Abstract
We previously reported that a neonatal administration of diethylstilbestrol or 17β-estradiol affected the mammary carcinogenesis induced by 7,12-dimethylbenz[a]anthracene (DMBA) in rats. The aim of this present study was to investigate the effects of 4-n-octylphenol (OP), a weak estrogenic disruptor, on the induction of mammary carcinomas (MC) and benign proliferative lesions (PL) induced by DMBA in rats. All female rats were administered at 0, 0.1, 10, 100, and 1,000 μg OP once at birth, given 10 mg DMBA at 50 days after birth, and thereafter underwent necropsy at 351 days after birth. All male rats were given 10 mg DMBA at 28, 42, and 56 days after birth, and 0, 10, 100, and 1,000 ppm OP fed from day 70-153; they underwent necropsy at 153 days after birth. Neonatal single administration of OP in female rats showed no effects such as persistent estrus, anovulatory ovaries, or PL. A slight increase in numbers of rats with MC occurred at the highest dosage. Feeding a large dose of OP for a long period in male rats induced atrophy of testes and slightly increased numbers of affected MC but not increased numbers of males while it showed no effects on PL. These results suggested that administration of a large dose of OP for a long period may have had a minimal effect on mammary carcinogenesis in male rats.
In humans, the incidence of breast cancer has been increasing worldwide, 18, 25 and it is essential for its prevention to accurately determine the cause. In general, the risk of developing breast cancer is mainly related to daily diet, lifestyle, environment, and genetics. The relationship of breast cancer risk to environmental agents, such as chemical exposure, has been widely studied. 1, 4, 16 Exposure to certain chemicals and hormone-mimicking or endocrine disruptors are suspected to contribute to the increasing incidence of breast cancer, as well as precocious puberty in the United States. 6 We previously reported that a single administration of diethylstilbestrol (DES), 19 a synthetic estrogen with strong estrogenic activity and a known endocrine disruptor, and 17β-estradiol (E2), 7 a natural estrogen, during the neonatal period affected the induction of mammary carcinomas (MC) induced by 7,12-dimethylbenz[a]anthracene (DMBA) in rats.
We have reported that rat mammary dysplasia induced by DMBA is morphologically similar to that in humans and is characterized by gross cysts and solid masses with microscopic features, including fibrotic adenosis and acinar adenosis. 12, 13, 28, 29, 31 Mammary dysplasia, fibrotic adenosis, and acinar adenosis are relevant to benign proliferative lesion (PL), fibroadenoma, and lobular hyperplasia in Fisher rats. 3, 14
Recently, we reported that the continuous administration of DES during exposed neonatal periods could affect the incidence and progression of MC and PL in rats. 14, 31 However, it remains to be determined whether the administration of weak estrogenic endocrine disruptors, such as 4-n-octylphenol (OP), which exists in daily life and has weak estrogenicity, 2, 15, 24 affects the mammary tumorigenesis.
Octylphenols, compounds derived from nonylphenol, are estrogenic endocrine-disrupting agents. They have been detected in the soils of rivers and may cause adverse effects in certain fish and shellfish species, being passed along the biological food chain. They stimulate the replication of estrogen receptor (ER)-positive human mammary carcinoma cells and also increase the uterus weight and height of the endometrial epithelium in ovariectomized rats. 8
In the present study, OP was administered to female rats once at birth, and a large dose of OP was fed to male rats to investigate its effect on the induction of mammary tumors induced by DMBA.
Materials and Methods
Animals
The animals were inbred Sprague-Dawley (SD) male and female rats, maintained in a filtered air laminar flow at the Institute of Laboratory Animal Sciences, Frontier Science Research Center, Kagoshima University, Kagoshima, Japan. The animals were given a commercial diet (CE-2, CLEA Inc., Tokyo, Japan) and tap water ad libitum. The room temperature was maintained at 25°C ± 2°C and relative humidity at 55% ± 10%, with a 12-hour light/dark cycle. The use of animals in this research complied with all relevant guidelines set by Kagoshima University.
Study designs
Experiment A (female)
All female rats were divided into 6 groups at birth. Rats in groups I (n = 23), II (n = 19), III (n = 18), IV (n = 24), V (n = 17), and VI (n = 31) were subcutaneously administered OP (Aldrich Chemical Company, Inc., Milwaukee, WI, USA) dissolved in 0.05 ml sesame oil at 0, 0.1, 1, 10, 100, and 1,000 µg/rat, respectively, once at birth. At 21 days after birth, all rats were weaned. At 50 days after birth, all groups were given 10 mg DMBA (Wako Pure Chemical Industries Ltd., Osaka, Japan) dissolved in 1 ml sesame oil by gastric intubation. All DMBA-administered animals, except those killed during the observation period, were examined once weekly by palpation to detect mammary tumors during 50 to 351 days after birth. The estrus cycles of all animals were examined during 29 to 100 days after birth by a vaginal smear test. At 351 days after birth, all surviving animals underwent necropsy (Fig. 1). Ovaries, uterus, and pituitary and adrenal glands were weighed.
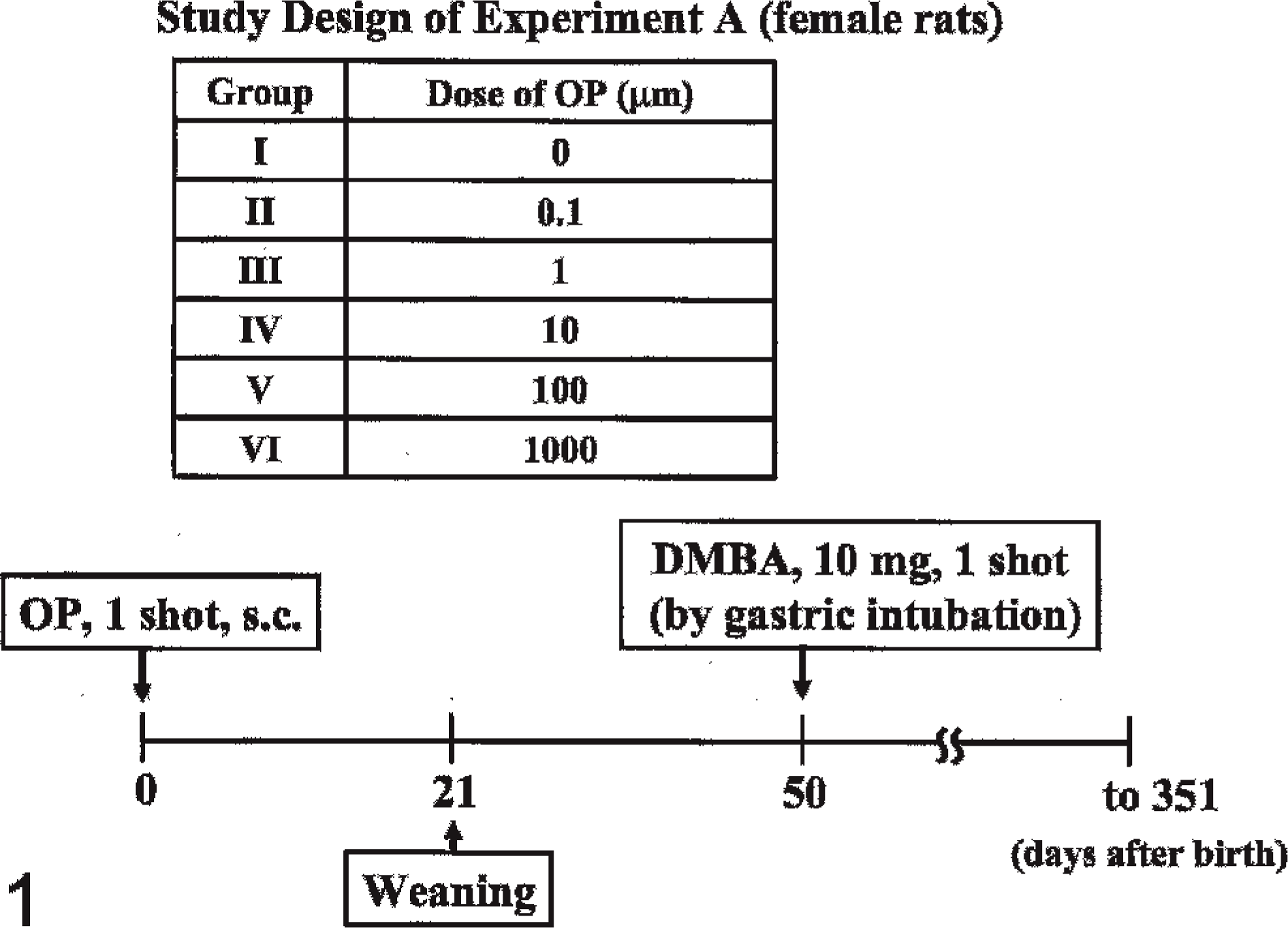
Protocol of Experiment A used in this study.
