Abstract
The expression of Ki67, BCL-2, and COX-2 was investigated in 53 canine cutaneous mast cell tumors (MCTs) by immunohistochemistry and quantitative real time polymerase chain reaction (qPCR) to evaluate their prognostic significance and the association with the histologic grading and the mitotic index (MI). MCTs were graded according to the Patnaik grading system and the novel 2-tier grading system proposed by Kiupel. The numbers of mitotic figures/10 high-power fields (MI) were counted. Both grading systems were significantly associated with prognosis. The Patnaik grading was of limited prognostic value for grade 2 MCTs, with 23% being associated with mortality. The concordance among pathologists was strongly improved by the application of the 2-tier grading system, and 71% of high-grade MCTs were associated with a high mortality rate. MI and Ki67 protein expression were significantly associated with grading and survival. No significant association between BCL-2 protein expression and either grading system or health status was observed. BCL-2 mRNA expression was significantly higher in grade 2 than in grade 1 MCTs, while no statistically significant differences were detected between low- and high-grade MCTs. The increased BCL-2 mRNA level was significantly associated with increased mortality rate. The COX-2 protein expression was detected in 78% of the MCTs investigated. However, neither association with the tumor grade nor with the health status was observed. COX-2 mRNA was significantly up-regulated in MCTs compared to surgical margins and control skin tissue, but it was neither associated with tumor grade nor with survival.
Canine cutaneous mast cell tumor (MCT) is a common neoplastic disease in dogs, accounting for 7% to 21% of all cutaneous neoplasms. Canine MCTs have variable biologic behaviors, ranging from solitary benign masses that can be cured with surgery alone, to systemic and potentially fatal metastatic disease.67,72
The prognostic significance of several markers for canine MCT, including the histological grading,5,49,50,52 the mitotic index,14,40,52,57 the staining for argyrophilic nucleolar organizer regions (AgNORs),6,61,65,70 and immunohistochemistry for proliferating cell nuclear antigen (PCNA)1,61,65,70 as well as for Ki671,61,70 have been previously investigated. No single factor accurately predicts the biological behavior or the response to treatment in dogs affected by MCTs, and clinical factors, including location, tumor size, breed, sex, and clinical stage, can influence the outcome. 72
Numerous histopathological grading systems have been proposed to prognosticate canine MCTs.5,26,50 Among them, the classification proposed by Patnaik et al 50 is the most frequently used system, but it is influenced by subjective interobserver variations and intratumoral heterogeneity.47,48 As a result, various attempts have been undertaken to use more objective methods to assess the grade of MCTs. To improve concordance among pathologists and provide better prognostic significance, a 2-tier histologic grading system has been recently proposed. 33 The diagnosis of high-grade MCT is characterized by any one of the following criteria: at least 7 mitotic figures in 10 high-power fields (HPFs), at least 3 multinucleated cells in 10 HPFs, at least 3 bizarre nuclei in 10 HPFs, karyomegaly. 33
The prognostic importance of the mitotic index (MI) for dogs with cutaneous MCT has been evaluated in different studies.14,40,52,57 Data reported by Romansik et al 57 stated that MI correlates directly with tumor grade; furthermore, the median survival time for dogs with a MI ≤ 5 was significantly longer (70 months) than for those with a MI > 5 (2 months), regardless of grade. 57 However, the study conducted by Elston et al 14 states that a stratification in 3 groups with cut-points at 1 and 7 mitoses per 10 HPFs is superior to the stratification previously proposed by Romansik et al. 57 Nevertheless, the MI is an objective measure and may help to limit interobserver variation in the histological evaluation of MCTs. A problem that remains to date is that the various studies have proposed different cut-off values and thus the data are still difficult to apply in a diagnostic setting.
Ki67 is a nuclear protein that is expressed in all active phases of the cell cycle but it is not present in noncycling cells.18,63 The relative number of Ki67-positive cells is used to determine the proliferation index or the relative number of cells actively involved in the cell cycle (growth fraction).18,44,63 A high Ki67 expression in MCTs was associated with increased mortality, the rate of local recurrence, and metastasis.1,61,70 Since Ki67 emerged to be a prognostic factor that is independent of histological grade,61,70 Ki67 immunohistochemistry can be used as an objective prognostic marker in MCTs.
Members of the BCL-2 family of proteins play important roles in regulating cell survival and apoptosis. 78 The BCL-2 family includes pro-apoptotic members and anti-apoptotic proteins such as BCL-2 and BCL-xL that inhibit apoptosis by blocking the release of cytochrome c. 34 Overexpression of BCL-2 and BCL-xL can predispose to neoplastic transformation causing aberrant accumulation of cells;36,75 in fact, an increased expression of BCL-2 and BCL-xL has been found in a variety of different cancers.4,15,31,32,60 In many neoplastic cells, it has been shown that a high expression of BCL-2 and BCL-xL correlates with resistance to conventional chemotherapy.15,31,68 In vitro studies have demonstrated that survival of murine and human mast cells also depends on BCL-2 and BCL-xL.3,8,42,76,80 A recent study provides evidence that in patients with mastocytosis, the expression of BCL-2 protein in mast cells was significantly enhanced compared to controls. 23 Although the mechanism by which BCL-2 family proteins promote neoplasia is not fully understood, it is well accepted that enhanced expression of BCL-2 and BCL-xL allows cells to live longer and thus to accumulate genetic alterations.4,78 No data are available about the possible role of BCL-2 in canine MCTs progression and prognosis.
Cyclooxygenase-2 (COX-2) is a member of the family of cyclooxygenase enzymes that catalyze the conversion of arachidonic acid to prostaglandin G2 (PGG2). PGG2 is further metabolized to a variety of other prostaglandins (PG) and thromboxanes (TXA). 66 The role of COX-2 in neoplasia has already been investigated in depth.16,17,25,66 Specific mechanisms whereby COX-2 contributes to the development of malignancies include the inhibition of cellular apoptosis, promotion of tumor angiogenesis, increased tumor cell motility and invasiveness, immunomodulatory activity, and the conversion of procarcinogens to carcinogens. 17
COX-2 protein expression has been associated with prognosis in human breast carcinoma, 10 malignant mesothelioma, 13 squamous cell carcinoma, 30 and chronic myelogenous leukemia. 19 Overexpression of COX-2 has been previously identified in a variety of canine neoplasms, including intestinal carcinoma, 41 transitional cell carcinoma of the bladder (TCC), 28 squamous cell carcinoma, 51 mammary carcinoma, 11 and renal cell carcinoma, 29 even though the prognostic significance of COX-2 expression in these cancers has not been clearly established. In both humans and dogs, these findings prompted the investigation of the use of nonsteroidal anti-inflammatory drugs (NSAIDs) therapeutically. NSAIDs emerged to be as efficacious as conventional chemotherapy in dogs with TCC and oral squamous cell carcinoma, even though these drugs are not COX-2-specific and the levels of COX-2 expression in these tumors is variable.35,62
Previous in vitro experiments have demonstrated rapid and transient up-regulation of COX-2 gene in activated mast cells. 56 However, the findings of a previous immunohistochemical study suggested that canine MCT do not express COX-2 protein in any appreciable fashion. 24
In the present study, Ki67, BCL-2, and COX-2 protein and gene expression have been investigated in canine cutaneous MCTs by immunohistochemistry (IHC) and quantitative real time RT-PCR (qPCR), in order to evaluate their prognostic significance and their association with other well-accepted prognostic markers, such as the histologic grading and the MI.
