Abstract
Cutaneous round cell tumors in goats present a diagnostic challenge. In this article, we provide a description of caprine cutaneous mast cell tumors (MCT) and histiocytomas, and report on the validation of anti-human antibodies to CD117/KIT and Iba1 by immunohistochemistry on a range of caprine tissues. Cells immunolabeled for CD117/KIT included resident mast cells in normal lung and skin, interstitial cells of Cajal (intestine), and neuronal cell bodies (brain). Cells immunolabeled for Iba1 included resident macrophages in many tissues including normal lung, dendritic cells (hemolymphatic tissues), Kupffer cells, and microglia. Of 5 cutaneous MCT, only one had metachromasia of cytoplasmic granules; however, neoplastic cells of all 5 MCT had positive immunolabeling for CD117/KIT. The CD117/KIT immunolabeling pattern was predominately focal paranuclear in 3 cases, and cytoplasmic or membranous in 1 case each. Two histiocytomas were identified and had strong positive immunolabeling for Iba1 but not CD117/KIT. All 7 cutaneous round cell tumors described herein occurred in goats less than 4 years of age; the 2 cutaneous histiocytomas were in goats less than 14 months of age. Neither of the cutaneous histiocytomas recurred within 24 months of surgical removal.
Domestic goats remain important food animals for both milk and meat production and are also increasingly kept as pets and used in urban landscaping due to their grazing capabilities. Overall goat populations were estimated to increase from 2.30 million to 2.64 million in the United States and from 751 million to over 1 billion animals globally from 2000 to 2017. 5 With a larger population and the resulting need for advanced veterinary care for companion animals or valuable breeding stock, knowledge regarding neoplasia within the goat population has become more important. Previous studies from the Oregon Veterinary Diagnostic Laboratory (OVDL) reported on the overall prevalence of different types of neoplasia among submissions of goat necropsies and biopsies and provided detailed classification of the caprine lymphoma subtypes observed in this population. 11,14
In addition to lymphoma, the previously published studies had identified 7 round cell tumors that did not have an identifiable, proliferative lymphocyte population, as indicated by absence of immunolabeling for CD3 and CD79a by immunohistochemistry (IHC). 11,14 Interestingly, these round cell tumors occurred in the skin of goats under 4 years of age. For these tumors, differential diagnoses included cutaneous histiocytoma, mast cell tumor (MCT), plasmacytoma, double-negative/null lymphoma, and amelanotic melanoma. 11 Reports of a single case each of cutaneous MCT and cutaneous histiocytoma have been published before 2,23 but did not contain information regarding immunophenotypic characterization of the neoplastic cell population.
The tyrosine kinase CD117/KIT is a type III, transmembrane receptor that is expressed on mast cells, some hematopoietic stem cells, interstitial cells of Cajal of the gastrointestinal tract, melanocytes, and neoplasms of these cells and others. 17 CD117/KIT plays a role in signal transduction of its ligand stem cell factor, including the development and function of mast cells, and is conserved across a wide range of species. 13,24 Immunolabeling for CD117/KIT is widely used in the identification of cell types, classification of neoplasms, and the characterization of tyrosine kinase expression patterns in MCT. 17 Three distinct cell-associated labeling patterns have been described in cutaneous MCT of dogs, cat, and horse and may have prognostic relevance in the dog and cat. 4,12,21,26
The ionized calcium-binding adapter molecule (Iba)-1 is a calcium-binding protein that is expressed in the cytoplasm of tissue macrophages, dendritic cells, and microglial cells. 8 While expression is increased in the activated cell state, it can be readily detected in resting cells of formalin-fixed paraffin-embedded tissues by immunohistochemistry and bright field microscopy. 1,3 Immunolabeling for Iba1 is widely used to characterize leukocyte infiltrates in inflammatory responses or neoplastic conditions as well as in the identification and characterization of histiocytic neoplasms. 9,19,27,29 Commercially available antibodies raised against human Iba1 have shown cross-reactivity in tissues of dogs, cats, and horses. 15,16,22,27
Herein, we provide descriptions and images of the histomorphologic and immunophenotypic characteristics of cutaneous MCT and histiocytoma in goats. Furthermore, we provide evidence in support of the prior, provisional diagnosis of MCT and cutaneous histiocytoma in the skin of goats using IHC for CD117/KIT and Iba1, respectively. We report on the validation of 2 anti-human antibodies for immunolabeling of CD117/KIT and Iba1 in caprine tissues using an array of normal caprine tissues; and in MCT (CD117/KIT only) and histiocytoma (CD117/KIT and Iba1). Finally, we provide a brief update on prevalence of tumor types among caprine neoplasms submitted to the OVLD.
