Abstract
Tumor hypoxia has been associated with increased malignancy, likelihood of metastasis, and increased resistance to radiotherapy and chemotherapy in human medicine. Hypoxia-inducible factor-1 (HIF-1) is a key transcription factor that is induced by tumor hypoxia and regulates the pathways involved in cellular response and adaptation to the hostile tumor microenvironment. HIF-1 induces transcription of different proteins, including Ca-IX and Glut-1, which are considered endogenous markers of chronic hypoxia in solid tumors in humans. In this study, sections from 40 canine sarcomas (20 histiocytic sarcomas and 20 low-grade soft-tissue sarcomas) were immunostained for these markers. Expression of Glut-1 was scored based on percentage of positive staining cells (0 = <1%; 1 = 1%–50%; 2 = >50%) and intensity of cellular staining (1 = weak; 2 = strong); Ca-IX was scored based on percentage of positive cells (0 = <1%; 1 = 1%–30%; 2 = >30%). Intratumoral microvessel density was measured using CD31 to assess intratumoral neoangiogenesis. Histiocytic sarcomas showed statistically significant higher Glut-1 immunoreactivity and angiogenesis than did low-grade soft-tissue sarcomas. Intratumoral microvessel density in histiocytic sarcomas was positively associated with Glut-1 immunoreactivity score. These findings suggest a potential role of hypoxia in the biology of these tumors and may provide a base for investigation of the potential prognostic use of these markers in naturally occurring canine tumors.
Hypoxia is a pathological condition in which the entire body (generalized hypoxia) or a region of the body (tissue hypoxia) is not supplied with adequate oxygen. Tissue hypoxia can be due to vascular and/or metabolic problems. Neoplastic tissue is often subjected to hypoxia, as its vascularization is structurally and functionally abnormal. In addition, the oxygen-carrying capacity of the blood may be reduced in patients with cancer due to anemia. Most solid human tumors have been shown to contain regions of acute and chronic hypoxia. 7,48
Tumor hypoxia is associated with increased malignancy as it promotes genetic instability, increased likelihood of metastasis, and resistance to radiotherapy and chemotherapy. 20 Hypoxia is considered a poor prognostic indicator in a wide range of human cancers (eg, breast, cervix, and bladder cancer and soft-tissue sarcomas [STS]), and expression of hypoxia-inducible proteins is associated with aggressive behavior and treatment resistance. 3,8,24,26
Malignant behavior and treatment resistance in hypoxic tumors are mainly mediated by hypoxia-inducible transcription factors (HIFs). Under low oxygen tension in hypoxic cells, HIFs are stabilized and activated to regulate the pathways involved in cellular adaptation to hypoxia. 31
These include genes that promote glucose uptake and facilitate glycolysis (glucose transporter-1), influence pH (carbonic anhydrase-IX), stimulate angiogenesis (vascular endothelial growth factor [VEGF], matrix metalloproteinases, erythropoietin and its receptors, angiopoietin 1 and 2), and regulate apoptosis (Bcl-xL). 10,28,31,35
As part of the HIF pathway, the proteins glucose transporter 1 (Glut-1) and carbonic anhydrase-IX (Ca-IX) have been identified as endogenous markers of chronic hypoxia in solid tumors and have been shown to have a prognostic value in several human cancers. 44
Glut-1 is a membrane-bound glycoprotein that regulates basal glucose uptake. It is ubiquitously expressed in normal tissue, and it is expressed at higher levels in a number of tumors. Hypoxia induces a reduction of mitochondrial adenosine triphosphate (ATP) production and increases cellular glycolytic ATP production (Pasteur effect), leading to an increase in glucose transport and consumption (Warburg effect). The latter has been considered beneficial for the survival of tumor cells, conferring a growth advantage in a relatively hypoxic tumor environment. 45 Glut-1 expression has been inversely correlated with metastasis-free survival in women with advanced carcinoma of the cervix, 3 and it has been proven to be a negative prognostic factor in human STS. 16,43
Ca-IX is a transmembrane glycoprotein. It is a zinc metalloenzyme that catalyzes the reversible hydration of carbonic dioxide to carbonic acid, thus playing a role in pH regulation. Ca-IX expression is a predictor of poorer prognosis in human STS. 32,36 Similarly, Ca-IX has been associated with poor prognosis and poor response to chemotherapy and radiotherapy treatment in cervical carcinomas, 22 as well as poor prognosis of carcinomas of the head and neck, 38 lung (non–small cell), 27 nasopharynx, 23 and breast (invasive). 46
