Abstract
Many subtypes of low-pathogenicity avian influenza (LPAI) virus circulate in wild bird reservoirs, but their prevalence may vary among species. We aimed to compare by real-time reverse-transcriptase polymerase chain reaction, virus isolation, histology, and immunohistochemistry the distribution and pathogenicity of 2 such subtypes of markedly different origins in Mallard ducks (Anas platyrhynchos): H2N3 isolated from a Mallard duck and H13N6 isolated from a Ring-billed Gull (Larus delawarensis). Following intratracheal and intraesophageal inoculation, neither virus caused detectable clinical signs, although H2N3 virus infection was associated with a significantly decreased body weight gain during the period of virus shedding. Both viruses replicated in the lungs and air sacs until approximately day 3 after inoculation and were associated with a locally extensive interstitial, exudative, and proliferative pneumonia. Subtype H2N3, but not subtype H13N6, went on to infect the epithelia of the intestinal mucosa and cloacal bursa, where it replicated without causing lesions until approximately day 5 after inoculation. Larger quantities of subtype H2N3 virus were detected in cloacal swabs than in pharyngeal swabs. The possible clinical significance of LPAI virus-associated pulmonary lesions and intestinal tract infection in ducks deserves further evaluation.
Keywords
In recent years, strains of high-pathogenicity (HP) avian influenza (AI) virus have emerged in poultry and, secondarily, in humans, thus causing much concern worldwide. All strains of the virus affecting domestic animals or humans ultimately derive from low-pathogenicity (LP) strains circulating in wild birds, particularly waterfowl. 19 Yet, despite intensive virological studies in North America and Eurasia, 28 gaps remain in our knowledge about the dynamics of these LP strains in their natural hosts, in terms of their tissue distribution and potential replication sites; the lesions that they cause, which in turn may affect host behavior; and their shedding patterns. A better knowledge of these parameters of infection in individual birds is critical to fully understand the pathogenesis and epidemiology of different strains of LPAI virus. This information may in turn contribute to the design of more precise models of infection and more efficient surveillance programs as this virus circulates in populations of wild birds.
All 16 hemagglutinin (H) subtypes and all 9 neuraminidase (N) subtypes of AI virus have been detected in wild bird reservoirs, and dabbling ducks, particularly Mallard (Anas platyrhynchos), have been found to be infected with AI virus more frequently than other birds. 28 Some subtypes are also thought to predominate in certain groups of birds, such as H13 and H16 in gulls. 20,28,36,37 However, the reasons for this are poorly understood, and it is also not known whether gull-specific subtypes of AI virus can infect and cause lesions in tissues of waterfowl. Improved knowledge about the ability of different subtypes of AI virus to infect different groups of birds or move among different species would contribute to a better understanding of the dynamics of this virus in wild bird populations.
Except for an outbreak among Common Terns (Sterna hirundo) in South Africa in 1961, 2 strains of AI virus carried by wild birds generally are not known to cause disease in these birds and, until recently, there were no reports of mortality due to AI virus in wild waterfowl. Prior to the emergence of the highly pathogenic H5N1 subtype in Southeast Asia, strains of AI virus that were known to cause severe disease in poultry consistently failed to induce clinical disease experimentally in ducks. 7,31,32 Aside from 1 recent study of natural infection of wild Mallards by LPAI virus, 8 it is not known whether subtypes of LPAI virus can cause gross or microscopic lesions in these birds following exposure to the virus and, if so, how these lesions develop and regress over the course of infection. In poultry, morbidity and mortality can vary substantially among birds within a flock, even with LPAI viruses. 25,46 It is therefore possible that some strains of AI virus circulating in populations of wild waterfowl are able to induce mild clinical signs and lesions in some birds that may be sufficient to result in significant behavioral changes, such as delayed migratory patterns. 42
In this study, we wished to compare differences in character of host-virus interaction in Mallards between an LPAI virus of the subtype H2 known to infect this and other waterfowl species in the wild and an LPAI virus of the subtype H13, which has very rarely been isolated from ducks. 1,26 To this effect, we experimentally infected Mallards with 1 of 2 LPAI virus strains—an H2N3 strain isolated from a free-living Mallard and an H13N6 strain isolated from a free-living Ring-billed Gull (Larus delawarensis)—and followed their replication and associated lesions by sequential gross and microscopic examination, immunohistochemistry (IHC), detection of viral nucleic acid by real-time reverse-transcriptase polymerase chain reaction (RRT-PCR), and virus isolation in tissue culture.
Materials and Methods
Virus propagation
Subtype H2N3 (A/Mallard/New Brunswick/1/2006) was isolated in August 2006 at the Erasmus Medical Centre, Rotterdam, The Netherlands, from a free-living hatch-year Mallard captured in a marsh in New Brunswick, eastern Canada. A stock virus of A/Gull/Ontario/680-6/2001 (H13N6) was isolated in 2001 at the Canadian Food Inspection Agency, Winnipeg, from a free-living Ring-billed Gull captured on Lake Ontario, central Canada. Stocks of each viral subtype were propagated twice in 10-day-old specific-pathogen-free embryonated chicken eggs, and infectious allantoic fluid (1010.5 median embryo infective doses [EID50]/ml for H2N3, 109.5 EID50/ml for H13N6) was harvested and stored at –70°C for use as inoculum. On the inoculation day, inocula of each viral subtype were serially diluted in phosphate-buffered saline (PBS) to obtain a final titer of 1 × 108 EID50/1.5 ml.
Animal Inoculation and Sampling
Mallards were captive-bred (Dierenhandel Hoogendoorn, Stolwijk, The Netherlands) and housed in an animal shed indoors after hatching. At 22 weeks of age, 4 groups of experimental ducks, 8 birds in each group (4 male, 4 female), were housed in 4 negatively pressurized isolator units (glove boxes). One group of 4 control ducks (2 male, 2 female) was housed in a Plexiglas box in a separate room. Blood samples were taken from all 36 birds at that time to determine the presence of antibodies against influenza virus H subtypes 1 to 13 by the hemagglutination inhibition test. 29 Food and water were provided ad libitum. All birds were acclimated for 10 days in their respective boxes prior to experimental infection. Birds were weighed on days –10, –7, and –4 and pharyngeal and cloacal swabs collected on day –7. On day 0, 16 birds in 2 glove boxes (H2N3 group) were each inoculated under isoflurane anesthesia with 1 × 108 EID50 of H2N3 virus in 1.5 ml PBS, half of the inoculum intratracheally and half intraesophageally. The 16 birds in the 2 other glove boxes (H13N6 group) were similarly inoculated with 1 × 108 EID50 of H13N6 virus in 1.5 ml PBS. The 4 control birds (control group) received only PBS. The body temperature of 3 male ducks inoculated with H2N3 virus was measured by telemetry every 15 minutes by means of a transponder (IMAG, Wageningen, The Netherlands) inserted in their coelomic cavity at 20 weeks of age. Birds were weighed and pharyngeal and cloacal swabs collected daily from days 0 to 7 and every 2 to 3 days afterward. On each occasion, prior to these procedures, the undisturbed birds were observed carefully for evidence of clinical signs. On each of days 1, 3, 6, and 17, 4 birds from the H2N3 group and 4 birds from the H13N6 group (equal numbers of sexes) were euthanized by exsanguination under anesthesia with isoflurane; 1 of the 3 ducks from the H2N3 group with an intracoelomic transponder was killed on day 6, and the 2 other ducks were killed on day 17. Two control birds were euthanized on each of days 3 and 6. To avoid cross-contamination, precautions were used between handling individual birds by changing the table cover and disinfecting gloves and dissection instruments with 70% ethanol. All experimental procedures were approved by an independent Animal Care and Use Committee of the Erasmus Medical Centre, University of Rotterdam.