Materials and Methods
Case Identification
Cases of neoplasia in goats diagnosed at the OVDL were identified as previously described. 14 However, we expanded our search for caprine cases with a diagnosis of “neoplasia” to include submissions received from 2012 through the end of April 2020. All submissions were either routine surgical biopsy submissions or fixed tissues collected during necropsies, either in the field or at the OVDL. All tissues were fixed in 10% neutral buffered formalin for at least 24 hours, or longer for any specimens that showed evidence of incomplete fixation (such as red to pink central tissue or red blood). Formalin-fixed samples from both surgical biopsy and necropsy cases were processed to paraffin-embedded blocks and stored at room temperature. Tissues used for validation of the anti-human antibodies to CD117/KIT and Iba1 were harvested within 3 hours of death from a 2-day-old, female Boer goat (2.3 kg) as part of a routine postmortem examination. Only tissues without gross or histologic lesions were included in the generation of normal caprine tissue blocks; 27 samples from 22 tissues were combined into 3 composite blocks. All tissues used in this project were accessed according to OVDL’s policies and procedures.
Histologic and Histochemical Examination
Routine hematoxylin and eosin (H&E) stained sections (3–5 µm thick) and special stains, as needed, were created using manual staining (special stains) or a linear, automated slide stainer (Thermo Shandon GLX) and examined by bright field microscopy. Sections (3 µm thick) of 7 non-lymphoma round cell tumors, all occurring in the skin, were stained with toluidine blue or Giemsa as previously described. 4 Metachromasia, typical of mast cell tumors, was defined as clearly visible, deep purple, cytoplasmic granules on high power (400×) bright field microscopy. Negative metachromatic staining was defined as the absence of such cytoplasmic granules.
Immunohistochemistry
We validated rabbit polyclonal anti-human antibodies against CD117/KIT (Dako; catalog #A4502) and Iba1 (Wako Chemical; catalog #019-19741) for use on goat tissues by immunolabeling of a wide array of morphologically normal caprine tissues including tissues containing cells known to express CD117/KIT, Iba1, or both in other host species. 8,17,22,27 These tissues included cerebral cortex (superficial and deep), cerebral white matter, cerebellar cortex, cerebellar white matter, brainstem, eye, small and large intestine, tongue, esophagus, reticulum, rumen, abomasum, heart, lung, liver, spleen, lymph node, kidney, urinary bladder, uterus, ovary, thyroid gland, and skeletal muscle.
