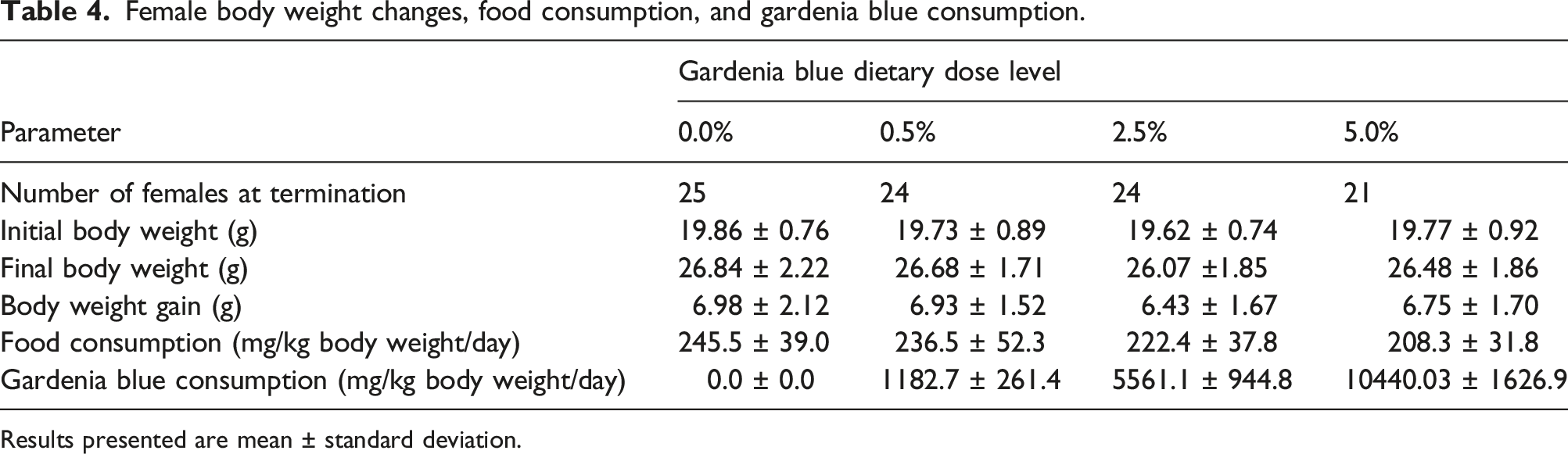
Results presented are mean ± standard deviation.
Tissue weights
Male absolute organ weights.
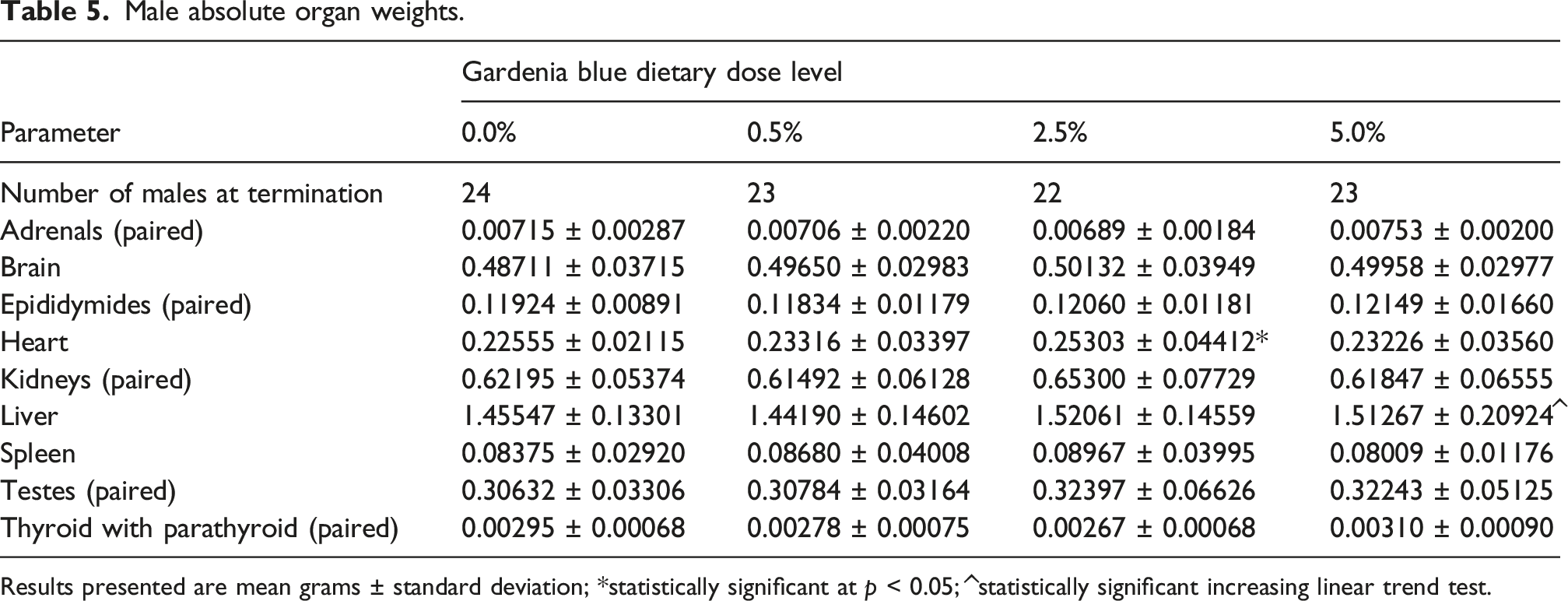
Results presented are mean grams ± standard deviation; *statistically significant at p < 0.05; ^statistically significant increasing linear trend test.
Female absolute organ weights.
