Abstract
Highly pathogenic H5N1 avian influenza viruses were isolated in 9 large-billed crows that died in Kyoto and Osaka prefectures in Japan from March to April in 2004. We studied 3 of the 9 crows using standard histologic methods, immunohistochemistry, and virus isolation. The most prominent lesions were gross patchy areas of reddish discoloration in the pancreas. The consistent histologic lesions included severe multifocal necrotizing pancreatitis, focal degeneration and necrosis of neuron and glial cells in the central nervous system, and focal degeneration of cardiac myocytes. All of these tissues contained immunohistochemically positive influenza viral antigens. The virus was isolated from the brain, lung, heart, liver, spleen, and kidney of the crows examined. Thus we concluded that highly pathogenic avian influenza virus was associated with clinical disease, severe pathologic changes, and death in the 3 crows.
Keywords
Highly pathogenic avian influenza (HPAI) is caused by infection with influenza A viruses of the family Orthomyxoviridae. All 16 hemagglutinin (H1–H16) and 9 neuraminidase (N1–N9) influenza A subtypes have been isolated from avian species,
5,
21
but HPAI viruses have been restricted to subtypes H5 and H7. It appears that HPAI viruses emerge only after the low pathogenic viruses have crossed from aquatic wild birds to terrestrial poultry.
1
However, the first reported isolation of an avian influenza virus from wild birds was the HPAI H5N3 subtype virus obtained in 1961 from common terns (
Regarding the influenza A virus infection in the
In this report, we describe the pathologic changes and viral tropism of the HPAI virus infection that produced fatal disease in native large-billed crows that were closely associated with the outbreak of HPAI in a layer chicken farm in Kyoto, Japan.
Materials and Methods
Case history
From the end of December 2003 to March 2004, outbreaks of HPAI occurred in the western part of Japan, involving 3 chicken farms and a group of chickens raised as pets. The H5N1 influenza A viruses isolated from each outbreak were genetically closely related and highly pathogenic to chickens. 14
The third outbreak of HPAI occurred in a layer chicken farm in Kyoto prefecture in the middle of February (Fig. 1) At this farm before control measures against HPAI were started, the carcasses of chickens were thrown into the compost facility by farm workers. It was found that numerous crows gathered at the compost place, and some of them ate the carcasses of chickens, although the exact number of crows that had been in contact with the virus-infected chickens was unknown. From the end of February to the end of June, 2,355 wild birds, including 307 crows, found dead in Kyoto prefecture were examined with an influenza A virus antigen detection kit or virus isolation using tracheal swabs by the Animal Hygiene Service Center in Kyoto prefecture. HPAI viruses (H5N1) were isolated from 7 large-billed crows between March and April. Other wild birds tested negative. HPAI viruses (H5N1) were also isolated from 2 large-billed crows found dead or prostrate in Osaka prefecture in March. All 9 virus-positive crows were obtained within 30 km of the infected chicken farm (Fig. 1). A/crow/Kyoto/53/2004 and A/crow/Osaka/102/2004 isolated from tracheal samples were highly pathogenic to chickens and genetically closely related to other Japanese isolates of chicken origins. 14
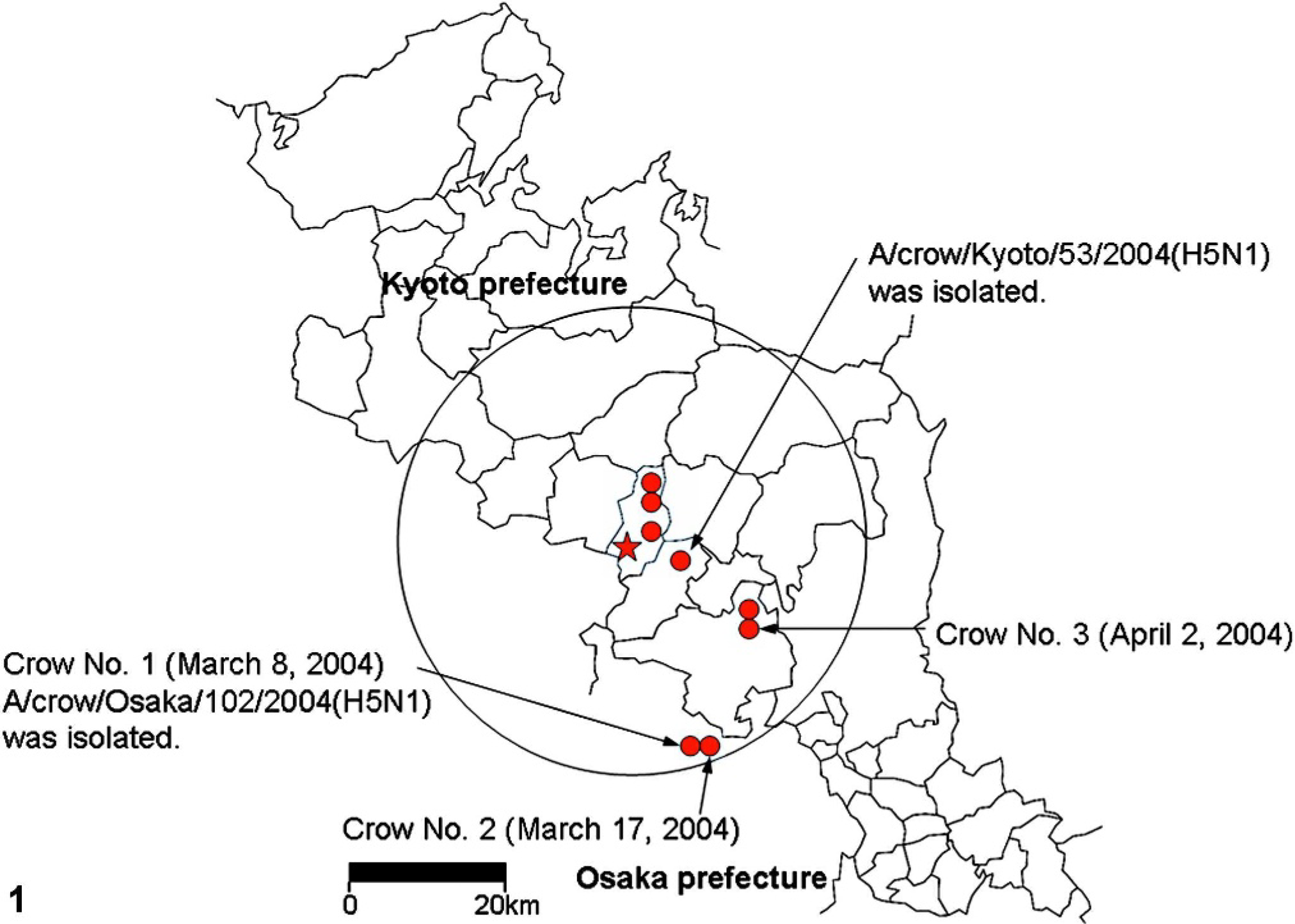
Map of Kyoto prefecture showing the location of the crows found dead or prostrate infected with HPAI H5N1 virus from March to April 2004. ★ = The layer chicken farm in which the outbreak of HPAI occurred in February 2004. The crow found dead at the farm on 3 March 2004 was the first case in which the HPAI H5N1 virus was isolated. • = HPAI H5N1 viruses were isolated from the crows found dead or prostrate at these sites. All 9 crows positive for the HPAI virus were found within 30 km of the infected farm (circle of black line).