Immunolabeling for CD117/KIT was performed at the OVDL and included the morphologically normal caprine tissues, all 7 cutaneous round cell tumors described in this study, and a canine MCT. All formalin-fixed, paraffin-embedded tissues were sectioned at 4 to 5 μm. Sections were collected on charged slides, baked at 60 °C for 1 hour, and rehydrated (2 changes of xylene, 2 changes of 100% ethanol, 1 change of 80% ethanol and water). High-temperature antigen retrieval was performed in a microwave pressure cooker (Viking Tender cooker) using Dako Target Retrieval solution (catalog # s1699) for 10 minutes after pressure was reached. The pressure cooker was slowly vented and the container with the slides allowed to cool for 20 minutes at room temperature. Slides were placed on the Dako Autostainer and washed in Tris-buffered saline, 0.1% Tween 20 Detergent (TBST; Biocare Medical; catalog #TWB945M) followed by 3% H2O2 (Sigma-Aldrich; in TBST) 10 minutes, and Dako serum-free protein block (catalog #x0909) for 10 minutes. The primary polyclonal rabbit anti-human CD117/KIT antibody (Dako; catalog #A4502) was diluted 1:50 in Dako antibody diluent (catalog #s3022) and applied for 30 minutes at room temperature followed by MaxPoly-One polymer HRP rabbit (MaxVision Biosciences; catalog #MP03) for 10 minutes at room temperature. Slides were rinsed of in TBST and the chromagen Nova Red (Vector Laboratories; catalog #SK-4800) was applied for 5 minutes. Slides were then washed in distilled water and counterstained with hematoxylin (Dako; catalog #s3302) diluted 1:10 in distilled water for 5 minutes, rinsed in distilled water, rinsed in TBST to blue, dehydrated in alcohol to xylene, coverslipped in mounting medium (Dako; catalog #CS703), and dried. Sections of a previously diagnosed canine mast cell tumor were used as methods control tissue for the IHC assay. Normal caprine skin (mast cells) and intestine (interstitial cells of Cajal) served as control tissues for specificity. Negative controls for each slide were generated by substituting Biocare Medical Polymer Negative Control Serum (Mouse and Rabbit, catalog #NC499L) for the primary antibody solution.
Immunolabeling for Iba1 was performed at the Animal Health Diagnostic Center within the College of Veterinary Medicine at Cornell University. Portions of the 2 putative cutaneous histiocytomas were removed from the original diagnostic paraffin block to create a composite block. Unstained sections of the cutaneous histiocytoma composite block and the 3 composite blocks of morphologically normal caprine tissue were submitted with the request that the slides be immunolabeled for Iba1 and returned with appropriate positive and negative controls. Immunohistochemical labeling using an automated platform was performed (Bond-Max IHC/ISH platform, Bond polymer refine DAB kit). Rabbit polyclonal anti-human Iba1 antibody (Wako Chemical; catalog #019-19741) was applied to unstained, paraffin-embedded sections. Sections were dewaxed (Bond dewax solution; Leica Biosystems; catalog #AR9222) and heat epitope retrieval (Bond epitope retrieval system; Leica Biosystems; catalog #AR9640) was performed for 10 minutes. A 5-minute-block with 3% hydrogen peroxide (Leica Biosystems; catalog #DS9800) followed. The Iba1 antibody was applied for 15 minutes at a dilution of 1:3000 followed by Leica Bond Polymer refine detection (catalog #DS9800) for 10 minutes. 3′,5′-Diaminobenzidine (DAB) was applied for 5 minutes (Leica Biosystems; catalog #DS9800) and counterstained with hematoxylin for 5 minutes. Iba1 was validated in a bovine tissue microarray to determine the immunoreactivity. Incubation of serial sections of tissue with an irrelevant species and isotype-matched antibody served as negative controls; canine lymph node was used as a methods control; normal caprine brain (microglia), liver (Kupffer cells), and lung (macrophages) were used as positive control tissue for specificity.
Positive immunolabeling was defined as either dark red (CD117/KIT) or dark brown (Iba1), fine punctate to homogenous chromogen in the cytoplasm and/or along the cell membrane. Very faint, homogenous, red or brown labeling without a specific cyto-anatomical location was interpreted as “nonspecific” settling of antibody and/or chromogen (background labeling). Based on previously published studies using the same antibodies on tissue sections from dogs and cats, we anticipated positive immunolabeling of resident mast cells, interstitial cells of Cajal, and neurons in the brain for CD117/KIT and microglia in the brain and macrophages/histiocytes/dendritic cells in a variety of tissues for Iba1.