Experiment B (male)
At 21 days after birth, all rats were weaned. Ten mg DMBA dissolved in 1 ml sesame oil was administered to all male rats 3 times at 28, 42, and 56 days after birth by gastric intubation, because the induction of mammary tumors in male rats requires pulse doses of DMBA, as described previously. 22 All rats were divided into 4 groups at 70 days after birth. Rats in groups I (n = 12), II (n = 13), III (n = 13), and IV (n = 12) were fed OP mixed in diet at 0, 10, 100, and 1,000 ppm, respectively, from 70 days after birth. All DMBA-administered animals, except those killed during the observation period, were examined once weekly by palpation to detect mammary tumors during 56 to 153 days after birth. At 153 days after birth, all surviving animals underwent necropsy (Fig. 2). All palpable mammary masses were weighed, and the longest diameter and shortest diameter of the surface were measured; the size (cm2) was calculated by the longest diameter × the shortest diameter of the surface. Testes, epididymis, accessory gonad (including seminal vesicle and prostate), and pituitary and adrenal glands were weighed.
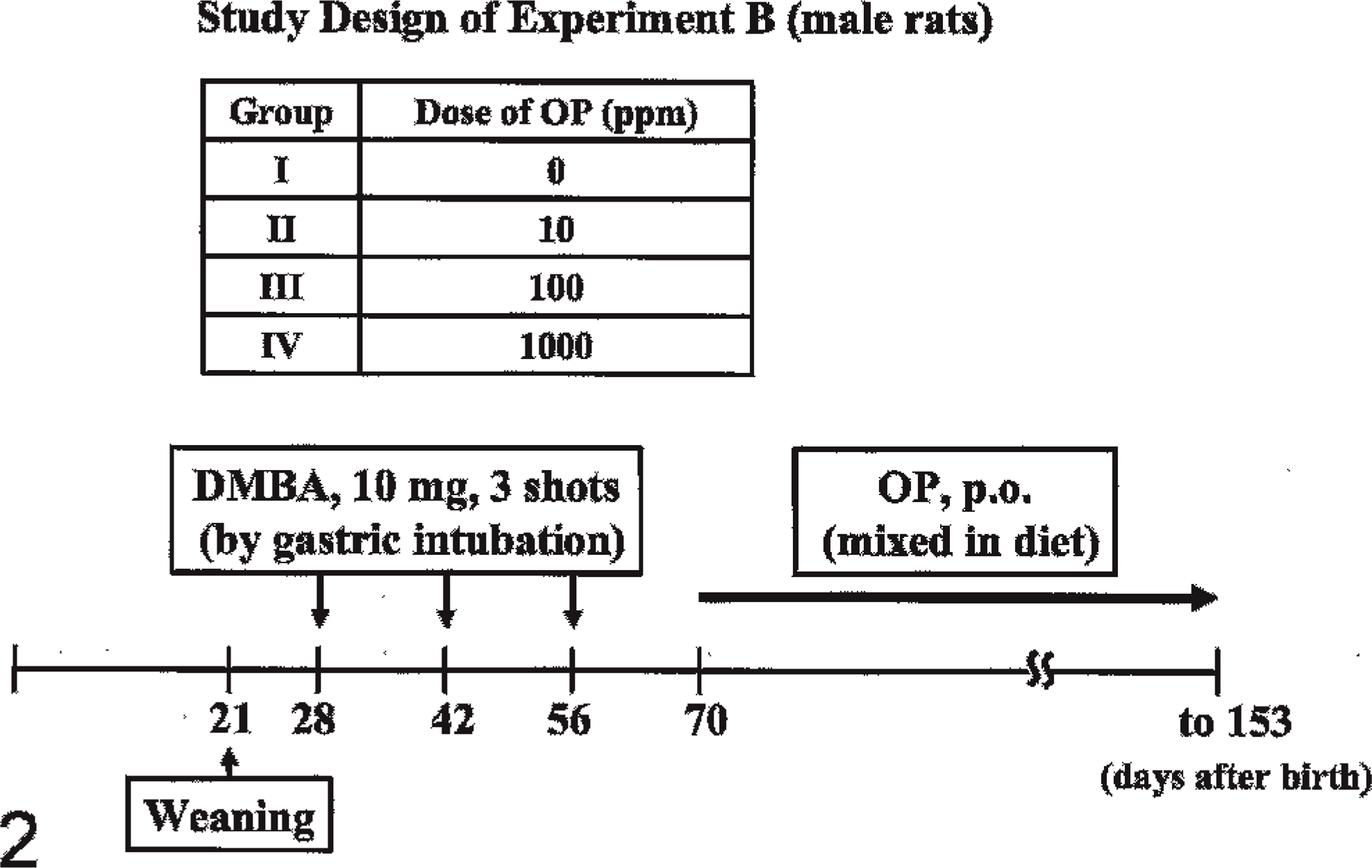
Protocol of Experiment B used in this study.
Histopathologic examination
All palpable mammary masses, ovaries, testes, epididymis, and accessory male reproductive tissues were fixed in 10% phosphate-buffered formalin, dehydrated, and embedded in paraffin. The widest cut surface of mammary tumors and ovaries, the cross-cut surface of testes, and the other organs were sectioned at 5 µm, stained routinely with hematoxylin and eosin, and then examined histopathologically.
Statistics
Mean differences were evaluated by Student's t-test. Data are shown as mean ± SD. Statistical analyses were performed using Dr. SPSS II program for Windows. The incidences (percentages) were tested using a fourfold contingency table (chi-square test). P values <.05 were considered to be statistically significant.
Results
Pathology of mammary masses induced by 7,12-dimethylbenz[a]anthracene
Single or multiple mammary masses, diagnosed as MC (Fig. 3) or PL, were seen in rats, and no metastasis was recognized. These masses were localized in all areas (L1-6 and R1-6) of the mammary glands. Mammary carcinomas were briefly subclassified into papillary type, solid type, or secretary type in this study, as described previously. 9, 20 Proliferative lesions consisted of solid masses, including fibroadenoma and lobular hyperplasia, and gross cysts in this study, as described previously. 12– 14, 28, 29, 31 Fibroadenoma, by far the most common benign neoplasm of the rat mammary gland, 3 consisted of rich hyalinized stroma and developed acini (Fig. 4). Lobular hyperplasia consisted of large lobular structures composed of acini. The lumen of the acini were dilated and filled with acidophilic secretions (Fig. 5). Gross cysts were soft, yellow, and oval and filled with milky fluid. Microscopically, gross cysts were lined by a single layer of flattened or cuboidal epithelium (Fig. 6).
