Abstract
Domestic cats are susceptible to infection with highly pathogenic avian influenza virus H5N1, resulting in pneumonia and in some cases, systemic spread with lesions in multiple organ systems. Recent transmission of the 2009 pandemic H1N1 influenza virus from humans to cats also resulted in severe pneumonia in cats. Data regarding the susceptibility of cats to other influenza viruses is minimal, especially regarding susceptibility to low pathogenic avian influenza viruses from wild birds, the reservoir host. In this study, the authors infected 5-month-old cats using 2 different North American shorebird avian influenza viruses (H1N9 and H6N4 subtypes), 3 cats per virus, with the goal of expanding the understanding of avian influenza virus infections in this species. These viruses replicated in inoculated cats based on virus isolation from the pharynx in 2 cats, virus isolation from the lung of 1 cat, and antigen presence in the lung via immunohistochemistry in 2 cats. There was also seroconversion and lesions of patchy bronchointerstitial pneumonia in all of the cats. Infection in the cats did not result in clinical disease and led to variable pharyngeal viral shedding with only 1 of the viruses; virus was localized in the alveolar epithelium via immunohistochemistry. These findings demonstrate the capacity of wild bird influenza viruses to infect cats, and further investigation is warranted into the pathogenesis of these viruses in cats from both a veterinary medical and public health perspective.
Although previously thought resistant to disease due to influenza virus infection, the domestic cat has proven to be naturally susceptible to infection resulting in disease with not only highly pathogenic avian influenza virus H5N1 (HPAI H5N1) but also 2009 pandemic H1N1 influenza viruses. Transmission of HPAI H5N1 virus to domestic and wild felids has been demonstrated naturally and experimentally, resulting in a range of clinical disease—from systemic infection with systemic lesions and death to only subclinical or asymptomatic infection with seroconversion.2,6–9,15,16 In natural and experimental cases of HPAI H5N1 virus in cats that resulted in death, lesions include severe bronchointerstitial pneumonia, with bronchiolar necrosis and alveoli filled with edema, hemorrhage, and necrotic cells and some cases with hyaline membranes in alveoli and type II pneumocyte hyperplasia.7,8,15 Additionally, systemic lesions of hepatic necrosis, adrenocortical necrosis, lymphoid necrosis, encephalitis, myocardial necrosis, necrosis of enteric nervous plexus, and tubulonephritis with intralesional localization of influenza virus antigen via immunohistochemistry have been reported.7,15,16
Infection with influenza virus in domestic felids is a zoonotic concern given their close relationship with humans; domestic cats have also recently been shown to develop severe disease from 2009 pandemic H1N1 influenza virus.10,17,19 Cats naturally infected with the 2009 pandemic H1N1 influenza virus had severe clinical disease in some cases, with severe pneumonia resulting in dyspnea; however, experimentally infected cats had only mild to minimal signs of disease (lethargy, anorexia) despite significant pulmonary lesions.10,19 Infection results in a bronchointerstitial pneumonia, characterized by bronchiolar necrosis with intraluminal fibrin, macrophages and edema, alveolar hyaline membranes, sloughed pneumocytes, type II pneumocyte hyperplasia, and macrophages filling alveoli, but relatively little inflammation in the bronchi.10,19 There are no reports of systemic lesions in cats infected with 2009 pandemic H1N1 virus. Cat to cat transmission of HPAI H5N1 virus has been demonstrated in zoo felids and experimental infections of domestic cats, as well as with experimental infection of cats with 2009 pandemic H1N1 virus.8,15,18,19
Few studies have investigated the capacity of the vast number of influenza virus isolates to infect cats, especially those from wild birds, a reservoir host for avian influenza viruses (AIVs). Limited earlier studies have experimentally infected cats with a single low pathogenic avian influenza virus (LPAIV) from poultry as well as a few human influenza viruses and showed shedding in cats in absence of disease.5,14 Recent serosurvey data have suggested that domestic cats are not only susceptible to 2009 pandemic H1N1 influenza virus but also other circulating seasonal human influenza viruses. 12 Given cats’ susceptibility to both HPAI H5N1 virus and 2009 pandemic H1N1 influenza virus, further investigation into susceptibility and potential disease from wild bird influenza virus infections in cats is warranted. In this study, we implement an experimental cat infection using 2 different LPAIVs (H1N9 and H6N4 subtypes) that were isolated from North American shorebirds with the goal of deepening our understanding of AIV infections in cats and assessing the potential for this domestic species to act as a vector for these influenza virus isolates. Additionally, our study provides full examination of not only shedding and seroconversion, but also shows development of microscopic lesions with localization of antigen via immunohistochemistry, which has not been performed with LPAIV in cats. The 2 isolates used in this study were selected from a screen of 28 wild bird AIV isolates in BALB/c mice; the selected isolates exhibited robust pulmonary replication in the BALB/c mouse model in our lab. 4 Our results show that domestic cats are susceptible to infection with these shorebird AIVs, leading to infections that are asymptomatic despite development of bronchointerstitial pulmonary lesions and pharyngeal shedding in the case of one of the isolates. Our findings further support that cats are susceptible to a variety of influenza viruses and that these viruses readily replicate deep in the respiratory tract. Additionally, cats could not only serve as an intermediate route of transmission of avian influenza viruses to humans, but also may provide a novel opportunity for avian influenza virus reassortment.