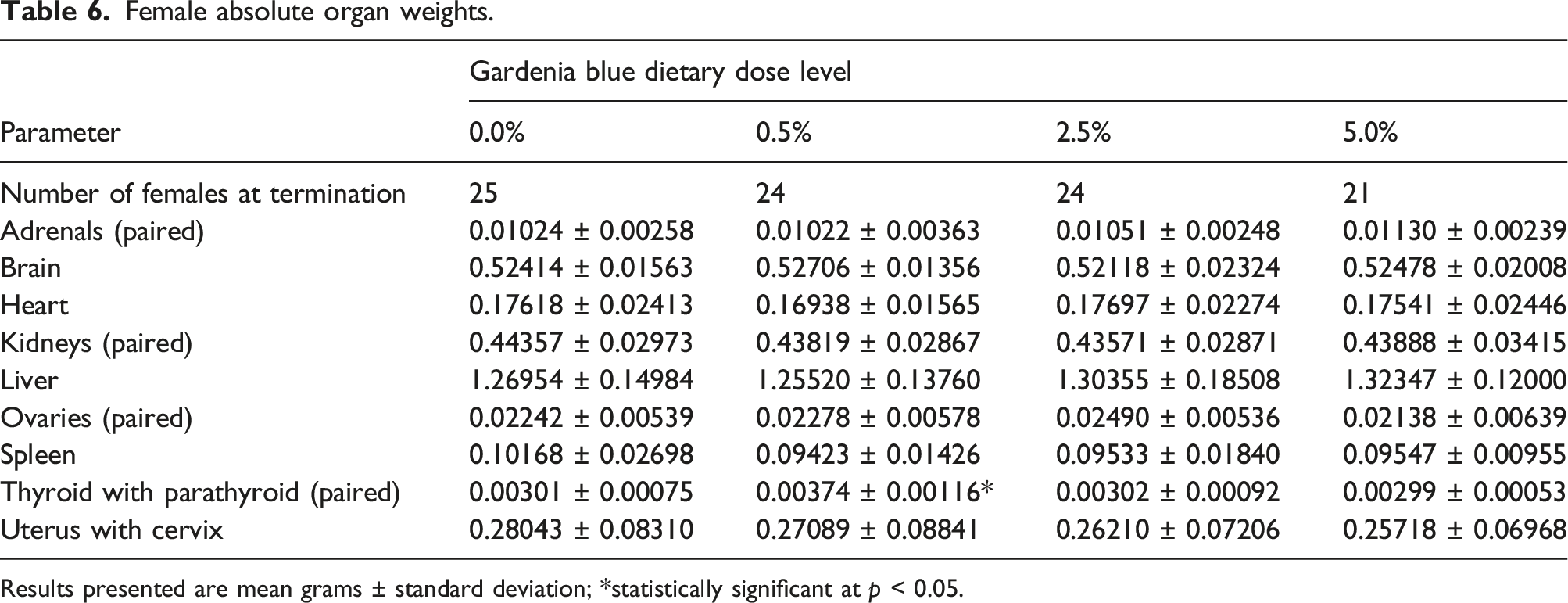
Results presented are mean grams ± standard deviation; *statistically significant at p < 0.05.
Male relative organ weights.
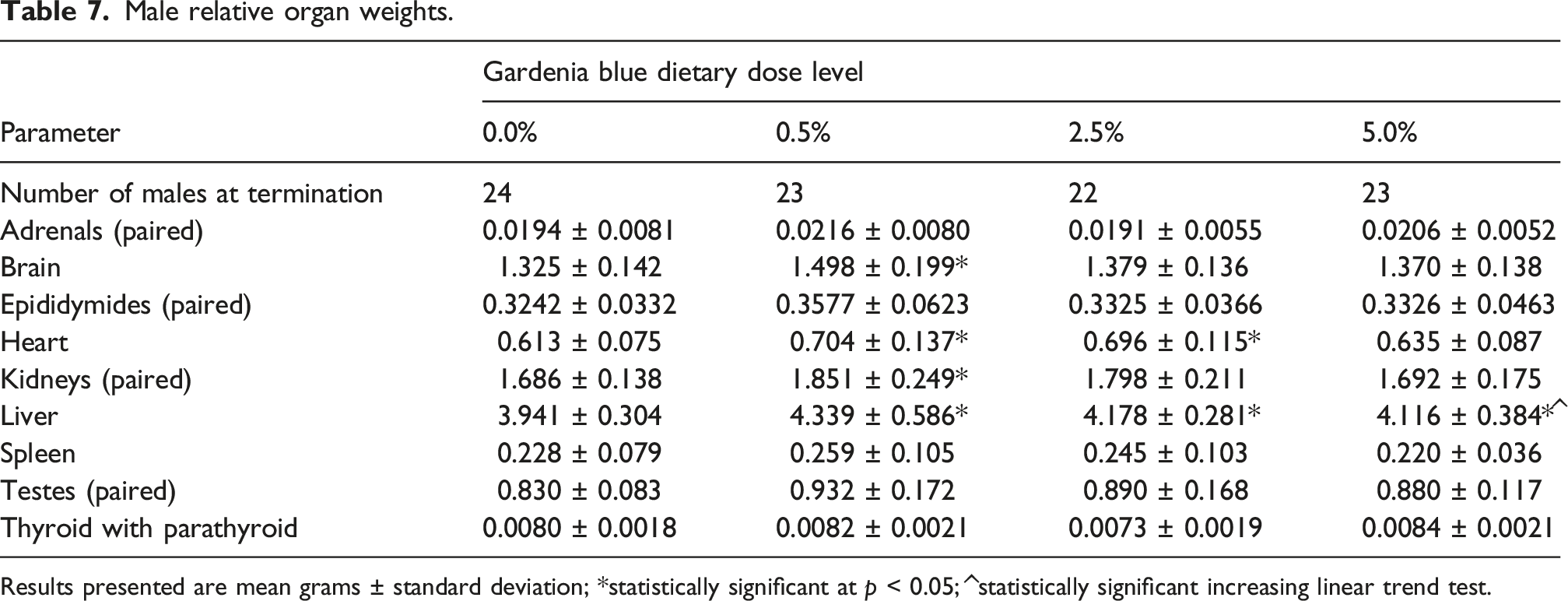
Results presented are mean grams ± standard deviation; *statistically significant at p < 0.05; ^statistically significant increasing linear trend test.
Female relative organ weights.
