Abstract
Better understanding of mast cell tumors (MCTs) in miniature pigs is needed to guide diagnosis and establish clinical significance. We characterized the gross pathology, histopathology, histochemical staining, and KIT immunoreactivity of cutaneous MCTs in a retrospective descriptive study of 11 miniature pigs (
Mast cell proliferative diseases are rarely reported in pigs, and include cutaneous mast cell tumors (MCTs),6,8–10,14 cutaneous and systemic mastocytosis,
2
and mast cell leukemia with systemic mastocytosis.1,12 Mast cell diseases have been described most often in commercial swine at slaughter; only 2 cases of mast cell leukemia and 2 cases of cutaneous MCTs have been described in miniature (pot-bellied) pigs (
To diagnose poorly granulated MCTs, toluidine blue or Giemsa stains are often applied. In previous reports of pigs, the discernment of cytoplasmic granules typical of neoplastic mast cells has often been difficult with H&E stain and occasionally even with toluidine blue and Giemsa stains.3,6,8,14 Thus, we compared the utility of these stains for the definitive diagnosis of MCTs in miniature pigs.
Cutaneous MCTs have been reported in many domestic species, including dogs, cats, horses, cattle, and ferrets,
7
and are most common and best described in dogs. Two histologic grading systems (Patnaik,
11
Kiupel
5
) with prognostic significance have been defined in dogs, using parameters such as infiltration of deeper tissues, presence of necrosis, mitotic count, granularity, anisokaryosis, and multinucleation. Immunohistochemistry (IHC) for the KIT protein (CD117) has also been used for diagnosis and assessment of prognosis in canine cutaneous MCTs.4,13 KIT is a receptor tyrosine kinase encoded by the
