Abstract
The objectives of this study were to investigate the normal histological localization of aquaporin (AQP) 5 protein in the lacrimal and nictitating membrane glands and to compare this localization in healthy and keratoconjunctivitis sicca (KCS) dogs. Lacrimal and nictitating membrane glands of 5 healthy Beagles and nictitating membrane glands of 5 KCS dogs (3 Beagles and 2 mongrel dogs: 0–13 years) were used for the present study. The owners of the KCS dogs did not consent to perform biopsies of the lacrimal glands. The localization and distribution of AQP5 protein were investigated by an immunohistochemical technique. In immunohistochemical staining, AQP5 was localized in the apical site of acinar epithelial and ductal epithelial cells from both the lacrimal and nictitating membrane glands in healthy dogs. However, AQP5 was not detected in the 5 KCS dogs. These results for immunohistochemical AQP5 localization might correlate with the deficiency in tear secretion found in KCS dogs.
Keywords
The lacrimal glands of the canine orbit and nictitating membrane produce the aqueous portion of the trilaminar preocular tear film, which is integral to the protection and nutrition of the cornea. These secretions also contribute to ocular surface immunity. The lipid layer produced by the meibomian gland stabilizes the tear film, preventing evaporation, and goblet cells in the palpebral conjunctiva elaborate mucin, which allows the aqueous tear fluid to spread evenly over the cornea. 1
Simultaneous dysfunction in the lacrimal and nictitating membrane glands causes keratoconjunctivitis sicca (KCS) disorder. KCS is caused by a pathological reduction in the aqueous component of the tear film and defined as an Schirmer tear test (STT) value <10 mm/min and concomitant ocular surface pathological changes or lesions or something besides pathology. 7 KCS dogs suffer chronic inflammatory and vision-threatening ophthalmic disease due to the deficient water layer in their eyes. 4 The most general cause of KCS is local immune-mediated disease in the lacrimal and nictitating membrane glands. This has been determined on the basis of histopathology of tear-producing glands and observed clinical responses to immunomodulators. 4 The most common lesion associated with the autoimmune-mediated adenitis is multifocal, mononuclear cell infiltration with varying degrees of fibrosis. In about 50% of the cases in KCS dogs, the morphologic changes are mild to modest and not so disruptive as to explain the lack of glandular function despite the chronic and absolute deficiency of tears. 4 The nictitating membrane gland plays a greater role in both the quantity and quality of tear secretion. Surgical removal of the nictitating membrane gland causes KCS in 6 weeks to 7 months, and although the STT reading may return to near normal in 12 months, the quality of the tear film and corneal surface is significantly altered. 7
KCS in dogs resembles Sjögren syndrome in humans, an autoimmune disease characterized by dry eyes and mouth. It has been reported that the distribution of aquaporin (AQP) 5 in the lacrimal gland of Sjögren syndrome patients is altered histologically. 9 AQPs are a family of water-channel proteins, currently 13 in number, that are widely distributed in mammalian tissues. 5 AQPs, present in the plasma membrane of epithelial, endothelial, and other cell types, transport water rapidly. The interstitial-to-luminal transport of sodium and chloride across the acinar epithelium is the driving force for osmotic water flow through AQPs. AQP3 and 5 are present in the nictitating membrane glands of dogs. 3 The aim of this study is to investigate the immunohistochemical localizations of AQP5 in the lacrimal and nictitating membrane glands in healthy and KCS dogs.
To perform this investigation, lacrimal and nictitating membrane gland tissues were collected from 5 healthy dogs and 5 KCS dogs whose conditions were caused by local immune-mediated disease (Table 1). The 5 healthy dogs consisted of Beagle dogs. Their ages ranged from 1 to 2 years. Before sampling from the healthy dogs, each animal’s STT values were confirmed to be over 15 mm/min. The 5 clinical cases of KCS occurred in 3 Beagles and 2 mongrel dogs. From the KCS dogs, part of the nictitating membrane gland was collected under general anesthesia after obtaining the owner’s consent. Samples of the lacrimal glands from KCS dogs were unavailable since their owners did not consent to perform the biopsy. Their ages ranged from 0 to 13 years. The criterion for determining local immune-mediated KCS was an STT value <10 mm/min and infiltration of lymphocytes of conjunctiva near the nictitating membrane glands. Secondary KCS cases were not included in the present study.