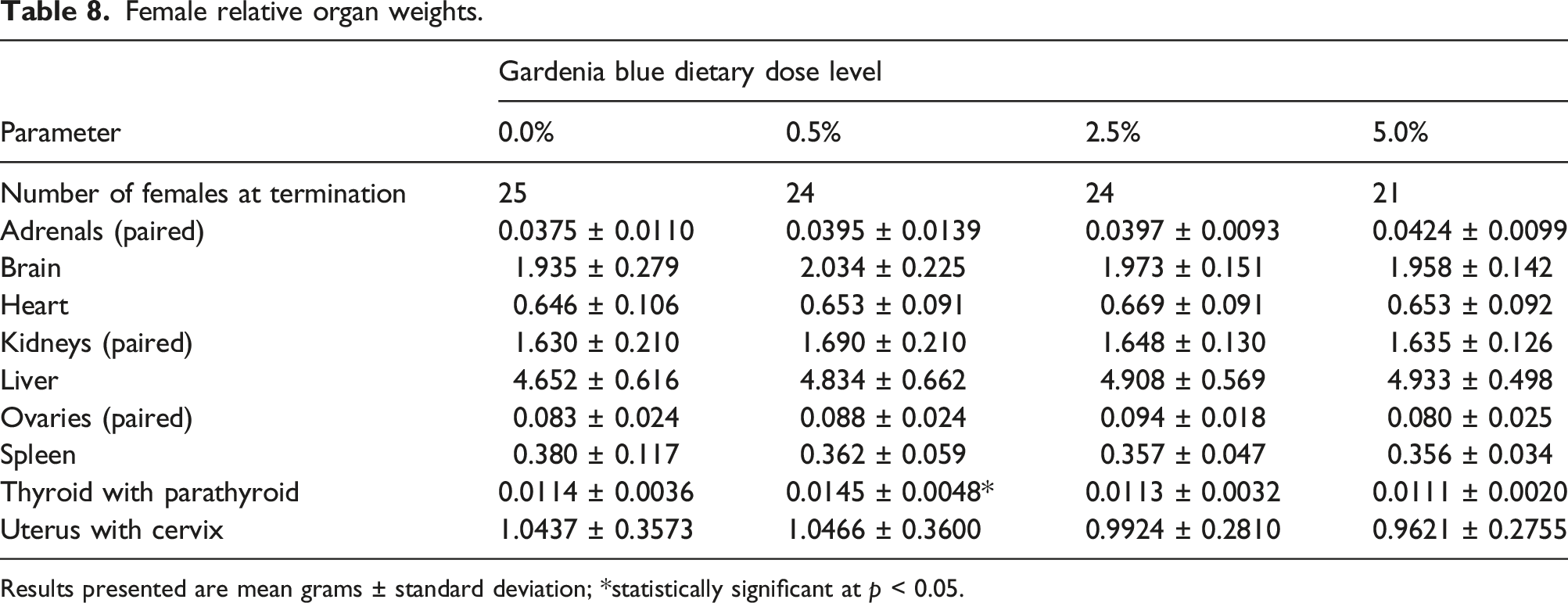
Results presented are mean grams ± standard deviation; *statistically significant at p < 0.05.
Clinical pathology
Male complete blood count and coagulation data.
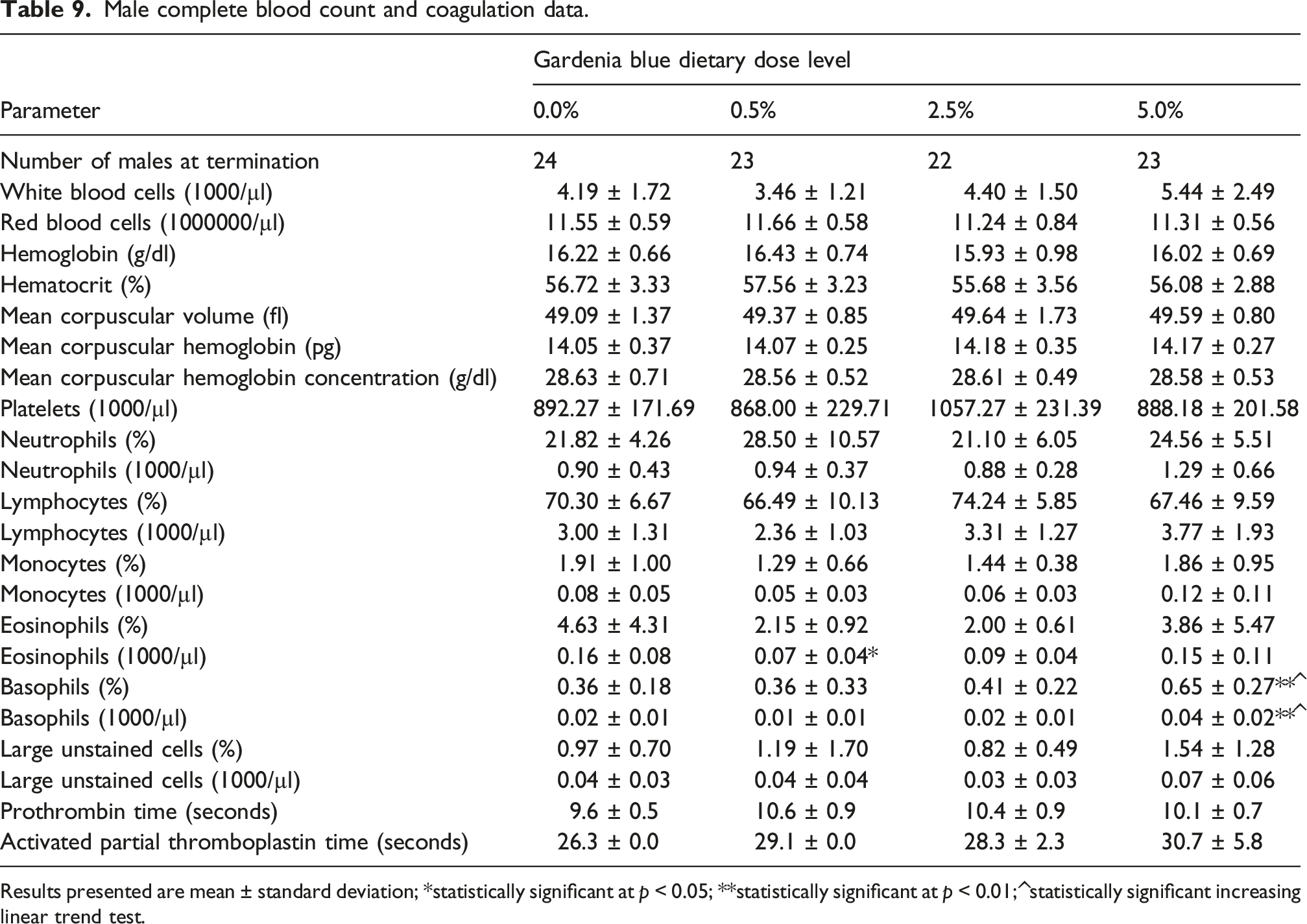
Results presented are mean ± standard deviation; *statistically significant at p < 0.05; **statistically significant at p < 0.01; ^statistically significant increasing linear trend test.