Materials and Methods
Viruses
Avian influenza viruses used in this study originated from ruddy turnstones (Arenaria interpres) and were originally isolated from cloacal swabs in 9-day-old embryonated chicken eggs (ECE) incubated at 37°C for 72 hours. Viruses were minimally passaged (3 or fewer passages) in ECEs. A PFU titer was established for each virus via plaquing isolates on Madin-Darby canine kidney cells with a 1.2% Avicel overlay (FMC biopolymer, Philadelphia, PA), incubating for 72 hours at 37ºC, 5% CO2, then removing the overlay, fixing cells and visualizing plaques with crystal violet. Select isolates were screened in a previous study in BALB/c mice. 4 Two isolates that exhibited efficient pulmonary replication and induced pulmonary lesions in BALB/c mice were selected for in vivo studies in cats. Isolates included A/Ruddy Turnstone/DE/650625/02 (H6N4) (abbreviated RT/625) and A/Ruddy Turnstone/DE/650645/02 (H1N9) (abbreviated RT/645).
Cat Infections
The study included 6 5-month-old domestic cats (Liberty Research, Inc, Waverly, NY); all were seronegative to circulating H1N1 and H3N2 influenza viruses as determined by hemagglutination inhibition assay. Sera samples collected from each cat prior to infection were treated with receptor-destroying enzyme (RDE) and tested for serum antibodies with a hemagglutination inhibition assay using 0.5% chicken red blood cells as previously described. 21 Viruses were diluted to contain 4 agglutinating units in sterile PBS. Additionally, pre and post inoculation sera were tested for serum antibodies by microneutralization assay using experimental viruses RT/625 and RT/645 as previously described, starting at a 1:20 dilution. 1 All cats had a pre inoculation sera microneutralization titer of less than 1:20 against these experimental LPAIVs. Cats were housed in open caging (Allentown, Allentown, NJ) in ABSL-3 facilities. A subcutaneous temperature transponder (BMDS) was implanted in each cat for identification and temperature measurement. Cats were observed twice daily by animal care staff and once daily by the veterinarian conducting the study (EA Driskell). Disease was not expected in inoculated cats; therefore, no clinical scoring system was applied to the study. Studies were conducted under guidelines approved by the Institutional Animal Care and Use Committee of the University of Georgia and the Centers for Disease Control and Prevention.