HIF-induced proteins are also key to tumor angiogenesis, the formation of new blood vessels necessary to sustain tumor growth. A number of key proangiogenic factors are HIF inducible, including VEGF, erythropoietin (Epo), platelet-derived growth factor, and matrix metalloproteinases. The expression of angiogenic factors (eg, VEGF and Epo-EpoR) 1,13,19,29 and their relationship to intratumoral neovascularization through intratumoral microvessel density (IMD) measurement have also been investigated in human cancer. 15,21,50
Despite the considerable human literature on tumor hypoxia, the expression of HIF-inducible proteins Glut-1 and Ca-IX in animal tumors and their potential prognostic and therapeutic significance have only been marginally examined. 34,39,40 However, evidence of a strong effect of tumor hypoxia on tumor metabolism has been demonstrated in canine mammary carcinomas, 34 and coexpression of Epo and EpoR has been correlated with hypoxia in a variety of spontaneous canine tumors, suggesting that naturally occurring companion animal tumors are potential models for investigation of tumor hypoxia. 41
The aim of this study was to investigate and quantify the expression of Glut-1 and Ca-IX in histiocytic sarcomas (from Flat-coat Retriever dogs), which are well known to have aggressive behavior and to carry a poor prognosis, 12 and low-grade canine STS (from various breeds), which are usually locally aggressive but rarely metastatic. 14 In addition, we compared IMD values between low-grade STS and histiocytic sarcomas and assessed the correlation of these with Glut-1 and Ca-IX expression.
Materials and Methods
Tissue Samples
A total of 40 formalin-fixed, paraffin-embedded canine tumor tissues were selected from the archive of the Cambridge University Veterinary School (CUVS) pathology service. The selection included 20 histiocytic sarcomas (11 splenic and 9 localized subcutaneous) and 20 low-grade STS (Trojani et al 47 grading system). All the tumor samples selected were reviewed by 2 of the authors (E.A. and F.C.-C.) to confirm the diagnosis. The histiocytic sarcomas were all from Flat-coat Retrievers, whereas the low-grade STS were from dogs of various breeds.
Immunohistochemistry
Serial sections of 3 μm were cut from the appropriate blocks and mounted on positively charged slides (Snowcoat; Surgipath Europe Ltd., Peterborough, UK). Sections were dried overnight at 50°C in an incubator before processing using the Dako PT link module (Dakocytomation, Carpinteria, CA). This allows for deparaffinization, rehydration, and antigen retrieval in a combined 3-in-1 procedure. Sections were then immersed in a preheated working solution (pH 9.0). Once the temperature had reached 97°C, the sections were bathed for 20 minutes, left to cool to 65°C, and immediately rinsed in buffer (Dako Envision wash buffer) at room temperature. An automated immunohistochemistry system (Dako Autostainer) was used to process the prepared tissues. Endogenous peroxidase activity was inhibited using Dako Envision Flex, peroxidase-blocking solution. The immunohistochemical antibodies used were Glut-1 (rabbit anti–human polyclonal 1:250; Sigma-Aldrich, Dorset, UK), Ca-IX (rabbit anti–human polyclonal, 1:2000; Abcam, Cambridge, UK), and CD31 (mouse anti–human monoclonal, 1:50; Dakocytomation). All these antibodies had been reported previously for canine tissue. 34,39,40 Peroxidase activity was demonstrated using diaminobenzidine solution for 10 minutes, and slides were counterstained with Envision Flex hematoxylin (Dakocytomation) for 5 minutes before rinsing, dehydrating, clearing, and covering. Canine renal carcinoma tissue was used as a positive control tissue for Ca-IX and canine spinal cord for Glut-1, although Glut-1 expression in red blood cells provided an internal control. Rabbit Ig (Dakocytomation) diluted to the same concentration as the primary polyclonal antibodies was used as a negative control. Antibody diluent was used as the negative control for CD31. For all antibodies, immunopositivity was present in the positive control sections, but it was not detected in negative controls.
Immunoreactivity Scoring Systems
To score the immunoreactivity in each stained section, we randomly selected microscopic fields, excluding areas of necrosis and fields containing less than 60% of neoplastic cells. To achieve random selection, we scanned the slides from the top left corner to the bottom right.
The scoring system used for assessment of Glut-1 immunoreactivity has been previously described. 39 In short, the percentage of positively stained tumor cells was assessed as the average of 5 to 10 randomly selected high-powered fields (400×) as follows: 0 = less than 1% of positive tumor cells, 1 = 1% to 50% positive tumor cells, and 2 = more than 50% positive tumor cells. The intensity of cellular staining was scored as follows: 1 = weak positive staining and 2 = strong positive staining. A product of the 2 scores was used to calculate the final immunoreactivity score. When the product was 1 to 2, the final combined score was 1, and when it was 3 to 4, the final combined score was 2.