Male clinical chemistry data.
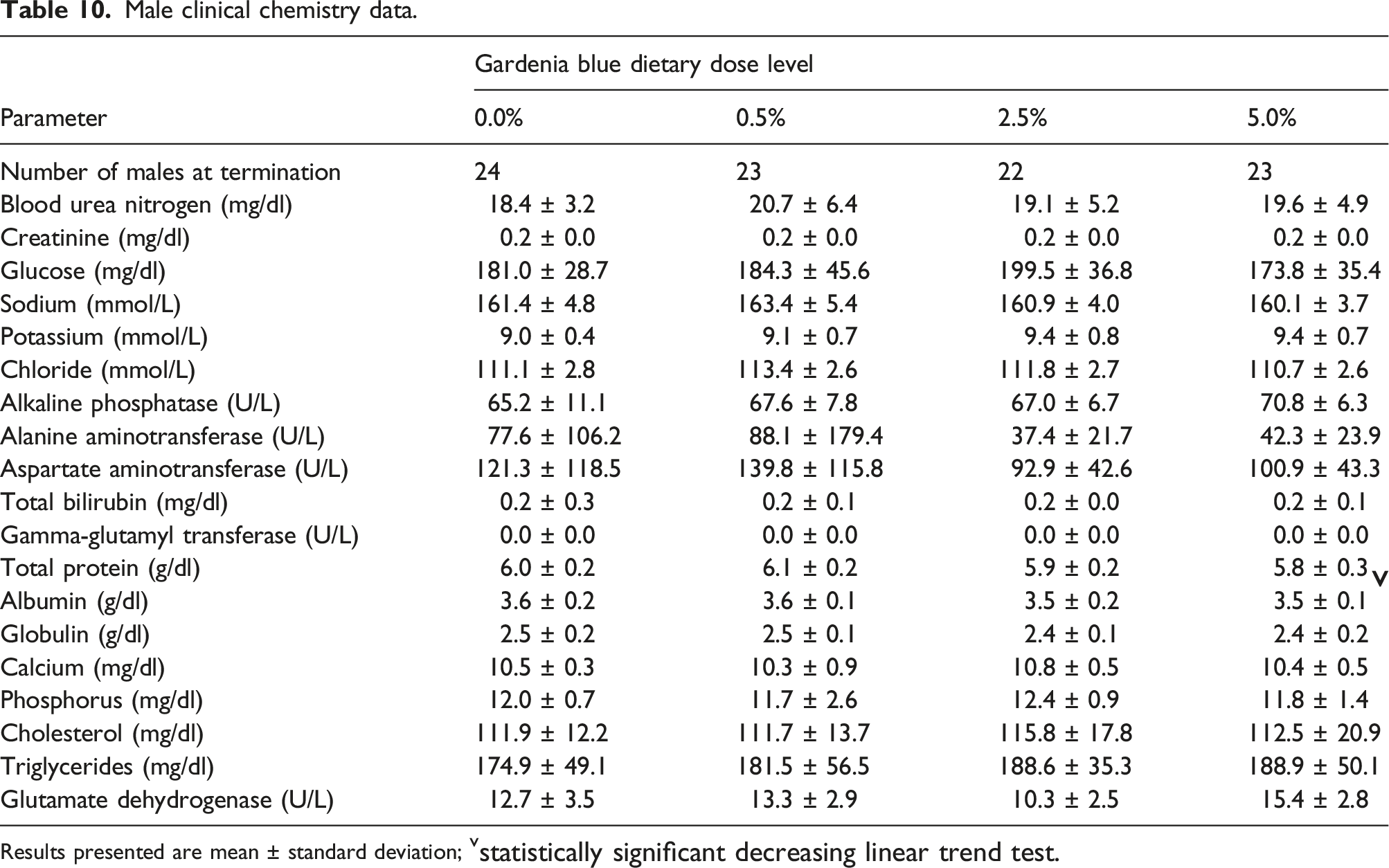
Results presented are mean ± standard deviation; vstatistically significant decreasing linear trend test.
Male urinalysis data.