Results
Cases
Between 2012 and 2018, 51 tumors from 49 goats were received at the OVDL (Supplemental Table S1). The prevalence of neoplasia by tupmor type in recent submissions to the OVDL was similar to that previously reported. 11,14 The exception was an increase in tumors of the reproductive tract, mostly leiomyosarcomas. Seven of 152 (4.6%) tumors submitted from goats since 1987 were diagnosed as non-lymphoma round cell tumors; 5 of these cases were diagnosed as MCT and 2 cases as cutaneous histiocytomas.
Breeds with cutaneous MCT included Nubian (
Histologic and Histochemical Examination
Histomorphologic, histochemical, and immunohistochemical characteristics of each tumor are described in Table 1 and Supplemental Table S2.
Clinical and Pathologic Findings in Non-Lymphoma Round Cell Tumors in Caprine Skin.

Abbreviations: RBC, red blood cell; N/a, not available.
a Nuclear size compared to the diameter of a red blood cell [×RBC]).
b Mitotic count was determined by examining ten 400× high-powered fields (2.37 mm2).
MCT
On H&E-stained sections, the masses diagnosed as MCT were well-demarcated, with occasional infiltrative clusters of round cells along the periphery. Tumor cells were arranged in dense sheets and had medium-sized, central, round nuclei with coarsely stippled chromatin and single nucleoli (Figs. 1, 2). Cellular features varied, ranging from large cells with a histiocytoid appearance (
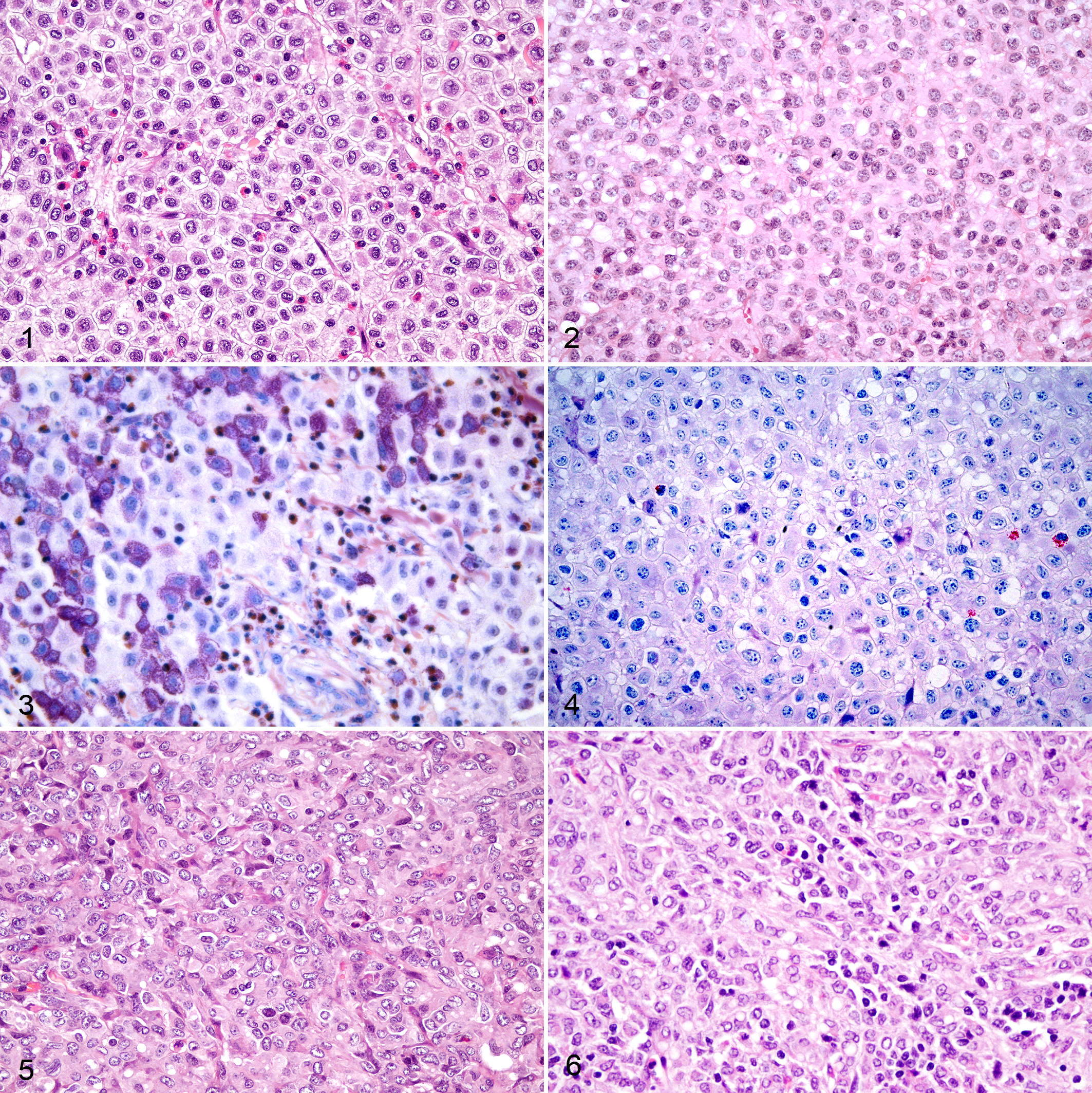
Mast cell tumors, skin, goat.
Histiocytomas
On H&E-stained sections, masses diagnosed as histiocytoma were well-demarcated with mild peripheral infiltrative rows of neoplastic cells (Figs. 5, 6). The neoplastic cells in both masses were arranged in dense sheets expanding dermal collagen. These cells had variably distinct cellular borders, abundant finely vacuolated cytoplasm with no visible granules, large oval nuclei with coarse chromatin and single nucleoli, marked anisokaryosis, and moderate nuclear pleomorphism. There were occasional binucleate and multinucleate cells. The mitotic count in 10 consecutive 400× HPF (2.37 mm2) was 14 and 24, respectively. Neither tumor had appreciable numbers of