Clinical Characteristics of Keratoconjunctivitis Sicca

STT, Schirmer tear test.
The frozen samples were homogenized in RIPA buffer. The submandibular gland of a rat was used for the positive control, and the lens from a healthy dog was used for negative control. 3 The protein solution was separated by 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), transferred to a PVDF membrane, and then subjected to Western blot analysis. The membranes were blocked with 5% blocking reagent (ECL Blocking Agents; GE Healthcare Bio-Sciences Corp., Piscataway, New Jersey) overnight at 4°C. Then, the membrane was incubated with rabbit anti-rat AQP5 polyclonal antibody (catalogue No. ab78486; Abcam plc, Cambridge, UK) (1:5000) for 1 hour at room temperature. The membranes were incubated for 1 hour at room temperature with diluted horseradish peroxidase–conjugated secondary antibody (catalogue No. NA9340V; GE Healthcare Bio-Sciences Corp.) (1:5000). Specific bands were visualized by chemiluminescence (ECL; GE Healthcare Bio-Sciences Corp.) and detected by a CCD imager (LAS-4000 film; Fujifilm Corporation, Tokyo, Japan).
Samples of the lacrimal and nictitating membrane gland from healthy dogs and nictitating membrane gland from KCS dogs were fixed with 4% paraformaldehyde (PFA) for 24 hours at 4°C and then embedded in paraffin. Thereafter, the sample blocks were sectioned at 3 μm and mounted onto