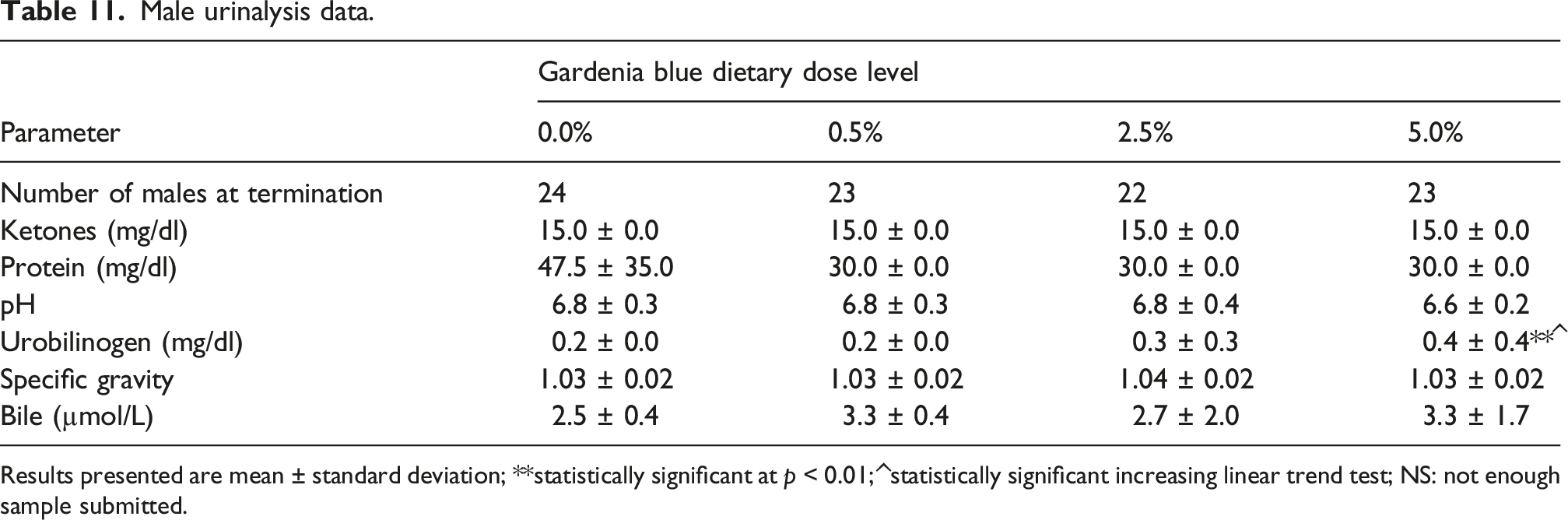
Results presented are mean ± standard deviation; **statistically significant at p < 0.01; ^statistically significant increasing linear trend test; NS: not enough sample submitted.
Female complete blood count and coagulation data.
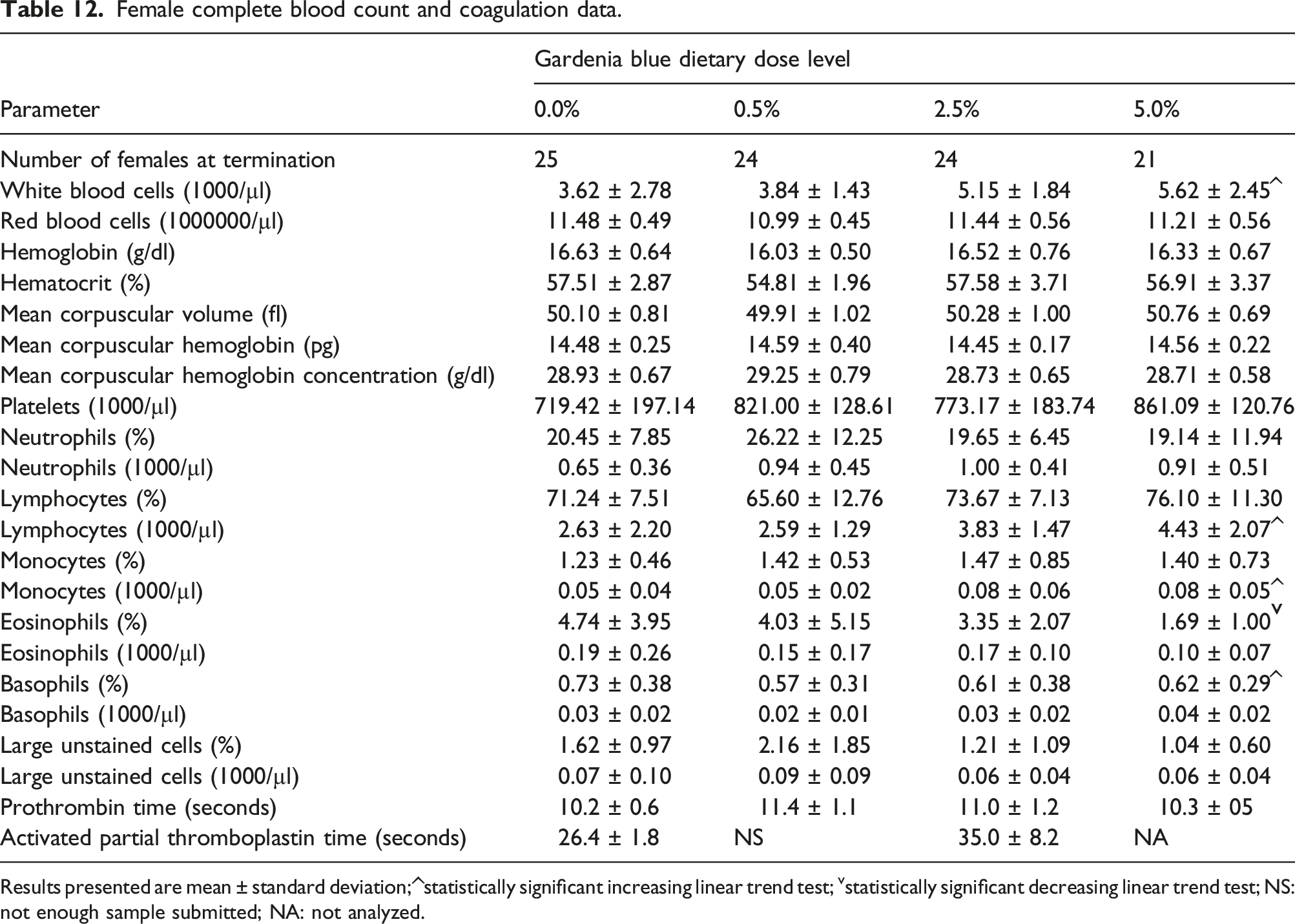
Results presented are mean ± standard deviation; ^statistically significant increasing linear trend test; vstatistically significant decreasing linear trend test; NS: not enough sample submitted; NA: not analyzed.
Female clinical chemistry data.