Materials and Methods
This prospective study was performed on a heterogeneous population of confirmed cases of spontaneous canine cutaneous mast cell tumors, undergoing follow-up for over a year. For all patients, age, breed, gender, number, and anatomical location of the tumor and data about therapeutic treatment were collected. Cases were included in the study based on the meeting of the following inclusion criteria: (1) All dogs underwent surgical excision of the entire tumor and the diagnosis was confirmed by histological examination; (2) adequate formalin-fixed paraffin embedded tissues for immunohistochemistry, as well as aliquots from the central part (core) of each tumor, stored in RNAlater® solution, for RNA extraction were available; (3) complete follow-up data, including local or distant recurrence, metastasis, and cause of death, were recorded for a minimum period of 12 months. Follow-up time was defined as the time from date of diagnosis to date of the last follow-up or death.
The histological grading was assigned following Patnaik system 50 and the novel 2-tier grading system, recently proposed by Kiupel et al, 33 by consensus of 3 pathologists (MV, AC, AV). The number of mitotic figures/10 HPFs (MI) was counted.
Immunohistochemistry
Immunohistochemistry was carried out by an automated immunostainer (Bond – maXTM, Menarini Italia, Italy). Four-µm sections were deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. The heat-induced antigen retrieval was performed in 10 mM citrate buffer (pH = 6.0) at 98°C. Endogenous peroxidases were neutralized by incubating the sections in 3% H2O2 in PBS (phosphate buffered saline); sections were incubated with primary antibodies against Ki67 (MIB-1 mAb, 1:50, DAKO, Carpinteria, CA), BCL-2 (pAb, 1:250, Sigma Aldrich, Suffolk, UK), and COX-2 (SP21, mAb, 1:50, Thermo Scientific, Fremont, CA) proteins, for 30 minutes at room temperature. The HRP Bond polymer refine detection system and the 3,3 diaminobenzidine tetrahydrochloride (DAB) were used as detection system and chromogen, respectively. Sections were then counterstained with Mayer’s hematoxylin. The specificity of the immunolabeling was verified by incubating sections with PBS instead of the specific primary antibody. As positive controls, sections from a lymphoma for Ki67, skin for BCL-2, and kidney for COX-2 were selected from dog tissues and included in each immunohistochemical run. All positive controls showed: nuclear immunolabeling of the neoplastic cells for Ki67, cytoplasmic immunolabeling of the basal cell of the epidermis and adnexae for BCL-2, and cytoplasmic immunolabeling of the cells of the macula densa for COX-2. For each sample, a total of 500 cells were counted and the positivity for Ki67 and BCL-2 was expressed as the average number of immunopositive cells per 100 cells, evaluated in 5 randomly selected HPFs (× 400) (Ki67 and BCL-2 index, respectively); for COX-2, a separate score staining for distribution and intensity was given to all positive-staining tumors. A COX-2 staining distribution score from 0 to 3 was assigned to all tumors, where 0 = 0%, 1 = < 30%, 2 = 30% to 60%, 3 = > 61% of positive cells in 5 randomly selected 400 × fields. The COX-2 staining intensity was defined as the strength of the signal in positive-staining tumors, with 0 = no signal, 1 = weak signal, 2 = moderate signal, and 3 = strong signal. The COX-2 staining index was obtained by multiplying the COX-2 staining distribution and intensity scores.
Gene Expression
Aliquots from the core of the tumor (up to 100 mg) and from 3-cm-wide surgical margins (SM, n = 30) were aseptically collected during surgical intervention and immediately stored in RNAlater® solution (Applied Biosystems, Foster City, CA), at –20°C, until use. Skin samples from control dogs (pathogen-free Beagle generously provided by GlaxoSmithKline Research Centre, Verona, Italy, and adult dogs not suffering from neoplasm and euthanized in veterinary clinics of Padua for serious traumas) were also collected.
Total RNA was isolated using TRIzol® reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. Samples were then purified with a classical phenol-chloroform extraction step. Total RNA concentration and quality (260/280 and 260/230 nm absorbance ratios) were determined using Nanodrop ND-1000 spectrophotometer (Nanodrop Technologies, Wilmington, DE) and denaturing gel electrophoresis (Sigma Aldrich, Munich, Germany).
To generate cDNA, two micrograms of total RNA and the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA) were used according to the manufacturer’s procedure. Complementary DNA was then stored at –20°C until use.
Canis lupus familiaris mRNA sequences of selected genes (see Table 1) were obtained from GenBank and Ensembl Genome Browser Web sites (http://ncbi.nlm.nih.gov/ and http://www.ensembl.org/). Primer pairs and probes were chosen using the UPL Assay Design Centre Web service (Roche Diagnostics, Basel, Switzerland; https://www.roche-applied-science.com/sis/rtpcr/upl/index.jsp) and default parameters. Primer pairs were designed to span an exon-exon junction to avoid genomic DNA amplification, and primers sets were subjected to primer test analysis (http://eu.idtdna.com/analyzer/Applications/OligoAnalyzer/Default.aspx) to exclude dimer synthesis. Each set of primers was optimized in the 300 to 600 nM range. Agarose gel electrophoresis confirmed the amplification of a single amplicon of the expected size. The sequence, the length, and the concentration of primer pairs; the amplicon size; and the human UPL probe used for the amplification are shown in Table 2.
Target and Housekeeping Genes Selected for Quantitative Real Time PCR (qPCR) Analysis: Description and GenBank or Ensembl Genome Browser Accession Number.
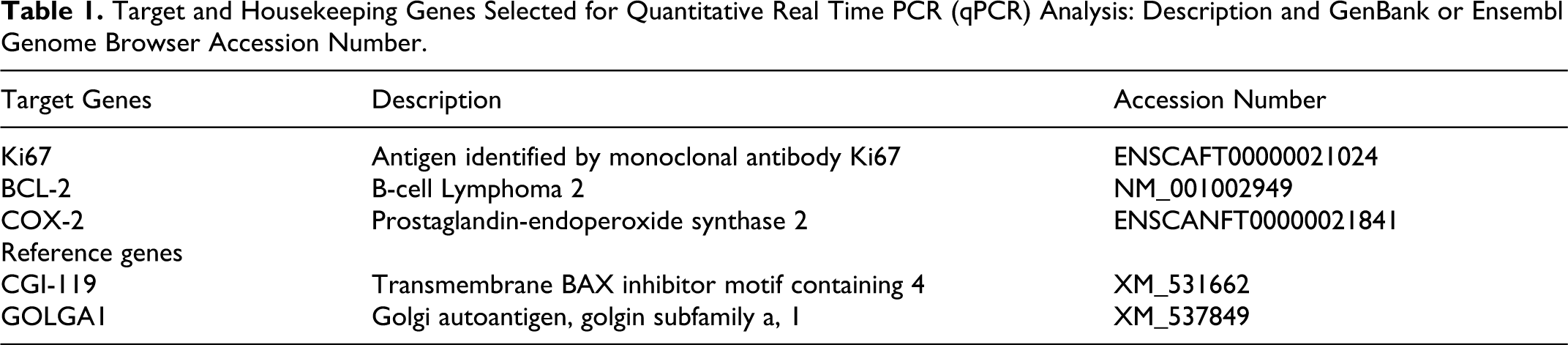
Primers and Human UPL Probes Used for Quantitative Real Time PCR (qPCR) Amplification of Target Genes.
Abbreviations: F, forward primer; R, reverse primer.