Histopathology
Pathologic examinations were performed on the 3 crows from which HPAI H5N1 viruses were isolated (crow Nos. 1–3; See Fig. 1). Crow Nos. 1 and 2 were sampled on 8 March and 17 March 2004, respectively, in Osaka prefecture. Crow No. 3 was sampled on 2 April 2004 in Kyoto prefecture. Tissue samples from the brain, lung, heart, pancreas, liver, kidney, spleen, proventriculus, gizzard, duodenum, adrenal gland, and testis were fixed in 10% neutral phosphate buffered formalin (NBF), routinely processed, and stained with hematoxylin and eosin (HE) for histopathologic examination. The vertebral column, including cervical, thoracic, and lumbar vertebrae, of crow No. 3 was decalcified with 10% formic acid in 10% NBF for 24 hours. These tissues were cut transversely, fixed again in 10% NBF, and processed as above to examine the spinal cord.
The virus-negative crows found dead in Kyoto prefecture from February to June 2004 were not histopathologically examined. Thus, 3 crows (crow Nos. 4–6) that had been protected from emaciation by a wild animal shelter in Chiba prefecture were used as controls for the histopathologic changes. Crow No. 4, a large-billed crow, was protected on 28 May 2002 with severe ocular trauma and died on 19 November 2002 after severe emaciation. Crow No. 5, a carrion crow, was protected on 18 March 2004 with ananastasia and died on 15 April 2004. Crow No. 6, a large-billed crow, was protected on 29 April 2004 with comminuted fracture of left humerus and died on 14 May 2004. No HPAI occurred in Chiba prefecture in 2002 or 2004. Virus isolation was not performed on crow Nos. 4–6. Tissues were fixed in 10% NBF and processed as described above.
Immunohistochemistry
A duplicate section mounted on silane-coated glass slide was heated by autoclave at 121°C for 5 minutes in 1 mM ethylenediaminetetra-acetic acid solution (pH 8.0) 16 and applied with monoclonal antibody specific for type A influenza virus matrix protein (Clone number: GA2B; Isotype: IgG1; Serotec Ltd., Oxford, UK). The primary antibody was allowed to incubate for 30 minutes at room temperature and then detected by the application of horseradish peroxidase anti-mouse Ig conjugate (Histofine, simple stain; Nichirei Inc., Tokyo, Japan). Diaminobenzidine (DAB) and aminoethylcarbazole (Nichirei Inc.) served as the substrate chromogen, and hematoxylin was used as a counterstain. Sections of tissues from normal and influenza virus-infected chickens 14 served as negative and positive controls, respectively. In addition, a monoclonal mouse antibody directed against West Nile/Kunjin virus (Cone number: 3.67G; Isotype: IgG1; Chemicon International, Inc., Temecula California) and phosphate-buffered saline (PBS) were used as substitutes for the primary antibody and served as isotype and omission controls, respectively. Neither of them showed positive reaction to any tissues from crow Nos. 1–6.
Virology
After necropsy, tissues from the brain, lung, heart, liver, spleen, and kidney of crow Nos. 1 and 2 were frozen, stored at −80°C, and assessed for levels of infectious virus. Tissue homogenate (10% w/v) in PBS containing antibiotics was prepared. Serial tenfold dilution of suspensions was inoculated in 0.1-ml amounts onto chicken kidney cell monolayers, 4 per dilution, and observed daily for cytopathic effects for 5 days. The homogenates were also inoculated into the allantoic cavity of 10-day-old embryonated eggs. Inoculated eggs were incubated for 5 days at 37°C, and the allantoic fluids were tested for hemagglutination activity.
Results
Clinical findings
Crow No. 1 showed weakness when captured by a citizen and died 2 days after capture, but no specific neurologic sign was recognized. The other 2 crows (crow Nos. 2 and 3) were found dead.
Gross findings
Before necropsy, no gross lesion was noted in the skin of the 3 crows. The most prominent lesions were observed in the pancreas of crow Nos. 1 and 2, which showed multifocal reddish discoloration on their surface (Fig. 2). The pancreas of crow No. 3 had severe postmortem autolysis. The lungs showed moderate (crow Nos. 1 and 3) and severe (crow No. 2) diffuse congestion. Crow No. 3 had a moderate splenomegaly. No significant gross lesions were noted in other organs, including brains. Crow Nos. 1 and 3 were male; the sex of crow No. 2 was not identified.
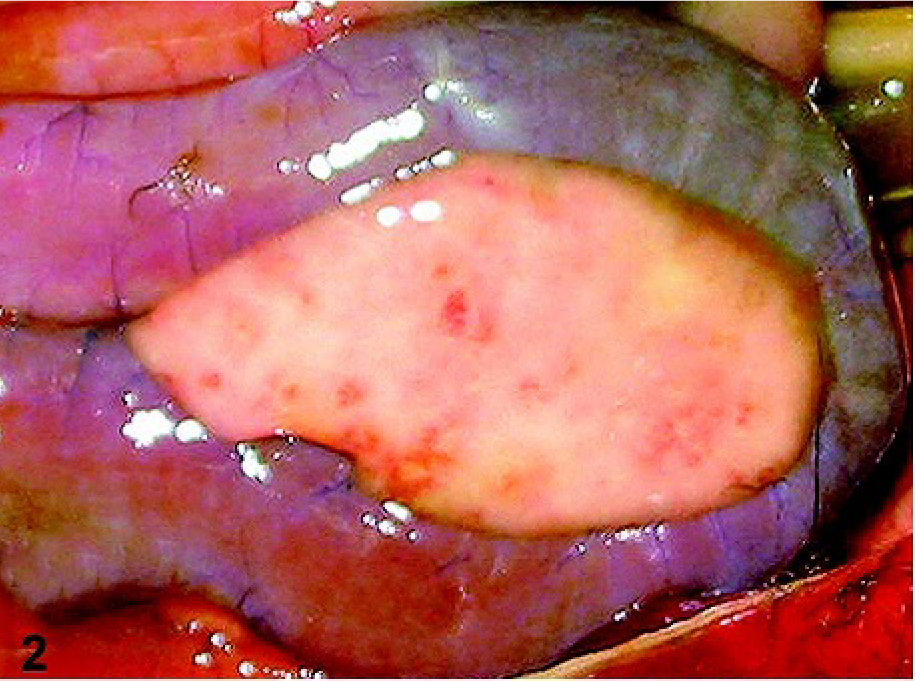
Pancreas; crow No. 1. Multifocal reddish discoloration is present in the pancreas.