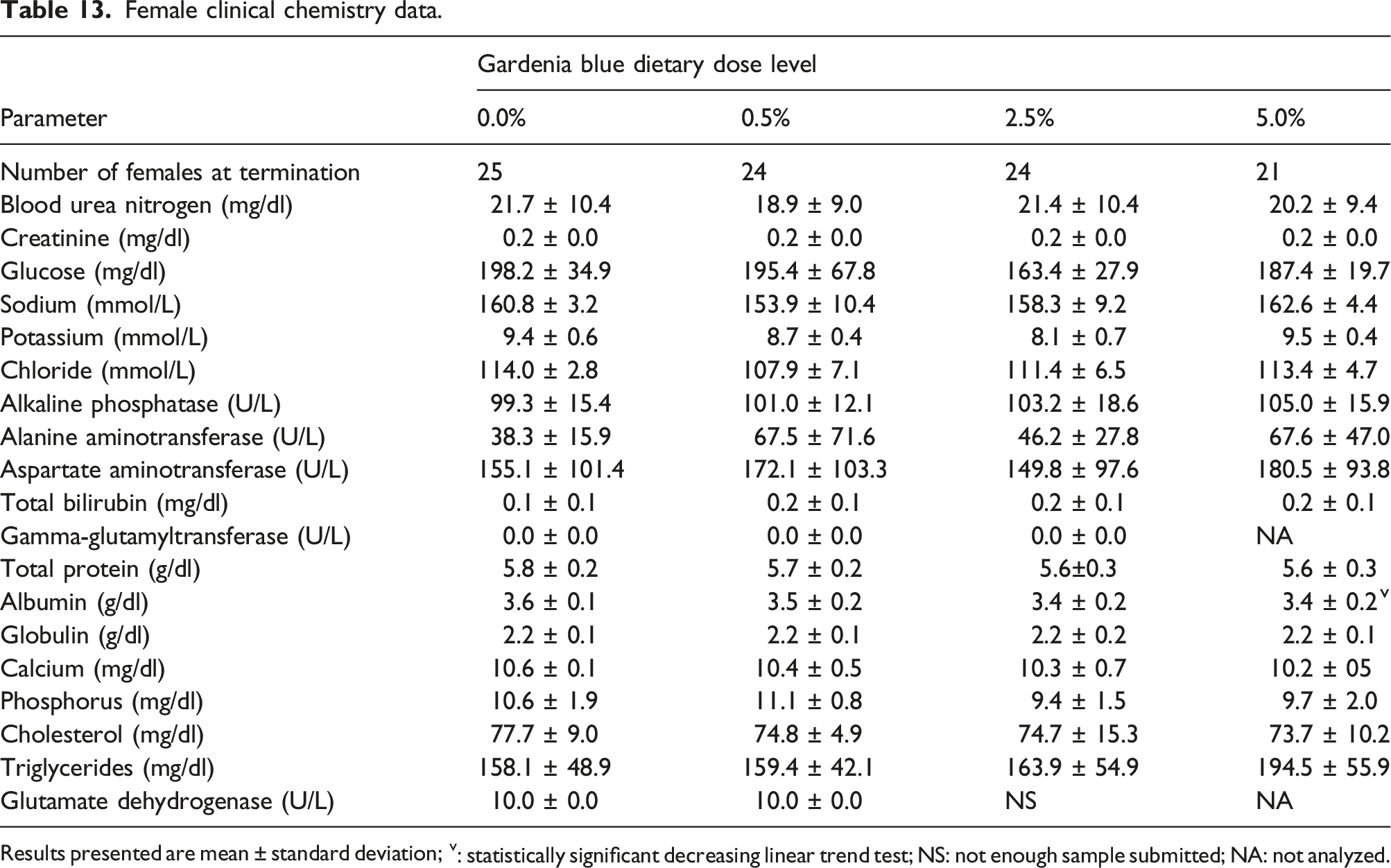
Results presented are mean ± standard deviation; v: statistically significant decreasing linear trend test; NS: not enough sample submitted; NA: not analyzed.
Female urinalysis data.
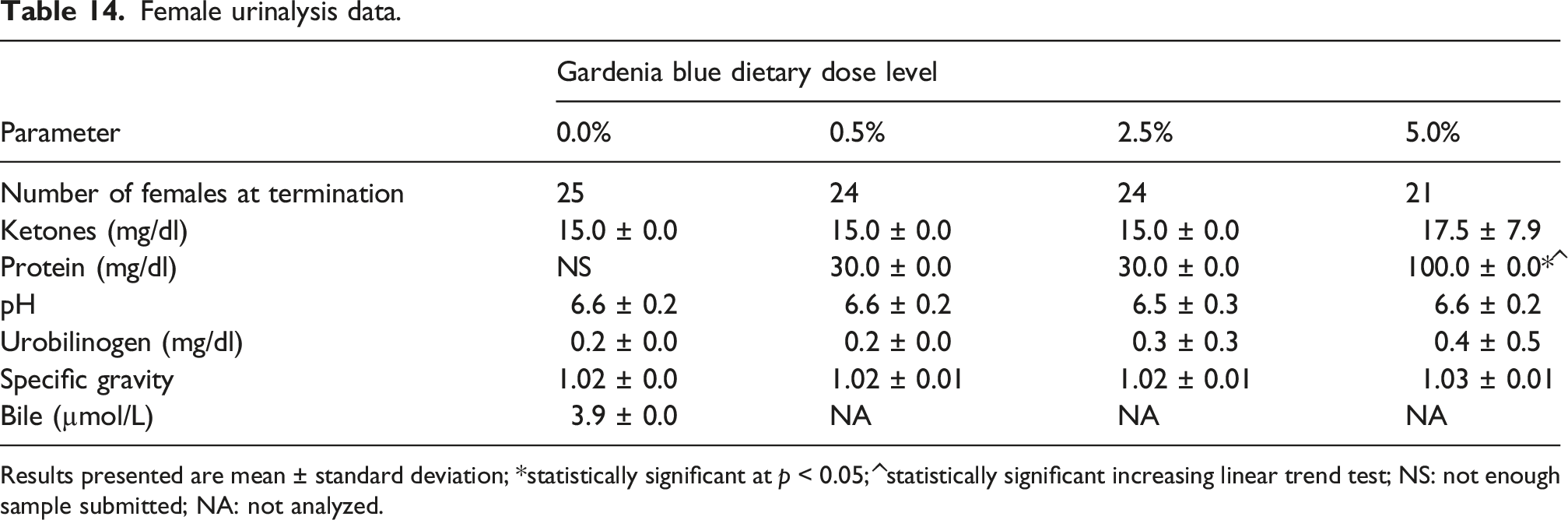
Results presented are mean ± standard deviation; *statistically significant at p < 0.05; ^statistically significant increasing linear trend test; NS: not enough sample submitted; NA: not analyzed.