adhesive slides. After being deparaffinized, the endogenous peroxidase in the samples was inhibited by 1% hydrogen peroxide for 30 minutes and blocked with 2.5% blocking reagent (Block Ace; Dainippon Sumitomo Pharma Co., Ltd., Tokyo, Japan) in Tris-buffered saline (TBS). AQP5 staining was performed using the primary antibody, rabbit anti-rat AQP5 polyclonal antibody (1:400) (catalogue No. ab78486; Abcam plc), overnight at 4°C. The bound anti-AQP5 antibody was revealed by staining with peroxidase-antiperoxidase complex (Histfine, catalogue No. 414181; Nichirei Corporation, Tokyo, Japan), and the fixed peroxidase was visualized using diaminobenzidine. Finally, counterstaining was performed with hematoxylin.
All experiments complied with the guidelines of the ethics committee of Nippon Veterinary and Life Science University.
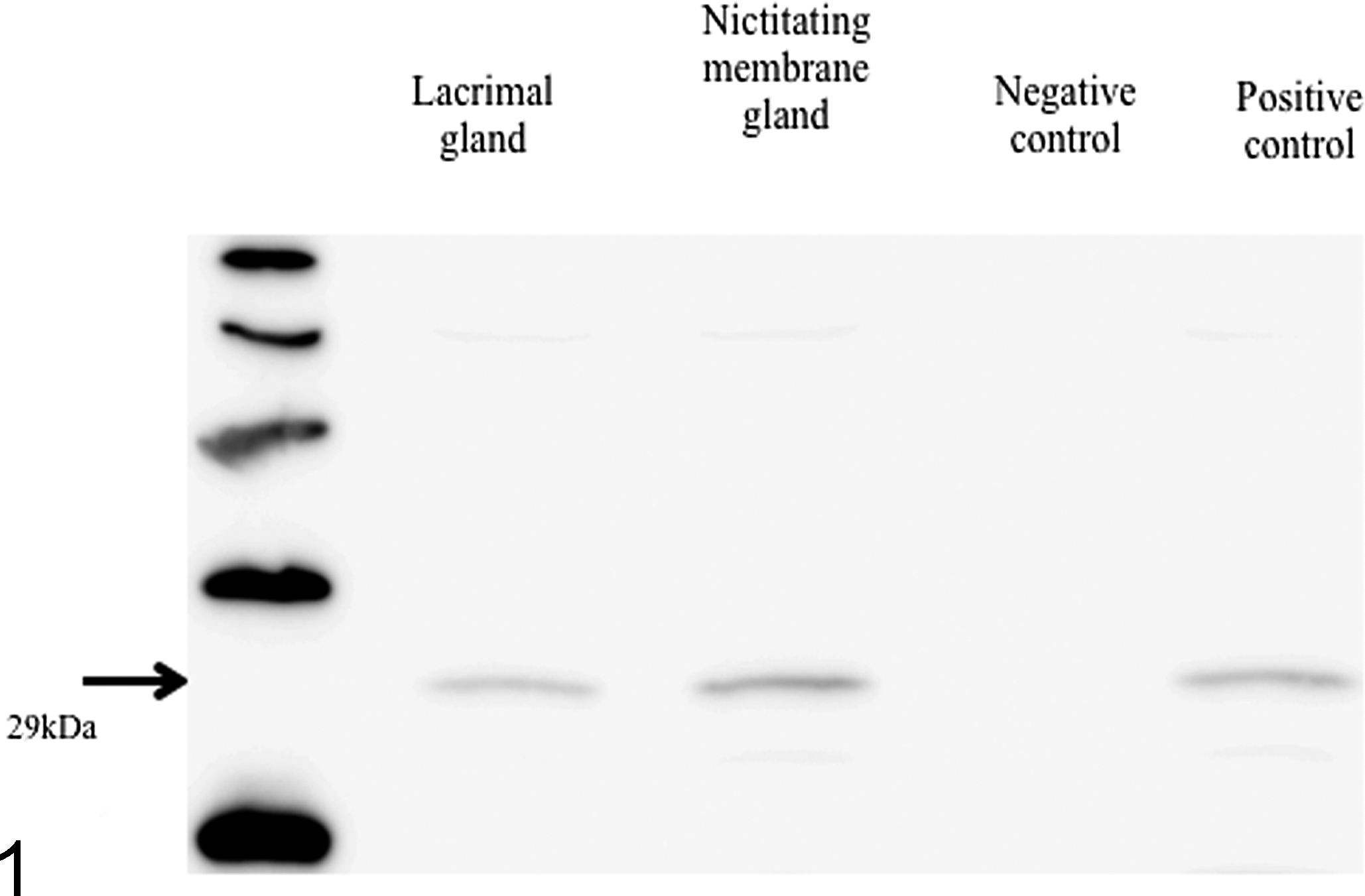
The extracts from the lacrimal and nictitating membrane glands were separated on a 12% SDS-PAGE gel by electrophoresis, transferred to a PVDF membrane, and then incubated with rabbit anti-AQP5. Clear single bands from every sample without negative control were detected at about 29 kDa (Fig. 1).
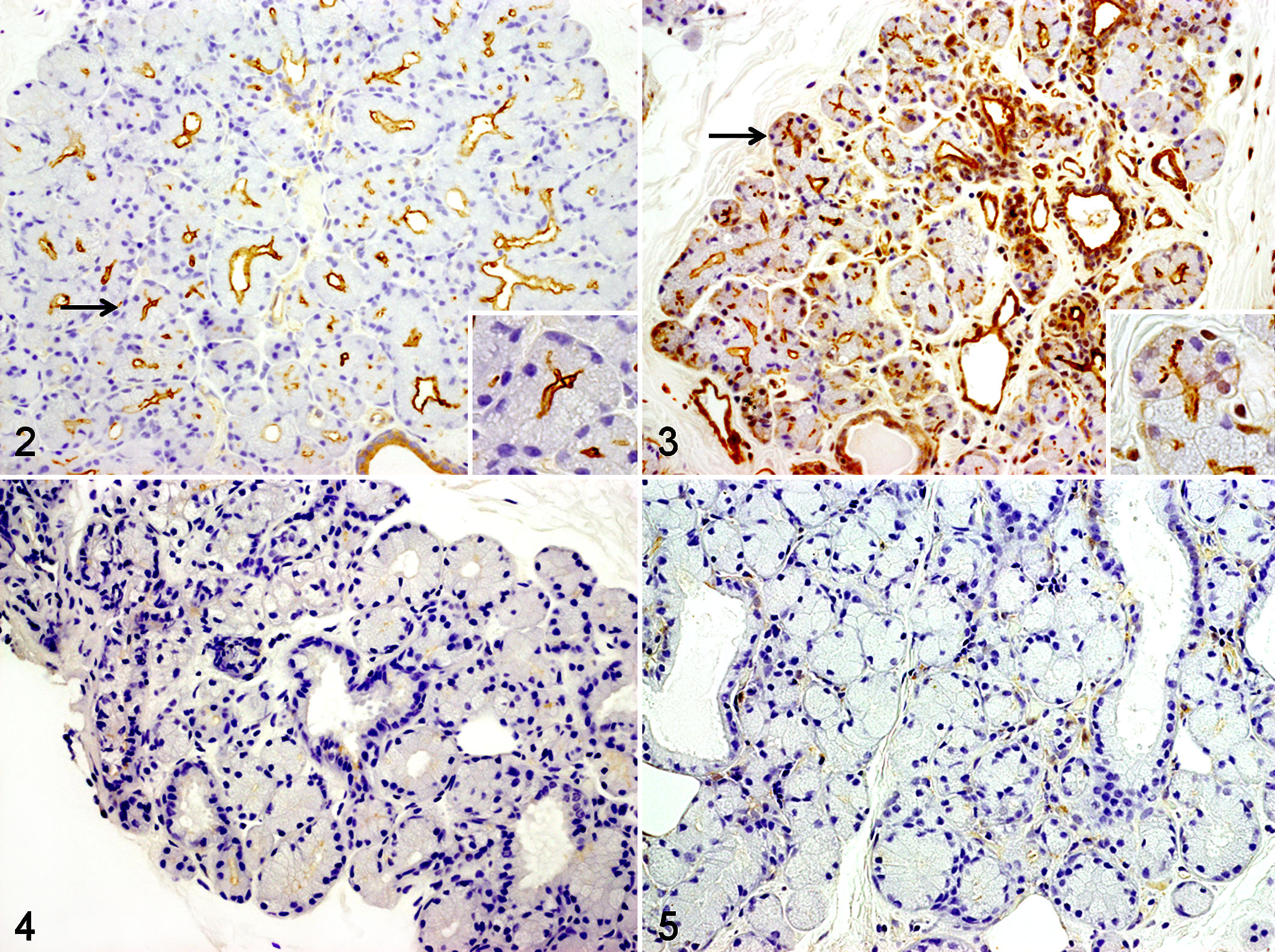