Histologic findings
Severe multifocal to coalescing necrotizing pancreatitis was present in the pancreas of crow Nos. 1 (Fig. 3) and 2 (Table 1). The coagulative necrotic foci contained granular or homogeneous eosinophilic nuclei and granular eosinophilic cytoplasm of exocrine cells and had mild to moderate infiltration of heterophils. The interlobular ducts of the pancreas of crow No. 1 had necrotic epithelial cells with karyorrhexis and fragmented cytoplasm. No significant change was detected in the endocrine cells of the pancreatic islets from crow Nos. 1 and 2. There was congestion in the blood capillaries and small veins in the pancreas of crow Nos. 1 and 2, and a slight hemorrhage was present in the necrotic area. Some neurons in the interlobular connective tissues of the crow No. 2 pancreas had karyorrhexis and karyolysis. Other neurons had swollen nuclei filled with eosinophilic granular materials. Immunohistochemically (Table 2), a large number of acinar epithelial cells and interlobular ductal epithelial cells contained viral antigens (Fig. 4). Not only in the pancreas but also in the other organs, immunolabeling showed that the nuclei were stained more intensely than the cytoplasm and the viral antigen–positive materials were mostly granular.
Histopathologic changes in large-billed crows infected with HPAI H5N1 virus.
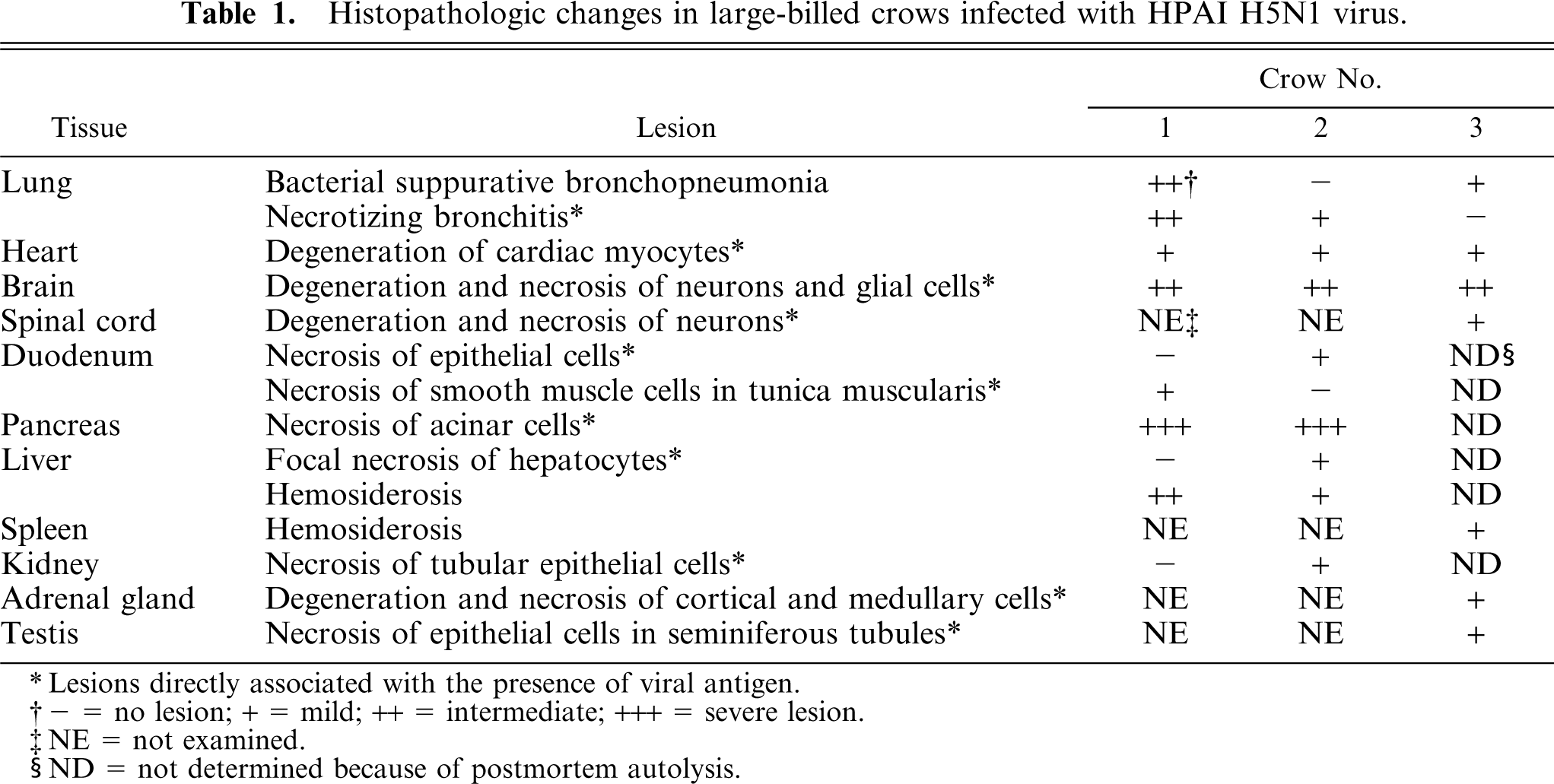
Lesions directly associated with the presence of viral antigen.
− = no lesion; + = mild; ++ = intermediate; +++ = severe lesion.
NE = not examined.
ND = not determined because of postmortem autolysis.
Distribution of viral antigen-positive cells in large-billed crows infected with HPAI H5N1 virus.