Macroscopic and microscopic observations
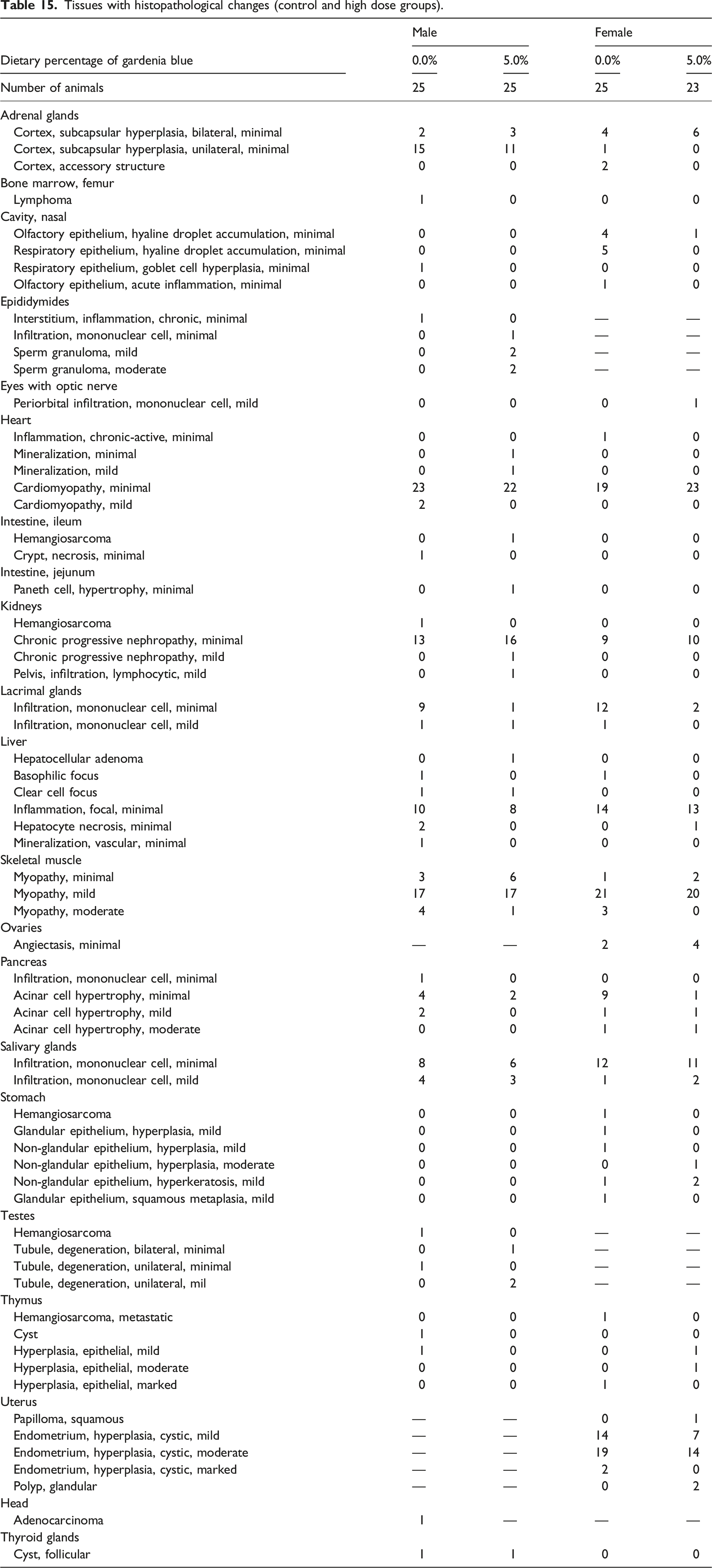
The only noteworthy macroscopic changes in gardenia blue-treated males or females versus corresponding controls were dose-related gross blue discoloration of the gastrointestinal tract and mesenteric lymph nodes and dose-related darkening of the kidneys. Additionally, sporadic incidences of mass lesions, tissue discolorations, and organ enlargements occurred across control and gardenia blue dose groups and were not considered test article-related.
Tissues with histopathological changes (control and high dose groups).