In the lacrimal glands of the healthy dogs that had normal tear secretion, AQP5-positive signals were observed in the apical membrane of acinar epithelial and ductal epithelial cells (Fig. 2). Similar to the case in the lacrimal gland, AQP5-positive signals were observed in the apical membrane of acinar epithelial and ductal epithelial cells in nictitating membrane glands (Fig. 3). The apical membrane of acinar epithelial cells showed stronger positive signals than the membrane of ductal epithelial cells. In contrast with the healthy dogs, the 5 KCS dogs showed remarkably decreased AQP5-positive signals at the apical site of the membrane of acinar epithelial and ductal epithelial cells in the nictitating membrane glands (Figs. 4, 5).
In Western blotting, a clear single band from the dog samples was detected at about 29 kDa, which was the approximate predicted weight of the dog’s AQP5 protein. These results suggested that affinity-purified anti-AQP5 would react against the dog sample. The presence of this band raises the possibility that AQP5 exists in the lacrimal and nictitating membrane glands of the dog.
Immunohistochemical staining using affinity-purified anti-AQP5 showed clear regions for AQP5 in the apical membrane of the acinar epithelial and ductal epithelial cells in the mouse lacrimal gland. 2 Similar results in dogs confirmed that AQP5 was localized at corresponding sites in these animals. However, in dogs, AQP5 tends to show a stronger localization at the apical site of the membrane of acinar epithelial cells than in the membrane of ductal epithelial cells.