Body temperatures from each of the 3 ducks with intracoelomic transponders in the H2N3 group were averaged daily after inoculation and compared with the average temperature for each duck prior to the inoculation day (days –6 to –1, inclusively) using the Wilcoxon signed-ranked test. The slopes of the weights for each duck were calculated by group (H2N3, H13N6, and control) for the preinoculation period (days –10, –7, –4, and 0) and for the first 5 days postinoculation (days 1, 2, 3, 4, and 5 or until the duck was euthanized). A Wilcoxon signed-ranked test was applied to the pre- and postinoculation slopes. Weight measurements taken before the inoculation were averaged for each duck to produce individual “before” values, and similarly, daily weight measurements taken after the inoculation were averaged for each duck to produce “after” values. Wilcoxon signed-ranked tests were used to compare the paired values (“before” and “after”) from each duck by group.
Virus Detection
Pharyngeal and cloacal swabs were collected in 1.5 ml of virus transport medium (Hank’s balanced salt solution containing 10% glycerol, 200 U/ml penicillin, 200 μg/ml streptomycin, 100 U/ml polymyxin B sulfate, 250 μg/ml gentamycin [MP Biomedicals, Zoetermeer, The Netherlands]) and frozen at –70°C. The following tissue samples were collected from each bird at the time of necropsy and frozen at –70°C: trachea, lung, air sac (partition between cranial and caudal thoracic air sacs on left and right sides), proventriculus, duodenum, small intestine at the level of the Meckel’s diverticulum, distal ileum, ceca, colon, cloacal bursa, liver, pancreas, and kidney. Tissue samples from experimental birds were subsequently weighed and homogenized in 3 ml of virus transport medium with a homogenizer (Kinematica Polytron, Lucerne, Switzerland).
RNA isolation and examination by RRT-PCR for the presence of the matrix gene segment of AI virus were performed as described previously for human influenza viruses and modified for avian influenza viruses. 27,44 RNA was isolated using a MagnaPure LC system with the MagnaPure LC Total nucleic acid isolation kit (Roche Diagnostics, Almere, The Netherlands). Amplification and detection were performed on an ABI 7000 with the TaqMan EZ RRT-PCR Core Reagents kit (Applied BioSystems, Nieuwerkerk aan den IJssel, The Netherlands). The samples were prepared and processed in parallel with several negative (transport medium) and positive (culture supernatant of viral isolate) control samples in each run. For virus isolation, 10-fold serial dilutions of the pharyngeal and cloacal swabs collected from experimental birds starting on day 1 and of homogenized tissue samples from these birds were inoculated in tissue culture (Mardin-Darby canine kidney [MDCK] cells) and the results expressed as 50% tissue culture infective dose (TCID50)/ml or TCID50/g of tissue, respectively, in triplicate as described previously. 33
For all samples, a cycling threshold (Ct) of 40 or higher on RRT-PCR analysis and a titer <100.8 TCID50/ml or g were considered negative for the presence of AI virus. The areas under the curve (AUC) of the viral excretions from the H2N3 group, measured by RRT-PCR and virus isolation from pharyngeal and cloacal swabs, were calculated for each duck and compared using the Wilcoxon signed-ranked test. A Cox proportional hazards model, with shared frailty, was used to analyze the duration of virus detection by RRT-PCR and virus isolation in pharyngeal and cloacal swabs of H2N3-infected birds as time elapsed after inoculation. The frailties were shared with each duck to account for the correlated observations from swabs within the same duck. Model diagnostics included the verification of the assumed proportional hazards using Shoenfeld and scaled-Shoenfeld residuals, checking for outliers with deviance residuals, and assessing goodness of fit with Cox-Snell residuals. 9 Statistical significance was set at P < .05.
Histology and Immunohistochemistry
Necropsies were performed according to a standard protocol, and the following tissues were collected for histologic and immunohistochemical examination: brain; nasal ethmoid; pharyngeal mucosa (caudal to choanae); trachea; left primary bronchus; left lung (transverse section in its mid-region); air sac (as for virus isolation); esophagus; proventriculus; duodenum; 4 intestinal segments, each approximately 7 cm long (2 consecutive segments between the distal region of the duodenum and Meckel’s diverticulum and 2 consecutive segments between Meckel’s diverticulum and colon); Meckel’s diverticulum and intestine just proximal to it; distal ileum; ceca; colon; cloacal bursa; spleen; liver; gallbladder; pancreas; cranial and caudal regions of the left kidney; left adrenal; gonad; and oviduct. A section of right lung was also taken when gross lesions were present. The 7-cm-long segments of intestine were prepared in the form of Swiss rolls. 26 The bursa of some male birds had regressed markedly and was lost in the trimming process. After fixation in 10% neutral-buffered formalin for 4 to 12 days, all tissue samples were embedded in paraffin, sectioned at 3 μm, and stained with hematoxylin and eosin (HE) for microscopic examination. Duplicate sections were stained for the detection of influenza A viral antigen. For this, the sections were incubated for 1 hour at room temperature with a primary antibody against the viral nucleoprotein (HB65; American Type Culture Collection, Manassas, VA) at a concentration of 2.5 to 5 μg/ml, as described previously, 34 with the following modifications: tissue sections were preincubated with 0.1% protease for 10 minutes at 37°C (Sigma, St Louis, MO). Binding of the primary antibody was detected using a peroxidase-labeled goat–anti-mouse IgG2a (Southern Biotech, Birmingham, AL). Peroxidase was revealed using 3-amino-9-ethyl-carbazole (AEC; Sigma Chemicals, Zwijndrecht, The Netherlands), resulting in a bright red precipitate. Lung tissue of a cat experimentally infected with influenza virus A/Vietnam/1194/2004 (H5N1) was included as a positive control. Isotype-matched and omission controls were included as negative controls. Tissue samples were labeled as positive for the influenza A viral antigen even if only a single or very few cells showed distinct staining of their nucleus. Microscopic anatomical terms used to describe the distribution of pulmonary lesions were based on Hodges. 16 In particular, a pulmonary lobule is defined here as the respiratory parenchyma surrounding a parabronchus (tertiary bronchus) and composed of air capillaries and blood capillaries.