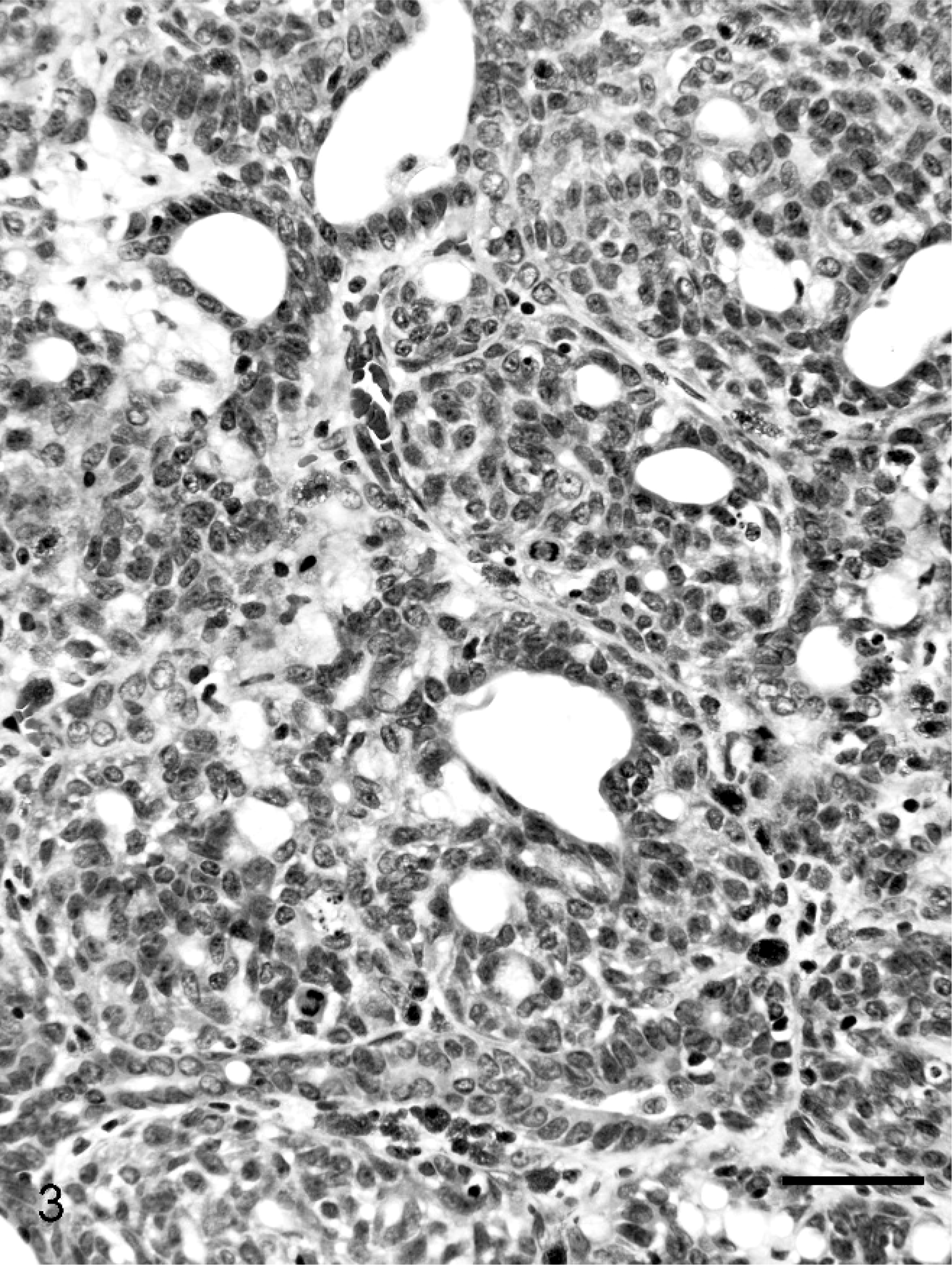
Mammary carcinoma in group I (Experiment A). Tubular proliferation, solid pattern, mitoses, apoptosis, and prominent nuclei are seen. Bar = 50 µm. HE stain.
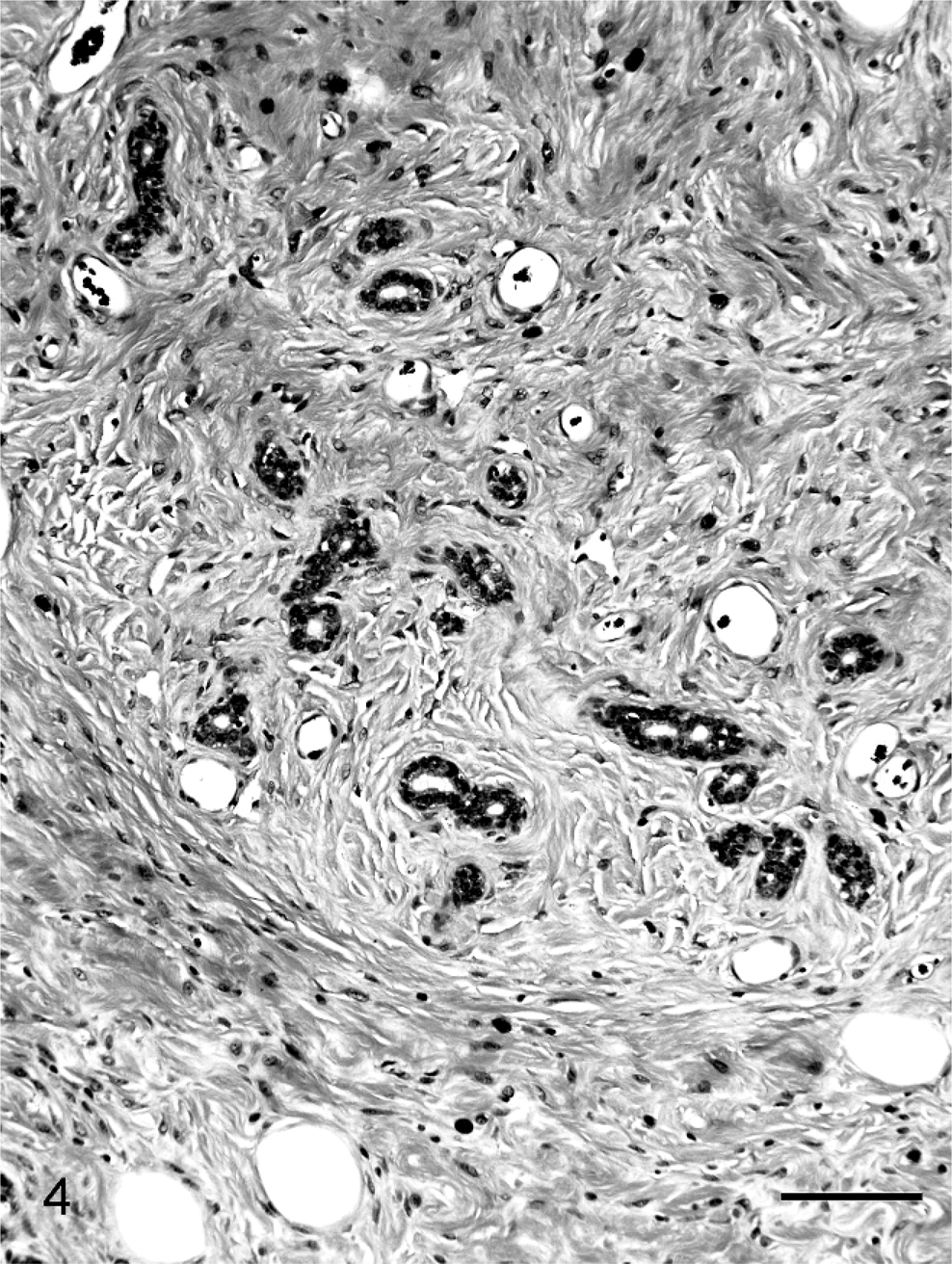
Fibroadenoma in group I (Experiment A). Fibroadenoma consists of rich hyalinized stroma and developed acini. Bar = 100 µm. HE stain.
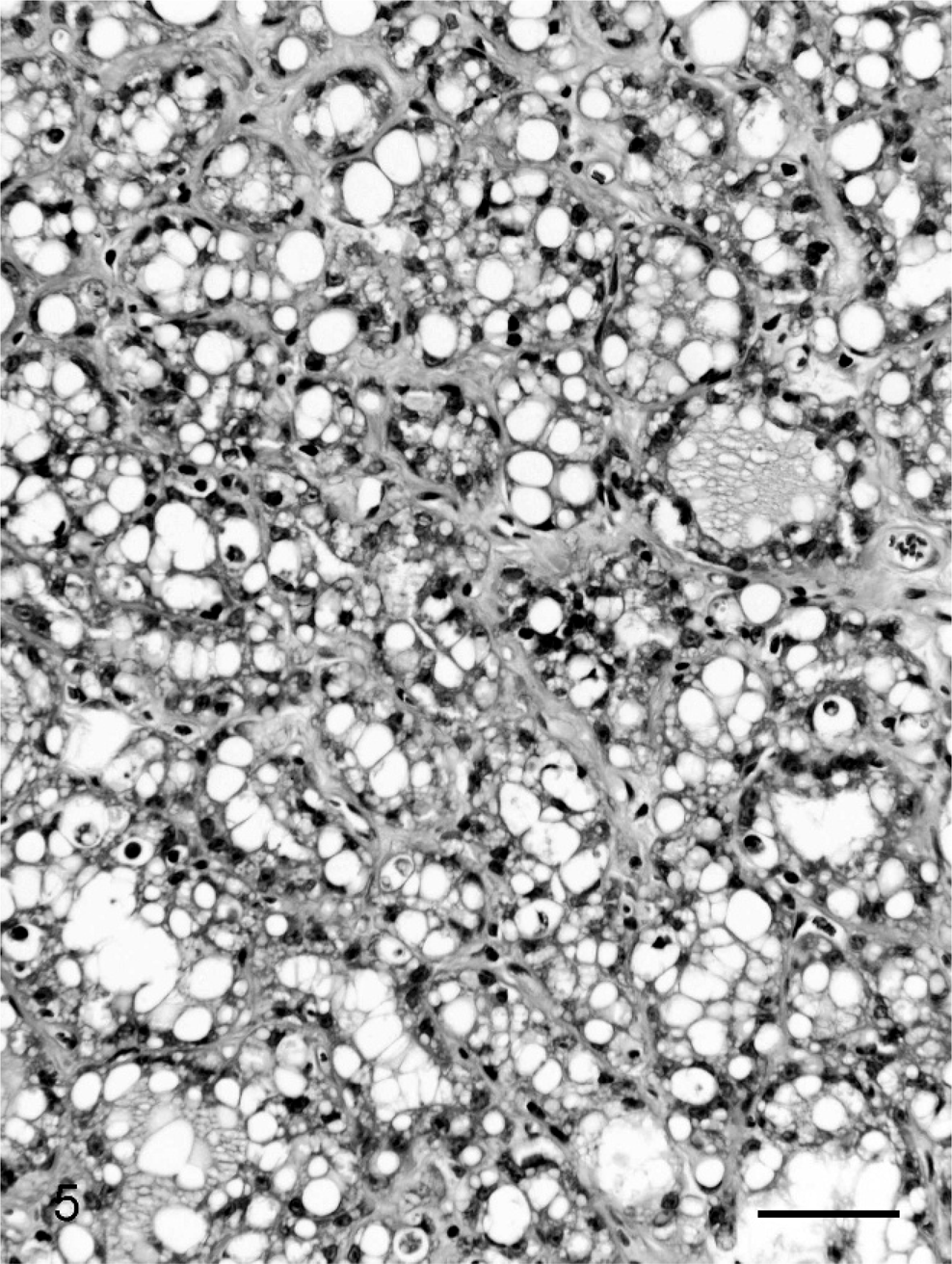
Lobular hyperplasia in group I (Experiment A). Lobular hyperplasia consists of large lobular structures composed of acini. The lumen of the acini were dilated and filled with acidophilic secretions. Bar = 50 µm. HE stain.