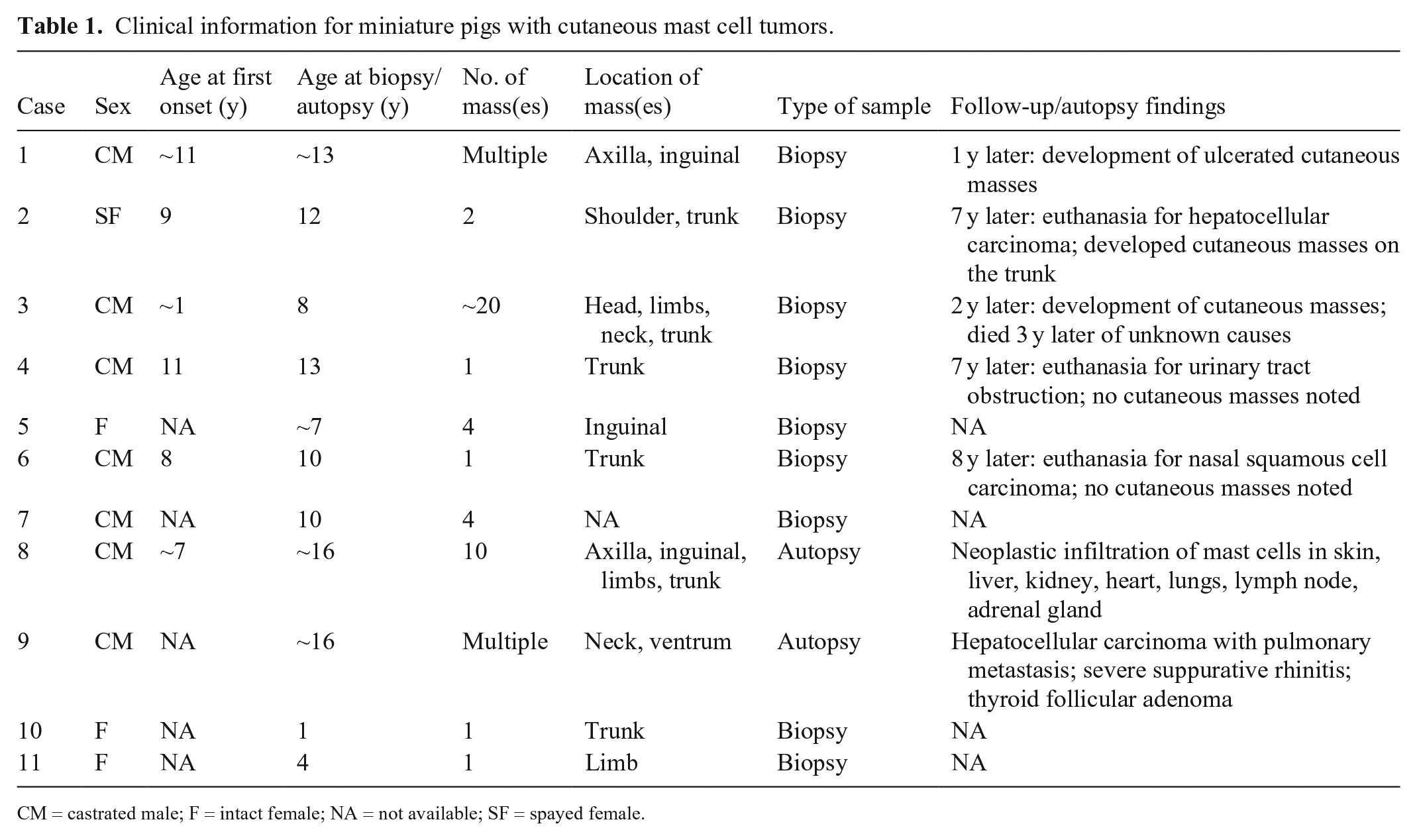
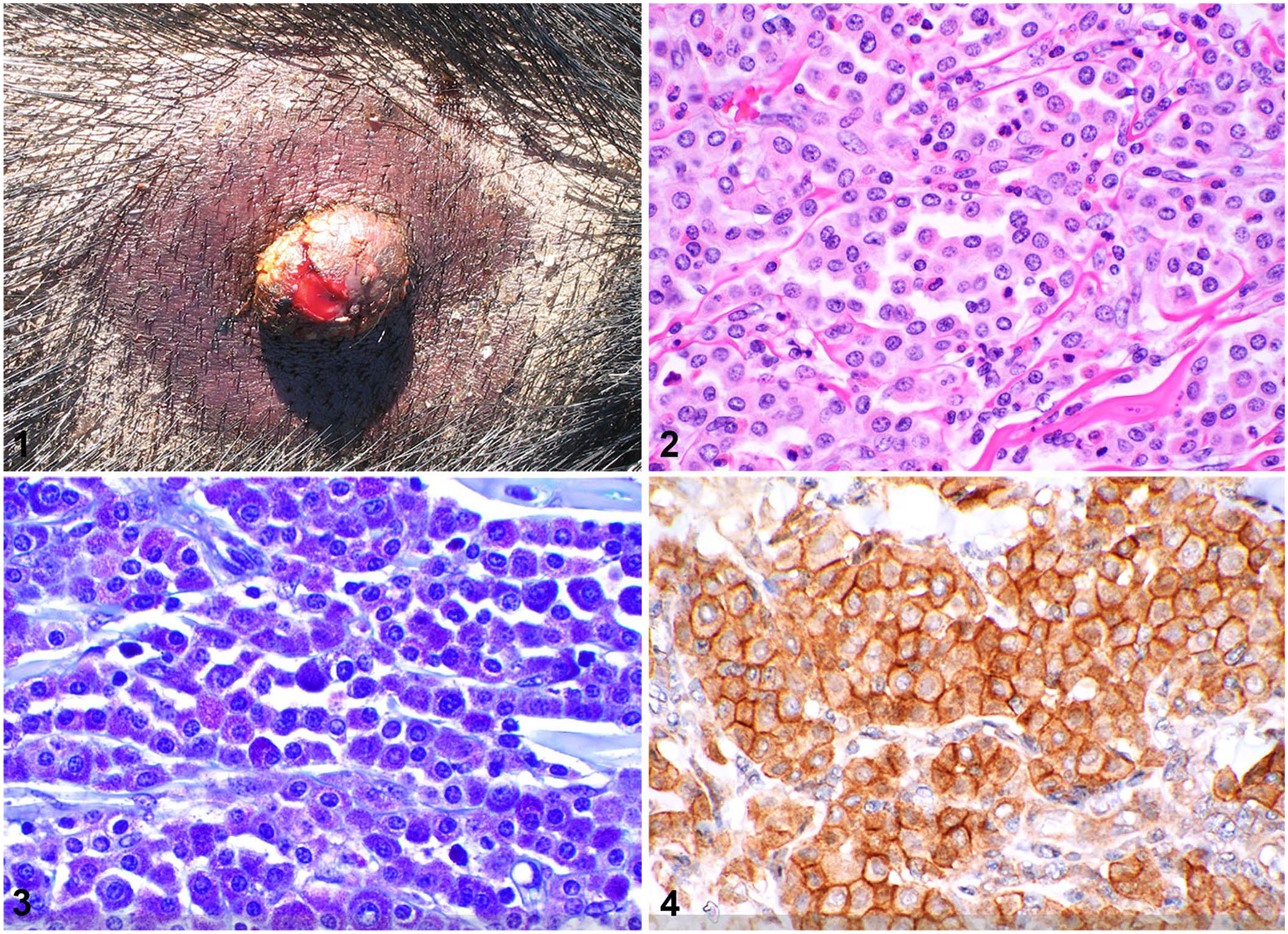
We included all miniature pigs with a histologic diagnosis of cutaneous MCT found by searching the diagnostic database at the North Carolina State University, Veterinary Hospital (Raleigh, NC, USA) and the Pennsylvania Animal Diagnostic Laboratory System from 2003–2019. Cases included those with available paraffin blocks and/or histology slides. Most (9 of 11) of these cases were observed in one author’s clinical practice (K. Mozzachio). The diagnosis was confirmed by 2 board-certified veterinary pathologists (K. Mozzachio, K.E. Linder) using the histologic criteria published for MCTs in other domestic animals. 3 Eleven miniature pigs with cutaneous MCTs were identified (Table 1) and included 7 castrated males and 4 females (3 intact, 1 spayed), 1–16-y-old. The exact ages of 5 pigs, adopted from rescue organizations, were unknown and were estimated from the medical records. Clinically, cutaneous masses were multiple (7 of 11) or solitary (4 of 11) domed masses or plaque-like thickenings that were slow-growing (Fig. 1); however, in cases with multiple masses, not all were evaluated. Cutaneous masses ranged from ~0.5 cm diameter to 5 × 4 cm and were most frequently located on the trunk (6 of 10 pigs), limbs (3 of 10), and inguinal region (3 of 10); skin location was unavailable for 1 case. Local recurrence after surgical removal was reported in 2 cases.