Results
Clinical Findings
Serum samples collected from the 36 birds prior to experimental infection were all negative for the presence of antibodies to influenza virus H subtypes 1 to 13 by the hemagglutination inhibition test. No clinical signs were observed in any of the experimental or control groups. Up to the day of inoculation, both H2N3 and H13N6 groups had, on average, positive body weight slopes, indicating increase in body weight. During the first 5 days postinoculation, the H13N6 group showed a significantly higher body weight slope (P = .005), indicating a faster increase in body weight. In contrast, the H2N3 group showed a significantly lower body weight slope (P = .049) after inoculation and was on average negative, indicating a lower increase in body weight and some weight loss. This was corroborated by the average weights of the birds in the H2N3 and H13N6 groups before and after inoculation: the ducks in the H2N3 group showed no significant difference in weights before and after inoculation (P = .393), whereas ducks in the H13N6 group gained significantly more weight, on average approximately 20.2 g, after inoculation (P = .041). The control ducks showed no significant difference in body weight slope (P = .095) or in average body weight (P = .353) before and after sham inoculation. In the 3 ducks from the H2N3 group with an intracoelomic transponder recording body temperature, there was a nonsignificant trend (P = .109) on day 1 whereby the average body temperature was 0.4°C higher than during the 6 days prior to inoculation.
Virus Detection
All pharyngeal and cloacal swabs collected from experimental and control birds on days –7 and 0 were negative by RRT-PCR. All pharyngeal and cloacal swabs collected from control birds from day 1 to day 6 were negative by RRT-PCR, except for the pharyngeal swab of 1 bird euthanized on day 3 (Ct = 35.5), which was ascribed to contamination of unknown source.
In the H2N3 group, AI virus was demonstrated in pharyngeal swabs of most birds from days 1 to 10 by RRT-PCR and from days 1 to 4 by virus isolation, declining thereafter (Fig. 1a,b). In cloacal swabs, AI virus was demonstrated in slightly more than half of the birds on day 1 by RRT-PCR and by virus isolation; it was demonstrated in most birds from days 2 to 10 by RRT-PCR and from days 2 to 5 by virus isolation (Fig. 1a,b). By day 12 and beyond, pharyngeal and cloacal swabs from only a small proportion of the birds were positive by RRT-PCR, and none were positive by virus isolation. Based on the AUC, there were larger quantities of virus detected from cloacal swabs than from pharyngeal swabs using RRT-PCR (P = .009) and virus isolation (P = .007). Based on the shared frailty model, it was estimated that after the first detection, the hazard rate of viral detection using virus isolation was 3.2 times greater with cloacal swabs than with pharyngeal swabs (P = .037). In other words, after the first detection, virus isolation in tissue culture was approximately 3 times more likely to continue to detect the presence of virus from cloacal swabs than from pharyngeal swabs. No such statistically significant difference was found between cloacal and pharyngeal swabs by using RRT-PCR.
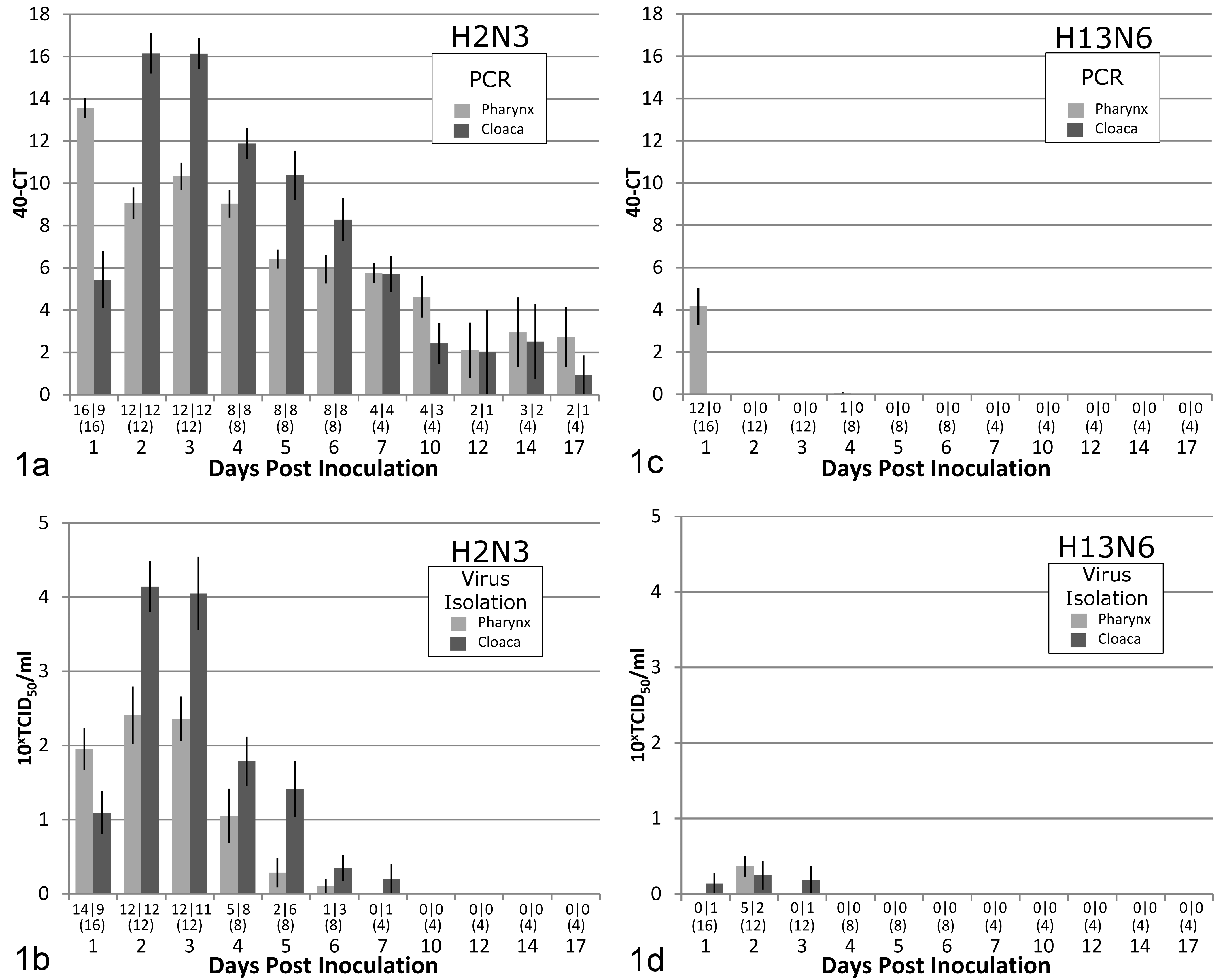
Bar graph with standard error estimates of results of viral detection in pharyngeal and cloacal swabs taken from Mallard ducks inoculated on day 0 with subtypes H2N3 and H13N6 of low-pathogenicity avian influenza (LPAI) virus and followed during the experimental study period (17 days). (a, c) Real-time reverse-transcriptase polymerase chain reaction (PCR) (expressed as 40 [minimum cycling threshold (Ct) value considered negative] – actual Ct value). (b, d) Virus isolation (10x × 50% tissue culture infective dose per ml [TCID50/ml], with a titer <100.8 TCID50/ml considered negative). Numbers above each day represent number of birds with positive pharyngeal swab/number of birds with positive cloacal swabs (total number of birds tested on that day). Each bar represents the average value for all birds tested on that day.