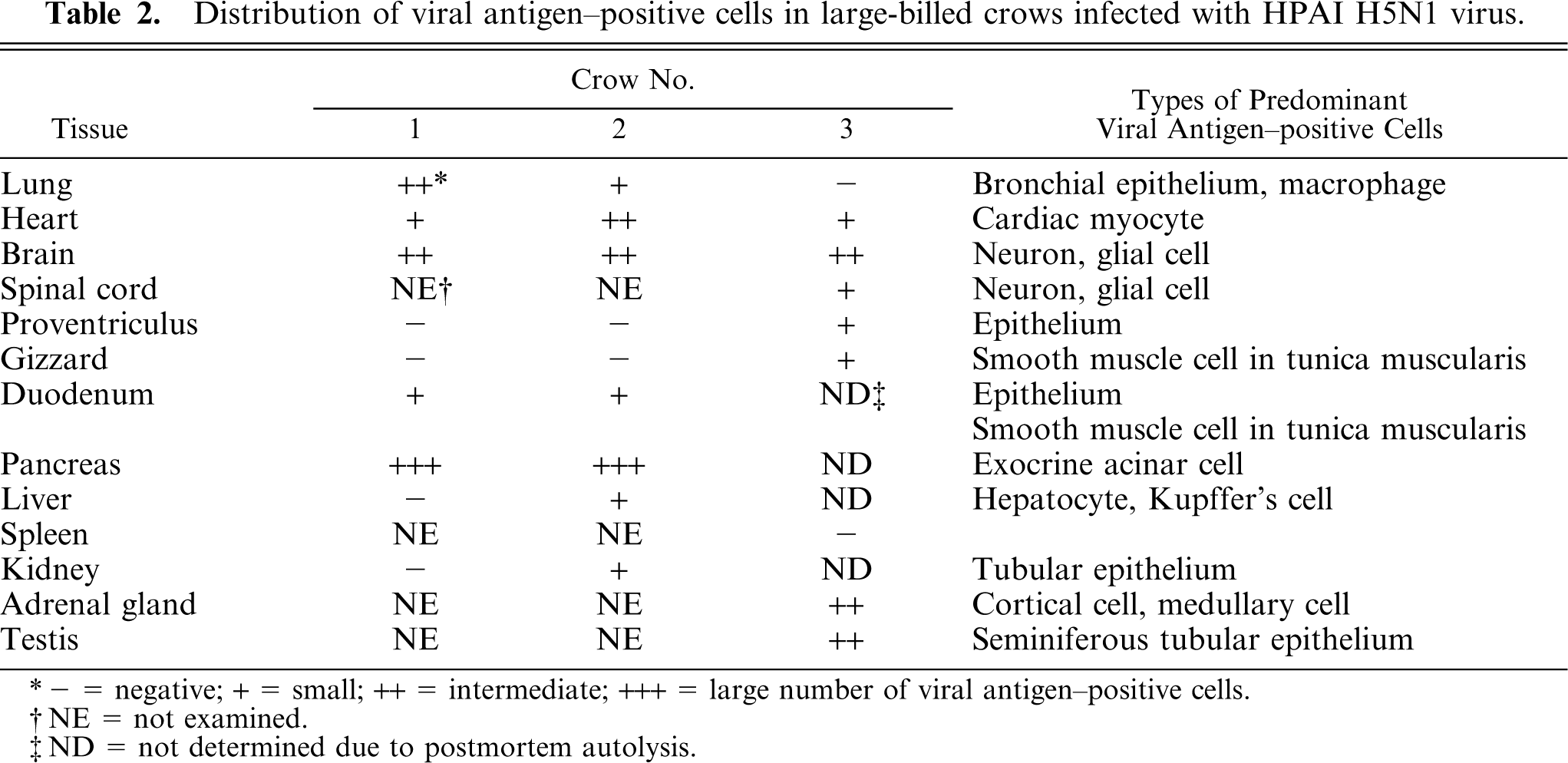
−= negative; + = small; ++ = intermediate; +++ = large number of viral antigen-positive cells.
NE = not examined.
ND = not determined due to postmortem autolysis.
Pancreas; crow No. 1. Multifocal to coalescing necrotic foci are surrounded by vacuolar degenerated pancreatic acinar cells. HE. Bar = 100 µm.
Pancreas; crow No. 1. Viral antigen is present in the degenerative and necrotic acinar cells and in the sloughing interlobular ductal epithelial cells (lower right). Immunohistochemistry for influenza A matrix protein. DAB chromogen, hematoxylin counterstain. Bar = 100 µm.
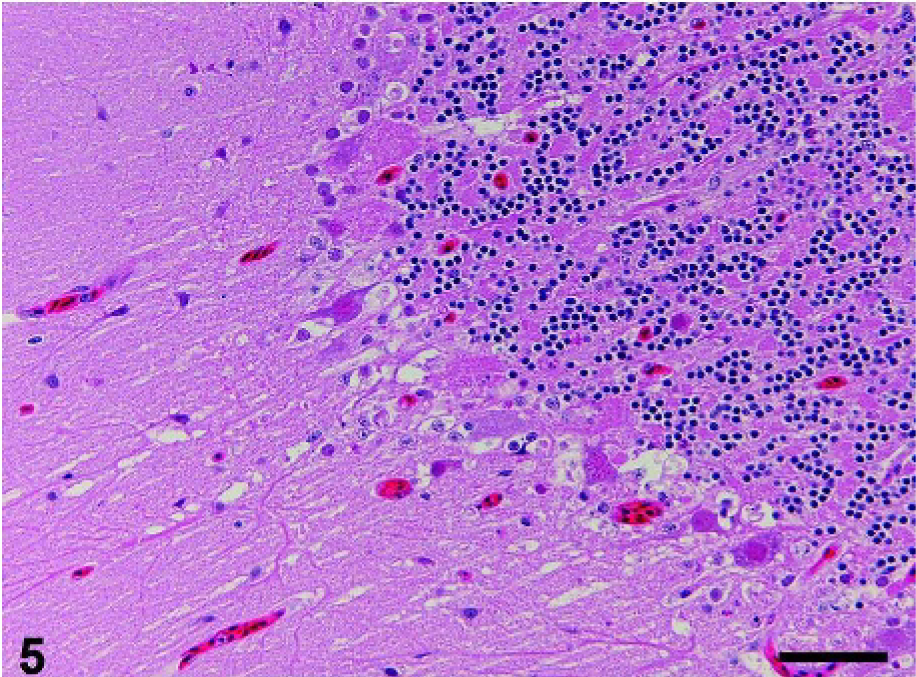
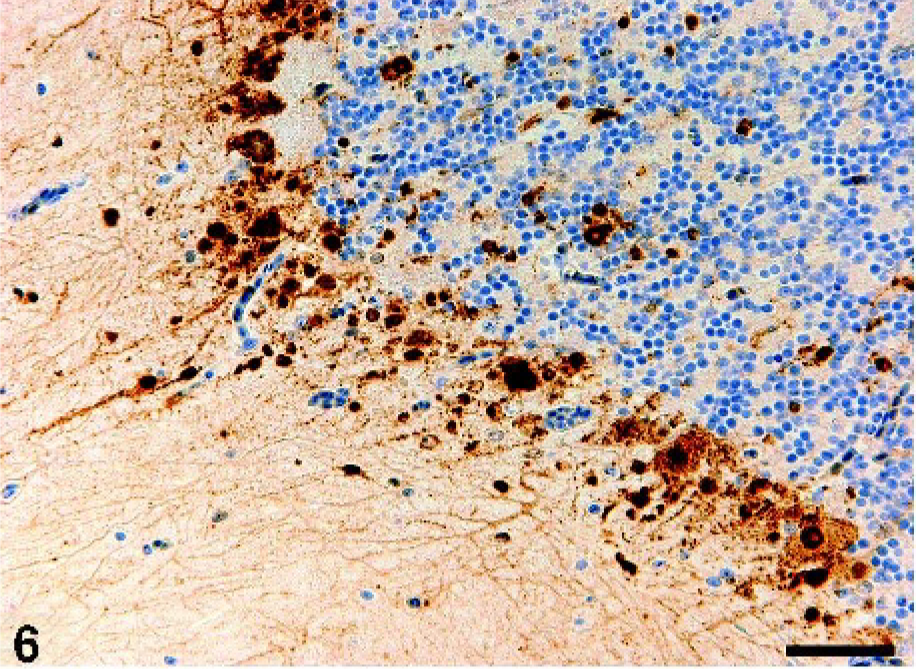
In the central nervous system, the foci of degeneration and necrosis of neurons and glial cells were randomly distributed in the cerebrum, cerebellum (Fig. 5), and brainstem of crow Nos. 1–3. In the foci, some neurons contained swollen nuclei filled with eosinophilic granular materials. Glial cells with karyorrhexis and karyolysis were also present in the foci. Fibrin was deposited in some blood capillaries and venules in the cerebrum of crow Nos. 1 and 2. Mild infiltration of macrophages was present in the meninges of the cerebrum (crow Nos. 1 and 3) and brainstem (crow No. 1). Viral antigens were detected in the degenerative neurons and glial cells in the cerebrum, cerebellum (Fig. 6), and brainstem in crow Nos. 1–3. Ependymal cells in the cerebrum of crow No. 3 also had viral antigens.