For each of the viruses used, 3 cats were anesthetized with acepromazine (Boehringer Ingelheim Vetmedica, St. Joseph, MO), butorphanol (Fort Dodge Animal Health, Fort Dodge, IA), and dexmedetomidine (Pfizer Animal Health, Monmouth Junction, NJ) and intratracheally inoculated with 106 PFU of virus in allantoic fluid diluted in 500 µL of sterile PBS. Temperature, weights, complete blood counts, and nasal, pharyngeal, and rectal swabs were sampled on days 0, 1, 3, 5, and 7 post inoculation (pi) under sedation with acepromazine and butorphanol. On day 7 pi, all cats were sedated and euthanized. A necropsy was performed with gross examination of thoracic and abdominal viscera, brain, and nasal turbinates. Lung and trachea were sampled and frozen at –80°C for virus isolation. Sections of cranial lung lobes, caudal lung lobes, trachea, tracheobronchial lymph nodes, heart, thymus, spleen, liver, kidney, stomach, small intestine, colon, pancreas, mesenteric lymph nodes, adrenal gland, nasal turbinates, and brain were fixed in 10% neutral buffered formalin for histopathology and immunohistochemistry. One section from each of the following lung lobes were examined on each cat: cranial left lobe, cranial right lobe, caudal left lobe, caudal right lobe, and accessory lobe. Virus isolation was performed for all swabs and fresh lung and trachea. Swabs (in 2 mL PBS) and tissues were frozen at –80°C for later virus isolation. Tissues were then homogenized in 1 mL PBS, clarified by centrifugation, and samples from clarified tissue homogenate and PBS from swabs were inoculated into 9-day-old ECEs (4 ECEs per sample) and incubated 72 hours for virus isolation. Complete blood counts were performed using Vet Scan analyzer (Abaxis, Union City, CA).
Histopathology and Immunohistochemistry
All tissues collected were routinely processed for light microscopy. Briefly, sections of formalin-fixed, paraffin-embedded tissues were deparaffinized and stained with HE or processed for immunohistochemistry. Immunohistochemistry was performed on lung, trachea, and tracheobronchial lymph node for all cats. Immunohistochemistry was performed on an autostainer (Dako Cytomation, Carpinteria, CA) with DAB chromagen (Dako Cytomation) or Vulcan fast red chromagen (Biocare Medical, Concord, CA). Tissues were blocked with Dako Biotin Blocking System (Dako Cytomation). To detect viral antigen, a goat polyclonal antibody to the nucleoprotein of influenza A virus (Biodesign International, Sako, Maine; diluted 1:10,000) and rabbit biotinylated anti-goat IgG as a secondary antibody (Vector Laboratories, Burlingame, CA) were used. For double labeling, a mouse monoclonal antibody for cytokeratins AE1/AE3 (Biogenex, San Ramon, CA; diluted 1:200) with Biogenex Multilink kit with HRP label (Biogenex) and a mouse monoclonal antibody to the nucleoprotein of influenza A (Biodesign International; diluted 1:1000) with Biogenex Multilink kit with alkaline phosphatase label (Biogenex) were used. Hematoxylin counterstain was used for all immunohistochemistry. Sections of formalin-fixed, paraffin-embedded heart from a chicken infected with H5N1 influenza virus incubated with the goat polyclonal or mouse monoclonal primary antibodies for influenza virus antigen served as a positive control. The same tissue (chicken heart) was used as a negative control for the single labeling for influenza A by replacing the primary antibody with isotype-matched control antibody. For the double labeling immunohistochemistry, lung sections from a domestic cat that died due to trauma were treated with the same protocol as double-labeled experimental tissues served as negative controls.
Statistical Analysis
Leukocyte counts were log transformed and statistically analyzed using repeated measures ANOVA (Stata version 11.0) to examine differences between viral groups and days pi. Degrees of freedom for F tests of repeated measures factors were adjusted using the Greenhouse-Geisser estimate of epsilon to correct for any departures from the sphericity assumption. All testing assumed a two-sided alternative hypothesis, and P values < 0.05 were considered significant.
Results
Clinical
Cats inoculated with either RT/625 or RT/645 had no clinical evidence of disease; all cats remained alert and playful with minimal weight change. For RT/625 inoculated cats, no weight loss occurred post-inoculation compared to pre-inoculation weights, and for RT/645 there was an average of 3% weight loss on day 1 pi, but an average weight gain for days 3 through 7 pi. There was no significant group, day, or group by day interaction effect on counts of total leukocytes, lymphocytes, monocytes, or granulocytes (Table 1).
Total Leukocyte Counts in Cats Inoculated With RT/625 (H6N4) or RT/645 (H1N9) Avian Influenza Viruses.
aReference ranges from Schalm’s Veterinary Hematology, 6th ed. for 16- to 17-week-old kittens.