The Ca-IX immunoreactivity scoring system used is a variation of a previously reported system. 4 Sections were scored as the average of 20 fields at 200×. The percentage of positive cells was scored as follows: 0 = less than 1% positive cells, 1 = 1% to 30% positive cells, and 2 = more than 30% positive cells.
Intratumoral Microvessel Density
Subcutaneous histiocytic sarcomas and low-grade STS sections were stained with CD31 antibody, and IMD was quantified based on the method by Weidner et al. 49 Only subcutaneous histiocytic sarcomas were included, to compare between tissues with similar vascular network. Vascular “hot spot” regions, defined as regions of high vascular density within the tumor, were identified at 100× magnification. Within these areas, individual microvessel counts were made on a 200× field, and any brown-staining endothelial cell or endothelial cell cluster, clearly separate from an adjacent microvessel, was considered a single, countable microvessel.
Statistical Analyses
A Wilcoxon signed ranked test was used to assess the differences between Glut-1 and Ca-IX immunoreactivity score on a case-by-case basis for all tumor types. The procedure consisted of statistical assessment of the deviation from median = 0 on the differences between Glut-1 and Ca-IX immunoreactivity score for each case. The difference in immunoreactivity score between different tumor types was assessed with a Mann-Whitney test. To assess the differences in IMD (CD31 staining), the data were first subjected to normality testing. The data were not normal, and the Kruskal-Wallis test was chosen to evaluate differences between groups. The differences were considered statistically significant at P < .05 for all the tests described above.
Results
Ca-IX and Glut-1 Expression
All tumor sections were examined for Glut-1 and Ca-IX immunoreactivity, and the results are summarized in Table 1. Glut-1 staining pattern was membranous and occasionally cytoplasmic (Figs. 1, 2). Glut-1 immunopositivity was more intense at the periphery of areas of necrosis, which was more frequent in sections from histiocytic sarcomas. Ca-IX staining pattern was cytoplasmic and generally weak (Figs. 3, 4).
Frequencies of Glut-1 and Ca-IX Immunoreactivity Score Results for Canine Sarcomas.
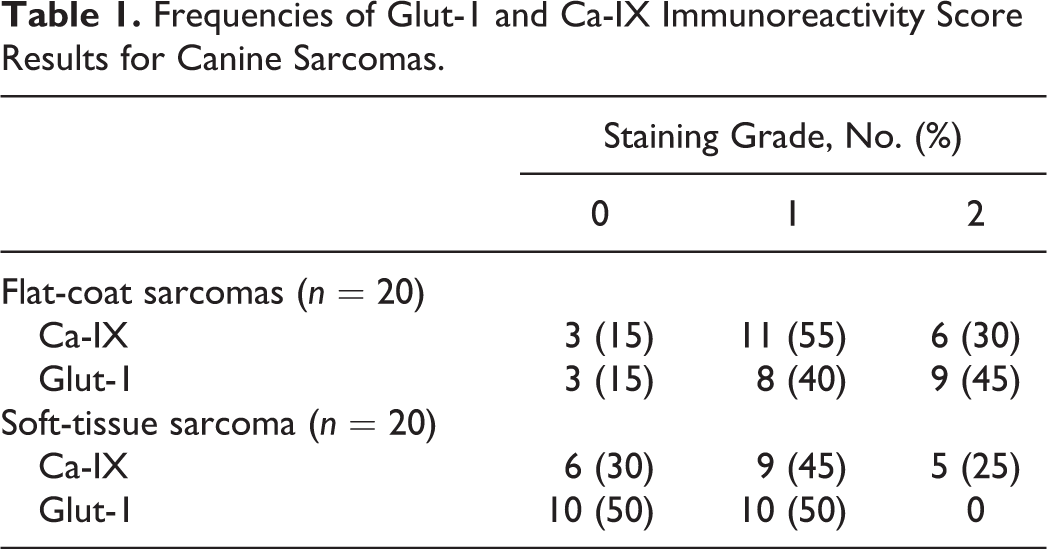
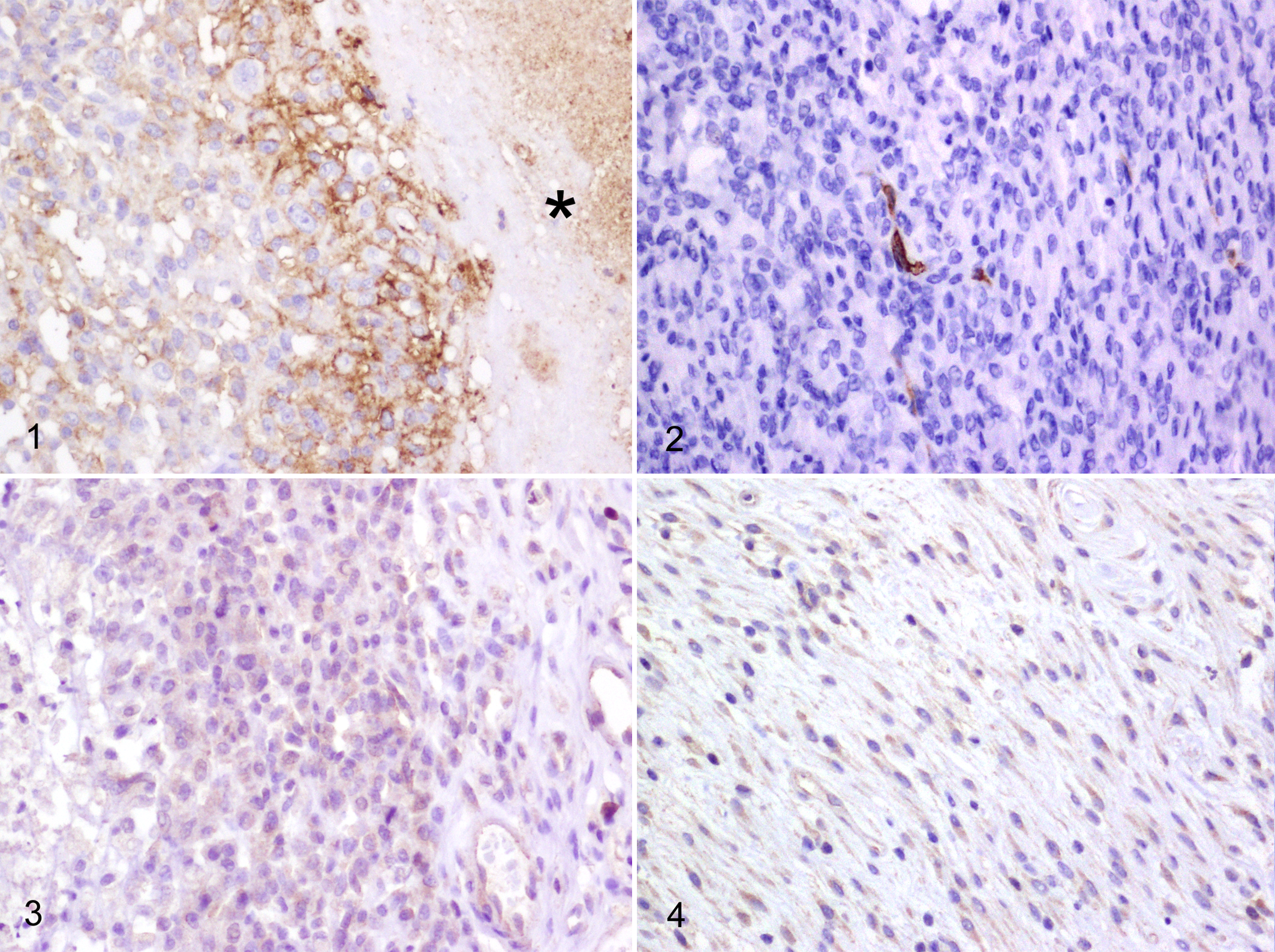
Regarding differences in immunoreactivity between the 2 markers evaluated on a case-by-case basis (Table 2), there were no significant differences in both histiocytic sarcomas and STS.
Differential Immunoreactivity Score of Glut-1 and Ca-IX in Canine Sarcomas.