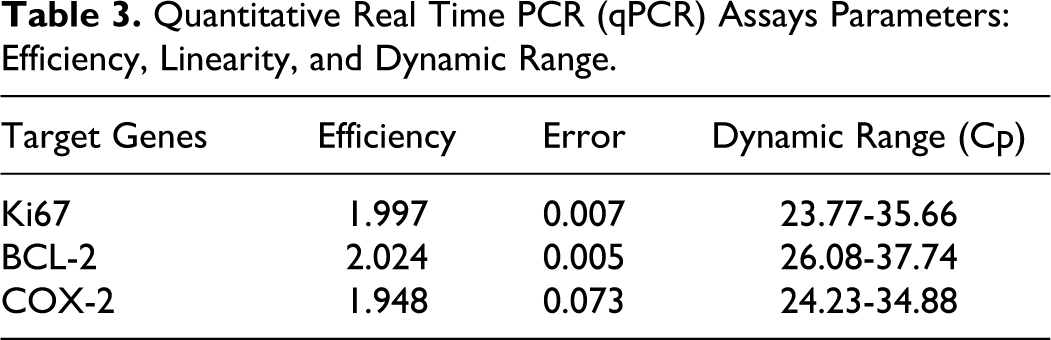
For the measurement of qPCR efficiency, calibration curves were obtained after the amplification of decreasing amounts of a cDNA pool serially diluted at four-fold intervals. qPCR efficiencies close to 2 and error values < 0.2 were obtained (Table 3). Canine CGI-119 and GOLGA1 were chosen as reference genes for the absence of tissues- and pathological states–dependent differences in the mRNA expression, as previously reported. 2
Quantitative Real Time PCR (qPCR) Assays Parameters: Efficiency, Linearity, and Dynamic Range.
qPCR was performed on 2.5 μL of 50-fold diluted cDNA (about 5 ng) in a final volume of 10 μL by using 1X LightCycler® 480 Probe Master containing 100 nmol/L of the selected human UPL probe and LightCycler 480 Instrument (Roche Diagnostics, Basel, Switzerland) with standard PCR conditions. Resulting data were analyzed and quantified with the LightCycler 480 software release 1.5.2 (Roche Diagnostics, Basel, Switzerland), using the second derivative maximum method. 55 The messenger RNA relative quantification (RQ) was performed by using the ΔΔCt method. 38 Tumor samples and surgical margins values were finally expressed as fold-changes (AU), such as normalized to the RQ mean value of control skin samples.
Statistical Analysis
All statistics were carried out using STATA 9.2 (StataCorp LP, College Station, TX).
The MCT was considered as the statistical unit. Bivariate analyses were used to process the data; because of the relatively small sample size, mainly associated to the mortality, multivariable analysis came out to be difficult to apply and results did not often converge. Variables such as age, sex, breed, and adjuvant therapy were not considered in this study. Given the low size of the G3 MCTs (n = 3) for the Patnaik grading system, the analysis was performed considering G1 versus G2 and G1 versus G2 + G3.
The association between prognosis (ie, mortality) and the histological grading was assessed by the Fisher’s exact test.
The distributions of values of MI, protein, and gene expression of Ki67, BCL-2, and COX-2, compared with both the histological grading systems and the outcome, were evaluated performing the Mann-Whitney test; in addition, the evaluation of the homogeneity of variances across the groups was carried out by the nonparametric test proposed by Brown and Forsythe, 7 replacing the mean with the median.
In order to categorize the parameters Ki67, BCL-2, and COX-2 and, therefore, identify a cut-off, ROC curve analysis was applied considering the mortality as reference variable; the area under curve (AUC), as an estimate of the performance of the test, and sensitivity (Se), specificity (Sp), positive and negative predictive values (PPV and NPV, respectively) were calculated. For MI, the cut-off = 5 was used. The direction and the strength of the associations were reported as the odds ratio with the 95% confidence interval (CI).
The Mann-Whitney test was used to compare mRNA expression in MCTs with control skin samples. The comparison between mRNA expression in MCTs and surgical margins was performed using the Wilcoxon signed-ranks test.
The Spearman’s Rho index was calculated to evaluate the correlation between protein and mRNA expression of Ki-67, BCL-2, and COX-2.
The survival time was investigated by means of Kaplan-Meier survival analysis; the two grading systems and the different categorized parameters were considered as stratification variables. The log rank test was used to compare the survival among groups.
Results
The time course of the study covered a period of 5 years (2006 to 2010), and a total of 53 MCTs from 51 dogs that met the inclusion criteria were enrolled in the study. Both male (18) and female (30) dogs were included in the study. The gender was not available for 3 cases. The mean age at surgery was 7 years (range, 3-13 years). The following breeds were represented: crossbred (14), Boxer (9), Labrador retriever (10), English Setter (3), Golden retriever (3), Cocker Spaniel (2), Dogo Argentino (2), Dachshund (1), Boston Terrier (1), Bouvier des Flandres (1), Bulldog (1), Cane Corso (1), Dalmatian (1), and Pitbull (1). For 1 dog the breed was not available. Two dogs were affected by two MCTs that were surgically excised at the same time. Both dogs were still alive at the end of the study period (24 and 17 months of follow-up time, respectively), and no local or distant recurrence of the tumor was observed.
The mean follow-up time was 18 months (range, 3-36). Thirty-eight patients were still alive at the end of the study period (follow-up range, 12-36 months); 2 of them received adjuvant chemotherapy. Three dogs showed development of an additional MCT at a site distant from where the original mass was observed and were still alive at the end of the study. Eight dogs showed clinical signs of MCT recurrence at the initial tumor site and died or were euthanized (2 dogs) for progressive MCT disease. All of these dogs received adjuvant chemotherapy. The mean survival time was 8.5 months (range, 3-13 months). Five patients died for other diseases, not correlated with MCT.
Histological Grading and MI
Twenty-eight MCTs were classified as grade 1 (G1), 22 as grade 2 (G2), and 3 as grade 3 (G3), according to Patnaik system. The full concordance among pathologists was 100% for the diagnosis of G3, 70% for the diagnosis of G2, and 75% for G1. In the remaining cases, the grade was attributed by the consensus of at least two pathologists.
Overall, 8 dogs died for progressive MCT disease; the mortality rate was 0% in G1 MCTs, 23% in G2 MCTs (5 cases), and 100% in G3 MCTs (3 cases). Considering the survival analysis stratified by Patnaik grading system (Fig. 1), G2 MCTs showed a survival probability of 0.91 (n = 21, 95% CI, 0.68-0.97) and 0.85 (n = 17, 95% CI, 0.61-0.95) at 6 and 12 months, respectively, stabilizing at 0.72 (n = 10, 95% CI, 0.44-0.87) after 15 months. The survival curves of G1 and G2 MCTs were significantly different (P = 0.006). The three G3 MCTs presented 3, 6, and 9 survival months, respectively.