Virology
Despite absence of clinical disease, all cats seroconverted to either RT/625 or RT/645. For RT/645, virus was not isolated from either antemortem swabs or postmortem tissues, but all cats seroconverted by day 7 pi determined by microneutralization assay (Table 2). For RT/625, all cats seroconverted and virus was isolated from the lung of 1 of 3 cats on day 7 pi. Additionally, 2 out of 3 cats inoculated with RT/625 had evidence of pharyngeal shedding based on positive virus isolation from pharyngeal swabs taken on days 3 and 5 pi for one cat and on days 3, 5, and 7 pi for another cat (Table 2). Virus was not isolated from the tracheal tissue or nasal or rectal swabs for RT/645- or RT/625-inoculated cats.
Viral Infection and Shedding in Cats Inoculated With RT/625 (H6N4) or RT/645 (H1N9) Avian Influenza Viruses.

aVirus detected by virus isolation in embryonated chicken eggs.
bPercent lung affected with inflammation estimated from equivalent microscopic sections between cats for both cranial and caudal lung lobes.
cMicroneutralization titer.
Histopathology and Immunohistochemistry
No gross lesions were observed. Microscopic lesions were primarily restricted to the lung in both RT/625- and RT/645-inoculated cats and were usually more extensive and severe in the cranial lung lobes, possibly a result of the inoculation method. Most cats had only a small percentage of lung involvement microscopically, although up to 50% pulmonary involvement of examined sections was present in a few of the cats (Table 2). In the lung on day 7 pi, bronchioles were filled by primarily macrophages and few neutrophils, often with adjacent alveoli filled with macrophages (Figs. 1, 3, 4). There was bronchiolar epithelial regeneration as affected bronchioles were lined by large, plump epithelial cells (Fig. 1). Frequently, vessels were surrounded by thick cuffs of lymphocytes (Fig. 2). Clear evidence of alveolar involvement was present, with prominent, extensive type II pneumocyte hyperplasia of alveolar epithelium (Figs. 3, 4). No lesions were observed in the intrapulmonary bronchial or bronchiolar glands.
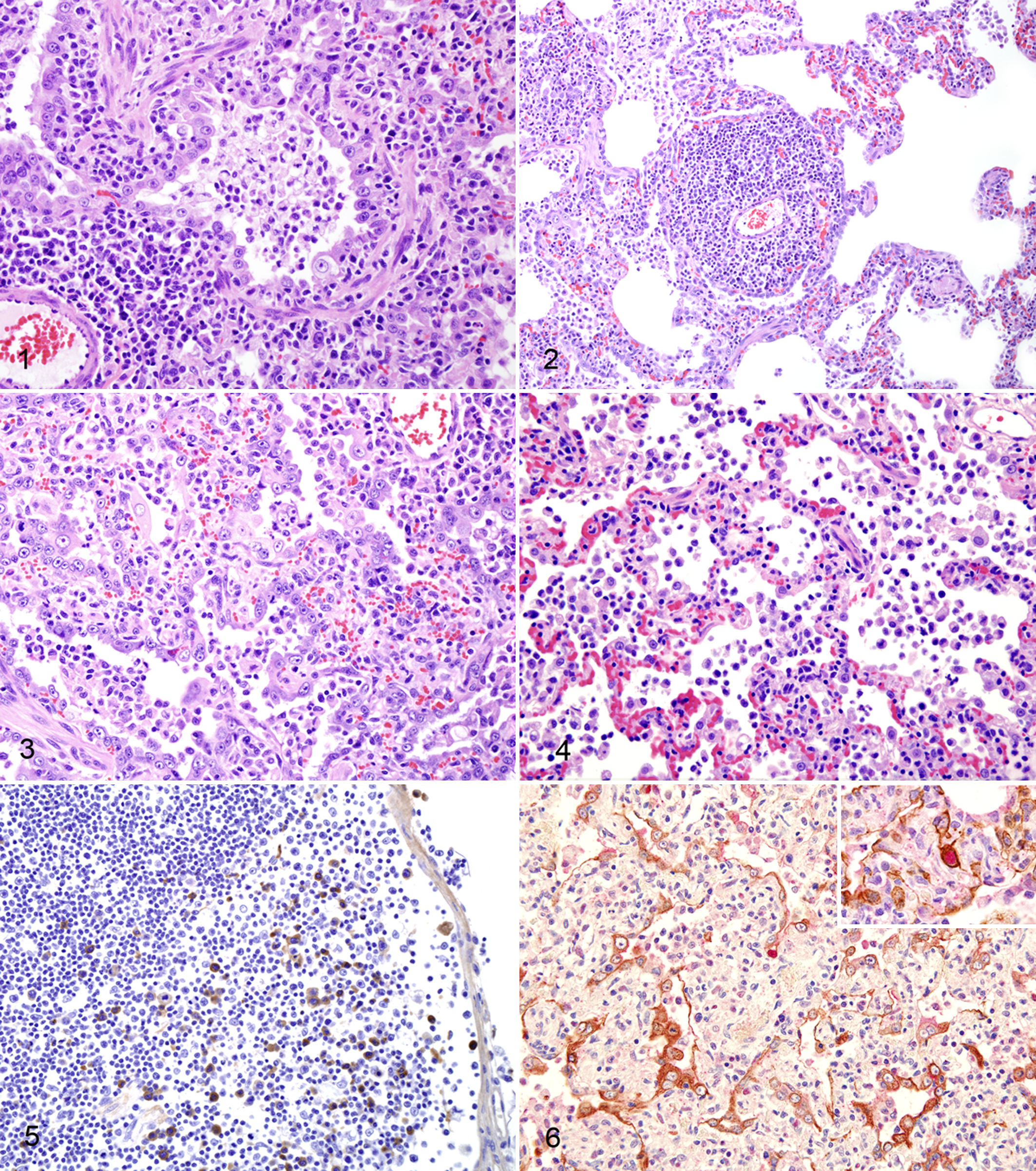