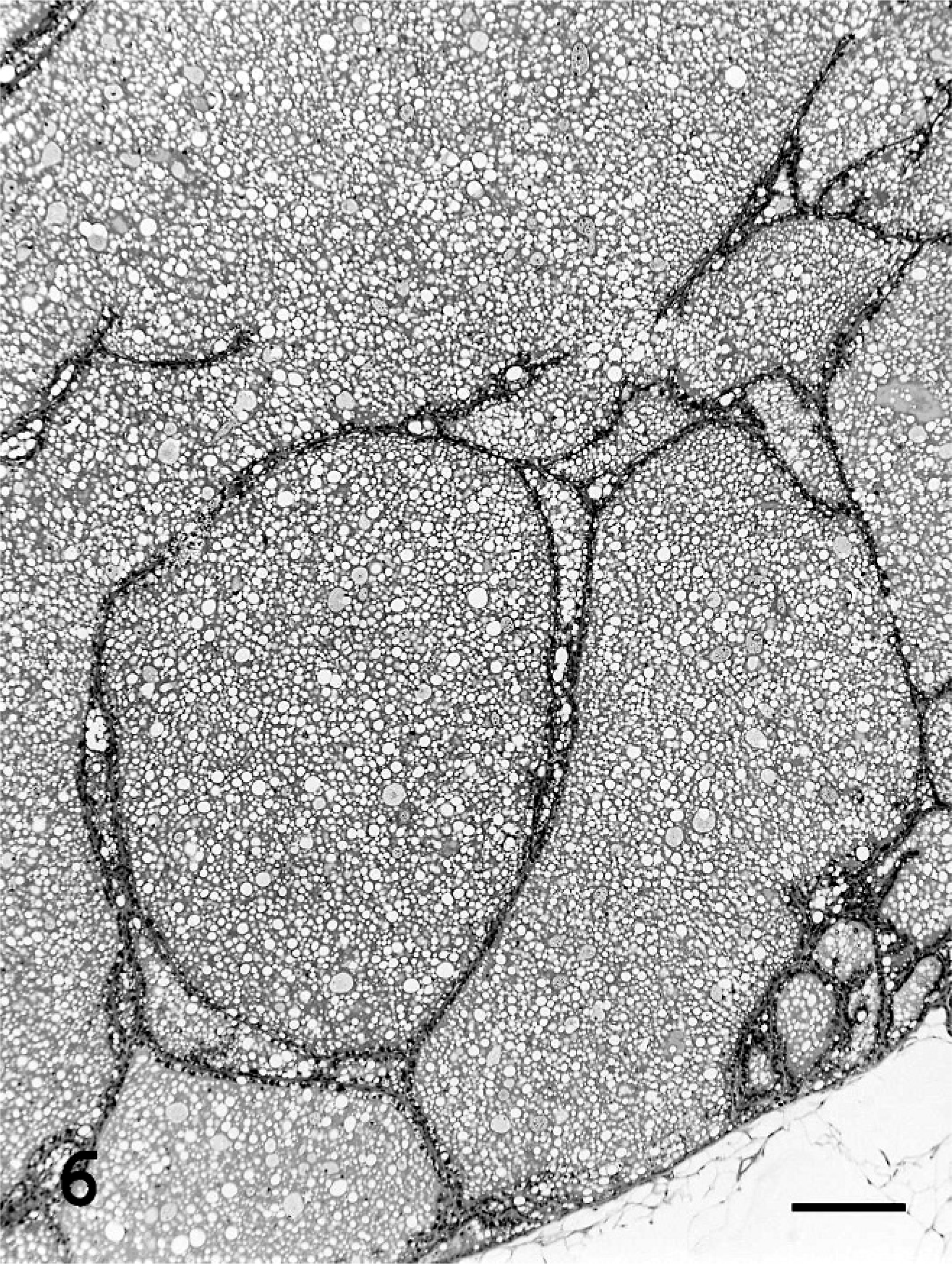
Gross cysts in group I (Experiment A). Gross cysts are filled with milky fluid and lined with a single layer of flattened or cuboidal epithelium. Bar = 200 µm. HE stain.
Experiment A (female)
In all groups administered OP, there were no significant changes in the day of the opening of the vagina and incidence of persistent estrus (PE). Corpus lutea (CL) were also seen grossly and microscopically in the ovaries of all rats. At necropsy, there were no significant changes in the body weight (BW) or ovaries, uterus, pituitary and adrenal gland weights in any group administered OP. There were no significant changes in the MC incidence in any group administered OP, whereas the number of MC per rat in group VI at 351 days after birth was slightly higher than in group I (control). In PL, there were no significant changes in the incidence and number in any group administered OP (Table 1). Proliferative lesions consisted of fibroadenomas, lobular hyperplasia, and gross cysts.
Effects of neonatal single administration of 4-n-octylphenol on the endocrine disrupter and mammary tumorigenesis in female rats (Experiment A).*
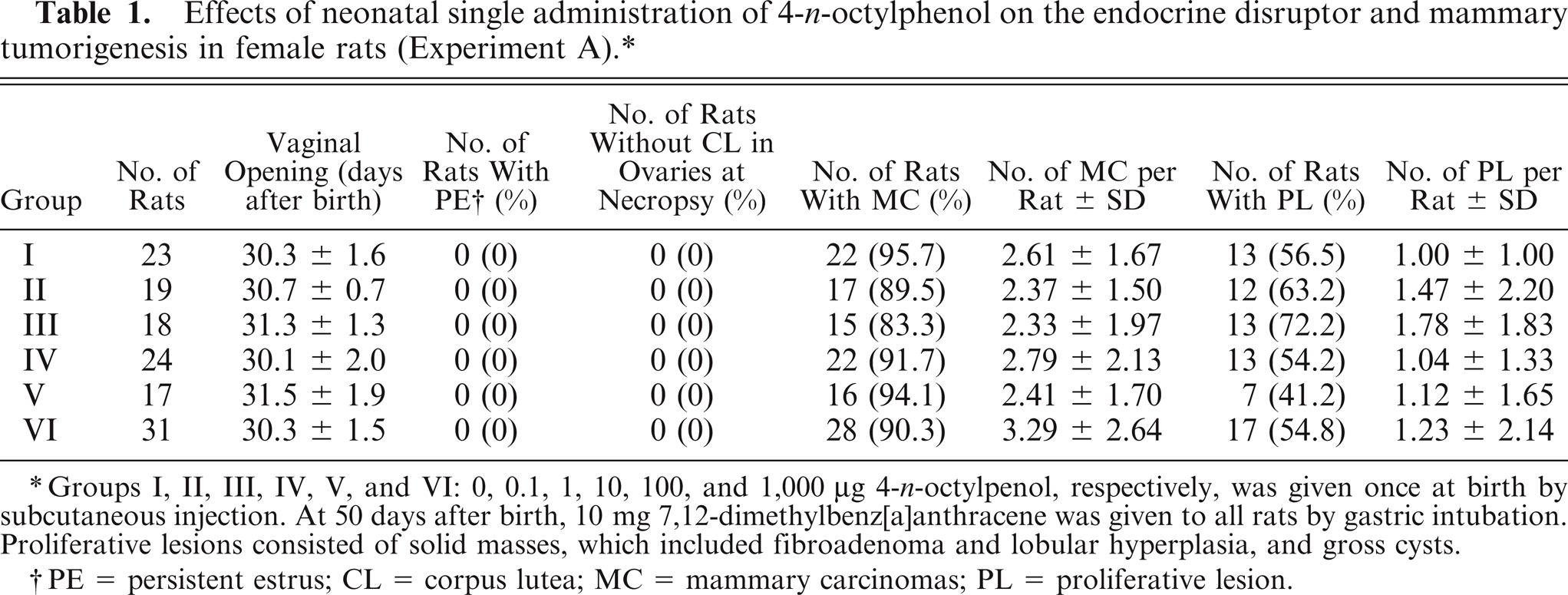
Groups I, II, III, IV, V, and VI: 0, 0.1, 1, 10, 100, and 1,000 μg 4-n-octylpenol, respectively, was given once at birth by subcutaneous injection. At 50 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to all rats by gastric intubation. Proliferative lesions consisted of solid masses, which included fibroadenoma and lobular hyperplasia, and gross cysts.