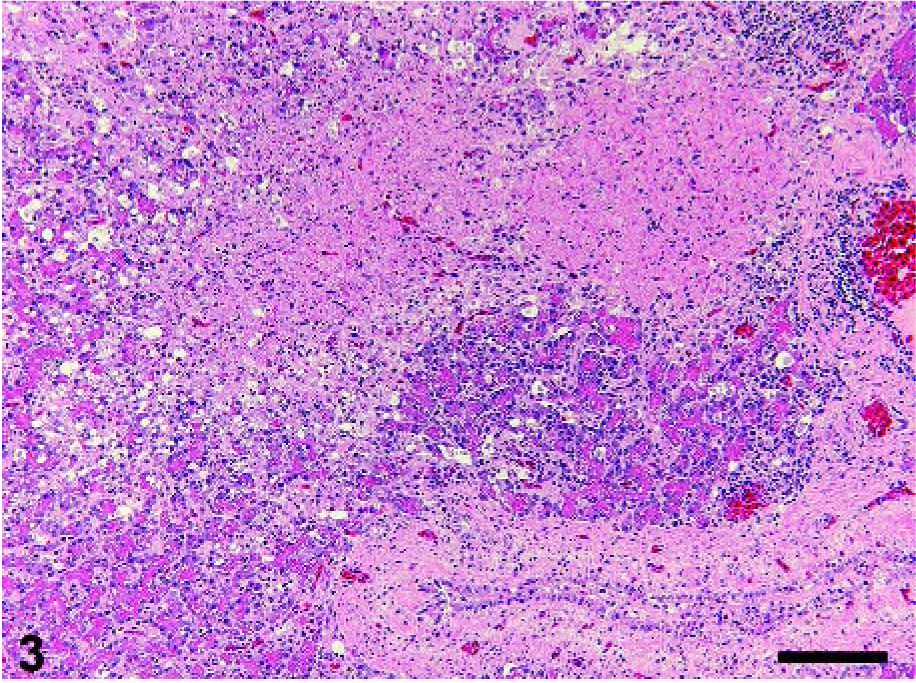
Cerebellum; crow No. 2. Purkinje cell layer has degenerative cells and is accompanied by edema. HE. Bar = 50 µm.
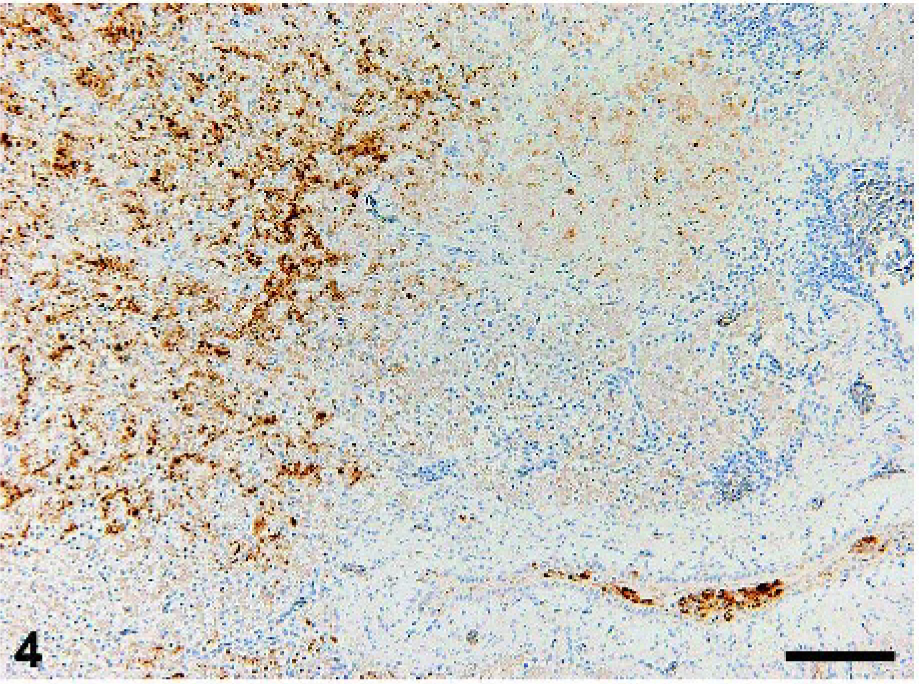
Cerebellum; crow No. 2. Viral antigen is demonstrated in the cell bodies and nuclei of Purkinje cells and their surrounding neurons and glial cells. Immunohistochemistry for influenza A matrix protein. DAB chromogen, hematoxylin counterstain. Bar = 50 µm.
The thoracic spinal cord in crow No. 3 had degenerative neurons with swollen nuclei that showed karyorrhexis and were filled with eosinophilic fine granules. Viral antigens were detected in the neurons and glial cells in the thoracic and lumbar spinal cords.
In the respiratory system, moderate focally extensive and mild multifocal bronchopneumonia with bacterial foci and heterophil infiltration was detected in the lungs of crow Nos. 1 and 3, respectively. The bacterial foci contained both gram-positive cocci and gram-negative bacilli. Moderate congestion in the blood capillaries and small vessels and mild fibrin deposits within these blood vessels and in the heterophil-infiltrated necrotic area were also present in the pneumonic lesions, in which viral antigens were present in the macrophages. In the lung of crow No. 2, severe congestion and moderate fibrin deposits were present in the blood capillaries and small vessels, leading to stenosis of air capillaries. Moderate multifocal (crow No. 1) and mild solitary (crow No. 2) necrotizing bronchitis with sloughed epithelial cells and heterophil infiltration were associated with viral infection (Fig. 7). The vial antigen-positive necrotic bronchial epithelial cells had karyorrhexis and degraded cell bodies. In the cranial thoracic air sac of crow No. 1 there were multiple foci of coagulative necrotic epithelial cells with karyorrhexis. Some necrotic and sloughing epithelial cells contained viral antigens. In addition, in a ganglion in the lung of crow No. 2, some neurons had swollen nuclei filled with eosinophilic homogeneous and fine granules. Others had atrophied nuclei with condensed amphophilic chromatin. Infiltrated lymphoid cells surrounded the ganglion. Viral antigens were detected in the degenerative neurons.