There was sinus histiocytosis present in the tracheobronchial lymph nodes of all inoculated cats. All RT/625- and RT/645-inoculated cats had strong intracytoplasmic immunoreactivity in pulmonary and tracheobronchial lymph node macrophages for the nucleoprotein of influenza A virus (NP) using immunohistochemistry, suggestive of phagocytosis of influenza antigen (Fig. 5). There were also rare alveolar cells with strong intranuclear and some intracytoplasmic immunoreactivity for the NP of influenza for both RT/625- and RT/645-inoculated cats (Table 2). These cells were confirmed as pneumocytes with double staining for the NP of influenza virus A and cytokeratin AE1/AE3 (Fig. 6). The morphology of these pneumocytes were flat, suggesting they were type I pneumocytes. No immunoreactivity for the NP of influenza was observed in type II pneumocytes.
Discussion
Natural outbreaks and experimental infections have demonstrated cats’ susceptibility to more virulent avian influenza viruses; in this experiment, we have expanded that range to include some circulating wild bird avian influenza viruses not previously thought to infect mammals. This further supports the growing evidence that cats are more susceptible to influenza virus infection than previously thought. In marked contrast to HPAI H5N1 virus and the 2009 pandemic H1N1 influenza virus infection in cats, cats experimentally infected with shorebird avian influenza viruses demonstrated no clinical signs or statistically significant differences in leukocyte counts, despite presence of pulmonary lesions. Though the lesions were marked in some areas of the lung on microscopic examination, there were grossly no lesions and the extent of the lesions was limited mostly to the cranial lung lobes. The absence of observable gross lesions, despite a high estimated percentage of affected area of some cranial lung lobe sections microscopically, was most likely due to the fact that the lesion was resolving when examined on day 7 pi. Microscopic changes observed at this point, including type II pneumocyte hyperplasia and only macrophages in air spaces, most likely would not manifest in obvious color or texture change as would acute or active changes that include edema, exudate, or hemorrhage. The overall percentage of lung affected as well as host response to the virus may both be factors contributing to the absence of clinical signs in infected cats. The factors of pathogenicity of lethal influenza virus cases in cats remains to be determined, as it is clear that some influenza viruses can replicate in the cat lung and cause lesions yet not result in clinically detectable disease.