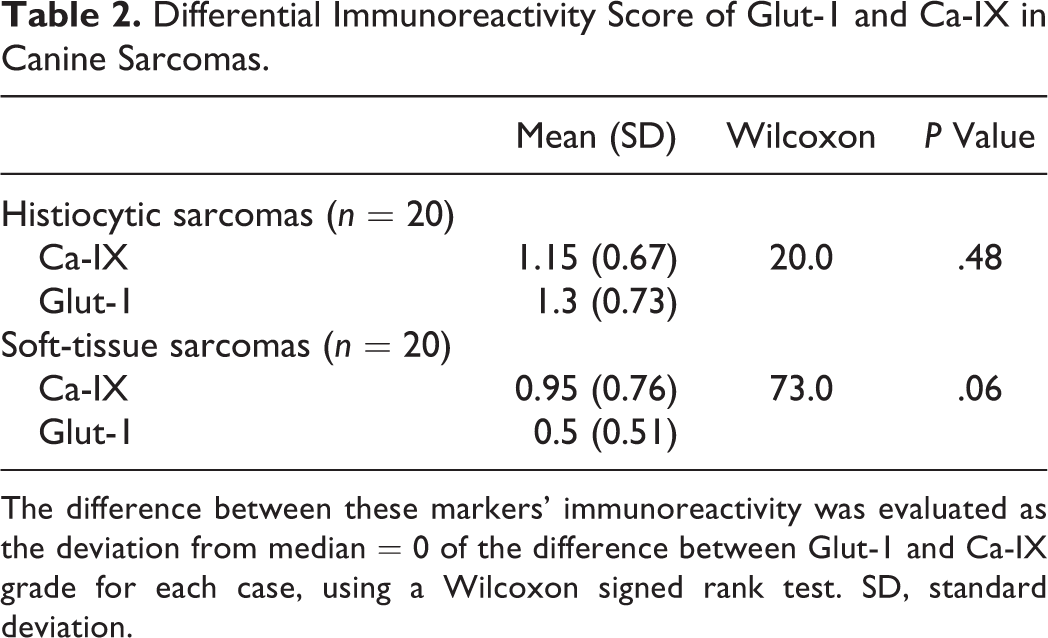
The difference between these markers’ immunoreactivity was evaluated as the deviation from median = 0 of the difference between Glut-1 and Ca-IX grade for each case, using a Wilcoxon signed rank test. SD, standard deviation.
The results of the comparison of immunoreactivity score between histiocytic sarcomas and low-grade STS are presented in Table 3. Significantly higher Glut-1 staining was noted in histiocytic sarcomas than in low-grade STS, in the absence of significant differences for Ca-IX staining.
Differences in Immunoreactivity Score of Glut-1 and Ca-IX Between Histiocytic Sarcomas and Low-Grade Soft-Tissue Sarcomas.
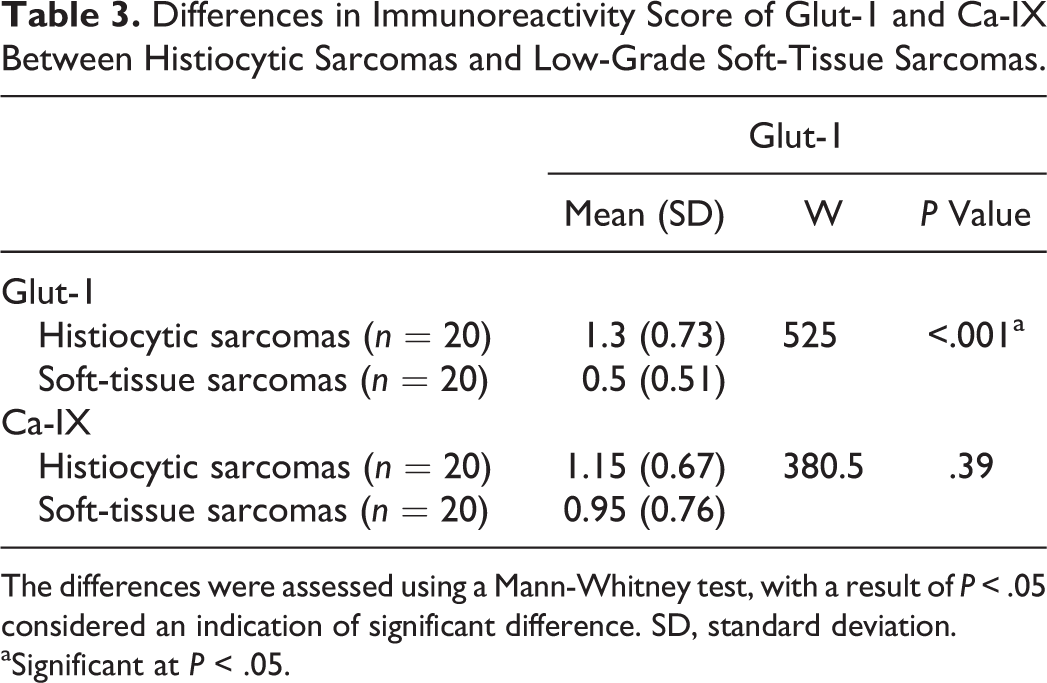
The differences were assessed using a Mann-Whitney test, with a result of P < .05 considered an indication of significant difference. SD, standard deviation.
aSignificant at P < .05.