Also in the H2N3 group, a majority of the tissues examined from the respiratory and digestive tracts by RRT-PCR were positive for AI virus on days 1 and 3 (Table 1a). The lowest Ct values were from the lung on day 1 and from the caudal half of the intestine and the bursa on day 3. The liver and kidney from most birds were also positive on days 1 and 3, although Ct values for these tissues were high. Results of virus isolation paralleled the RRT-PCR results: titers were highest in the lung on day 1 and in intestinal tissues and the bursa on day 3 (Table 1a). Virus was also isolated from the kidney of 2 of 4 birds on day 1. On day 6, tissues from the digestive tract and the bursa remained positive by RRT-PCR in most birds, but the Ct values had increased substantially. By virus isolation, intestinal tissue from few birds remained positive on day 6, and all tissue samples were negative on day 17.
Detection by Real-Time Reverse-Transcriptase Polymerase Chain Reaction (PCR) and by Isolation in Tissue Culture (Mardin-Darby Canine Kidney Cells) of 2 Subtypes of Low-Pathogenicity Avian Influenza (LPAI) Virus in Internal Organs of Mallard Ducks Experimentally Inoculated via the Trachea and Esophagus on Day 0.
Four birds were sampled on each of 4 days. A cycling threshold (Ct) of PCR ≥40 was considered negative (–) for the presence of LPAI virus. Legend for virus isolation (VI) in each of the 4 birds sampled on each day: white, no virus isolated (titer <100.8 × 50% tissue culture infective dose [TCID50]/g); yellow, titer 100.8 to 101.9 TCID50/g; blue, titer 102.0 to 103.9 TCID50/g; red, titer 104.0 to 107.6 TCID50/g.
aRange of (number of birds with) positive Ct values.
bTiter of virus isolation in each of 4 birds sampled.
cAt level of Meckel’s diverticulum.
In the H13N6 group, AI virus was demonstrated in pharyngeal swabs of 12 of 16 birds on day 1 by RRT-PCR and in 5 of 12 birds on day 2 by virus isolation (Fig. 1c,d). In cloacal swabs, AI virus was demonstrated in very few birds by virus isolation, but not by RRT-PCR, from days 1 to 3 (Fig. 1c,d). By RRT-PCR, samples of respiratory tissue from most birds were positive for AI virus on day 1; the colon, bursa, and kidney were also positive in most birds, although Ct values for these tissues were high (Table 1b). The lung was still positive in all birds euthanized on day 3 and in 2 of 4 birds euthanized on day 6. By virus isolation, only a few samples from the respiratory and intestinal tracts were positive on day 1 (Table 1b).
Macroscopic Findings
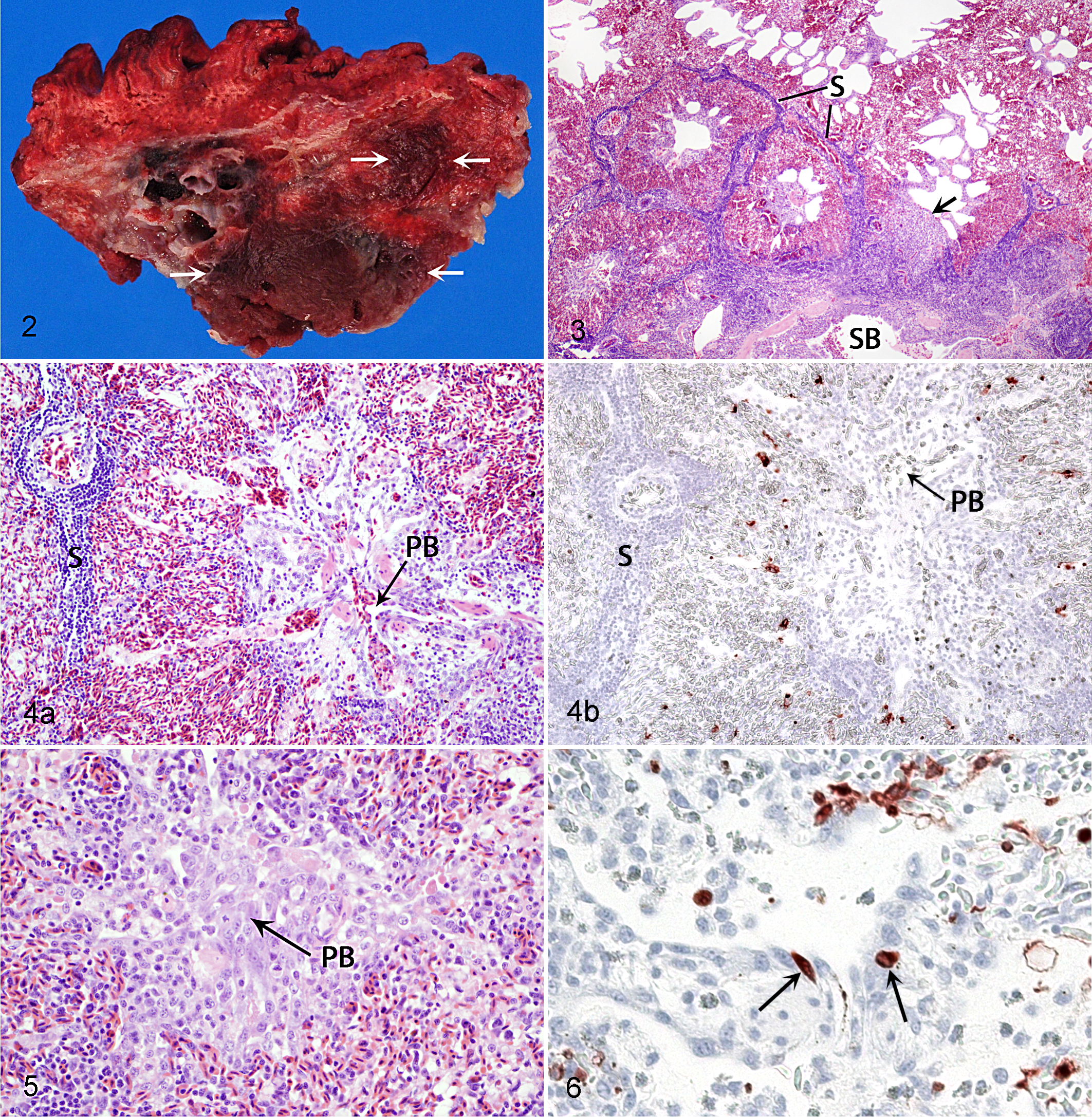
All birds were in good nutritional condition when necropsied. The only obvious lesions noted involved the lungs. On days 1 and 3 in all H2N3-infected birds and on day 3 in all H13N6-infected birds, the lateral region of 1 or both lungs had a well-demarcated area of congestion and presumed consolidation, involving approximately one-quarter or less of the parenchyma of individual organs (Fig. 2). A similar lesion affecting much smaller areas of the lungs was seen in 1 H2N3-infected bird and 2 H13N6-infected birds on day 6. No lesions were present in any of the infected birds on day 17 or in any of the control birds.