Clinical information for miniature pigs with cutaneous mast cell tumors.
CM = castrated male; F = intact female; NA = not available; SF = spayed female.
Cutaneous mast cell tumors in miniature pigs.
Nine tumor samples were surgical biopsies, and 2 were from autopsies. In 8 cases, paraffin-embedded blocks of tissue were available, and histologic sections were routinely stained with H&E, low-pH (0.5–1) toluidine blue, high-pH (2.5–3) toluidine blue, and Giemsa stains. In 3 cases, only previously stained slides were available. Toluidine blue stains were performed manually using a 0.5% toluidine blue solution dissolved in 0.5 N hydrochloric acid for the low-pH stain, and using a 0.5% toluidine blue solution dissolved in 10% ethanol for the high-pH stain. Giemsa stain was performed using an automated system (Artisan; Dako). IHC for KIT protein (CD117) was performed using an anti-KIT polyclonal rabbit antibody (A4502; Dako) at a 1:500 dilution with an automated stainer (Intellipath; Biocare). Tissue sections without the addition of primary antibody served as negative controls. Resident dermal mast cells served as internal positive controls for histochemical stains and KIT IHC.
At autopsy, one miniature pig with a history of cutaneous MCT recurrence had multiple (≥10) cutaneous masses, and histologic infiltration of neoplastic mast cells occurred in lymph nodes and multiple internal organs (liver, kidney, heart, lungs, adrenal gland). A second pig had multiple cutaneous masses noted as an incidental finding with a primary diagnosis of metastatic hepatocellular carcinoma at autopsy.
