Abstract
A 9-year-old cynomolgus monkey (Macaca fascicularis) infected orally with bovine spongiform encephalopathy (BSE) was presented for necropsy following euthanasia 4 years post infection (p.i.). This macaque R984 was exposed to a BSE dose that causes a simian form of variant Creutzfeldt-Jakob disease (vCJD) within 5 years p.i. in other macaques. All orally BSE-infected macaques developed a significant weight gain within the first 2 years p.i. compared with non-BSE-infected age- and sex-matched control animals, suggesting increased risk of type 2 diabetes (T2D). In contrast, macaque R984 developed rapid weight loss, hyperglycemia, and glucosuria and had to be euthanatized 4 years p.i. before clinical signs of vCJD. Pancreas histopathological evaluation revealed severe islet degeneration but, remarkably, no islet amyloid deposits were present. Immunostaining of pancreas sections for insulin and glucagon confirmed the loss of endocrine cells. In addition, prions were present in the adenohypophysis but not in other areas of the brain, indicating centripetal prion spread from the gut during the preclinical phase of BSE infection. Plasma glucose and insulin concentrations of macaque R984 became abnormal with age and resembled T2D. This unusual case of spontaneous T2D in the absence of islet amyloid deposits could have been due to early prion spread from the periphery to the endocrine system or could have occurred spontaneously.
A 9-year-old female cynomolgus monkey (Macaca fascicularis) R984 was dosed orally with 5 g of bovine spongiform encephalopathy (BSE) at the age of 5 years. Macaque R984 belonged to a group of 18 female macaques orally dosed with BSE as part of an European Union–supported BSE risk assessment study. 10 Cerebrospinal fluid (CSF) samples were collected at regular intervals for analysis of 14-3-3 protein (14-3-3p) as a biomarker of brain damage from all macaques. 10 All CSF samples of macaque R984 remained 14-3-3p negative, and there was no evidence of behavioral changes or ataxia throughout the observation period (data not shown). Therefore, macaque R984 represented a preclinical stage of prion disease. However, the animal developed severe hyperglycemia and glucosuria and displayed progressive weight loss, which necessitated euthanasia 4.1 years post infection (p.i.) before the expected entry into clinical phase of simian variant Creutzfeldt-Jakob disease (vCJD).