Histopathologic Findings
In all birds, lesions when present were confined to the lungs. On day 1, all H2N3-infected birds and 2 of 4 H13N6-infected birds had pulmonary lesions that were characterized by a lymphocytic, histiocytic, and heterophilic interstitial pneumonia and involved the lateral region of the organ, reaching medially to the intrapulmonary primary bronchus (IPPB) or its immediate tributaries of secondary bronchi (Fig. 3). More specifically, numerous inflammatory cells (mainly lymphocytes) and variable amounts of edema fluid infiltrated interlobular septa; there was marked congestion of the pulmonary lobules; interatrial septa surrounding parabronchial lumens were infiltrated by inflammatory cells (mainly macrophages and heterophils); and there was hypertrophy of the epithelium lining some parabronchial lumens (Fig. 4a). The mucosa of the IPPB and of secondary bronchi, at the interface between affected and normal regions of the organ, and the mesenchymal tissue surrounding these bronchi were infiltrated by a moderate to large number of inflammatory cells and were variably edematous.
On day 3, all H2N3- and H13N6-infected birds had pulmonary lesions that were similar in their nature and distribution to those on day 1. In addition, infiltration of lymphocytes in interlobular septa was now also involving the walls of small blood vessels; the pulmonary lobules were more cellular as a result of hypertrophy of epithelial cells of air capillaries and infiltration of macrophages and lymphocytes; the epithelium lining several parabronchial lumens was hypertrophic and also hyperplastic; and the mesenchyma surrounding the IPPB and secondary bronchi was more edematous. In some instances, hypertrophy and hyperplasia of the parabronchial epithelium and infiltration of inflammatory cells in surrounding interatrial septa had led to complete obstruction of parabronchial lumens (Fig. 5). Exudate, edema fluid, and cellular debris were now present in the lumens of parabronchi and, to a lesser extent, secondary bronchi in some birds.
On day 6, pulmonary lesions in all infected birds had regressed, and on day 17, the only pulmonary lesions consisted of mild multifocal interlobular and peribronchial lymphoid hyperplasia in the lateral region of the organ in 2 of 4 H2N3-infected birds and 3 of 4 H13N6-infected birds.
No pulmonary lesions were seen in 3 of 4 control birds. One of 2 control birds euthanized on day 3 had pulmonary changes similar to, but milder than, those in experimental birds and confined to a narrow band of tissue along the lateral border of the organ. This was not the control bird from which AI virus was identified in a pharyngeal swab by RRT-PCR.
Immunohistochemical Findings
Cells positive for AI virus nucleoprotein by IHC were confined to the lungs, air sacs, intestinal tract, and cloacal bursa of infected birds (Table 2). All control birds were negative. On day 1, the lungs of all 4 H2N3-infected birds and of 2 of 4 H13N6-infected birds contained many positive cells that were confined to the region of the lung, showing evidence of inflammation in the corresponding HE section. Most of these cells were within pulmonary lobules and seemed to involve the epithelium of air capillaries (Fig. 4b). A few positive cells could also be found in the epithelium lining some parabronchial lumens (Fig. 6). In 1 H13N6-infected bird, the epithelium of secondary bronchi adjacent to the affected region of the pulmonary parenchyma contained numerous positive cells as well. The air sacs of 2 of 4 H2N3-infected birds and of 2 of 4 H13N6-infected birds were positive, but with only a very few positive cells scattered along the surface epithelium. The intestinal tract of 1 of 4 H2N6-infected birds was positive, with most segments of intestine examined containing many positive villous epithelial cells. These cells could be found at all levels of individual villi, from their base to their tip, but tended to predominate at their tip; bright red precipitate, either in particulate form or as positively staining nuclei of mononuclear cells of undetermined identity, could also be found occasionally within the lamina propria adjacent to the positive epithelial cells.
Detection by Immunohistochemistry, Based on a Monoclonal Antibody Against the Viral Nucleoprotein, of 2 Subtypes of Low-Pathogenicity Avian Influenza (LPAI) Virus in Tissues of Mallard Ducks Experimentally Inoculated via the Trachea and Esophagus on Day 0.