PE = persistent estrus; CL = corpus lutea; MC = mammary carcinomas; PL = proliferative lesion.
Experiment B (male)
The daily intake of food per rat in group IV during 70 to 83 (P < .01) and 84 to 97 (P < .01) days after birth and in group II during 98 to 111 (P < .05) days after birth was significantly lower than in the controls (Fig. 7).
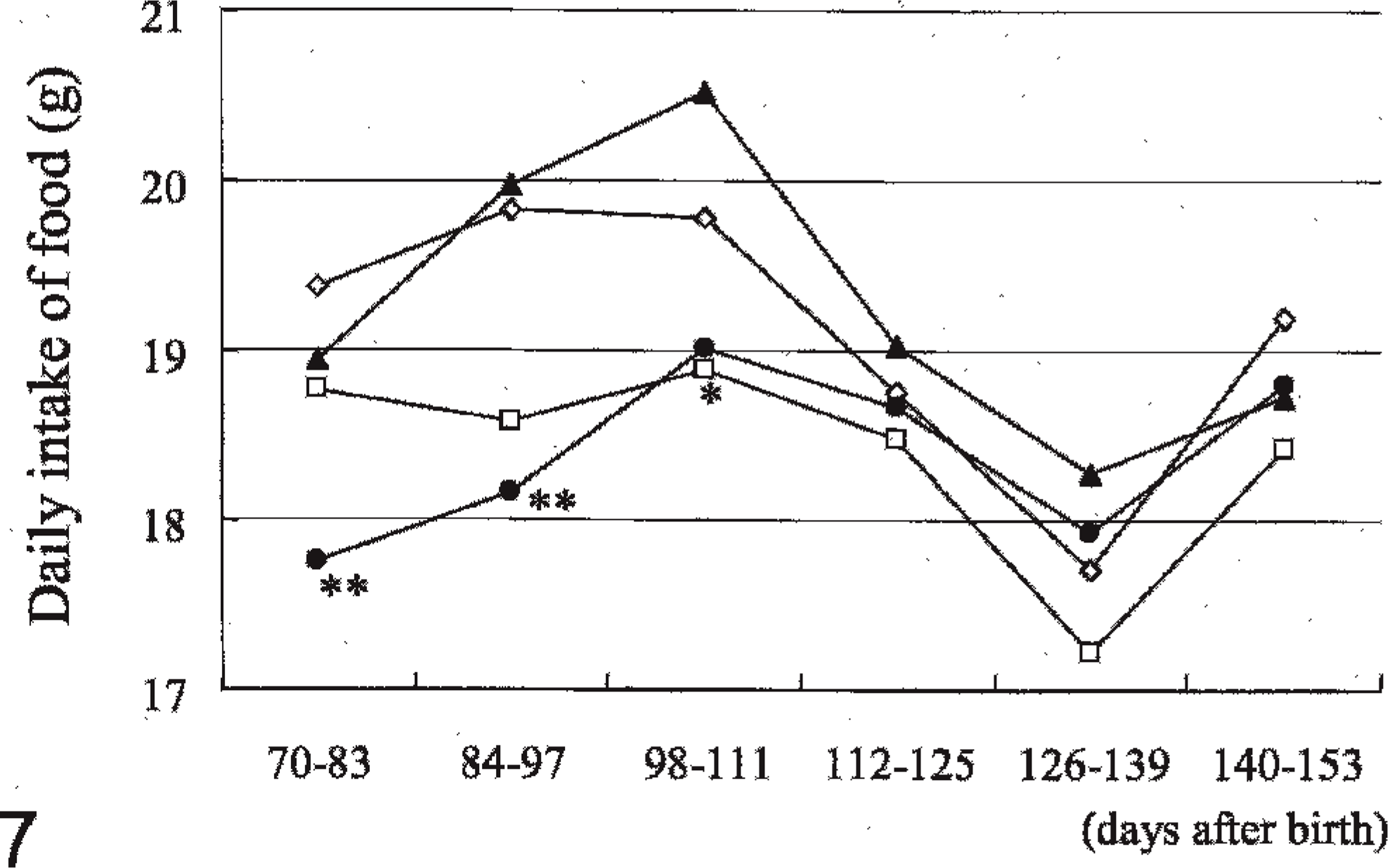
Mean diet volume (g) per rat per day (Experiment B). At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I, II, III, and IV were given 0, 10, 100, and 1,000, respectively, ppm 4-n-octylphenol from 70 days after birth. ∗P < .05, ∗∗P < .01: significantly different from Group I. —◊— Group I, —□— Group II, —▴— Group III, —•— Group IV.
At necropsy, the absolute body weight (AW) and the AW/BW of testes in group IV were significantly lower than in the controls (both P < .01), whereas there were no significant changes in the BW, AW, and AW/BW of epididymis, accessory gland (including seminal vesicles and prostate), and pituitary and adrenal glands in any group administered OP (Table 2). In testes, sperm were noted in all groups administered OP, and the incidence of rats with atrophy of seminiferous tubules, evidenced by the disappearance of spermatogonia, spermatocytes, and spermatids, in group IV (P < .05) was significantly higher than in the controls (Table 3, Figs. 8, 9). There were no histopathologic changes in the epididymis and accessory tissues.
Effects of feeding 4-n-octylpenol on the body and genital and endocrine organ weight at necropsy (Experiment B).*
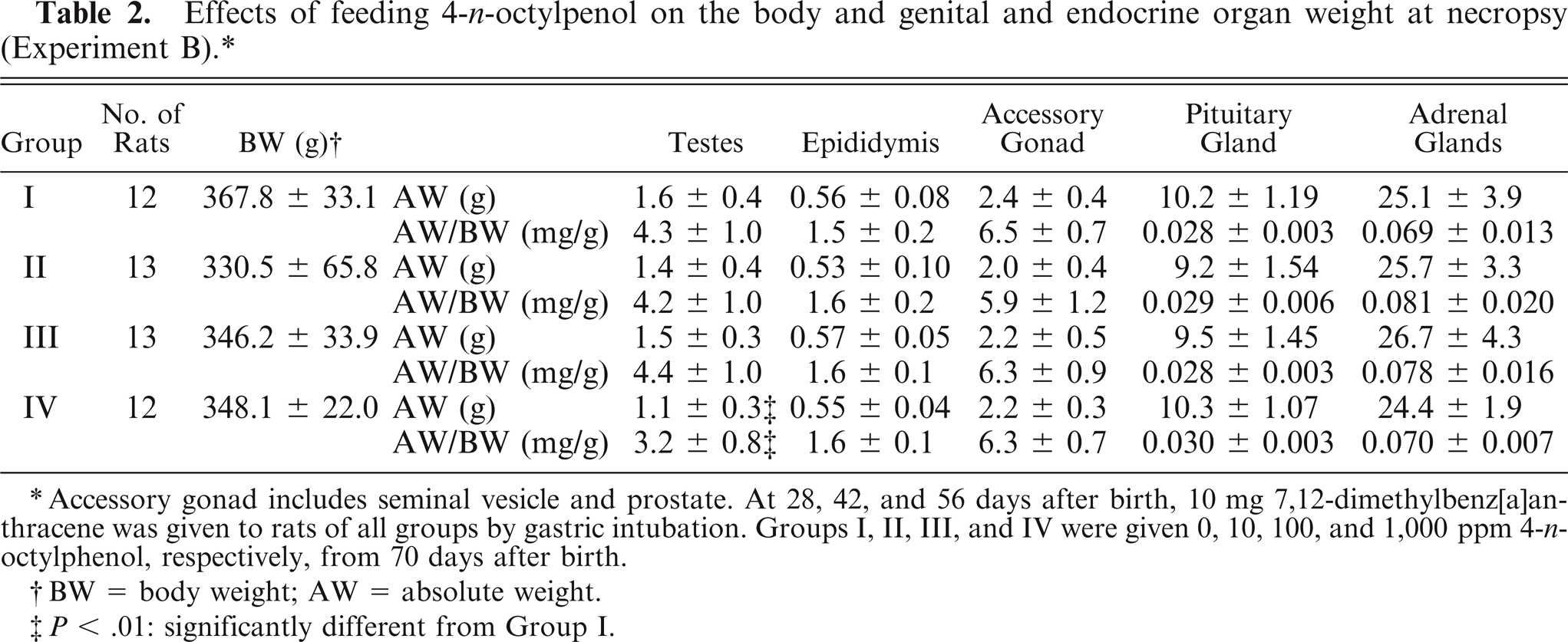
Accessory gonad includes seminal vesicle and prostate. At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]an-thracene was given to rats of all groups by gastric intubation. Groups I, II, III, and IV were given 0, 10, 100, and 1,000 ppm 4-n-octylphenol, respectively, from 70 days after birth.