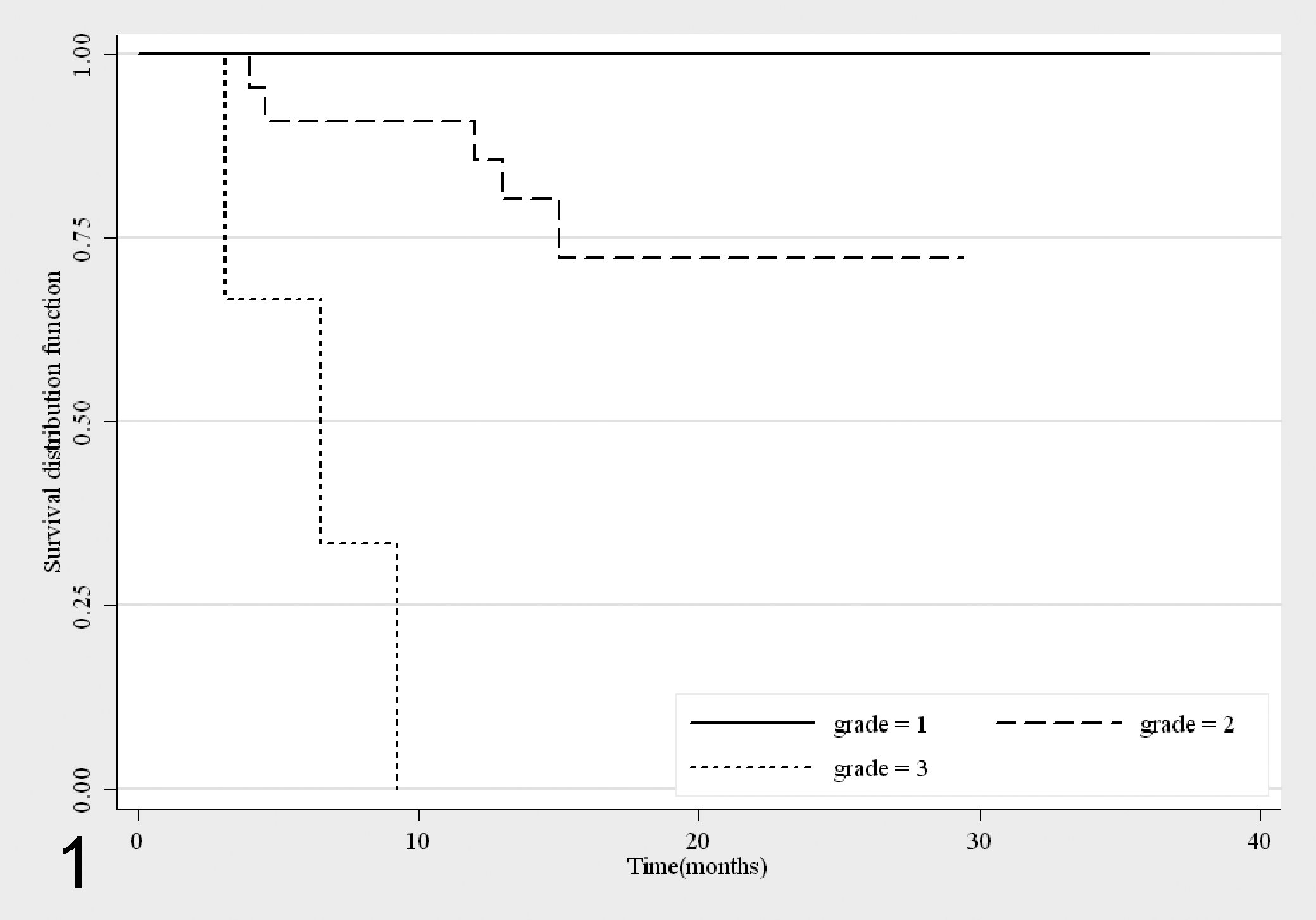
Survival curves for mortality due to mast cell tumors (MCTs), stratified by Patnaik grading system 50 (G1: n = 28; G2: n = 22; G3: n = 3).
By the 2-tier grading system, all the pathologists identified 46 MCTs as low (L) grade and 7 as high grade (H), and the mortality rate was 6% in the L grade (3 cases) and 71% in the H grade MCTs (5 cases). The survival probability at 12 months for L grade MCTs was 0.95 (n = 40, 95% CI, 0.83-0.98) and decreased to 0.92 from 15 months (n = 29, 95% CI, 0.77-0.97) until the end of the follow-up period. With regard to H grade MCTs, the 7 cases showed a quick decrease of the survival curve during the follow-up period, significantly different from L grade MCTs (P < 0.001) (Fig. 2).
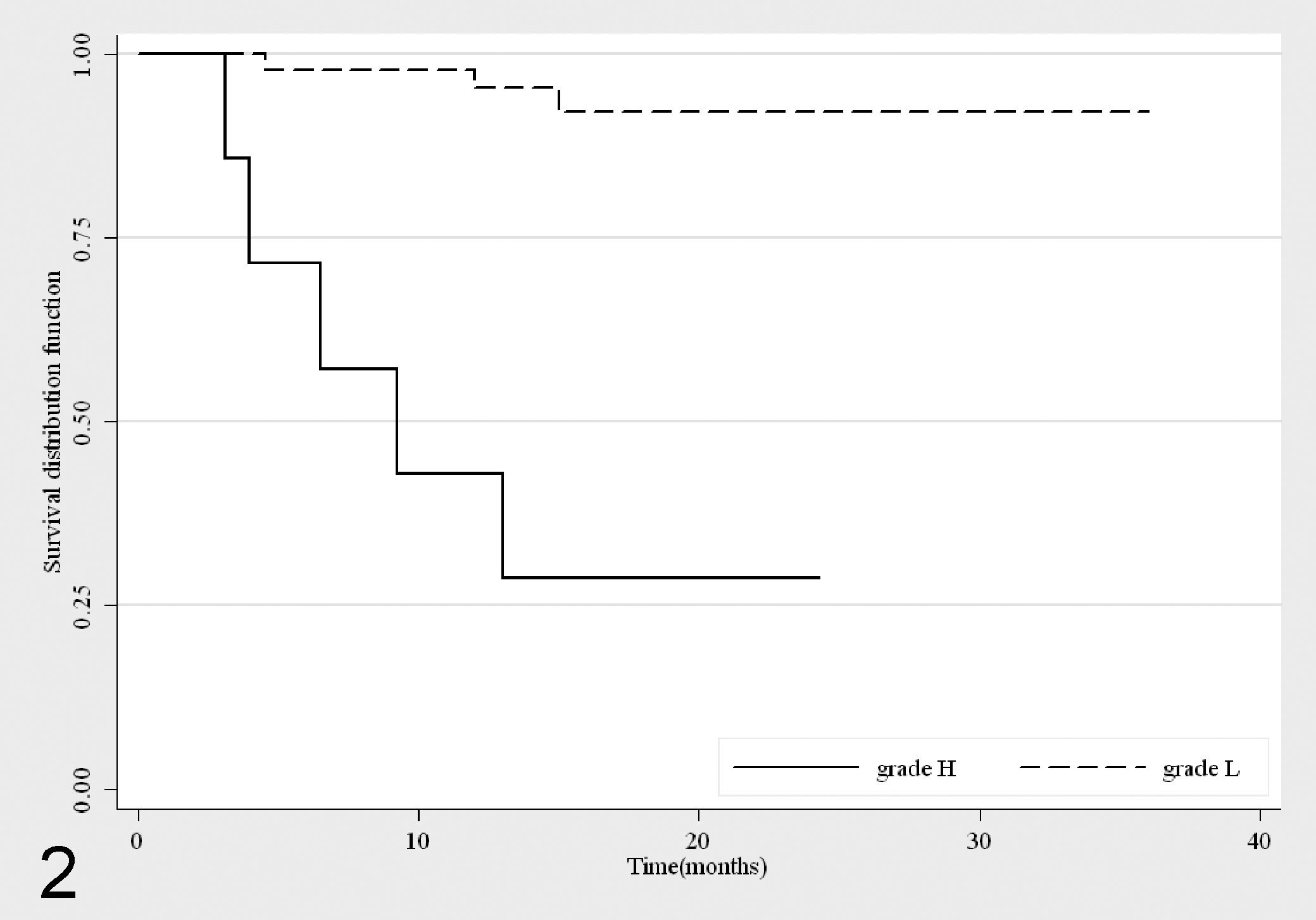
Survival curves for mortality due to mast cell tumors (MCTs), stratified by the 2-tier grading system 33 (L: n = 46; H: n = 7).