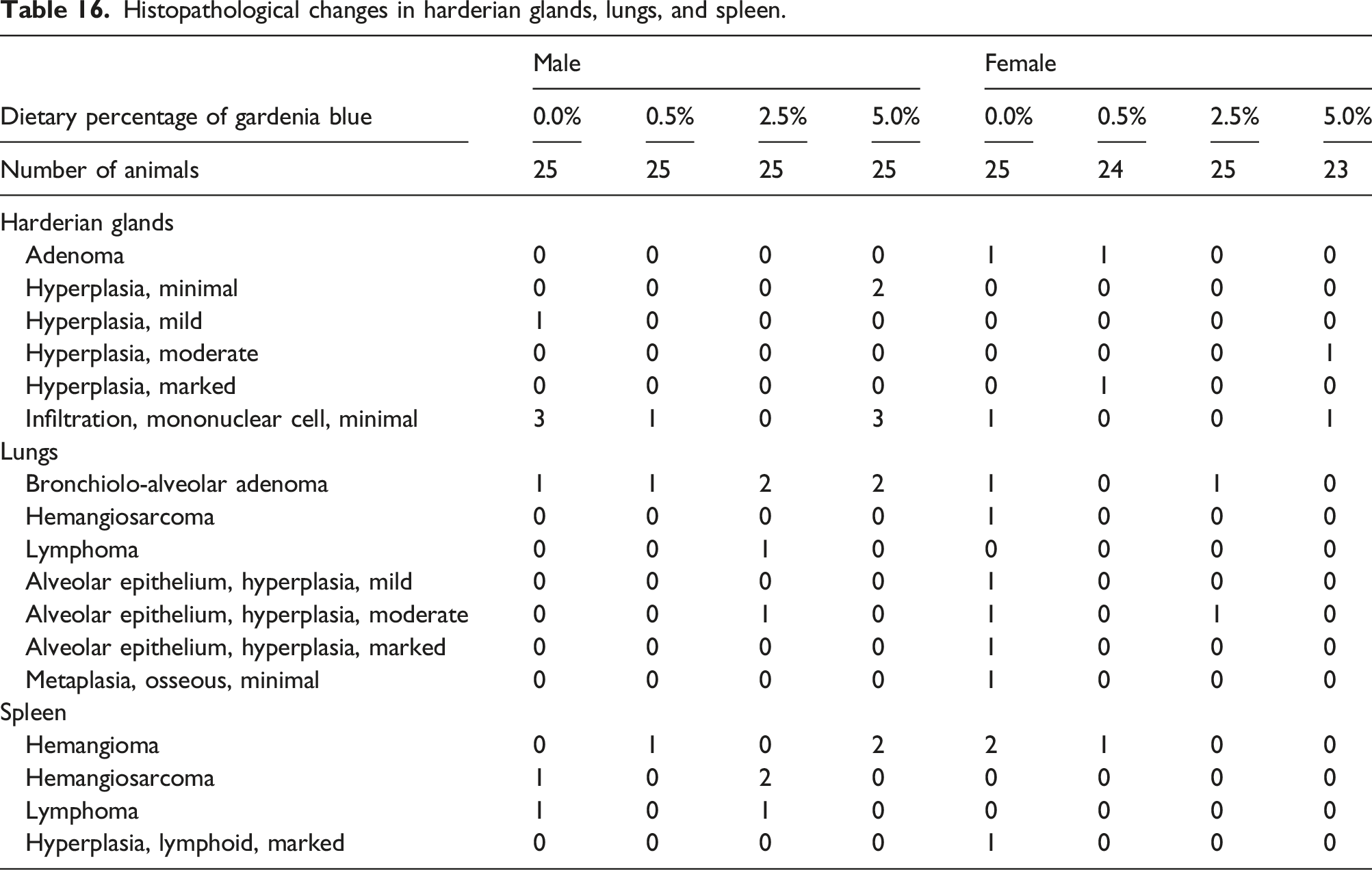
Histopathological changes in harderian glands, lungs, and spleen.
Discussion
The rasH2 model (CByB6F1-Tg (HRAS)2Jic) was originally developed to ascertain if in vivo expression of the human c-Ha-ras gene driven by its own promoter would induce tumors.37,38 Introduction of the rasH2 genetically manipulated mouse model for short-term carcinogenicity testing39,40 was quickly followed with interlaboratory comparative studies41–43 and its subsequent regulatory approval in the EU, Japan, and the U.S. as an acceptable bioassay alternative to the 2-years mouse carcinogenicity model.43–45 The rasH2 model has been well characterized with respect to spontaneous neoplastic and non-neoplastic lesions.46–48 Its originally defined bioassay study structure included a positive control, a high dose determined in a 30-days range-finding study, and two lower doses, resulting in general adoption of this model for genotoxic and nongenotoxic agents, notwithstanding some concerns and limitations of the model. 45 The rasH2 bioassay of gardenia blue reported here was carried out with a study design including a positive control, a maximum tolerated dose, and two lower doses.
In the present 26-weeks carcinogenicity study in transgenic rasH2 mice, no microscopic finding was noted that could be attributed to the dietary administration of gardenia blue. The range of neoplastic and non-neoplastic lesions reported in this study are well-documented and are all considered spontaneous/background findings, either specific to the transgenic rasH2 mice34,36–38,46,47,49,50 or common to inbred strains of mice.51,52 Additionally, incidences of neoplastic lesions observed in the control group in this study were consistent with historical control data published in the literature.34,39–41,53 There was no microscopic correlate to the treatment-related gross blue discoloration of internal organs (i.e. gastrointestinal tract and mesenteric lymph nodes). Additionally, there were no correlating histopathological changes to explain the statistically significant clinical pathology findings or the relative and absolute organ weight changes in both sexes. Consistent with recently reported gardenia blue toxicity studies,14–16 the present study with up to 5% dietary exposure of rasH2 mice to gardenia blue produced macroscopic tissue color changes that were not correlated with microscopic tissue changes and did not result in treatment-induced neoplastic or non-neoplastic lesions.
Footnotes
Author contributions
R Maronpot wrote the study protocol, the first manuscript draft, and edited the final submission. M Koyanagi, S Hayashi, M Nishino and M Iniwa reviewed and approved the study protocol and edited all draft versions of the manuscript. D Mahapatra conducted the gross and microscopic pathology and reviewed and edited the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R Maronpot is a paid consultant to San-Ei Gen (SEG) and monitored all aspects of the study and drafted the manuscript. M Koyanagi, S Hayashi, M Nishino and M Iniwa are employees of SEG, provided the funding to Inotiv for the study conduct, and reviewed and edited the manuscript. D Mahapatra is an employee of the contract laboratory where the study was done and served as the study pathologist who supervised the necropsy and diagnosed all tissue changes and reviewed and edited the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ministry of Agriculture, Forestry and Fisheries of Japan under the project for promotion of gardenia blue to an export environment.
Ethical approval
Ethical approval to conduct the animal studies for this project was obtained by the Inotiv Animal Welfare Ethics Committee prior to commencement of the study.
Informed consent
Patient data is not part of the current submission. Therefore, informed consent is not applicable.
Data availability statement
The datasets generated and/or analysed during the current study are available from Inotiv, Inc., Keystone Park Drive, Suite 200, Morrisville, North Carolina 27560 USA.