eosinophils. One tumor had small numbers of lymphocytes present (Fig. 6). These putative histiocytomas occurred in the skin over the ventrum (left) in a 14-month-old female Boer goat and in the right lower eyelid in a 7-month-old castrated male mixed breed goat. In both cases, tumor cells were present along the section margins, suggesting incomplete excision.
In sections stained with Giemsa and with toluidine blue, neither histiocytoma had metachromatic granules visible in neoplastic cells (Table 1; Supplemental Table S2). Mast cells with medium-sized nuclei and dense chromatin were apparent in the dermis away from the tumor bulk in Giemsa and toluidine blue stained sections from both cases and interpreted as normal resident cells.
Validation of Immunohistochemistry
Validation of Anti-human CD117/KIT Antibody for Use on Goat Tissues
In normal tissues from a goat unrelated to this study, large round cells with abundant, dark red cytoplasmic granules immunolabeled for CD117/KIT were interpreted as resident mast cells and were found in a variety of tissues including the interstitium of the lung (Fig. 7), splenic red pulp, submucosa of the digestive tract (rumen, small intestine, and large intestine), gut-associated lymphoid tissue, and dermis (Supplemental Table S3). Other cell types that labeled for CD117/KIT included interstitial cells of Cajal in superficial and deep ganglia of stomach and intestine (Fig. 8), neurons in the cerebrum and cerebellum including occasional Purkinje cells, ependymal cells (Fig. 9), primary oocytes of ovarian follicles (Fig. 10), and epithelial cells of uterine glands, all of which had diffuse cytoplasmic staining with exception of ependymal cells (only ciliated surface immunolabeled; Fig. 9). Neoplastic cells in a canine MCT immunolabeled predominantly along the cell membrane (CD117 pattern I; positive control for method; Fig. S1). Negative method controls (sections with primary antibody omitted) and specificity controls (tissues exposed to primary antibody but not expected to have cells expressing appreciable levels of CD117/KIT) showed no specific labeling (Supplemental Figs S2–S5; Supplemental Table S3).