BW = body weight; AW = absolute weight.
P < .01: significantly different from Group I.
Histopathologic findings in testis.*

At 28,42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I II, III, and IV were given 0, 10, 100, and 1,000 ppm 4-n-octylphenol, respectively, from 70 days after birth.
P < .05: significantly different from Group I.
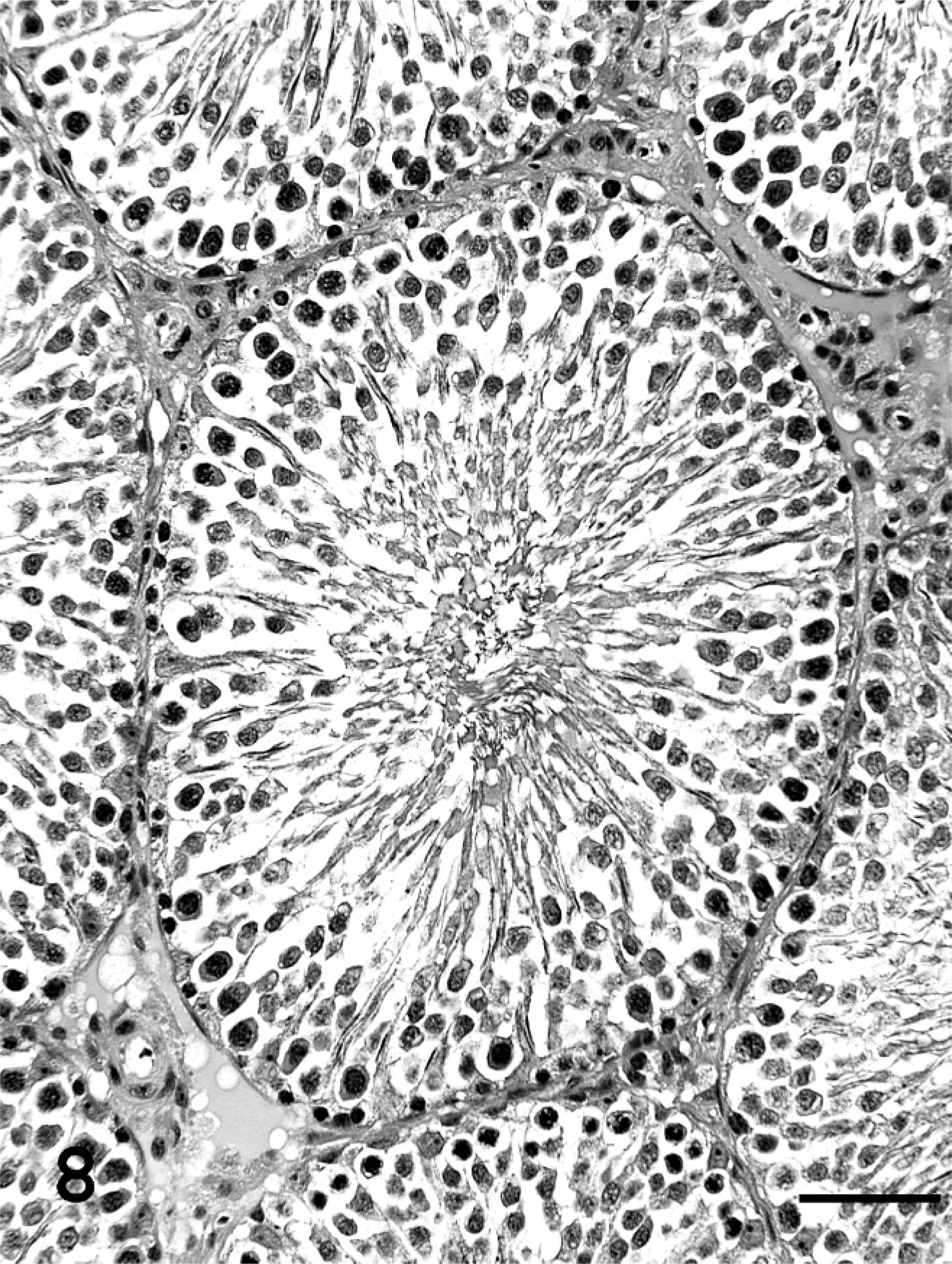
Cross-section of testis in group I (Experiment B). No abnormal changes were seen. HE stain. Bar = 50 µm.
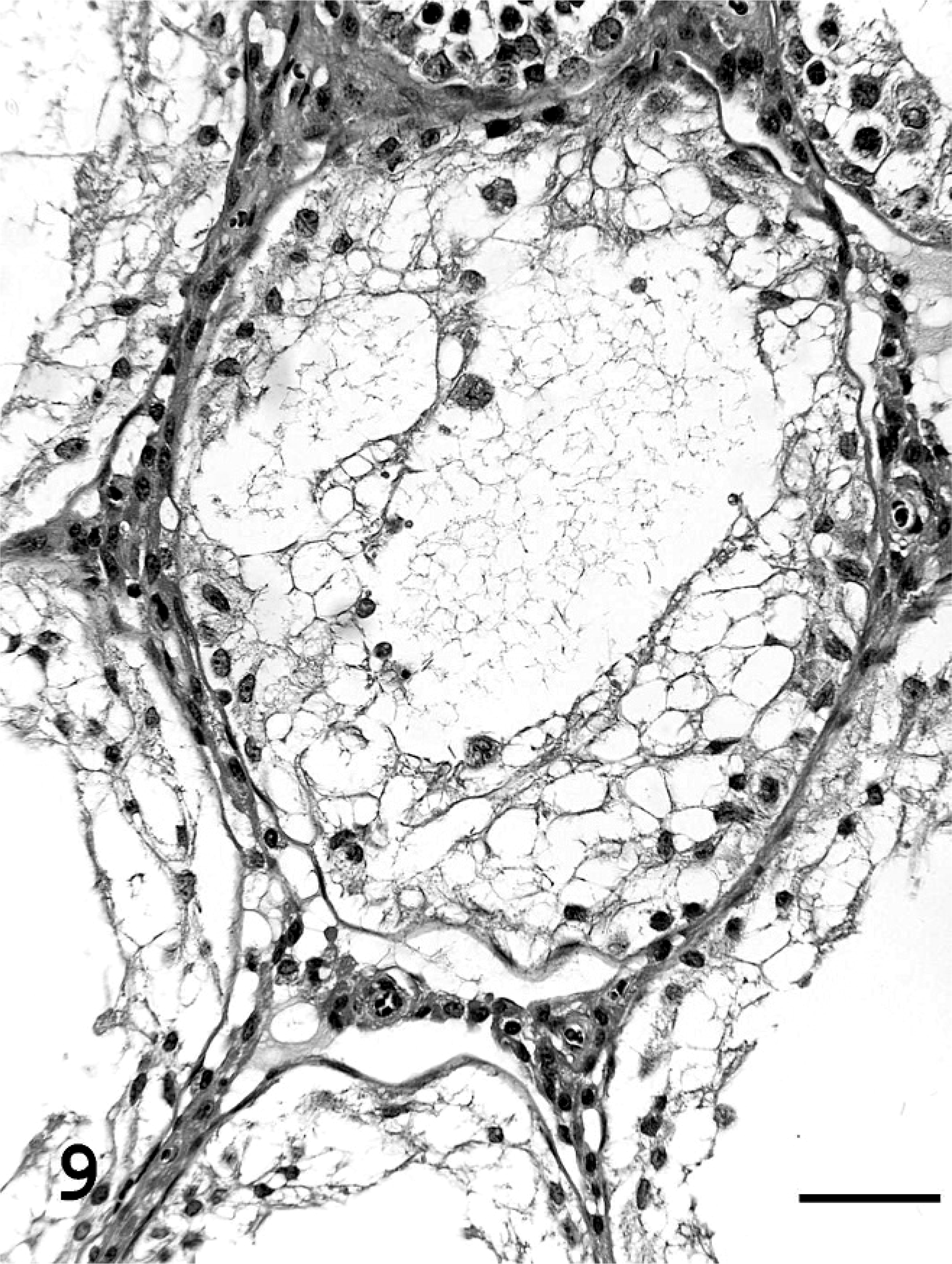
Cross-section of testis in group IV (Experiment B). Atrophy of seminiferous tubules was seen, which showed disappearing of spermatogonia, spermatocytes, and spermatids. Bar = 50 µm. HE stain.