Evaluation of IMD
The IMD for each sample was calculated as described for both low-grade STS and localized subcutaneous histiocytic sarcomas (Fig. 5). Histiocytic sarcomas had significantly higher IMD than low-grade STS (Kruskal-Wallis; P < .001).
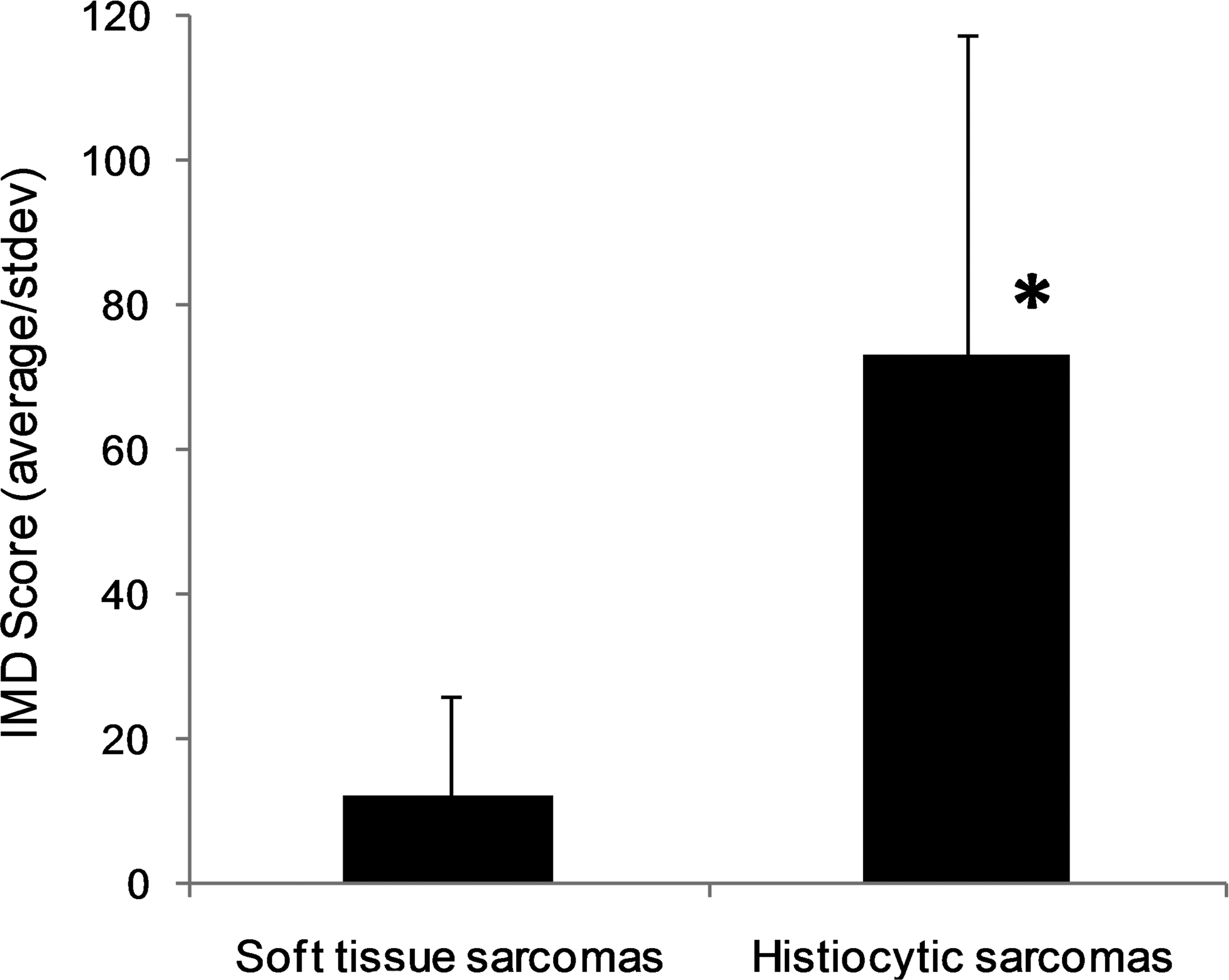
Comparison of intratumoral microvessel density (IMD) score in low-grade soft-tissue sarcomas (n = 20) and subcutaneous histiocytic sarcomas (n = 9). Error bars = standard deviation. *Significantly different at P < .001 (Kruskal-Wallis).
In the localized subcutaneous histiocytic sarcomas, the IMD values increased sequentially with Glut-1 and Ca-IX immunoreactivity scores, as shown in Fig. 6. This increase was significant for Glut-1 (Kruskal-Wallis; P = .002) and not significant for Ca-IX (Kruskal-Wallis; P = .21).