We chose to perform postmortem examination of cats on day 7 pi so viral shedding could be monitored. Additionally, this day was selected because cats infected with HPAI H5N1 virus have abundant pulmonary virus, pulmonary and systemic lesions indicative of viral damage, and peak viral shedding present at day 7 pi.8,15 However, other studies examining 2009 pandemic H1N1 influenza virus and subclinical cases of HPAI H5N1 virus in cats have shown viral infection and shedding waning by day 7 pi.9,19 We have examined both of these shorebird viruses in ferrets, and virus is cleared from the lung by day 7 pi. 3 Therefore, it is probable that the virus was nearly cleared in most of the cats by day 7 pi, as there is evidence of both viruses infecting the lung based on seroconversion and presence of microscopic lesions despite negative virus isolation from lung and paucity of positive immunoreactivity for the virus in the lung at this timepoint. Indeed, an experimental study of 2009 pandemic H1N1 influenza virus in cats demonstrated that 4 out of 4 cats had positive virus isolation from the lung and trachea day 4 pi, but only 1 out of 4 cats had positive virus isolation of the lung and trachea by day 7 pi. 19 Additionally, influenza antigen expression via immunohistochemistry was more prominent day 4 pi compared to day 7 pi in this study. 19
The function of the HA as a ligand for the host cell receptor seems to be a key factor for host range restriction due to a difference in receptor preference of AIVs versus human influenza viruses. Avian influenza virus strains bind to cell glycoproteins/glycolipids that have terminal sialyl-galactosyl residues with a 2-3 linkage [Neu5Ac(α2-3)Gal] and human influenza virus strains bind to terminal 2-6 linked glycoproteins/glycolipids [Neu5Ac(α2-6)Gal]. 11 Distribution of these receptors throughout the respiratory tract of different species is thought to be one factor in host range restriction, transmissibility, and virulence. 13 Experimental examination of viral attachment patterns of influenza virus in the respiratory tract of cats shows that seasonal human influenza viruses do not bind in the upper or lower respiratory tract of domestic cats. 20 In contrast, avian influenza viruses, including HPAI H5N1 virus, have a similar attachment pattern in the respiratory tracts of domestic cats and humans, with viral attachment more prominent in the lower respiratory tract to type II pneumocytes. 20 Additionally, these viruses have been demonstrated to bind non-ciliated bronchiolar epithelial cells in cats. 20 The attachment pattern described is similar to the findings in this study, with microscopic lesions and antigen location that are bronchiolar-centric with primarily alveolar involvement in the domestic cats infected with these wild bird AIVs. The described attachment pattern also fits with the observation that cats have historically been resistant to infection with human influenza viruses but are susceptible to HPAI H5N1 virus.
Other studies have demonstrated the capability of influenza virus to infect bronchiolar epithelium and alveolar epithelium in cats when there is opportunity for exposure deep in the respiratory tract.6,8,15,19 We have demonstrated that wild bird influenza viruses have a similar capacity to infect deep in the lung of cats, hemagglutinin subtype notwithstanding, with limited pharyngeal viral shedding. Domestic cats hunting and eating wild birds may be exposed to AIVs, though productive infection and shedding would likely depend on the isolate and dose of virus upon exposure. Additionally, the LPAIV reservoirs are birds in the order of Anseriformes and Charadriiformes, and domestic cats would more likely be hunting Passeriformes. Nonetheless, the capacity for cats to be infected with the viruses examined in this experiment further supports the ability of wild bird influenza viruses to infect cats without adaptation in domestic birds or other mammals; such events could lead to adaptation to domestic cats or reassortment in this species. Further investigation of the susceptibility of domestic cats to human and avian influenza viruses and elucidation of the pathogenesis of these different isolates in cats is warranted not only from a veterinary medical perspective but also from a public health point of view.
Footnotes
Acknowledgements
We wish to thank the Animal Resources personnel at the College of Veterinary Medicine, University of Georgia for excellent animal husbandry. We would also like to thank histotechnicians in the Department of Pathology at University of Georgia for their assistance, especially Abbie Butler for her outstanding immunohistochemistry support. Additional thanks go to Monique Silva de Franca and Jessica Mobley for superior assistance with animal work at the College of Veterinary Medicine, University of Georgia. We also thank Xiyan Xu and Alexander Klimov and the CDC (Atlanta, GA) for providing circulating influenza viruses as antigen sources to confirm seronegativity of study cats.
Declaration of Conflicting Interests
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by CDC grant 5U19CI00040102 and NIH contract HHSN266200700006C.