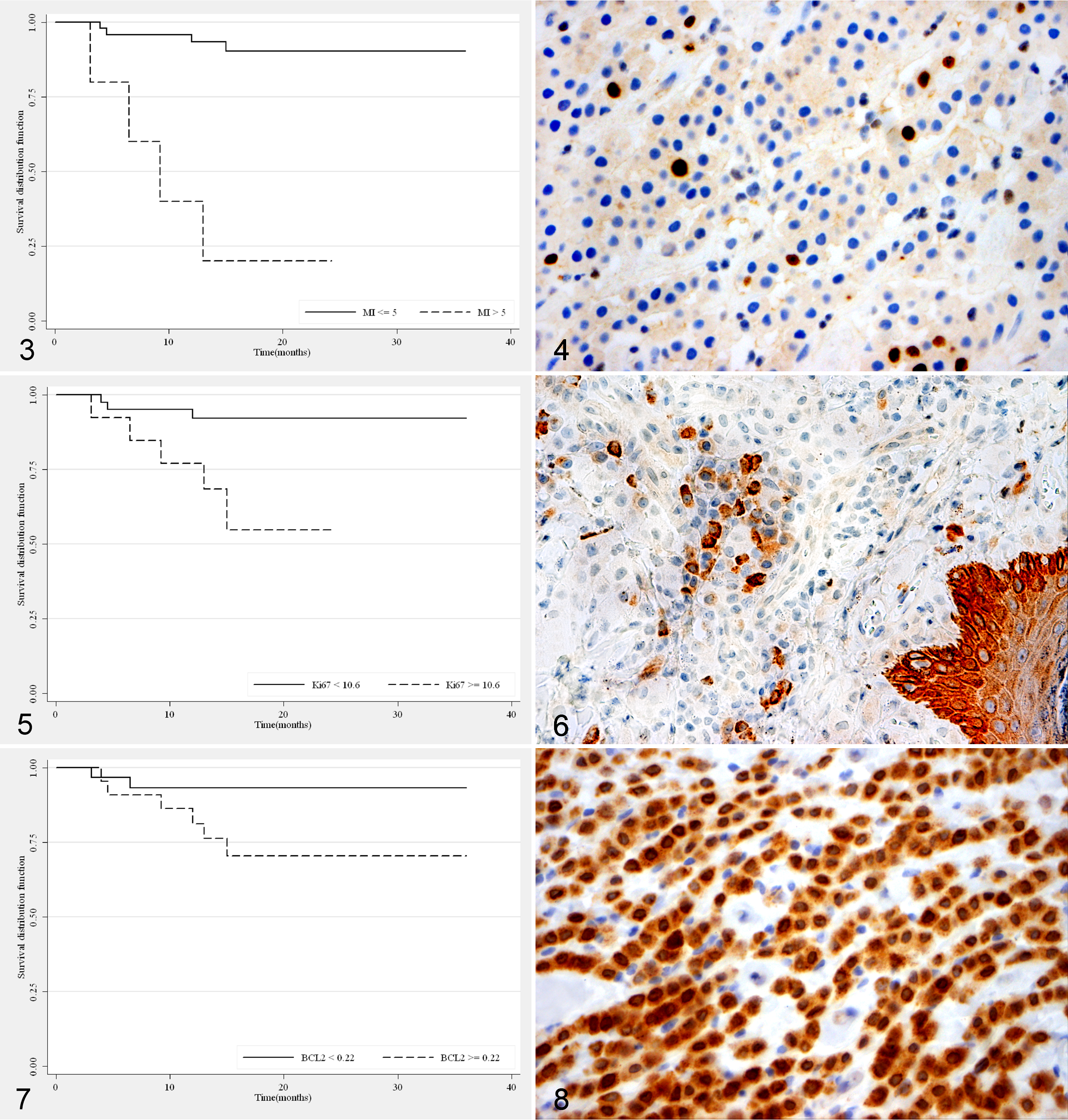
Both grading systems were significantly associated with prognosis (P < 0.001). Considering G2 and G3 altogether, the Patnaik grading showed a Se of 100% (95% CI, 63.1%-100%) and a Sp of 62% (95% CI, 46.5%-76.2%), while the 2-tier grading system had a Se of 62.5% (95% CI, 24.5%-91.5%) and a Sp of 95.6% (95% CI, 84.9%-99.5%). The Patnaik system had a PPV of 32.0% (95% CI, 14.9%-53.5%) and an NPV of 100% (95% CI, 87.7%-100%), while in the 2-tier grading system the PPV and NPV were 71.4% (95% CI, 29%-96.3%) and 93.5% (95% CI, 82.1%-98.6%), respectively. Combining the two grading systems, all G1 MCTs were classified as L grade, and similarly, all G3 MCTs were classified as H grade. The 82% of G2 MCTs were identified as L grade versus the 18% of H grade. MI was significantly associated with both grading systems (P < 0.001) and with prognosis (P = 0.001). The 53 tumors were divided into 2 groups on the basis of the MI: group 1 (n = 48) with MI ≤ 5 and group 2 (n = 5) with MI > 5. The mortality rate was significantly different between the two groups (P = 0.001): 92% of subjects with MI ≤ 5 were still alive while 80% of dogs with MI > 5 were dead at the end of the study period; furthermore, the two groups showed different survival curves (P < 0.001; Fig. 3). In our survey, the MI had a Se of 50% (95% CI, 15.7%-84.3%) and a Sp of 97.8% (95% CI, 88.2%-99.9%); the PPV was 80% (95% CI, 28.4%-99.5%) and the NPV was 91.7% (95% CI, 80.0%-97.7%).
Ki67
By immunohistochemistry, the nuclei of Ki67 positive cells were labeled brown, no immunolabeling was observed in the cytoplasm, and positive cells were uniformly distributed in the neoplastic tissue in all cases (Fig. 4). According to Patnaik grading system, the median value of the Ki67 index was 2.5, 6.0, and 20.6 for G1, G2, and G3 MCTs, respectively. A significant association between Ki67 protein expression and the grade was observed (P = 0.024) comparing G1 versus G2 + G3; a less pronounced difference was found comparing G1 versus G2 (P = 0.092). Interestingly, the variability of Ki67 protein expression in G1 group was significantly lower than G2 (P = 0.022) and G2 + G3 (P = 0.004). According to the 2-tier grading system, the Ki67 index was significantly lower in L grade than in H grade MCTs (P = 0.002). In addition, increased Ki67 counts were significantly associated with an increased incidence of MCT-related mortality (P = 0.049). The cut-off value that better discriminated MCTs associated with patient mortality was determined by the ROC curve’s analysis and was equal to 10.6. The area under the curve showed a value of 0.72 (95% CI: 0.52-0.92) as a measure of the global performance of the test. Using this cut-point, we correctly classified 79% of the 53 MCTs, and the Se and Sp were, respectively, 62.5% (95% CI, 24.5%-91.5%) and 82.2% (95% CI, 67.9%-92%). The PPV and NPV were 38.5% (95% CI, 13.9%-68.4%) and 92.5% (95% CI, 79.6%-98.4%), respectively. The probability of dying for a patient affected by a MCT with a Ki67 index ≥ 10.6 was 8 times higher than for a dog with a lower value (OR: 7.7, 95% CI, 1.16-57.22).