Follow-up information was available for 5 of 9 cases for which samples were obtained at biopsy. One pig is currently alive and has continued to develop new ulcerated cutaneous masses over 1 y following biopsy. Four pigs were either euthanized for a separate disease process (3 pigs) or died of unknown causes (1 pig) within 3–8 y after biopsy. Two of these pigs developed additional cutaneous masses; 2 pigs remained skin tumor free.
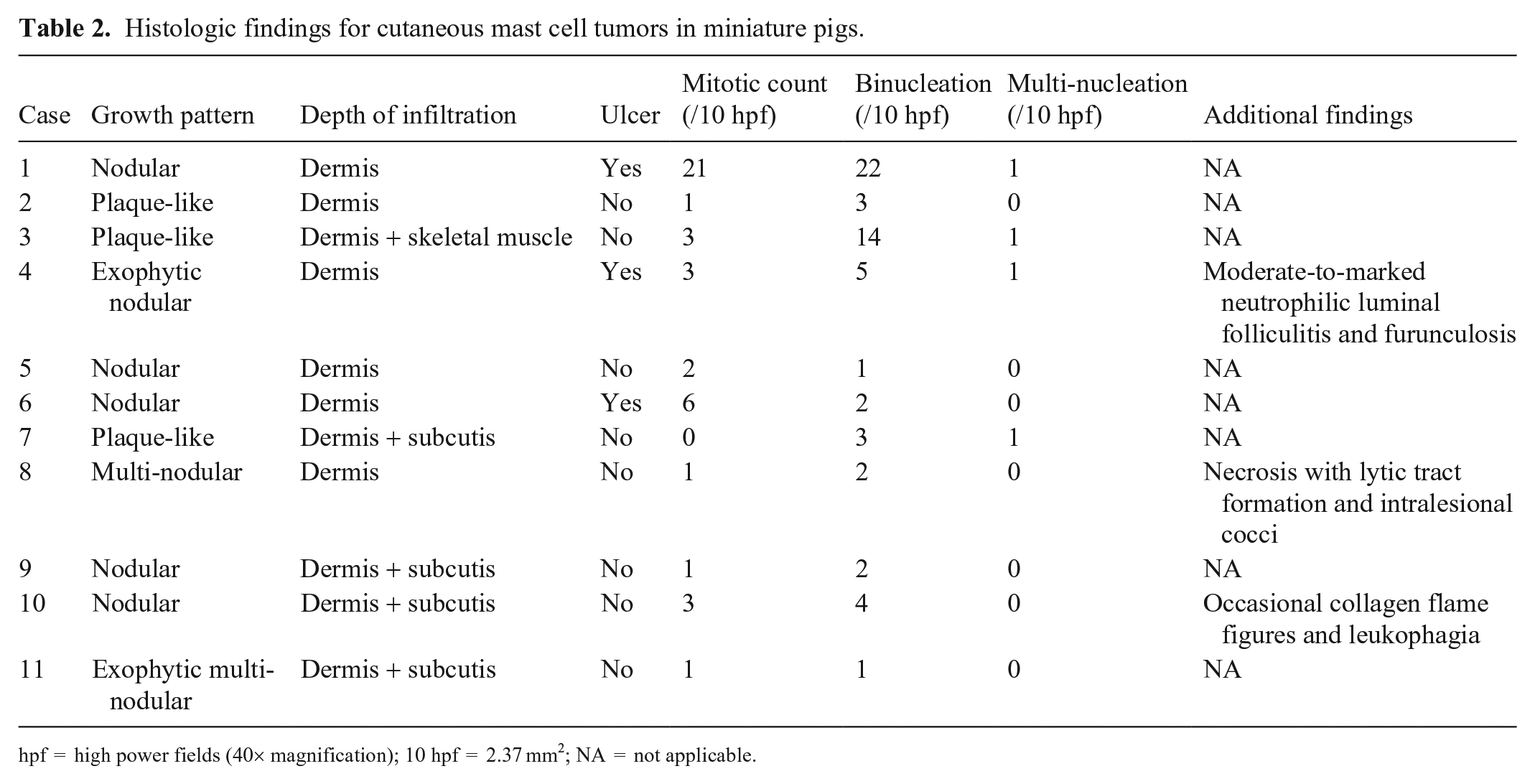
On histologic examination, MCTs (1 examined from each pig) were all based in the dermis with some extending into the subcutis (4 of 11) and underlying skeletal muscle (1 of 11; Table 2). Most of the tumors (8 of 11) were well-demarcated, but non-encapsulated, with a nodular (6 of 11) or multinodular (2 of 11) appearance; fewer (3 of 11) had a poorly demarcated plaque-like appearance spreading across the dermis (Suppl. Fig. 1). Neoplastic cells lacked epitheliotropism.