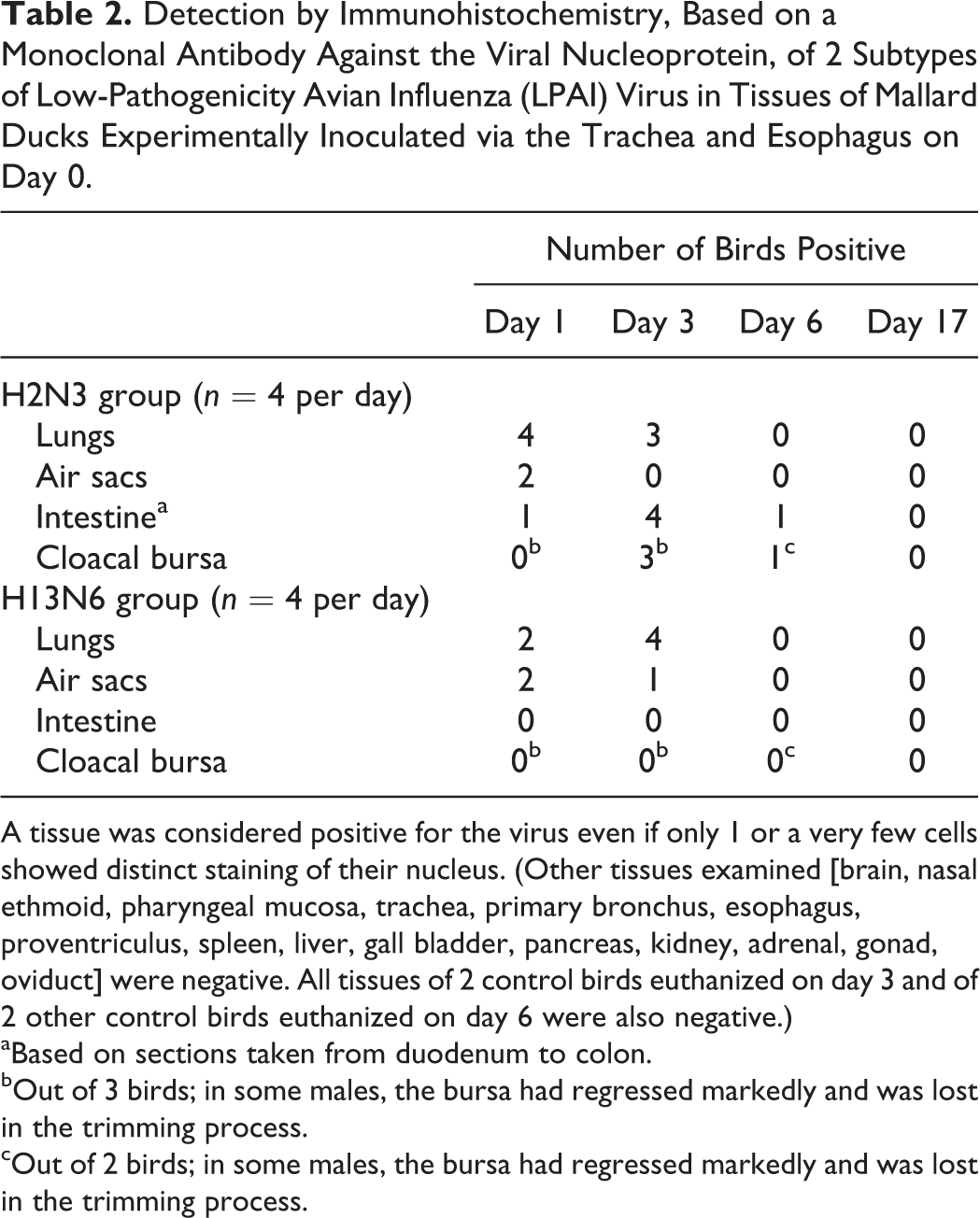
A tissue was considered positive for the virus even if only 1 or a very few cells showed distinct staining of their nucleus. (Other tissues examined [brain, nasal ethmoid, pharyngeal mucosa, trachea, primary bronchus, esophagus, proventriculus, spleen, liver, gall bladder, pancreas, kidney, adrenal, gonad, oviduct] were negative. All tissues of 2 control birds euthanized on day 3 and of 2 other control birds euthanized on day 6 were also negative.)
aBased on sections taken from duodenum to colon.
bOut of 3 birds; in some males, the bursa had regressed markedly and was lost in the trimming process.
cOut of 2 birds; in some males, the bursa had regressed markedly and was lost in the trimming process.
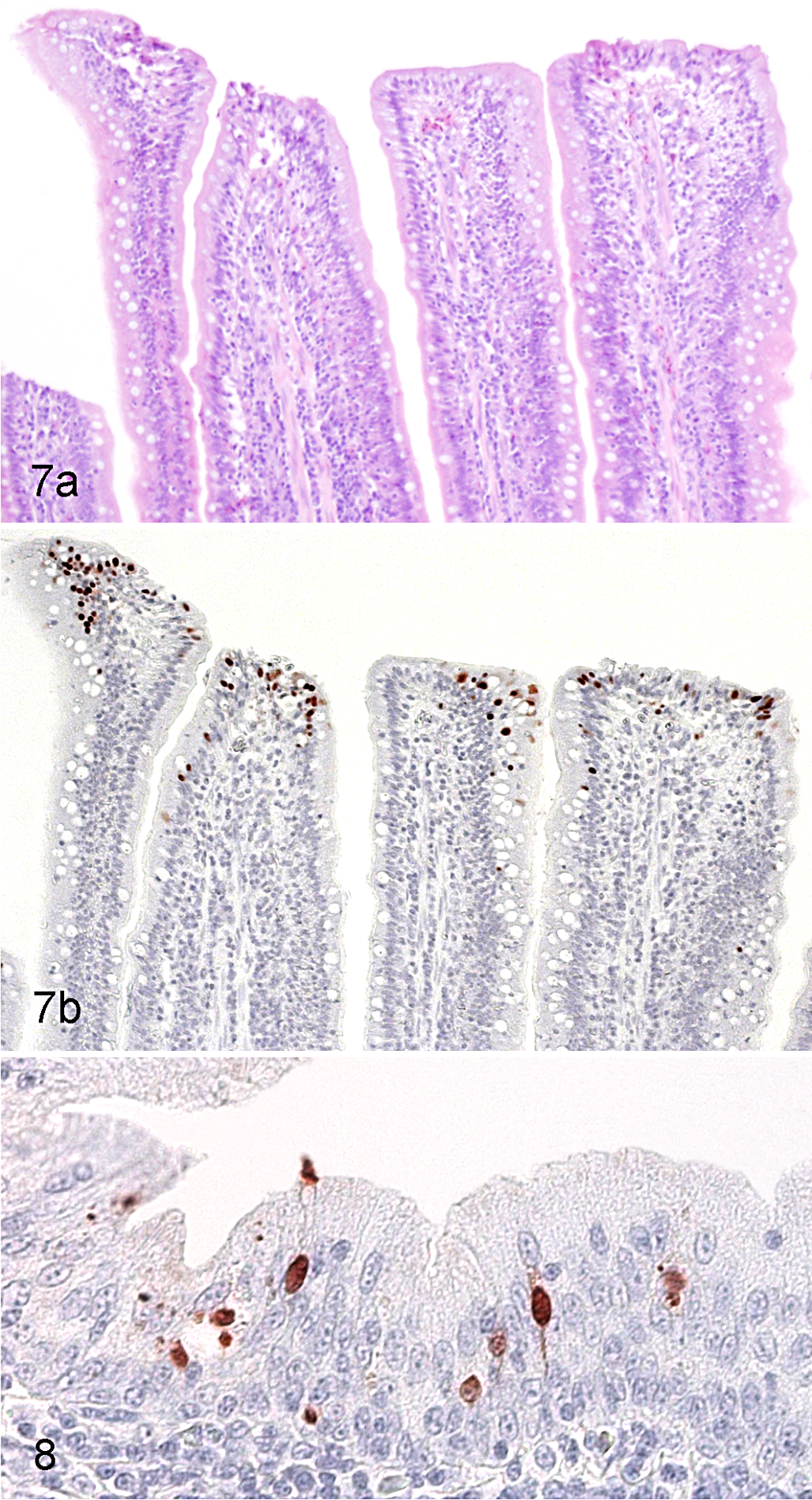
On day 3, the lungs of 3 of 4 H2N3-infected birds and of all 4 H13N6-infected birds were positive. As on day 1, the distribution of positive cells corresponded to that of inflammation, and these cells were found almost exclusively within pulmonary lobules. Their numbers, however, were distinctly less than on day 1. The air sacs of 1 of the 4 H13N6-infected birds were positive, but this was based on the presence of a single positive cell within the surface epithelium. The intestinal tracts of all 4 H2N3-infected birds were now positive. Most or all segments of intestine examined from these birds contained many positive epithelial cells, either isolated or in small groups, near or in the tips of mucosal villi, and often distributed in clusters of several villi along the mucosa. Despite the relative abundance of these cells, there were no accompanying lesions (Fig. 7). As on day 1, these positive epithelial cells were often associated with the presence of bright red precipitate in the adjacent lamina propria. Much less often, positive epithelial cells could also be found in small numbers in the epithelium at the base of villi and, rarely, within mucosal glands. The cloacal bursa of all 3 H2N3-infected birds from which this organ was available had many positive cells confined to the surface epithelium (Fig. 8). The intestinal tract and cloacal bursa of none of the H13N6-infected birds were positive.