Overall, 3 out of 40 MCTs with Ki67 index < 10.6 caused MCT-related mortality; 6-month and 1-year survival probabilities were 0.95 (n = 39, 95% CI, 0.81-0.98) and 0.92 (n = 34, 95% CI, 0.77-0.97), respectively. Five out of 13 MCTs with Ki67 index ≥ 10.6 caused MCT-related mortality; the survival probability observed was 0.92 (n = 13, 95% CI, 0.57-0.98) at 6 months and 0.77 at 12 months (n = 10, 95% CI, 0.44-0.92). The two survival curves were significantly different (P = 0.006) (Fig. 5). A moderate correlation between Ki67 protein and gene expression (Spearman’s Rho = 0.45) was also observed. No statistically significant differences in Ki67 gene expression were detected among control skin tissue, surgical margins (SM), and MCTs. Furthermore, no association between Ki67 mRNA and Patnaik grading system was observed. Considering the 2-tier system, in the H grade MCTs a wide variability in Ki67 mRNA levels was recorded (P < 0.001). Similarly, the gene expression data were not associated with the outcome.
BCL-2
The BCL-2 protein was detected in the cytoplasm of neoplastic mast cells and positive cells were uniformly distributed in the neoplastic tissue in all cases (Fig. 6). However, no significant association between BCL-2 protein expression and both the grading systems nor health status was recorded. A significant lower BCL-2 mRNA expression in neoplastic tissues compared with surgical margins (P < 0.001) and control skin samples was observed (P < 0.001). No correlation between gene and protein expression data was detected (Spearman’s Rho = –0.11). The BCL-2 mRNA expression was significantly higher in G2 than in G1 MCTs (P = 0.012), as well as in the group of G2 + G3 than in G1 (P = 0.014). On the contrary, no differences were detected between L and H grades. Notwithstanding, the H grade MCTs showed a significantly higher variability in the BCL-2 mRNA expression than L grade MCTs (P = 0.015).
Different distributions of BCL-2 mRNA data between alive and dead dogs were highlighted being the median value 0.17 AU and 0.32 AU, respectively (P = 0.024). Furthermore, a significant different variability was observed in the two groups (P = 0.028). The best identified cut-off of the BCL-2 mRNA expression was 0.22 AU (AUC: 0.74; 95% CI, 0.56-0.93), which allows to correctly classify 65% of the cases. The Se and Sp of BCL-2 mRNA were 75.0% (95% CI, 34.9%-96.8%) and 63.6% (95% CI, 47.8%-77.6%), while the PPV and NPV were 27.3% (95% CI, 10.7%-50.2%) and 93.3% (95% CI, 77.9%-99.2%), respectively. In terms of odds ratio, the probability of dying for a dog with BCL-2 mRNA value ≥ 0.22 AU was 5 times higher than for a dog with a lower value (OR: 5.25; 95% CI, 0.79-57.25). Overall, 2 out of 30 cases with BCL-2 mRNA value < 0.22 AU died because of their MCTs. Six-month and 1-year survival probabilities were 0.97 (n = 30, 95% CI, 0.78-0.99) and 0.93 (n = 25, 95% CI, 0.76-0.98), respectively. Six out of 22 cases with BCL-2 mRNA value ≥ 0.22 AU died because of their MCTs. Six-month and 1-year survival probabilities were 0.91 (n = 21, 95% CI, 0.68-0.97) and 0.81 (n = 17, 95% CI, 0.57-0. 92), respectively. The log-rank test provided a P value of 0.057 (Fig. 7).
COX-2
By immunohistochemistry the COX-2 protein was detected in the cytoplasm of neoplastic cells, in 78% of the MCTs investigated (Fig. 8). A strong association between the distribution and the intensity of the COX-2 staining was observed (Spearman’s Rho = 0.79). Notwithstanding, neither an association with the tumor grade nor with the health status was highlighted. COX-2 mRNA was significantly higher in MCTs compared with surgical margins (P < 0.001) and control skin tissue (P < 0.001). No correlation between gene and protein expression was observed. COX-2 mRNA levels were not associated with either the grading systems. A higher variability of the mRNA values was observed in H grade than in L grade MCTs (P < 0.001). The same distribution of COX-2 mRNA values between dead and alive dogs was observed.
Detailed results of Ki67, BCL-2, and COX-2 expression, according with both the grading systems and prognosis have been summarized in Tables 4 and 5.
Association Between Ki67, BCL-2, and COX-2 (Protein and mRNA Expression) and the Histologic Grades.
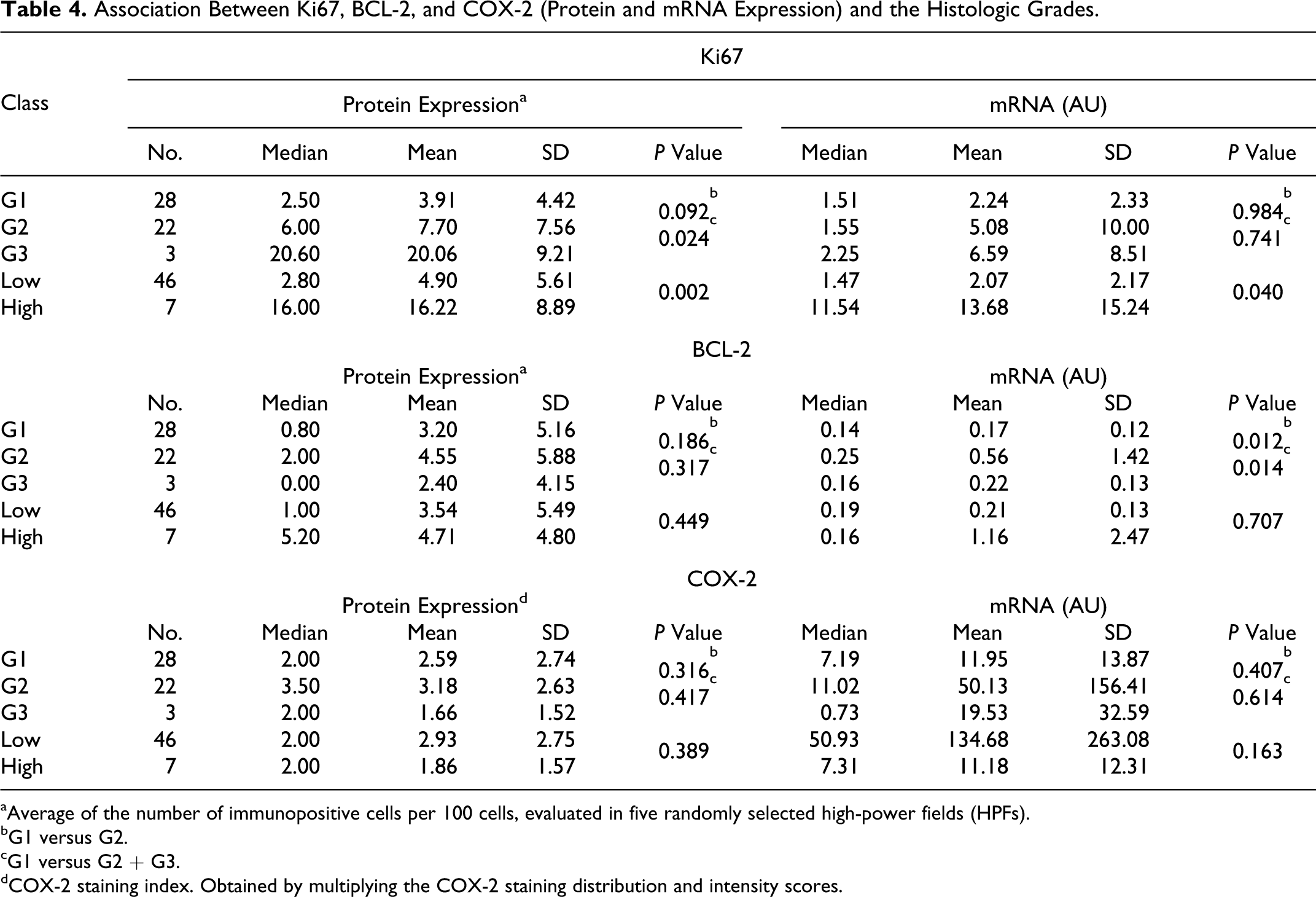
aAverage of the number of immunopositive cells per 100 cells, evaluated in five randomly selected high-power fields (HPFs).