Histologic findings for cutaneous mast cell tumors in miniature pigs.
hpf = high power fields (40× magnification); 10 hpf = 2.37 mm2; NA = not applicable.
Neoplastic mast cells were arranged in poorly to densely cellular sheets and rows supported by resident collagenous stroma. Neoplastic cells were often well-differentiated and were round with variable amounts of pale amphophilic-to-eosinophilic cytoplasm, fine-to-indistinct cytoplasmic granularity, and distinct cell borders (Fig. 2). Nuclei were round to occasionally slightly indented with stippled-to-coarse chromatin and occasionally had a single small nucleolus. Most cases (10 of 11) had minimal-to-mild nuclear pleomorphism. However, in case 1, moderate nuclear pleomorphism with frequent nuclear indentation and binucleation was present, occasionally with differing nuclear sizes within the same binucleate cell. Binucleate neoplastic cells were present in all 11 cases. Multinucleation with 3–4 nuclei was observed in 4 cases with no more than 1 multinucleate cell per 10 high power fields (hpf; 40×; 2.37 mm2). Most cases (10 of 11) had a low mitotic count (0–6 mitotic figures/10 hpf); case 1 had 21 mitotic figures per 10 hpf. All cases contained intermixed eosinophils, with 8 of 11 cases containing moderate-to-marked eosinophil infiltration (>50 eosinophils/hpf). Ulcers (3 of 11) were occasionally present over MCTs. Other features, each noted in a different single mass, included eosinophil collagen flame figures and occasional leukophagia, coagulative-to-lytic necrosis with intralesional cocci, and moderate-to-marked neutrophilic folliculitis and furunculosis.
Three histochemical stains (low-pH toluidine blue, high-pH toluidine blue, Giemsa) were performed to distinguish magenta-to-purple metachromatically stained cytoplasmic granules in neoplastic cells. Results were graded as negative (no metachromatic staining of granules), weak positive (subtle staining of granules or staining in <25% of neoplastic cells), or positive (strong staining of granules in ≥25% of neoplastic cells; Table 3, Fig. 3, Suppl. Figs. 2, 3).
Histochemical and immunohistochemical staining of cutaneous mast cell tumors in miniature pigs.

– = negative (lack of staining of granules in neoplastic cells); ± = weak positive (subtle staining of granules or strong staining in <25% of neoplastic cells); + = positive (strong staining of granules in >25% of neoplastic cells); NA = tissue blocks not available for staining.
1 = perimembranous labeling in >90% of neoplastic cells; 2 = focal perinuclear or stippled cytoplasmic labeling and loss of perimembranous labeling in >10% of neoplastic cells; 3 = diffuse cytoplasmic labeling in >10% of neoplastic cells.
Immunostaining using antibodies against the KIT protein (CD117) was observed in most neoplastic cells in 8 of 8 cases. The 3 patterns of immunolabeling reported in canine cutaneous MCTs 13 were observed: 4 of 8 cases had strong perimembranous immunolabeling in >90% of neoplastic cells (pattern 1), 1 of 8 cases had focal perinuclear immunolabeling with concurrent loss of perimembranous immunolabeling in 30–50% of the neoplastic population (pattern 2), and 3 of 8 cases had diffuse cytoplasmic immunolabeling in ~50% or more of the neoplastic population (pattern 3; Table 3, Fig. 4, Suppl. Fig. 4).