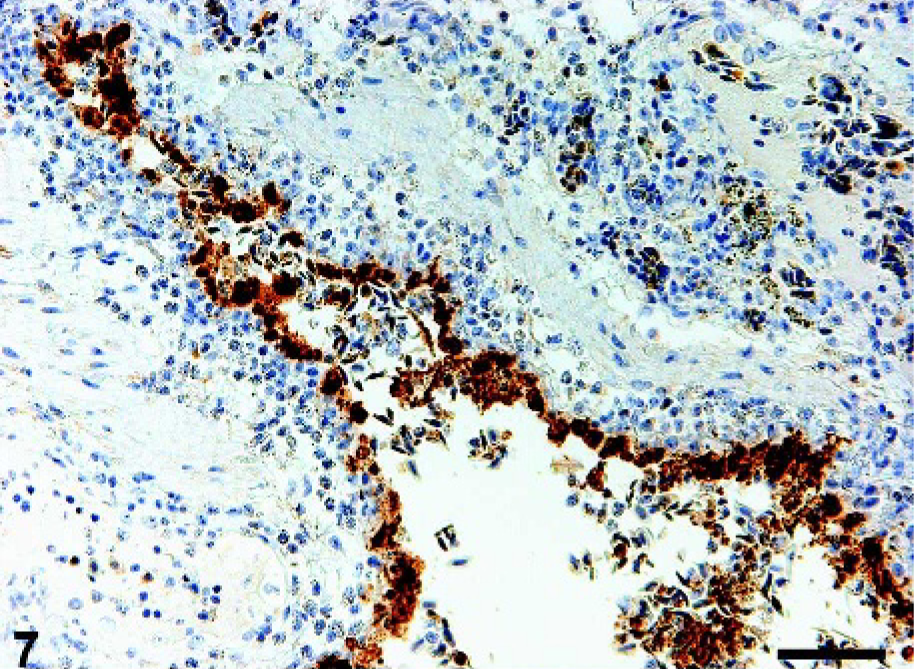
Lung; crow No. 1. The bronchus contains viral antigen–positive epithelial cells, some of which are sloughed into the bronchial lumen. Immunohistochemistry for influenza A matrix protein. DAB chromogen, hematoxylin counterstain. Bar = 50 µm.
The hearts of crow Nos. 1–3 showed mild multifocal degeneration of cardiac myocytes that were viral antigen–positive (Fig. 8). In crow No. 1, the degenerated cardiac myocytes had vacuolated cytoplasm and their cell bodies showed fragmentation and atrophy. In crow No. 2, some nuclei of the degenerative cardiac myocytes had eosinophilic granules that seemed to be viral antigen–positive. These degenerative cells were surrounded by a small number of heterophils. In crow No. 3, besides cardiac myocytes degeneration, the ganglion in the pericardium had viral antigen–positive neurons.
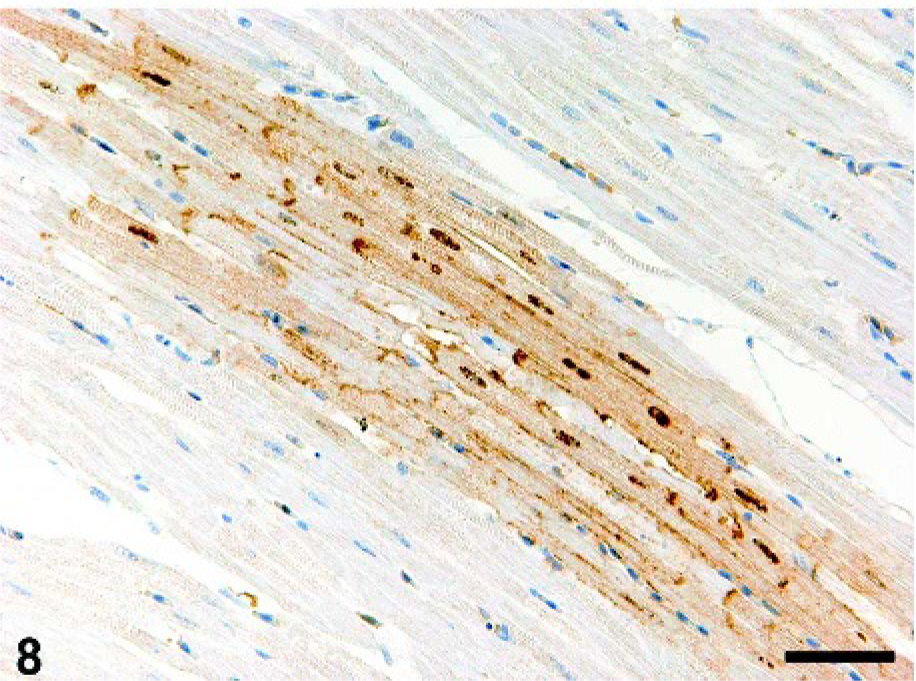
Heart; crow No. 2. Viral antigen is demonstrated focally in the myocardial fibers. Immunohistochemistry for influenza A matrix protein. DAB chromogen, hematoxylin counterstain. Bar = 50 µm.
In the digestive system, the proventriculus and gizzard of crow Nos. 1 and 2 had no viral antigen–positive cells. In crow No. 3, however, the epithelial cells in the deep proper gastric glands of the proventriculus and the smooth muscle cells in the tunica muscularis of the gizzard contained viral antigens. In the duodenum, a small number of the mucosal epithelial cells in crow No. 2 showed karyorrhexis associated with viral antigen localization. The tunica muscularis of the duodenum in crow Nos. 1 and 2 had a small number of viral antigen–positive smooth muscle cells. The viral antigen–positive smooth muscle cells of crow No. 1 showed mild hyaline degeneration and swelling. In the liver of crow No. 2, small necrotic foci of hepatocytes were randomly scattered. In the foci, some deeply eosinophilic hepatocytes showed atrophy and rounding with karyorrhexis and were associated with the presence of viral antigen. In the liver of crow Nos. 1 and 2, a moderate and small number of Kupffer's cells contained yellowish-brown hemosiderin within their cytoplasm, respectively. A small number of Kupper's cells in the liver of crow No. 2 were viral antigen–positive.
In the spleen of crow No. 3, a moderate number of macrophages phagocytized cell debris, and some of them contained hemosiderin. No apparent viral antigen was detected in the spleen.
In the kidney of crow No. 2, small necrotic foci of renal tubular epithelial cells had viral antigens (Fig. 9). The necrotic foci contained fragmented cell bodies and karyorrhexis of renal tubular epithelium.