The incidence of rats with MC in group IV at 105 (P < .01) and 112 (P < .05) days after birth was higher than in the controls (Fig. 10) but at day 351 there was not a difference in incidence across all groups. The number of MC per rat in group IV at 105, 112, 119, and 126 days after birth was slightly higher than in the controls (all P < .05) (Fig. 11). The mean day of detection (latent period) in group IV was earlier than in the controls (P < .05) (Table 4). Proliferative lesions, consisting of fibroadenoma, only occured in 2 group III males, but there were no significant differences in the incidence and number of tumors (Table 5).
Effects of feeding 4-n-octylphenol on the incidence and number of mammary carcinomas (MC) in male rats (Experiment B).*
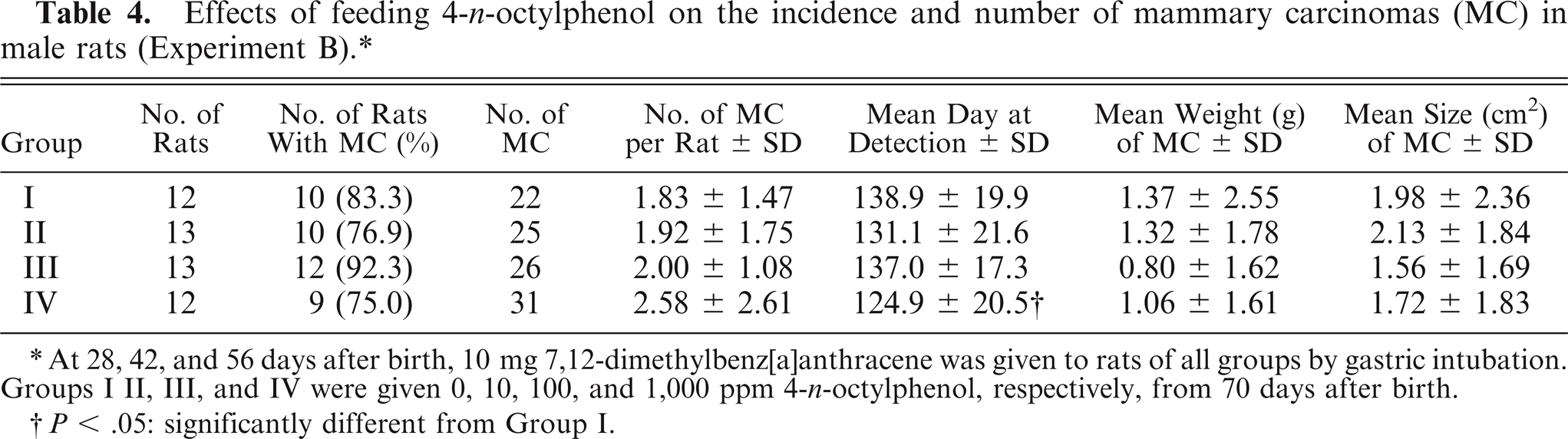
At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I II, III, and IV were given 0, 10, 100, and 1,000 ppm 4-n-octylphenol, respectively, from 70 days after birth.
P > .05: significantly different from Group I.
Effects of feeding 4-n-octylphenol on the incidence and number of fibroadenoma in male rats (Experiment B).*

At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I II, III, and IV were given 0, 10, 100, and 1,000 ppm 4-n-octylphenol, respectively, from 70 days after birth. Benign proliferative lesion consisted of only fibroadenoma.
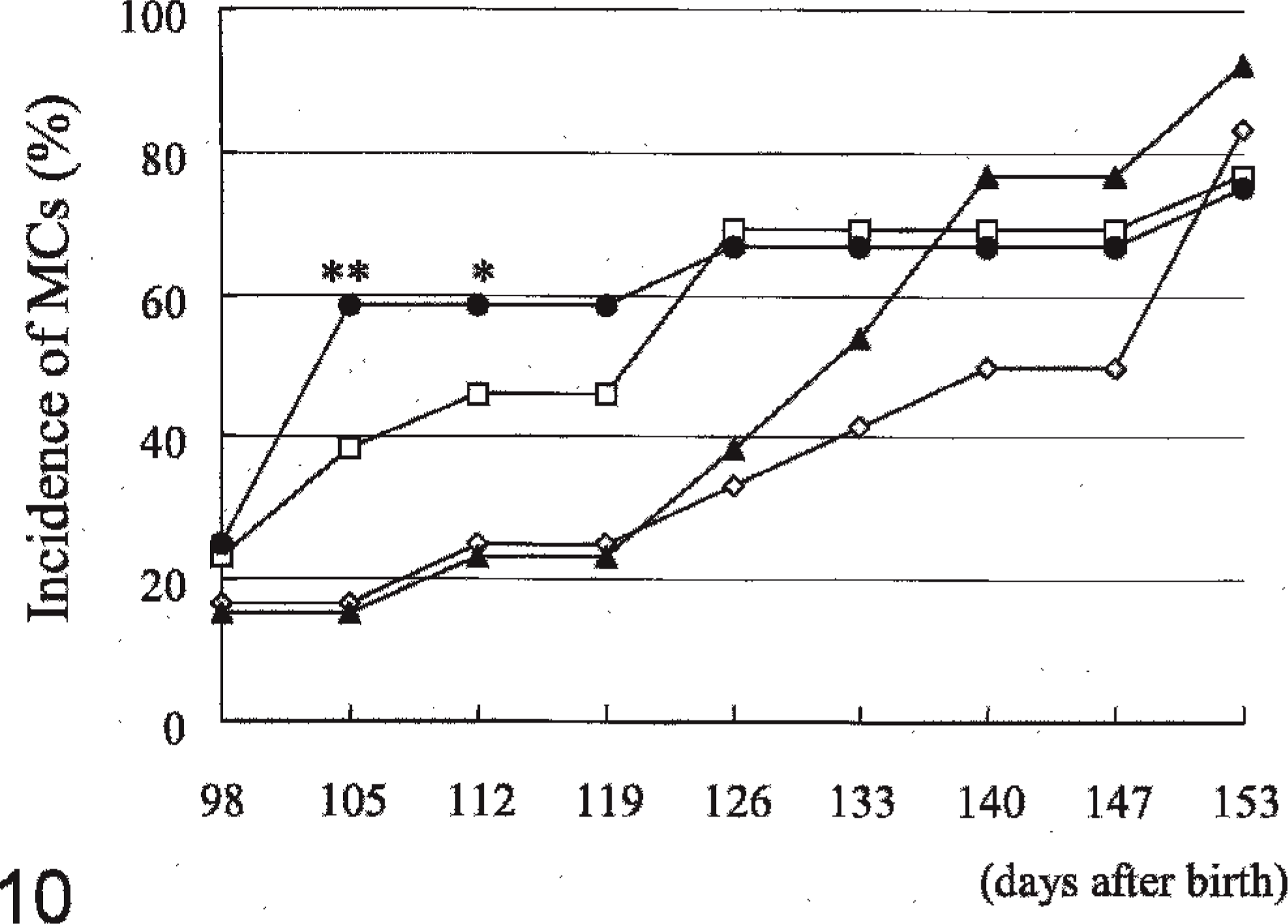
Effects of feeding 4-n-octylphenol on the incidence of rats with mammary carcinomas in Experiment B (male rats). At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I, II, III, and IV were given 0, 10, 100, and 1,000, respectively, ppm 4-n-octylphenol from 70 days after birth. ∗P < .05, ∗∗P < .01: significantly different from Group I. —◊— Group I, —□— Group II, —▴— Group III, —•— Group IV.