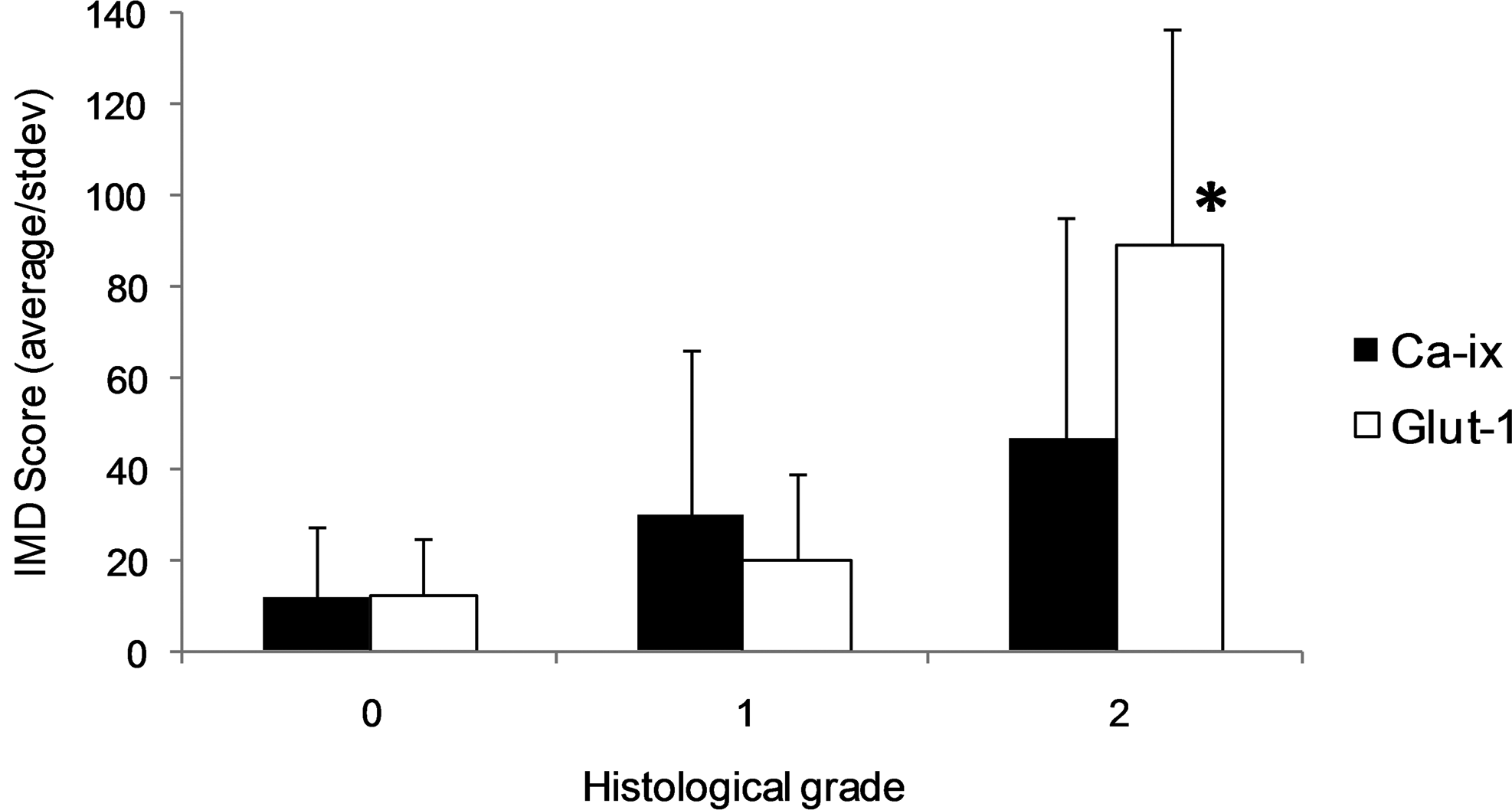
Intratumoral microvessel density (IMD) in low-grade soft-tissue sarcomas (n = 20) and subcutaneous histiocytic sarcomas (n = 9) relative to immunoreactivity score of Glut-1 and Ca-IX. Error bars = standard deviation. *Significantly different at P = .002 (Kruskal-Wallis).
Discussion
This study is the first to document the expression of the endogenous markers of chronic hypoxia Glut-1 and Ca-IX in naturally occurring canine histiocytic sarcomas and low-grade STS.