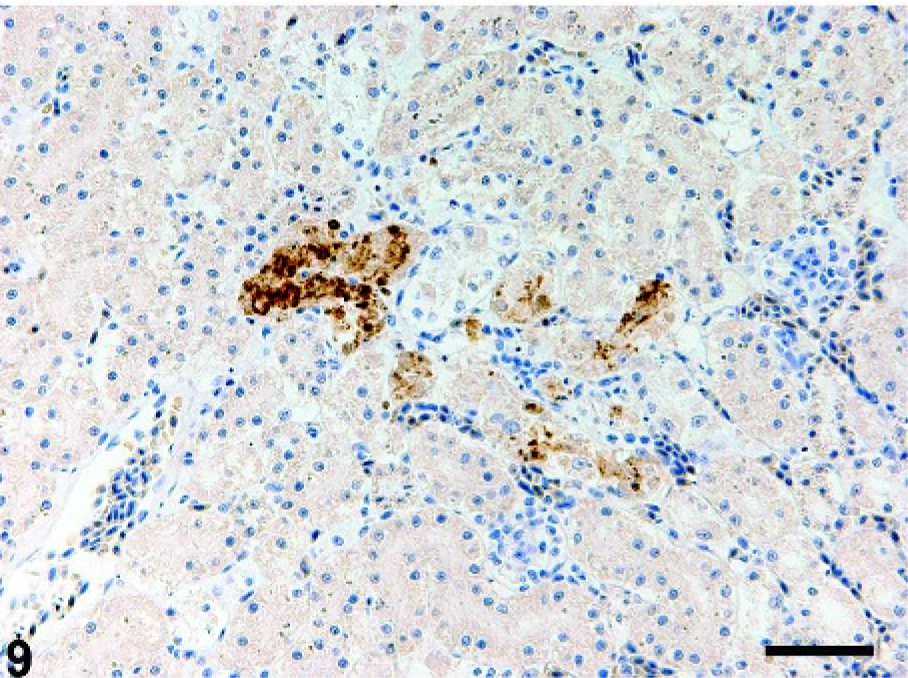
Kidney; crow No. 2. Tubular epithelial cells are positive for viral antigen. Immunohistochemistry for influenza A matrix protein. DAB chromogen, hematoxylin counterstain. Bar = 50 µm.
Both the cortical and medullary cells in the adrenal gland of crow No. 3 showed focal degeneration and necrosis. Most of the degenerated cells had karyorrhexis, and some of them had swollen nuclei filled with eosinophilic homogeneous and fine granules. Most of the degenerated viral antigen–positive cells appeared to be cortical cells. In the ganglion associated with the adrenal gland, a small number of the neurons had swollen nuclei filled with eosinophilic homogeneous and fine granules, and a moderate number of the neurons contained viral antigens.
The seminiferous tubules in the testis of crow No. 3 showed focal necrosis of epithelial cells that had deeply eosinophilic and fragmented cell bodies. Some of the epithelial cells were desquamated into tubular lumen. A moderate number of seminiferous tubular epithelial cells contained viral antigens. Because of necrotic changes, Sertoli cells and spermatogenic epithelium could not be clearly differentiated.
As shown in Table 3, the lung of crow No. 4 had a moderate, focally extensive bronchopneumonia with many bacterial foci. The kidney had moderate, multifocal hyaline droplet degeneration in the proximal tubular epithelia. Other tissues had no significant pathologic changes. Crow No. 4 may have died of secondary bacterial pneumonia caused by ocular trauma. In crow No. 5, the lung had a severe, multifocal to coalescing, necrotizing pneumonia caused by the fungus
Histopathologic changes in the crows that died in a wild animal shelter after they had been captured in a debilitated condition.
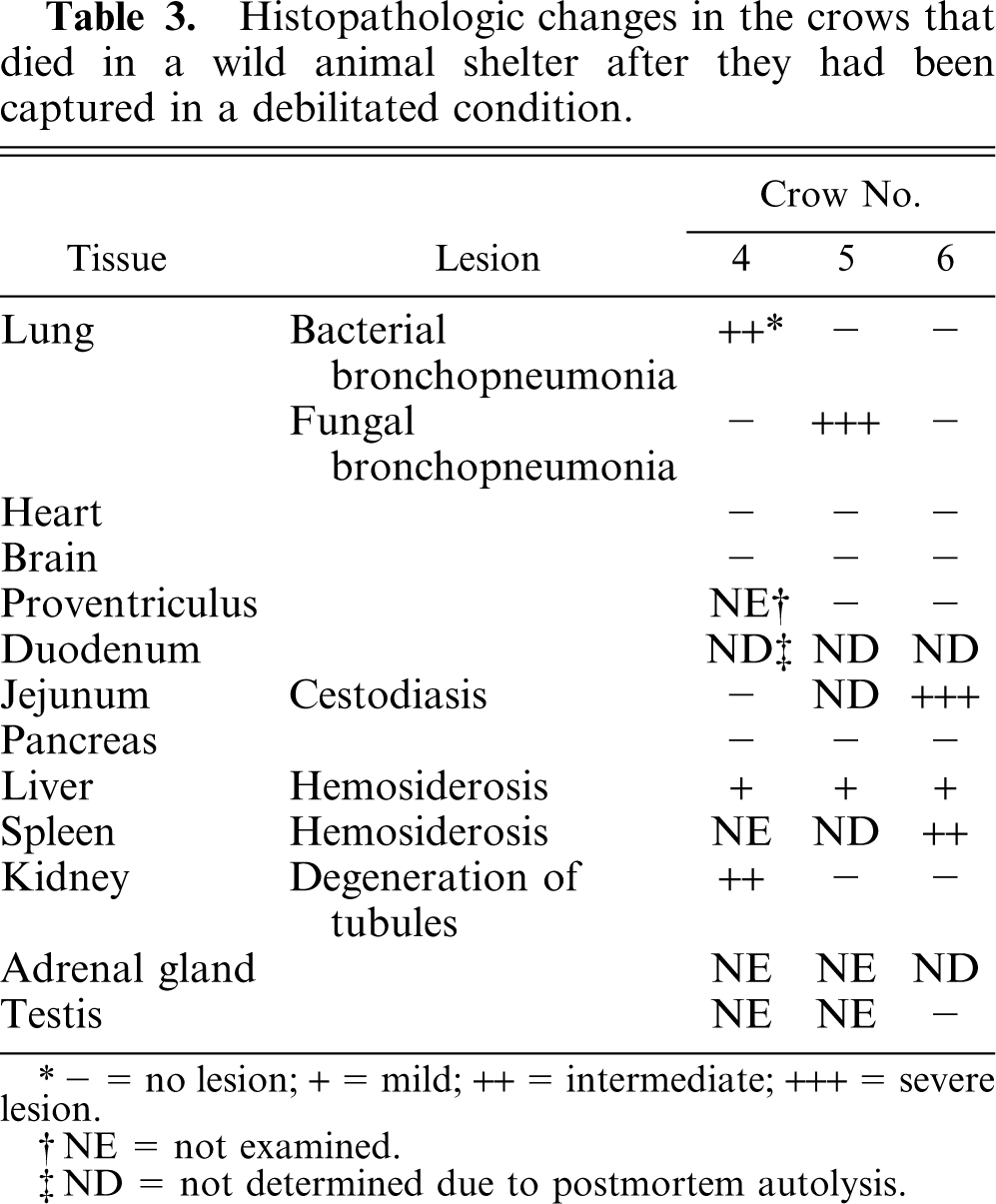
− = no lesion; + = mild; ++ = intermediate; +++ = severe lesion.