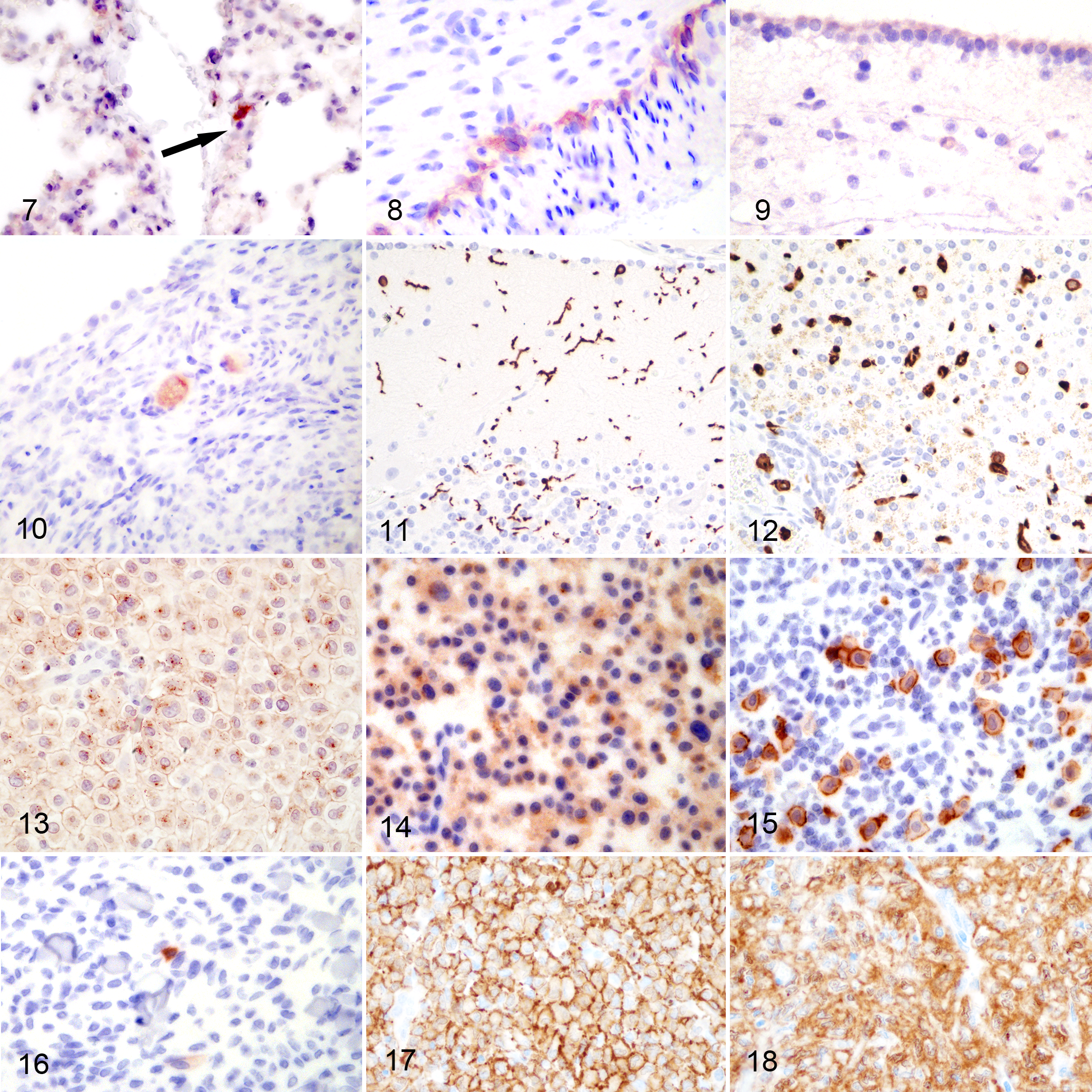
Normal tissues, goat. Immunohistochemistry for CD117/KIT.