Interestingly, the parotid gland, an exocrine gland similar to the lacrimal gland, has been shown to have a significantly decreased volume of saliva in transgenic knockout mice deficient in AQP5. 8 Lacrimal and parotid glands have shown the completely opposite AQP5 distribution patterns. 8 In mice lacrimal glands, AQP5 was localized more strongly in the ductal epithelial cells than in the apical membranes of acinar cells. However, in mice parotid glands, AQP5 was localized more strongly at the apical site of membranes of acinar cells than in the ductal epithelial cells. It is possible that increased AQP5 in parotid acinar cells may produce a large amount of saliva. 8
AQP5 was found to localize more strongly in the ductal epithelial cells than at the apical site of membranes of acinar cells in mice lacrimal glands. However, in dogs, the reverse was found, with AQP5 localizing more strongly at the apical site of membranes of acinar cells than in ductal epithelial cells in the lacrimal and nictitating membrane glands. The distribution of AQP5 in the lacrimal and nictitating membrane glands more closely resembles mice parotid glands than mice lacrimal glands. Since dogs secrete a much higher volume of tear fluid than mice, it is possible that AQP5 localization is responsible for such tear production.
The 5 KCS dogs studied here showed remarkably decreased AQP5 in the apical membrane of acinar epithelial and ductal epithelial cells in the nictitating membrane glands. These decreases of AQP5 distribution might correlate with deficiencies in tear secretion. AQPs are required when near-isosmolar fluid secretion is rapid. 10 In an observation using transgenic knockout mice deficient in AQP5, the secretion of saliva was found to be significantly decreased, whereas tear production was not decreased. 6,8 In mice, the rate of fluid transport per unit surface area of acinar epithelium in the lacrimal gland was estimated to be ∼30-fold less than that in the salivary gland. 10 Dogs produce a larger amount of tears than mice, and the distribution of AQP5 in the nictitating membrane gland of dogs resembles the saliva gland of mouse. Therefore, decreases in AQP5 distribution might correlate with deficiencies in tear secretion.
In humans, healthy distribution of AQP5 has been seen primarily at the site of the apical membrane of the acinar cells. In contrast, in Sjögren syndrome, there is markedly deficient labeling in apical membranes of lacrimal glands, and AQP5 protein diffuses in the cytoplasm. However, there is no reduction in the quantity of AQP5 protein, suggesting a defect in protein trafficking rather than in protein synthesis. 9 KCS in dogs presents similar symptoms to Sjögren syndrome in humans, but AQP5 distribution has been found to be more altered in KCS dogs than in Sjögren syndrome patients. The mechanism of this difference is unknown.
Expressional patterns of AQP subtypes affect the tear secretion levels and osmotic pressure in the eyes of mice. AQP5 may be the osmoregulator in tears in lacrimal gland ducts, maintaining an isotonic tear solution. A consistently isotonic tear film is very important for the ocular surface milieu, and hypertonic tear film is reported to favor KCS development and induction of apoptosis band inflammation in the cornea. 8 In the mouse lacrimal gland, AQP5 is reported to be localized at the apical site of the membranes of acinar and ductal epithelial cells, whereas AQP3 and AQP4 are localized in the contralateral basolateral membranes, AQP1 is in the surrounding microvascular endothelia, and AQP8 is in the ductal epithelial cells. 2,6 It is therefore probable that AQP plays a role in tear secretion in mice. In the dog nictitating membrane gland, AQP3 and AQP5 were shown to be present by Western blot analysis, but the distribution of AQP3 and AQP5 was not revealed. 3 AQP3 of the nictitating membrane gland in dogs is likely to localize in the contralateral basolateral membranes, similar to the case in mice. AQP5 might assume a more crucial role for tear secretion than AQP3 because AQP5 is localized at the luminal side.
It is possible that the other isoforms of AQP might be localized in the lacrimal and nictitating membrane glands of dogs. Further studies will be necessary to confirm the distribution of the other isoform and their possible roles in the physiological mechanism behind tear secretion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