In our case series, cutaneous MCTs were seen most commonly in middle-aged to older animals (>5-y-old) with earliest detection at 1-y-old. Previous reports, which mostly described mast cell neoplasia in younger pigs,3,6,8,9 may be related to the population studied (pigs for slaughter) as opposed to longer-lived pet miniature pigs. Cutaneous MCTs in miniature pigs often have a clinical presentation that is similar to MCTs in other domestic species, such as dogs, cats, horses, and ferrets.3,7 Most appear to be well-differentiated benign masses that can be solitary or multiple, occurring as papules, nodules, or plaques that are distributed on various areas of the body. Recurrence or development of additional masses after surgical excision can occur in a subset of cases. Less commonly, spread of neoplastic mast cells to lymph nodes and internal organs (liver, kidney, heart, lungs, adrenal gland) is observed, as seen in 1 of 11 cases in our study and in 1 previous case report, with spread to the kidney. 14 Whether the cutaneous lesions in these cases represent primary or metastatic neoplasia, or mastocytosis, is uncertain, but our findings indicate that at least some MCTs in miniature pigs can exhibit multiorgan involvement.
Histologically, cutaneous MCTs in miniature pigs are mostly well-differentiated and have some features in common. Most MCTs were fairly well-demarcated nodular to multinodular masses. Almost all cases had a low mitotic count (<7 mitotic figures/10 hpf [2.37 mm2]) with minimal-to-mild nuclear pleomorphism. Only one case had a higher mitotic count with more prominent nuclear pleomorphism. All tumors had interspersed eosinophils, similar to canine, equine, and bovine MCTs, but unlike feline MCTs in which intratumoral lymphocytes are observed more often than eosinophils.3,7 Unfortunately, prognostic correlation with histologic features was not possible given limited case follow-up.
Cytoplasmic granules are a key diagnostic feature of MCTs in all species. In our study, the miniature pig MCTs had faint-to-indistinct pale amphophilic-to-eosinophilic granularity rather than the typical blue-purple cytoplasmic granules observed in well-differentiated canine mast cells with H&E stain. A minority of cases lacked observable cytoplasmic granules. The difficulty of observing granules in porcine mast cell neoplasia has been described and could affect diagnosis.6,8,14 Toluidine blue or Giemsa stains can be used to highlight mast cell granules and aid in diagnosis; however, the degree of granule staining and diagnostic utility of the different stains varies by species, including for the pig.3,6,7,14 In our study, high-pH (2.5–3) toluidine blue stain was most effective at demonstrating granules that stained metachromatically, with some positive staining observed in almost all cases. Low-pH (0.5–1) toluidine blue and Giemsa stains were slightly less effective. Cytoplasmic granules could not be identified with histochemical stains in one case.
In our case series, KIT immunoreactivity was present in most neoplastic cells in all tested cases, even in the one case that lacked metachromatic granule staining with histochemical stains, and KIT is hence a promising diagnostic marker. Similar to canine cutaneous MCTs, the miniature pig MCTs in our study had various patterns of KIT immunolabeling. In dogs, patterns 2 (focal perinuclear) and 3 (diffuse cytoplasmic) are associated with increased local recurrence and decreased overall survival time.4,13 Of the 2 cases with reported local tumor recurrence in our case series, one had KIT pattern 1 whereas the other (in which neoplastic cells had spread to lymph nodes and internal organs) had KIT pattern 3. Two additional cases had KIT pattern 3: one with a solitary mass and no further development of masses, and one with incidental cutaneous masses noted at autopsy. Thus, a prognostic correlation with KIT expression pattern was not apparent in our case series. Further studies are needed to assess the prognostic significance of KIT expression in miniature pig cutaneous MCTs.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221079255 – Supplemental material for Cutaneous mast cell tumors in 11 miniature pigs: a retrospective study
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221079255 for Cutaneous mast cell tumors in 11 miniature pigs: a retrospective study by Brittany L. Rasche, Kristie Mozzachio and Keith E. Linder in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the North Carolina State University College of Veterinary Medicine, Histology Laboratory staff for assistance with processing and staining of histologic sections. Special thanks to the New Bolton Center of the Pennsylvania Animal Diagnostic Laboratory System, especially Dr. Susan Bender and Dr. Perry Habecker, for providing tissue blocks and access to pathology reports for 4 of the cases in our series.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