Validation of Anti-human Iba1 Antibody for Use on Goat Tissues
In normal tissues from a goat unrelated to this study, large stellate to fusiform to round cells with abundant, dark brown cytoplasm immunolabeled for Iba1 were interpreted as microglia (Fig. 11), macrophages/Kupffer cells (Fig. 12), dendritic cells, or Langerhans cells and were present in varying numbers in examined tissues including brain, spleen, digestive tract (esophagus, rumen, reticulum, abomasum, small intestine, and large intestine), gut-associated lymphoid tissue, lymph node, liver, trachea, lung, kidney and urinary bladder, skeletal muscle, heart, reproductive organs, and skin (Supplemental Table S3). All cells with positive immunolabeling for Iba1 had intense, diffuse to coarse granular, cytoplasmic immunolabeling. No other cell types were immunolabeled for Iba1. Dendritic cells in a canine lymph node had intense positive immunolabeling of the cytoplasm (positive control for method; Supplemental Fig. S6). Negative methods controls (sections with primary antibody omitted) and specificity controls (tissue exposed to primary antibody but not expected to have cells expressing appreciable levels of Iba1) showed no specific immunolabeling (Supplemental Figs. S7, S8; Supplemental Table S3).
Immunohistochemical Labeling of Tumors
MCT
In the MCT, the neoplastic round cells had fine, granular immunolabeling for CD117/KIT. Immunolabeling was classified as paranuclear, diffuse, or membranous.
12
Most MCT had multiple CD117/KIT labeling patterns, but all had one predominating pattern with more than 50% of the neoplastic cells exhibiting that pattern. A paranuclear distribution pattern, defined as a focal area of dense labeling within the cytoplasm adjacent to the nucleus with reduced membranous labeling, similar to CD117/KIT pattern II as reported in dogs, was most common (
Histiocytomas
The neoplastic cells in the histiocytomas, previously reported not to immunolabel for CD3 and CD79, 11,14 were not immunolabeled for CD117/KIT, although scattered cells interpreted as resident mast cells (as described above for Giemsa and toluidine blue stained sections) were present in the surrounding skin and served as internal positive specificity control (Fig. 16). Neoplastic round cells had moderate to intense immunolabeling for Iba1 that was membranous and to a lesser extent cytoplasmic (Figs. 17, 18). Other histiocytic cells (presumed to be macrophages) in the adjacent conjunctival submucosa of one sample also had intense, diffuse to coarse granular immunolabeling of the cytoplasm for Iba1. No immunolabeling was present in serial control sections incubated with nonspecific serum.
Discussion
CD117/KIT has been comprehensively evaluated as useful in diagnosing canine, feline, and equine MCT. 4,12,22 We suggest that IHC for CD117/KIT also has utility in the diagnosis of caprine MCT. The CD117/KIT labeling of resident mast cells, interstitial cells of Cajal, neurons, and uterine glands in morphologically normal caprine tissues are consistent with previous reports in humans, dogs, and cats. 13,17,22,26 This, in conjunction with the absence of immunolabeling of any other cell types or structures, suggests that the antibody used in our IHC assay is compatible for use in caprine tissues. A prior study reported no immunolabeling for CD117/KIT in a canine ovary with an oocyte and weak positive immunolabeling in a feline ovary. 18 CD117/KIT expression in subependymal astrocytes in adult macaques has been previously reported, but we were unable to find previous reports of CD117/KIT expression in the apical surface of ependymal cells. 30 One possible explanation is that the cilia of ependymal cells are immunolabeled. 10 However, we cannot definitively rule out cross-reactivity of the anti-human CD117/KIT antibody used here with a different protein on the apical surface of caprine ependymal cells.