NE = not examined.
ND = not determined due to postmortem autolysis.
Virology
Virus titration using chicken kidney cells and embryonated chicken eggs indicated that all the tissues examined in crow Nos. 1 and 2 contained infectious viruses. The brain and the lung of crow No. 1 had the highest virus titers of the organs examined (Table 4).
Viral titers in tissues of large-billed crows infected with HPAI H5N1 virus.∗
Virus titers are expressed log10TCID50 per 0.1 ml and log10EID50 per 0.1 ml.
Discussion
In this study, 3 wild large-billed crows naturally infected with HPAI H5N1 virus were examined by pathologic and virologic methods. This study demonstrated that the crows were susceptible to infection with the H5N1 virus and that the virus infection led to degenerative and necrotic lesions in the systemic organs, including the lung, heart, central and peripheral nervous system, alimentary tract, pancreas, kidney, adrenal gland, and testis. Viral antigen was demonstrated in these multiple tissues, suggesting the pantropic nature of this HPAI virus in the large-billed crow. Immunohistochemical analysis also revealed that the virus antigens were present not only in the central nervous system but also in the peripheral ganglia associated with the lung, heart, pancreas, and adrenal gland, suggesting that the virus may be highly neurotropic in the crow. The results of this study provide the first evidence for HPAI H5N1 virus–induced fatal disease in crows.
Perkins et al. 17, 18 indicated that gallinaceous species including chickens were highly susceptible to infection and disease with the A/chicken/Hong Kong/220/97 (chicken/Hong Kong) (H5N1) virus and that the pathogenesis of HPAI viral infection in these species relates to a combination of factors, including cardiovascular dysfunction caused by endothelial activation and disruption, leukocyte activation, and systemic cytokine release; cardiopulmonary failure; and/or multiorgan failure. In contrast, there is significant variation in the pathogenicity of the chicken/Hong Kong virus for different passerine species. 19 Viral antigen was demonstrated in multiple tissues of zebra finches and house finches, whereas it was demonstrated in only a small number of tissues from a minority of the sparrows and was not demonstrated in tissues from the starlings. In the large-billed crow Nos. 1–3 examined in this study, although viral antigen was demonstrated in multiple tissues, it was not detected in the vascular endothelium, suggesting that the HPAI H5N1 virus is less pathogenic for crows than for chickens. It has been postulated that in fatal human infections with avian influenza H5N1 virus, initial virus replication in the respiratory tract triggers hypercytokinemia complicated by the reactive hemophagocytic syndrome. 25 Although hemosiderosis was observed in the Kupffer's cells in the liver of crow Nos. 1 and 2 and the macrophages in the spleen of crow No. 3, the crows had no systemic hemophagocytic histiocytosis. In addition, mild and moderate hemosiderin depositions were observed in the Kupffer's cells of the livers of crow Nos. 4–6 and in the macrophages of the spleen of crow No. 6, respectively. The findings in the present study suggest that hemosiderosis was not specifically associated with H5N1 virus infection in the crow Nos. 1–3 and that the pathogenesis of HPAI virus infection in the crows is different from that of fatal human infections with avian influenza H5N1 virus. The most consistent lesions in the crows infected with HPAI H5N1 virus were the severe acute necrotizing pancreatitis and the moderate degeneration and necrosis of neurons and glial cells in the central nervous system. The crows may have succumbed to the severe dysfunction of these organs. Bacterial bronchopneumonia was observed in crow Nos. 1 and 3, but there was no histologic evidence of bacterial lesions in other organs or septicemia, suggesting that the bacterial infection in the lung was a secondary sequela to HPAI virus infection. In Kyoto prefecture, the other 6 crows that were positive for HPAI H5N1 virus isolation may also have died of the virus-induced disease as shown in this study. Alternatively, the possible presence of other organisms or other forms of stress may have played a role in the prostration of the crows, leading to the extensive proliferation of HPAI virus in the multiple organs.
Lipatov et al. 12 reported that, in the spinal cords and dorsal root ganglia from mice infected with highly pathogenic neurotropic variant of avian H5N1 influenza virus originally isolated from Hong Kong poultry, there were neurons with marginated chromatin and eosinophilic inclusions that partially or completely filled the nuclei. However, there is no description of intranuclear eosinophilic inclusion in the avian influenza. 23 In the present study, in crow Nos. 1–3, some neurons in the central and peripheral nervous systems had swollen nuclei filled with eosinophilic fine granular materials. The cardiac myocytes in crow No. 2 and the adrenal cortical cells in crow No. 3 also had similar degeneration. However, no clear marginated chromatin was observed. Immunostaining for viral antigens suggested that these degenerative cells contained influenza viral antigens. HPAI H5N1 virus infection in the crows may have resulted in extreme production of viral protein within the nuclei or severe degeneration of nuclear constituents in some neurons, which may have been visualized by HE staining as swollen nuclei filled with intranuclear eosinophilic granules. Although we examined tissue samples of brain, pancreas, and liver of crow Nos. 1 and 2 by transmission electron microscopy, we could not detect specific ultrastructural changes in these tissues that could be ascribed to HPAI virus infection. On the other hand, in crow Nos. 4–6, those nuclear changes found in crow Nos. 1–3 were not present in any tissues examined. These findings suggest that HPAI H5N1 virus infection in the crows leads to characteristic nuclear degeneration especially in the neurons as described above, although the pathogenesis of which remains to be examined.
During the recent outbreak of HPAI in Thailand in 2003–2004, it was demonstrated that 1) the H5N1 virus caused severe pneumonia in tigers and leopards at a zoo that fed on infected chicken carcasses; 9 2) tigers kept in captivity in a zoo could be infected by horizontal transmission; 24 and 3) domestic cats can be infected with H5N1 virus (A/Vietnam/1194/04) both by horizontal transmission and by feeding on virus-infected birds. 10 After the outbreak in the layer chicken farm in Kyoto prefecture in February 2004, 9 virus-infected crows were found dead or prostrate during the following month within 30 km of the infected farm. The crows may have become infected with the virus when they ate the dead infected chickens. There has been no evidence of transmission of the virus from the infected crows to farm chickens or to other wild birds. The presence of viral antigens in the respiratory, gastrointestinal, and renal tubular epithelium in the crows examined suggests that the crows may secrete the virus and play a part in transmitting the virus to the poultry and wild avian and mammalian species. Because of these possibilities, it is important that the poultry industry minimize the opportunity for crows to invade poultry farms and to contact poultry feed and water supplies.
Footnotes
Acknowledgements
This work was supported in part by Special Coordination Funds for Promoting Science and Technology from the Ministry of Education, Culture, Sports, Science and Technology, Japan. We thank Mr. M. Kobayashi and Ms. M. Shimada for preparing the histopathology sections.