On day 6, positive cells had disappeared from the lungs and air sacs of all birds in both infected groups. The intestinal tract of 1 of 4 H2N3-infected birds was positive, but this was based on the presence of a single cluster of 4 positive epithelial cells in the tip of a mucosal villus. The cloacal bursa of 1 of 2 H2N3-infected birds from which this organ was available for examination was positive, based on the presence of a few positive cells scattered within the surface epithelium. As on previous days, the intestinal tract and cloacal bursa of all H13N6-infected birds were negative. None of the tissues from any of the infected birds collected on day 17 was positive.
Discussion
This study provided an opportunity to compare the interaction with the host of 2 subtypes of LPAI virus of markedly different origins in experimentally infected hatch-year Mallards, since the H2 subtype is relatively common among wild waterfowl in North America and has also been isolated from gulls, whereas H13 is the most frequently detected subtype of LPAI virus in gulls but has very rarely been isolated from ducks. 1,11,20,24
Identical experimental protocols were used for both subtypes. However, comparison between results from RRT-PCR and those from virus isolation suggested that the MDCK cell line used for virus isolation was much less sensitive for culture of the H13N6 strain than for that of the H2N3 strain. The sensitivity of this cell line for isolating AI virus is also less than that of culture in embryonated chicken eggs. 12,27 Therefore, results of virus isolation obtained in this study may provide only a partial picture of the distribution of AI virus in the experimental birds.
Both subtypes were able to infect at least some organs of our experimental birds, based on results of RRT-PCR, virus isolation, and IHC. This is interesting, considering that the H13 subtype has very rarely been isolated from wild ducks and that in other studies, recovery of this subtype from experimentally infected ducks has been difficult. 15,20
The lungs and air sacs were permissive for viral infection and replication by the 2 strains used in the present study, as shown by IHC of these tissues on day 1. The respiratory tract of poultry and waterfowl is well recognized as a site of replication of LP and HP subtypes of AI virus in addition to the intestinal tract. 3,21,30,39 –41 As opposed to the H13N6 strain, however, the H2N3 strain also replicated well in the intestinal tract and cloacal bursa of infected birds. The preferred replication sites of the H13N6 subtype in gulls are not well defined. This subtype has been isolated from a combination of pharyngeal and cloacal swabs from various species of gulls, and subtype H13N6 was isolated from a high proportion of lungs collected from free-living 3-week-old Ring-billed Gulls in southern Ontario, Canada. 43 In the present study, virus was detected in the intestinal tract and cloacal bursa of a few H13N6-infected birds on day 1 by RRT-PCR and virus isolation, but not by IHC. The virus could have been present in the intestinal and bursal lumens as a result of ingestion of secretions from the respiratory tract or from the intraesophageal inoculum. Alternatively, it could have been within the epithelium of abdominal air sacs covering the serosal surfaces of these and other organs. 35 These air sacs may be more likely than other air sacs to be infected following intratracheal inoculation since they have a direct communication with the primary bronchi. 16 Based on previous studies, most H13 subtypes tested do not replicate in the intestinal tract of ducks experimentally infected by the oral or intratracheal routes. 5,20 It is not clear whether this failure of the virus to infect the intestinal tract of ducks is a result of its failure to recognize cell receptors in this organ or intolerance to the microenvironment of the proventriculus and gizzard of these birds during its passage through these organs. 5
Infection of the respiratory tract by both viral subtypes was brief and had generally subsided by day 5. Infection of the intestinal tract and cloacal bursa by H2N3 virus started and subsided slightly later than in the respiratory tract, as demonstrated by RRT-PCR, viral isolation, and IHC. A similar pattern was observed by Slemons and Easterday 38 in Pekin ducks experimentally exposed by aerosol to LPAI virus and by Swayne et al 40 in chickens inoculated intranasally with mildly and moderately pathogenic isolates of H5N2 virus. This delay in infection of the intestinal tract and cloacal bursa as compared with that of the respiratory tract may reflect the method of experimental infection used in the present study, which provided a more direct exposure to receptive cells in the lung. It may also reflect the difference in physical spread of the virus in air versus ingesta and secretions from the digestive tract.
In the process of infecting the lungs, both viral subtypes were associated with a locally extensive pneumonia accompanied by proliferation of the epithelial lining of parabronchi that typically affected the lateral region of 1 or both lungs. By IHC, AI virus nucleoprotein was demonstrated in numerous parenchymal cells, interpreted as epithelial cells lining air capillaries, and also in fewer epithelial cells lining parabronchi and secondary bronchi in the areas of pulmonary inflammation. However, we cannot exclude the possibility that a component of the diluted allantoic fluid other than LPAI virus may have contributed to some of the inflammatory response in the lungs. Similarly, the presence of mild pulmonary lesions in 1 of the 2 control birds inoculated only with PBS and euthanized on day 3 suggested a possible effect of sham inoculation.
The nature and distribution of the pulmonary lesions were similar to those described by Cooley et al 7 in experimental oral and intratracheal infection of Pekin ducks with 5 subtypes of AI virus, including virulent and avirulent strains. In particular, the abundance of lymphocytes and the rapidity with which they accumulated (within 24 hours following infection) was remarkable when compared with the inflammatory process in mammals. A similar phenomenon was observed in the wing web of chickens injected with a variety of agents that caused acute inflammation: lymphoid hyperplasia, particularly around small blood vessels, appeared as early as 6 hours after injection and was the most prominent cellular reaction at 36 hours. 4 Pulmonary lesions were also observed in free-living Ring-billed Gulls from which H13N6 virus was isolated, but the association between the presence of these lesions and virus isolation from the lungs was not statistically significant. 43 The reason for the apparent predisposition of the lateral region of the lungs to developing inflammatory lesions in experimentally infected birds in this study and in that of Cooley et al 7 is unclear, although according to Cooley et al, 7 the divergence of the large secondary bronchi from the caudal part of the primary bronchus could explain the preferential involvement of the caudal region of the lung in their study. No inflammation was seen in the air sac walls (more specifically, the partitions between cranial and caudal thoracic air sacs) in association with infected epithelial cells in the present study, perhaps because very few such cells were infected.