Outcome of the Oral BSE Infection
At necropsy, a surrogate marker for infectious prions called proteinase K–resistant prion protein (PrPres) 10 was detected by Western immunoblot 10 or paraffin-embedded tissue (PET) blot 10 in gut-associated lymphoid tissue (GALT) specimens, spleen, lumbar spinal cord segments, the adenohypophysis, and the trigeminal ganglions at both sides. However, PrPres deposits were not detected in the obex, brain stem, or other regions of the brain. Moreover, tissue sections of the pancreas were also PrPres negative (data not shown).
Microscopic Findings
Pancreas histopathological examination of the case revealed very small islets (Fig. 1a) compared with 6 age- and sex-matched uninfected healthy controls and BSE-infected nondiabetic macaques (Fig. 1b). This finding was consistent with severe insulin deficiency. A unique observation was the absence of islet amyloid deposits, which are typical for naturally occurring type 2 diabetes (T2D) in non-human primates (NHP). 5
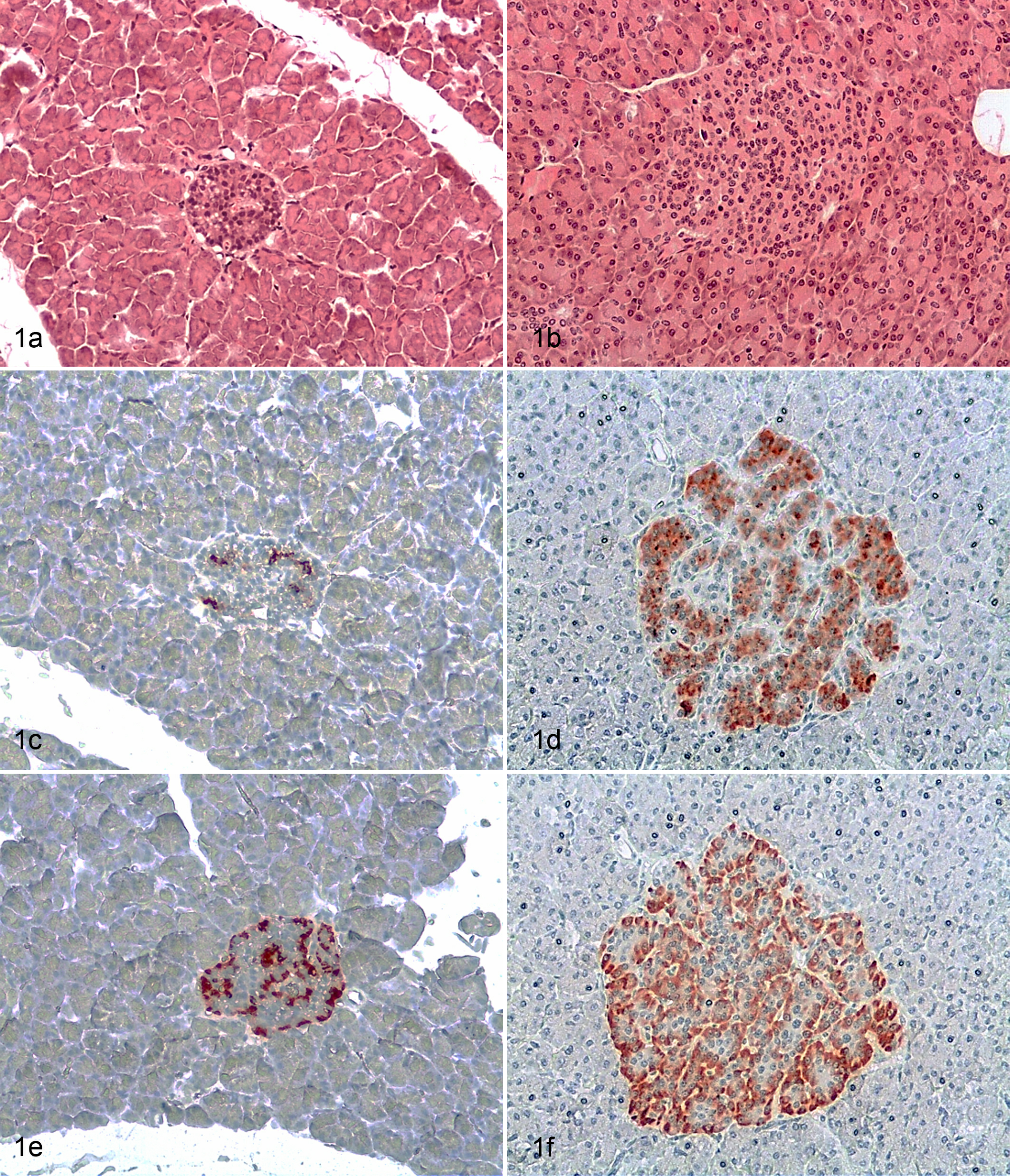
Pancreas; cynomolgus macaque. Islets size of the R984 macaque (a) compared with islets of a nondiabetic animal infected with bovine spongiform encephalopathy (BSE) (b). Immunostaining for insulin (β cells) (c) and glucagon (α cells) (e) of the R984 animal compared with islets of a nondiabetic BSE-infected macaque (d, f).
Immunohistochemistry
Insulin (β cells) and glucagon (α cells) immunostaining was performed as described (Hamburg, Germany). 7 Briefly, sections were incubated with DAKO polyclonal guinea pig anti-insulin antibody (1:50) and DAKO polyclonal rabbit anti-human glucagon antibody (1:4000) at room temperature for 2 hours. DAKO LSAB+ System-HRP kit was used for horseradish peroxidase labeling and DAKO AEC+ High Sensitivity Substrate Chromogen for staining. Immunohistochemistry analysis confirmed the loss of endocrine cells. The remaining islets contained few β cells (Fig. 1c), whereas α cells were more frequent (Fig. 1e). However, the number of β and α cells was dramatically reduced compared with age- and sex-matched uninfected healthy controls (Fig. 1d, f).
Metabolic Parameters
After oral BSE infection, macaque R984 gained 80% (2.8 kg) weight in the first 2 years followed by rapid weight loss (Fig. 2a). This abnormally rapid weight gain was also observed in the remaining 4 animals of the orally BSE-infected group.