In this study, 85% of the histiocytic sarcomas examined demonstrated a Glut-1 immunoreactivity score of 1 or 2. This suggests that Glut-1 is consistently expressed at a measurable level in a high percentage of these tumors. Interestingly, more abundant and intense Glut-1 immunoreactivity was noted in the neoplastic cells immediately adjacent to areas of necrosis within these sections, which supports the theory that hypoxia develops at the limit of oxygen diffusion from tumor capillaries. Previous studies have reported similar observations of increased Glut-1 expression in tumoral perinecrotic regions in both human and canine tumors. 2,39
The results also show a statistically significant difference in Glut-1 immunoreactivity of histiocytic sarcomas compared with low-grade STS, suggesting that hypoxia is more severe in histiocytic sarcomas. This is an interesting observation because in human STS, hypoxia has been associated with high histological grade 43 and a shorter disease-free survival. 36 Canine histiocytic sarcomas have been reported to have a metastatic rate of more than 90% and an average median survival time of 4 months, 18 whereas canine low-grade STS are reported to have a median survival of approximately 33 months following conservative surgery. 9
Intratumoral microvessel density measured by CD31 immunostaining was significantly higher in the subcutaneous histiocytic sarcomas than in the low-grade STS.
In humans, IMD determined by CD31 immunostaining has been associated with high metastatic rate and higher histological grade in solid tumours. 21 Similarly, canine soft-tissue sarcoma IMD values, measured by factor VIII and CD31 immunostaining, have been positively correlated with higher histological grade, necrosis scores, and mitotic scores. 30
The statistically significantly higher IMD in subcutaneous histiocytic sarcomas compared with low-grade STS is an interesting finding. Furthermore, in subcutaneous histiocytic sarcomas, the increase in IMD paralleled the increase in immunoreactivity of Glut-1.
Tumor cell adaptation to hypoxic conditions is mediated by activation of the HIF pathway, which, when activated by low oxygen tension, induces transcription of several different gene-regulating pathways, including angiogenesis, glycolysis, tissue invasion, and apoptosis, all of which contribute to tumor survival and progression.
Many of these HIF downstream products have been intercorrelated in human hypoxic tumors. 5,19,37 No such correlation is possible in the present study. However, considering that VEGF is one of the main downstream products of HIF and that VEGF has been shown to play a role in mediating angiogenesis in canine soft tissue sarcomas, 11 it would be of interest to further investigate the biological interrelationships among hypoxia-induced gene expression in naturally occurring tumors of companion animals.
Moreover, in view of the well-documented invasive and metastatic behavior of histiocytic sarcomas, 12,18 it is not unreasonable to hypothesize that in these tumors, hypoxia could be a driving factor of neoangiogenesis. The larger number of new blood vessels might facilitate tumor invasion, increase the possibility of hematogenous metastases, and therefore account for the more aggressive clinical behavior.
Regarding Ca-IX, there was no difference in immunoreactivity score between the 2 types of sarcomas. Ca-IX is a membrane protein that participates in a variety of physiological processes, including respiration and acid-base balance, calcification, and bone resorption. Regarding its expression and prognostic significance in sarcomas, Ca-IX has been shown to be a marker of poor prognosis in human STS 32 and central chondrosarcoma. 6 The prognostic significance regarding other malignancies is more controversial. Ca-IX expression has been demonstrated to be an indicator of poor prognosis in cervical, 22 head and neck, 38 non–small cell lung, 27 nasopharyngeal, 23 and invasive breast carcinoma, 46 but there are other studies showing no correlation in cervical, 33 head and neck, 17 and bladder carcinoma 25 or a correlation with good outcome in renal clear cell carcinoma. 42 The lack of correlation and co-localization of Ca-IX with Glut-1 in this study may indicate that this marker is also controlled by non–hypoxia-induced mechanisms in canine sarcomas. More work is needed to clarify this.
The limitations of the study are its retrospective nature, lack of survival data, and relatively small number of cases. The assessment of a potential prognostic value of these markers in canine sarcomas would require a larger, prospective study, including low-, intermediate-, and high-grade STS with clinical follow-up data. However, this study, which reports Glut-1 and Ca-IX immunoreactivity in canine histiocytic sarcomas and low-grade STS, reveals comparatively higher Glut-1 immunoreactivity and IMD in histiocytic sarcomas, which paralleled the increase in Glut-1 immunoreactivity. These findings suggest a potential role of hypoxia in the biology of these tumors and encourage further research on the role of hypoxia in canine neoplasia. Also, these results may provide a base for investigation of the potential prognostic use of these markers in naturally occurring canine tumors.
Footnotes
Acknowledgements
We thank Madeline Fordham for her assistance with the immunohisto-chemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