Lesions were not identified by Daoust et al 8 in the respiratory tract of wild Mallards naturally infected with LPAI virus, and the respiratory tract is thought to play a limited role in the replication, transmission, and ecology of AI virus in wild birds. 27 Nonetheless, the isolation of LPAI virus from oropharyngeal swabs of naturally infected wild waterfowl suggests the potential for primary infection of the respiratory tract in these birds. 10,30 This, in combination with potential depression of the immune system associated with the stress of migration, 45 could manifest itself as a pneumonia of clinical significance. Stress and, particularly, secondary bacterial infections are recognized as important factors contributing to the exacerbation of clinical signs and lesions associated with natural infection by LPAI virus in poultry. 37,41
Clinical signs were not observed in any of our experimental birds infected by either the H2N3 or H13N6 subtype. However, although both groups showed an increase in body weight before inoculation, this body weight gain decreased significantly during the period of virus shedding after inoculation in the H2N3 group but not in the H13N6 group. This measurable difference in nutritional condition between the 2 groups suggests that the H2N3 infection was more pathogenic for these birds than the H13N6 infection, perhaps due to the higher incidence of viral pneumonia in the H2N3 group than in the H13N6 group, the occurrence of intestinal infection in the H2N3 group but not in the H13N6 group, or both phenomena. Birds in the H2N3 group equipped with an intracoelomic transponder showed a slight (0.4°C) increase in body temperature on day 1, although this increase was not statistically significant. Interestingly, a small (0.5°C) but significant increase in body temperature was observed in Mallards infected orally with another LPAI virus (H7N7), coinciding with the early stage of viral shedding (days 1 and 2), although none of the birds exhibited clinical signs. 18
No lesion was associated with infection of the intestinal tract and cloacal bursa in H2N3-infected birds in this study, despite the relative abundance of infected cells, particularly in the intestinal villous epithelium. The pattern of distribution of infected cells in the intestinal mucosa and bursa mirrored that in wild Mallards naturally infected by LP subtypes of AI virus, including the presence of positive cells and particulate material in the intestinal lamina propria, suggesting participation by immunomodulating cells. 8 Viral shedding decreased markedly after 10 days, based on RRT-PCR results from pharyngeal and cloacal swabs; this corresponds to the period of shedding generally observed in other experimental infections. 14,22,38
The marked contrast in the occurrence of inflammation among the infected organs in H2N3-infected birds is notable. On the basis of the results of IHC, the pulmonary parenchyma, intestinal mucosal epithelium, and cloacal bursal epithelium all contained many infected cells at some point in the course of infection. Yet, inflammation was evident only in the lungs. Whereas the anatomical integrity of the intestinal and bursal mucosal epithelium could be confirmed microscopically, cellular details were insufficient to verify this in the pulmonary parenchyma. Breach of the epithelial barrier would be expected to induce an immune response mediated by the underlying dendritic cells. 17 Alternatively, in contrast to the epithelial surfaces of the airways and pulmonary parenchyma that are sterile, the intestinal and bursal mucosal surfaces are constantly exposed to microbial antigens and are therefore more likely to ignore antigens from microorganisms that are not perceived as an immediate threat to the integrity of the epithelial barrier. 6,17
Low levels of infections by both subtypes of LPAI virus were identified by RRT-PCR, but not by IHC, in various internal organs (proventriculus, liver, and/or kidney) from several experimental birds in the early stage of the experimental infection. Similarly, H13N6 virus was isolated from kidneys of naturally infected Ring-billed Gulls. 43 As previously suggested, the presence of the virus in the proventriculus could have resulted from ingestion of secretions from the respiratory tract or from the intraesophageal inoculum, and its detection in the kidneys could have been more specifically related to infection of the epithelium of abdominal air sacs covering this organ. Rarely, however, LP subtypes of AI virus have been found to infect tissues of chickens other than those of the respiratory and intestinal tracts. 23,39
Significantly larger quantities of virus were found by both RRT-PCR and virus isolation in cloacal swabs than in pharyngeal swabs of birds in the H2N3 group. Moreover, based on virus isolation from cloacal and pharyngeal swabs, the duration of viral excretion was longer from the cloaca than from the oral cavity. The results of virus isolation, in particular, would indicate a higher contamination of the environment by viable viral particles from the cloaca than from the oral cavity, a conclusion also reached by Hénaux and Samuel. 14 Many more influenza viruses have been isolated from cloacal samples than from the respiratory tract in wild ducks. 22 However, some subtypes are more likely to be isolated from oropharyngeal swabs than from cloacal swabs. 3,10,21,41 Although an indirect fecal-oral route involving ingestion of contaminated water is recognized as an important mode of transmission for AI viruses among wild waterfowl, 13 the relative abundance of cells positive for the virus in the pulmonary parenchyma by IHC in H2N3-infected birds in the present study suggests that transmission by aerosol remains a plausible alternative route of exposure to the virus.
There was good evidence by RRT-PCR, virus isolation, and IHC that the H2N3 subtype replicated better and was excreted in larger amounts in our experimental birds than the H13N6 subtype. Although the H13N6 subtype may be able to replicate in the lungs of Mallards, the relatively low amount of virus released into the environment by an infected bird would likely prevent it from being maintained within a population of free-living birds, which supports the observation that the H13 subtype has very rarely been isolated from ducks. 1,24
In conclusion, we confirm that the H2N3 virus is better adapted to Mallards than the H13N6 virus but that the latter virus is nonetheless able to cause a productive pulmonary infection and inflammation in Mallards. We also show that the H2N3 virus can cause extensive productive infection of epithelial cells of the intestinal mucosa and cloacal bursa without eliciting inflammation, in contrast to its effect on the pulmonary parenchyma. This presumably reflects the long co-evolution of the virus and its host in these particular organs, and it supports the importance of the fecal-oral route as a mode of transmission of LPAI virus among wild waterfowl. Finally, the significant decrease in body weight gain in Mallards associated with H2N3 virus–associated pneumonia and intestinal tract infection suggests the potential for negative effects on the health of wild birds infected by LP subtypes of AI virus.
Footnotes
Acknowledgements
We thank Dr John Pasick, Canadian Food Inspection Agency, Winnipeg, Manitoba, Canada, for supplying subtype H13N6 of AI virus and Leslie Reperant and Lonneke Leijten, from the Erasmus Medical Centre, Rotterdam, the Netherlands, for their assistance with laboratory procedures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