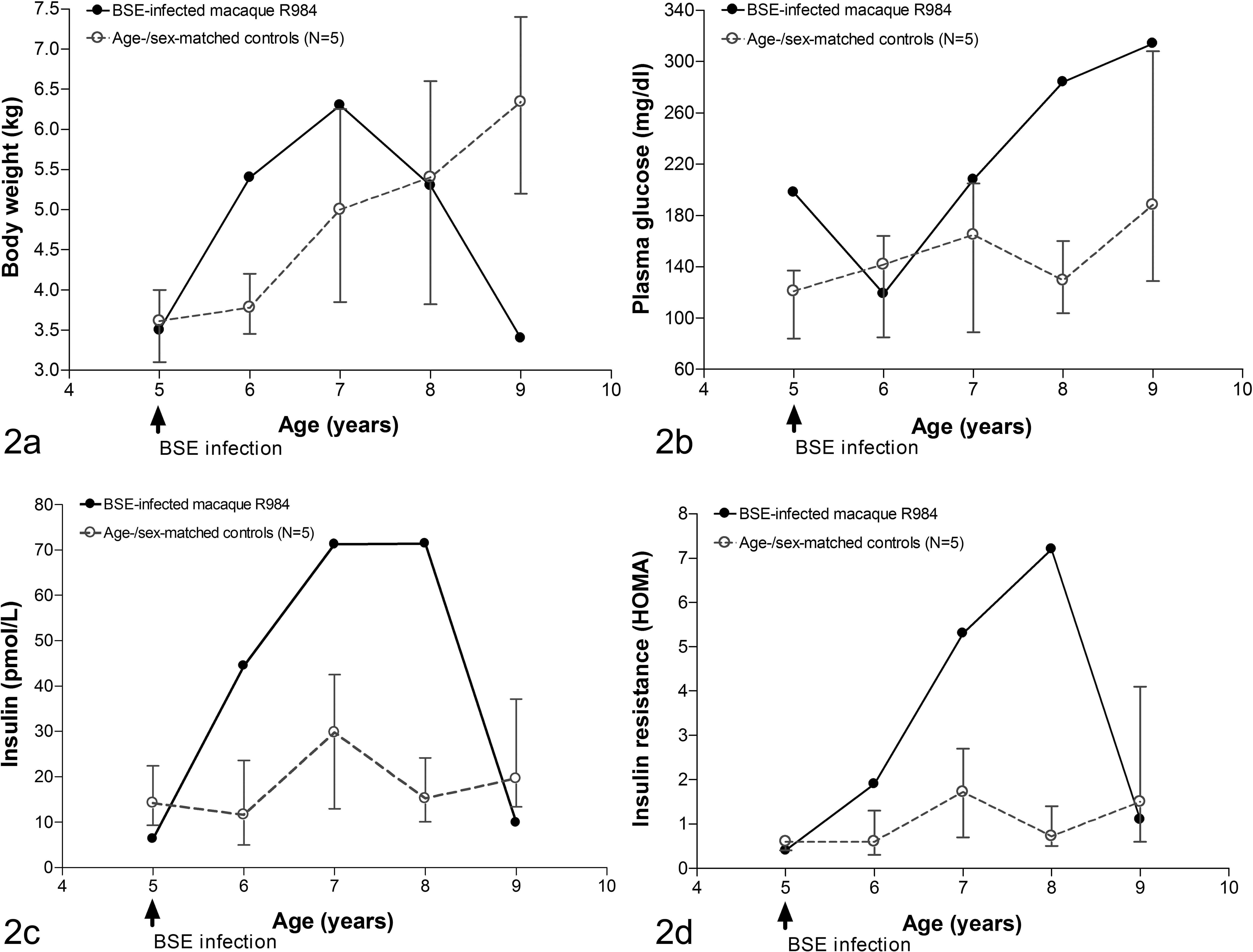
Longitudinal metabolic changes in the macaque R984 infected with bovine spongiform encephalopathy (BSE). (a) The course of body weight changes of the R984 and healthy macaques over the study period. (b) Fasting plasma glucose concentration. (c) Fasting plasma insulin concentrations. (d) HOMA-Index for insulin resistance. The data for the control animals are expressed as mean and range (maximum and minimum).
Fasting plasma samples, collected at ∼12-month intervals, from the case were analyzed for glucose using an Accu-Check Aviva glucometer (Roche Diagnostics, Mannheim, Germany). Fasting plasma glucose concentrations started to increase 2 years p.i., and by 4 years p.i. the animal was severely hyperglycemic (Fig. 2b). End of study triglyceride and cholesterol concentrations as well as α-amylase and lipase activities were analyzed using VetTest 8008 (Idexx Corp., Ludwigsburg, Germany). Triglyceride and cholesterol concentrations were higher in the diabetic macaque R984 (1.88 g/L and 3.75 g/L, respectively) than in non-BSE-infected macaques (0.52-1.05 g/L and 0.73-1.39 g/L, respectively); lipase and α-amylase activities were not different (data not shown).