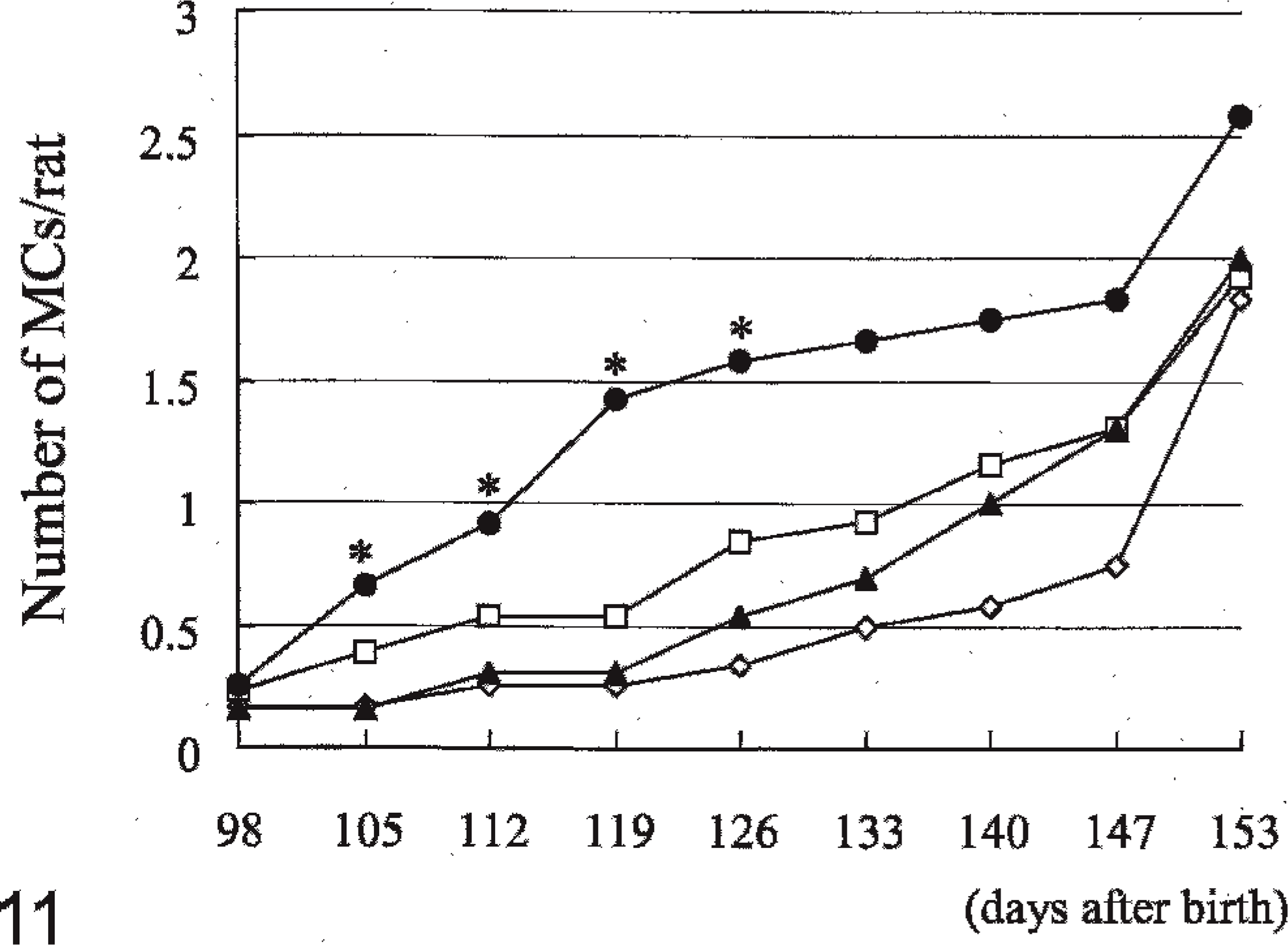
Effects of feeding 4-n-octylphenol on the number of mammary carcinomas per rats in Experiment B (male rats). At 28, 42, and 56 days after birth, 10 mg 7,12-dimethylbenz[a]anthracene was given to rats of all groups by gastric intubation. Groups I, II, III, and IV were given 0, 10, 100, and 1,000, respectively, ppm 4-n-octylphenol from 70 days after birth. ∗P < .05: significantly different from Group I. —◊— Group I, —□— Group II, —▴— Group III, —•— Group IV.
Discussion
Recently, we reported that a high dose of DES (100 µg) 19 and E2 (1,000 µg) 7 during the neonatal period induced PE and an absence of CL in female rats due to disturbance of the gonadotropin-secreting system in the hypothalamus; this resulted in the suppression of MC. Conversely, we reported that the administration of a relatively low dose of DES (1 µg) 19 and E2 (10 µg) 7 during the neonatal period increased the number of terminal end bud cells, resulting in a stimulatory effect on the initiation of MC in female rats. In the present study, a high dose of OP (1,000 µg) during the neonatal period did not induce PE and the absence of CL, and there were no statistically significant changes in the induction of MC and PL in any group administered OP, because the estrogenicity of OP was very weak. 8, 15, 24 Moreover, it was speculated that a single administration of OP at birth may not affect the induction of MC and PL in male rats because a single administration of 1,000 µg OP in male rats at birth resulted in neither decreased weights nor histopathologic changes in the testes, epididymis, or accessory gonad at 50 or 100 days after birth (unpublished data). However, it is possible that more exposure of OP may have affected the induction of MC in female rats. A high dose of OP slightly increased the number of MC in male rats in this study though the number of animals with MC was less than in DMBA controls.
In humans, carcinoma arising in the male breast is a rare occurrence, with a frequency ratio to breast cancer in the female of less than 1 : 100, but the risk factors are similar to those in women and include exposure to exogenous estrogens, increasing age, and obesity. 17, 21 Immunohistochemical studies have demonstrated a higher percentage of male breast cancers expressing ERs than occurs in women; about 80 to 93% of men have ER-positive tumors. 23 We previously reported that the induction of MC in male rats required pulse doses of DMBA and that about 50% of these MC were ER-positive tumors. 22 Moreover, we recently reported that the induction of PL in male rats required a higher dose of DMBA than that required for female rats. 9 It was hypothesized that exogenous estrogenicity of OP may directly affect the induction of mammary tumors induced by DMBA in male rats, because serum estradiol concentration of male rats is extremely low.
Although it is much more difficult to induce MC with chemical carcinogens in male rats than in female rats, it has been found that repeated pulse doses of hydrocarbon carcinogens elicit a high yield of MC in male SD rats. 10, 26, 27, 30 We previously reported that MC were induced in male rats by 3, 5, and 8 doses of 10 mg DMBA and that the incidence of MC with the 3 doses of 10 mg DMBA were relatively low. 22, 27 Therefore, a large dose of OP was administered to male rats for a long period after the 3 doses of DMBA to investigate effects on the promotion or progression of mammary tumors. In the present study, it is possible that weak estrogenicity of OP may have affected reduced food consumption because E2 reduced food consumption. 5 In turn, Huggins, Grand, and Brillantes previously reported that all female rats fed 20 mg DMBA at 50 days after birth had visible breast tumors within 110 days after birth. 11
It was reported that E2 decreased testis weight and induced seminiferous tubule degeneration due to hyperprolactemia. 5 In the present study, a high dose of OP decreased testes weight and induced atrophy of seminiferous tubules, probably due to the estrogenicity of OP. It was speculated that a high dose of OP would stimulate the promotion or progression of mammary carcinogenesis because of a shortened latent period of MC and that stimulation of the promotion or progression of MC may also be caused by the estrogenicity of OP. However, the estrogenicity of OP was much weaker than that of E2 because there were no histopathologic changes in the epididymis and accessory glands in the present study, while atrophy of epididymal tubules was reported in the epididymis when E2 was administered for 12 weeks in rats. 5 It was also hypothesized that testicular atrophy induced by a high dose of OP may have influenced MC growth but not incidence in the present study because risk factors for male breast cancer in humans include testicular atrophy after mumps orchtis, which causes decreased testicular function. 17, 21
These results suggested that the administration of a large dose of a weak estrogenic endocrine disruptor, such as OP, for a long period does not affect female mammary carcinogenesis nor male mammary tumor incidence though a weak influence on male mammary tumor growth may have occured.
Footnotes
Acknowledgements
This work was supported in part by the Kodama Memorial Fund Medical Research. We are grateful to Dr. M. Funato, Mr. T. Kodama, and Mr. M. Souda for their valuable technical assistance.