The consistent labeling of neoplastic round cells with typical cytological features combined with the presence of eosinophils indicates a diagnosis of MCT in 5 of these tumors. The lack of metachromatic granules is a common feature of some feline MCT. 25 The 3 CD117/KIT labeling patterns we observed are consistent with what is reported in MCT in other species. 4,12,18,26 Follow-up information on clinical outcomes were not available for these caprine MCT. However, a previously published case report of a cutaneous mast cell tumor in a 6-week-old, female, Nubian goat had no recurrence approximately 2 years after diagnosis. 2
Iba1 is widely used as marked for cells of the macrophage/histiocyte lineage in the characterization of inflammatory infiltrates. 1,8,9 This appears to be the first report of Iba1 immunolabeling in caprine tissues. The anti-human Iba1 antibody used in this study consistently and intensely labeled cells with morphological characteristics of microglia, macrophages, histiocytes, dendritic cells, Langerhans cells, and cells of the reticuloendothelial system in morphologically normal caprine tissues, in numbers and distributions patterns consistent with those reported for other species. 1,8,9 As no other structures or cells were labeled, we interpreted the immunolabeling as specific and the marker as a potentially useful tool in both diagnostic and research settings.
In the 2 cutaneous round cell tumors that were not immunolabeled for CD117/KIT, CD3, or CD79, the moderate to intense Iba1 immunolabeling of neoplastic cells confirms the diagnosis of cutaneous histiocytoma in these 2 goats. Both of these goats had a disease-free interval of 24 months (no further follow-up data were available). In dogs, cutaneous histiocytomas are benign proliferations that regress spontaneously. 6 This is also consistent with the previously reported cutaneous histiocytoma from a 14-month-old French Alpine buck, 23 in which a mass expanding the dermis of the scrotum had been removed with incomplete margins, but the remaining tumor regressed over the next 6 months.
Multinucleate cells, large cells, and high mitotic counts as well as cellular pleomorphism and anisokaryosis are all seen in canine cutaneous histiocytoma, especially before the onset of regression. These features can occur in MCT as well. Other differential diagnoses considered based on examination of H&E-stained sections were null or double-negative lymphoma (negative for CD3 and CD79a), plasmacytoma, and amelanotic melanoma. Null-cell lymphomas in dogs are commonly multicentric or diffuse and both null-cell lymphoma and amelanotic melanocytic tumors generally carry a poor prognosis with rapid progression in humans and dogs. 7,20,28 Immunolabeling for CD117/KIT has not been reported in neoplastic lymphocytes but has been reported in canine melanoma. Neither melanoma nor lymphoma in cats and dogs are immunolabeled for Iba1. 19 Eosinophilic infiltrates are not a common or prominent feature of melanomas in cats or dogs.
Lymphomas, MCT, and histiocytomas occur in the skin of goats and distinguishing these entities may require immunohistochemistry. Importantly, while all MCT had large numbers of eosinophils, metachromatic granules were usually absent in these caprine MCT. Therefore, commonly applied special stains to elicit metachromasia in neoplastic mast cells is of limited or questionable use in differentiating MCT from other cutaneous round cell tumors, including cutaneous histiocytoma, in goats. We suggest that immunohistochemistry for CD117/KIT may be required to definitively identify MCT in goats. From the data available for the 2 cases presented here, cutaneous histiocytomas in goats appear to have a good prognosis following excision. A misdiagnosis of null lymphoma or amelanotic melanoma could result in expensive, potentially unnecessary treatments or unwarranted euthanasia.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820988145 - Mast Cell Tumors and Histiocytomas in Domestic Goats and Diagnostic Utility of CD117/c-Kit and Iba1 Immunohistochemistry
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820988145 for Mast Cell Tumors and Histiocytomas in Domestic Goats and Diagnostic Utility of CD117/c-Kit and Iba1 Immunohistochemistry by T. William O’Neill and Christiane V. Löhr in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge and thank the histology laboratory at the New York State Animal Health Diagnostic Center, Cornell University College of Veterinary Medicine, and Dr Andrew Miller for their expertise and assistance with Iba1 immunohistochemistry that is presented in this report. We thank the staff in the histology laboratory at the OVDL for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Department of Biomedical Sciences, Carlson College of Veterinary Medicine, Oregon State University.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