Insulin and glucagon concentrations were measured using a human ultrasensitive insulin ELISA kit (Mercodia, Uppsala, Sweden) and a human glucagon ELISA kit (Phoenix Europe, Karlsruhe, Germany). Insulin concentrations increased during the first 3 years p.i. and decreased sharply shortly before the animal was euthanatized (Fig. 2c); glucagon remained stable (data not shown). The case also developed insulin resistance 2 years p.i. as defined by homeostasis model assessment (HOMA) 4 (Fig. 2d).
Diagnosis
Following the histopathology, immunohistochemistry, and biochemistry analyses, the diagnosis of this preclinical BSE case was insulin deficiency and diabetes due to severe islet degeneration. Moreover, the case showed an unusual pattern of prion spread and a PrPres-positive pituitary gland, whereas all other areas of the brain were PrPres negative.
Discussion
Cynomolgus monkeys and other NHP species are prone to develop diabetes that resembles T2D in humans. 5,8 Accordingly, during our long-term studies in 18 orally BSE-infected macaques and 6 mock controls, a number of infected and noninfected animals developed clinical and laboratory signs of diabetes (Strom et al., manuscript in preparation). At necropsy, amyloid plaques were detectable in pancreatic islets of all prediabetic and diabetic macaques but not in diabetic macaque R984. Macaque R984 presented clinically as a typical simian diabetes case except for complete absence of islet amyloid plaques. To our knowledge, a case of spontaneous diabetes in cynomolgus monkeys in without islet amyloid deposits has not been reported. In addition, macaque R984 showed evidence of a centripetal prion infection of the adenohypophysis.
Progression toward T2D is associated with obesity, insulin resistance, dyslipidemia, decreased β-cell mass, and the presents of islet amyloid polypeptide deposits. Islets of overt T2D diabetic NHPs are larger compared with healthy controls. However, islets of animals with overt diabetes are comprised of islet amyloid. Islet cell loss in the absence of islet amyloid deposition strongly suggests a different pathogenesis for spontaneous diabetes in this BSE-infected macaque.
Prions cause neurodegenerative diseases in humans and animals. 6 The main if not the sole component of the infectious agent is the misfolded pathogenic isoform of the cellular prion protein (PrPC). A proteinase K-resistant core of the pathogenic isoform (PrPres) is used as a surrogate marker for the clinical evidence of infectious prions, 1 and these PrPres fragments can be detected in spongiform encephalopathies. However, infectivity can also be detected in the absence of these PrPres fragments, suggesting the existence of so-called PK-sensitive prions (PrPsens). 2
Type 2 diabetes is strongly associated with obesity. Interestingly, this case report shows that oral BSE infection caused weight gain at a very early stage of the preclinical phase of infection, indicating that peripheral BSE infection could be a risk factor for obesity-linked development of spontaneous T2D. This confirms previous findings in rodent models and shows a possible link between an infectious agent and T2D. 9 Insulin resistance in all our diabetic animals was likely the result of the increased body mass along with lipotoxicity and/or inflammation (reviewed in Lumeng and Saltiel 3 and Wang and Kaltenboeck 9 ). We hypothesize that prion infection–induced obesity accelerated this process by a yet unknown mechanism.
Compared with rodents (Suppl. Fig. S1), cynomolgus macaques express significantly lower levels of PrPC in islets (Suppl. Fig. S2). In this view, infectious prions cannot replicate at a high rate in simian islets since prions use PrPC as a template for replication. This may explain why PrPres was not detectable in simian pancreatic tissues. Alternatively, a PK-sensitive form of prions 2 may be present in pancreatic islets.
There is no evidence that islet amyloid–free diabetes in macaque R894 was related to the detection of prions in the pituitary gland of this macaque during an early preclinical period of BSE infection. Hypothalamic dysfunction could have affected glucoregulation and contributed to the absence of islet amyloid, a possibility that requests further investigations.
In conclusion, we show that the loss of islet cells in spontaneous T2D can occur in the absence of islet amyloid deposits. In addition, there was evidence that BSE prions centripetally entered the pituitary gland during the preclinical phase of BSE infection in this single case.
Footnotes
Supplementary information available in online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