bG1 versus G2.
cG1 versus G2 + G3.
dCOX-2 staining index. Obtained by multiplying the COX-2 staining distribution and intensity scores.
Association Between Ki67, BCL-2, and COX-2 (Protein and mRNA Expression) and the Mast Cell Tumors–Related Mortality.
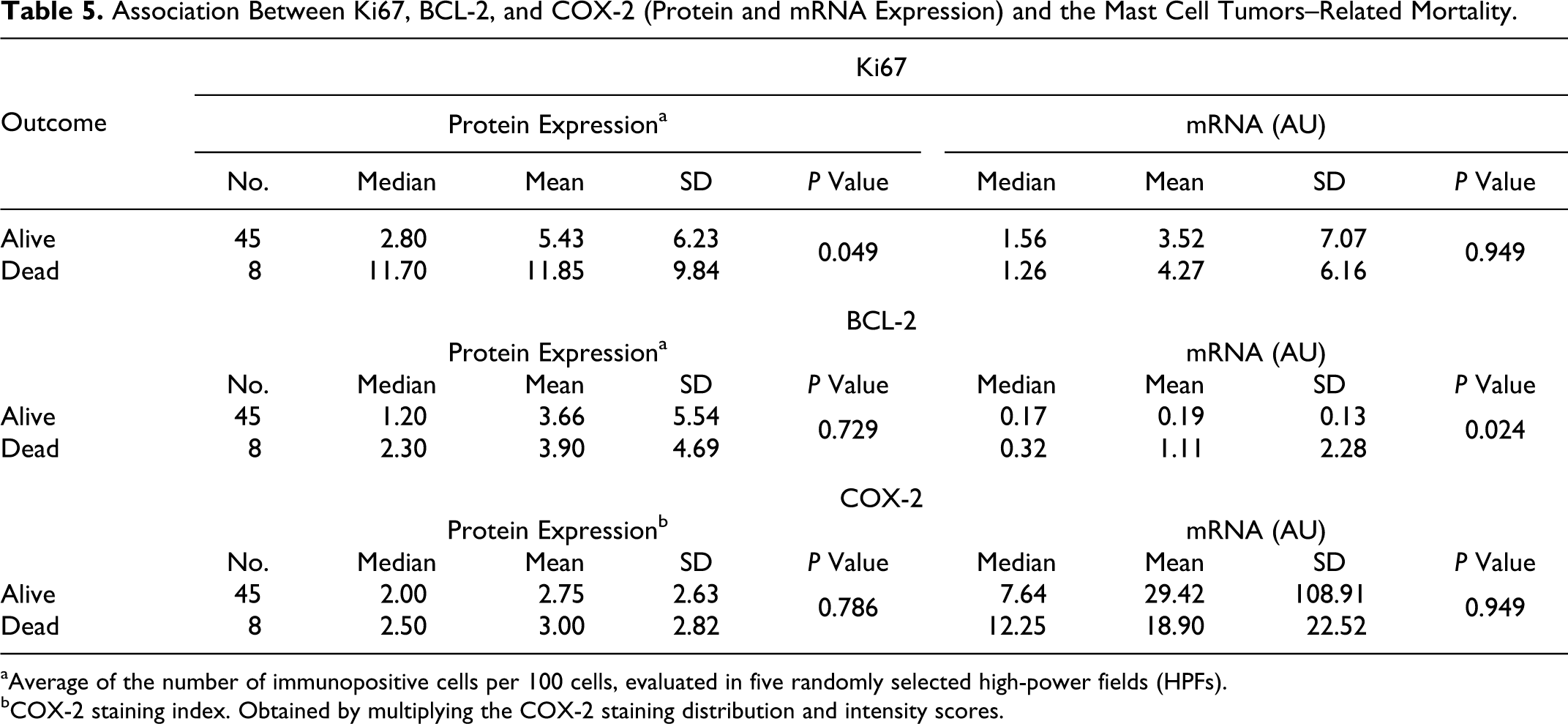
aAverage of the number of immunopositive cells per 100 cells, evaluated in five randomly selected high-power fields (HPFs).
bCOX-2 staining index. Obtained by multiplying the COX-2 staining distribution and intensity scores.
Discussion
In the present study, the prognostic significance of some markers involved in cell proliferation and apoptotic pathway have been investigated, considering both the protein and mRNA expression, in canine cutaneous MCTs.
Two different histologic grading systems have been applied, in order to compare their prognostic significance and the relationship with the expression of the markers investigated.
The Patnaik grading system, based on MI, localization, invasiveness, and cellular differentiation, emerged to be scarcely repeatable among different pathologists47,48 and of limited prognostic value for G2 MCTs, as these tumors behave unpredictably.64,71 In our survey, the most significant interobserver variation in grading was between G1 and G2 MCTs. All dogs with G1 MCT were still alive at the end of the study period, without evidence of tumor recurrence. On the contrary, all dogs with G3 MCT died for MCT recurrence and metastasis, with a mean survival time of 6 months (from 3 to 9). Five dogs out of 22 (23%) with a G2 MCT died for progressive MCT disease with a mean survival time of 10 months (from 4 to 15 months). These results are in accordance with previous published data, indicating that well-differentiated MCTs (G1) carry an excellent long-term prognosis, while anaplastic (G3) MCTs are characterized by poor long-term prognosis. 46 The behavior of intermediate (G2) MCTs is more difficult to predict. The majority of G2 MCTs are cured with wide surgical resection, 46 but between 5% and 22% metastasize.64,71 In order to eliminate the ambiguity of intermediate grade MCTs and to better predict the biological behavior, an alternative 2-tier grading system, based on the number of mitotic figures in combination with cellular atypia, has been recently proposed for cutaneous MCTs. 33 In our survey, the accordance between pathologists was strongly improved by the application of this 2-tier grading system. The cases classified as H grade MCTs showed a quick decrease of the survival curve during the follow-up period, significantly different from L grade MCTs; the mortality rate was 71% for H grade MCTs and 6% for L grade MCTs, with a PPV and NPV of 71.4% (95% CI, 29%-96.3%) and 93.5% (95% CI, 82.1%-98.6%), respectively. According with this new classification, H grade MCTs have a high risk of aggressive behavior, while low-grade tumors should be clinically monitored and further prognosticated with other prognostic markers. 33 In any case, other prognostic markers are strongly required to better define the prognosis of MCTs. Particularly, the MI and the Ki67 protein expression seem to have the highest prognostic value, as already observed in other studies.1,14,57,61,70 In our survey, MCTs with a MI > 5 had lower survival times and higher mortality rates than MCTs with MI ≤ 5. Furthermore, the probability of dying for a patient affected by an MCT with a Ki67 index ≥ 10.6 was 8 times higher than for a dog with a lower value. Previous studies have investigated the prognostic significance of Ki67 protein expression in MCTs, and different cut-off values have been proposed. Notwithstanding, different methods to count the Ki67 positive cells fraction have been used among different studies,39,59,61 leading to different cut-off values. Standardization of the Ki67 positive cells counting method among different pathologists should be helpful to determine the better cut-off value to help the clinician to assess the prognosis of MCTs.
The possible role of the anti-apoptotic molecule BCL-2 in canine cutaneous MCT progression has never been investigated before. In human patients affected by cutaneous mastocytosis, the expression of BCL-2 protein and mRNA were significantly enhanced if compared with healthy controls, suggesting that prolonged survival of mast cells may be associated with increased mast cell numbers. 23 In our study, a significant decrease of BCL-2 mRNA expression in neoplastic tissues compared with surgical margins and control skin samples was observed. This finding could be explained by the presence of different cells types in skin samples (SM and control skin), including non-neoplastic cells, such as the basal epithelium, that could constitutively express different levels of specific genes. On the contrary, samples collected from the core of the tumor did not include the epidermis. Thus, results of gene expression from surgical margins are mainly representative of skin expression, while those from tumor samples are mainly representative of neoplastic mast cells expression.
Among tumor samples, the mRNA levels were significantly higher in MCTs of grade 2 and 3 compared with grade 1, while no significant differences were observed between L and H grades. A possible explanation of this latter finding could be the relative low number of H grade MCTs as well as the high variability observed among the H grade MCTs. The gene expression appeared to be associated also with prognosis. Particularly, MCTs with BCL-2 mRNA amounts ≥ 0.22 AU had shorter survival time than those with lower values. Several studies on mast cell leukemia show a BCL-2 over-expression, pointing out the importance of this anti-apoptotic protein in mast cell homeostasis. 9 Other experiments reveal that upon stimulation via the aggregation of high-affinity IgE receptors (Fc∊RI), mouse mast cells synthesize the anti-apoptotic component A1 rather than BCL-2. 76 The role of survival/apoptosis-regulatory genes in mast cell activation has not been fully defined. There are some reports on the expression and regulation of BCL-2 in mast cells.8,42,79,80 Despite the wide-ranging ability of BCL-2 to promote cell survival, it is clear that in a number of circumstances, BCL-2 is not responsible for protecting cells from apoptosis.20,27 Therefore, it is possible that other members of the BCL-2 family may provide protective effects in some specific biological processes.
In our survey, no significant association between the BCL-2 protein expression and the histologic grade was observed. Furthermore, the expression of BCL-2 protein does not seem to be related with prognosis. These results could be linked to the high variability (SD, Tables 4 and 5) observed in each group of MCT (G1, G2, G3, L, H, alive, and dead), as well as to the relative low number of dogs with poor prognosis. Notwithstanding, looking at the median values of BCL-2 protein expression in different MCT groups, an increasing trend in G2 + G3 versus G1, in H versus L grades MCTs, as well as in dead versus alive dogs, has to be noticed. The lack of correlation between gene and protein expression data has been already reported in literature for other proteins12,69 and might be due to both post-transcriptional and post-translational mechanisms of regulation that involved BCL-2.58,74 A further explanation should be that mRNA relative quantification could be influenced by cells composition of the sample, which may include non-neoplastic cells, such as stromal, vascular, and inflammatory cells, that could constitutively express different levels of specific mRNAs. On the contrary, immunohistochemistry allows to localize and semi-quantify the protein exclusively in the neoplastic cells. In this respect, an approach that provides mRNA extraction and amplification from cells obtained through the laser capture micro-dissection technique might be useful in perspectives. Further investigations are in progress to better elucidate the role of BCL-2 family members in the pathogenesis of canine MCTs.
COX-2 seems to be involved in the processes of malignant transformation and tumor progression by affecting cell proliferation, mitosis, cell adhesion, apoptosis, immune-surveillance, and angiogenesis. 73 COX-2 protein expression has been described in several canine tumors.11,28,29,41,51 Even though the role of COX-2 and its derived metabolites in tumorigenesis appears clear, the prognostic significance of COX-2 expression in cancer has not been clearly established. Some studies reported that COX-2 immunohistochemical detection was correlated with poorer prognosis in canine and feline invasive mammary carcinomas37,43,53,54 and canine appendicular osteosarcomas. 45 Considering the evidence in supporting a role for COX-2 in oncogenesis, targeted inhibition of COX-2 might represent an interesting option for chemoprevention and/or chemotherapy. 77 In human oncology, COX-2 is recognized as a promising pharmacological target to prevent and treat several human tumors.21,22 NSAIDs have been shown to be as efficacious as conventional chemotherapy in dogs with TCC and oral squamous cell carcinoma.35,62
A previous immunohistochemical study stated that canine MCTs have no significant degree of COX-2 expression, suggesting a lack of efficacy of these tumors to COX-2–selective inhibitors. 24 In the present study, the COX-2 protein was detected by immunohistochemistry in the cytoplasm of neoplastic cells in 78% of the MCTs investigated. Furthermore, we observed a significant COX-2 mRNA up-regulation in MCTs toward surgical margins and control skin. These evidences suggest that COX-2 could play a role in MCTs oncogenesis and that COX-2 inhibitors may be useful in the prevention and/or treatment of canine cutaneous MCTs. However, determination of COX-2 expression levels in MCTs does not appear to be useful as a prognostic biomarker. Further investigations are needed to clarify the possible role of COX-2 in MCTs pathogenesis and to assess the use of NSAIDs as chemotherapeutic and chemopreventive agents in dogs.
In conclusion, this study provides novel information about the expression of biomarkers involved in neoplastic cells proliferation, survival, and apoptosis in canine cutaneous MCTs. Both protein and gene expression levels have been investigated by immunohistochemistry and qPCR in neoplastic tissues, and correlations with tumor differentiation and prognosis have been considered. The prognostic value of grading, MI, and Ki67 protein were confirmed. However, no single marker can accurately predict the biological behavior of canine MCTs.
Footnotes
Acknowledgements
The contribution of Dr Marina Martano to surgeries and collection of follow-up data, Dr Chiara Nassuato to immunohistochemical evaluation, Dr Rosa Maria Lopparelli to biomolecular analysis, and Dr Elisa Baioni to statistical analysis is kindly acknowledged. Moreover, we would like to thank the personnel of the Histopathology Laboratory of the Istituto Zooprofilattico Sperimentale delle Venezie for their skillful technical assistance, as well as Prof Ilaria Iacopetti and Drs Helen Poser, Barbara Carobbi, and Patrizia Cristofori for their contribution in providing part of tumor and control samples included in the present investigation.
Declaration of Conflicting Interests
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by a grant from the Italian Ministry of Health to the Istituto Zooprofilattico Sperimentale delle Venezie (RC IZSVE 09/06).
