Abstract
Clinical and histologic features of 26 cases of canine peripheral giant cell granuloma (formerly giant cell epulis) are reported. Two main histologic categories were evident: (1) “classic” peripheral giant cell granuloma, characterized by variable numbers of multinucleated giant cells (MNGCs) admixed with densely cellular mononuclear spindle-shaped cells in variable amounts of collagenous matrix, and (2) the “collision” peripheral giant cell granuloma, with features of both a peripheral giant cell granuloma and a fibromatous epulis of periodontal ligament origin. In the 16 dogs for which the outcome was known, 2 peripheral giant cell granulomas recurred after excision. No age or sex predilection was evident; however, lesions were more common in maxillary than in mandibular gingiva. In contrast to cats, peripheral giant cell granulomas in dogs behave like fibromatous epulides of periodontal ligament origin and seldom recur after excision. Positive staining with TRAP (tartrate-resistant acid phosphatase) of the MNGCs and a fraction of the mononuclear cell population is consistent with osteoclastic origin.
The term epulis is used inconsistently for localized exophytic gingival growths, both reactive and neoplastic, 8 and has been applied to various lesions, including fibromatous epulis of periodontal ligament origin (including the formerly classified fibromatous and ossifying epulis), acanthomatous ameloblastoma (formerly acanthomatous epulis), 6 and peripheral giant cell granuloma (formerly giant cell epulis). 5,25 According to the World Health Organization classification, 12 the diagnosis of epulis is limited to the fibromatous epulis of periodontal ligament origin, which, along with acanthomatous ameloblastoma, is relatively common in dogs and has well-established clinicopathologic features. 4,6,8,11,23 –25 However, little is known about the clinicopathologic features of the rare canine peripheral giant cell granuloma. 9,10,16,22,25
Histologically, peripheral giant cell granulomas consist of multinucleated giant cells (MNGCs) and mononuclear spindle-shaped cells in collagenous matrix with variable osteoid deposition, hemorrhage, hemosiderin, and ulceration of gingival mucosa. 5,8,13,17,18,21,22,25 Although human peripheral giant cell granulomas are generally considered reactive and non-neoplastic, the cause of these lesions is poorly understood. 13,17,18
Six reported cases of canine peripheral giant cell granuloma did not recur after surgical excision. 22,25 In comparison, canine fibromatous epulides of periodontal ligament origin rarely recur, whereas acanthomatous ameloblastomas are invasive and often recur after excision. 6,8,11,12,25 Interestingly, peripheral giant cell granulomas in cats have a faster growth and recurrence rate than fibromatous epulides. 5,19 The purpose of this study is to report the clinical and histologic features of 26 cases of peripheral giant cell granuloma in dogs.
Material and Methods
Case materials were reviewed from 26 dogs with oral masses diagnosed as giant cell epulis (23/26), spindle cell sarcoma with giant cells (2/26), or oral tumor with giant cell epulis in the differential diagnosis (1/26). The biopsy specimens had been submitted to the Laboratory of Pathology and Toxicology at the University of Pennsylvania School of Veterinary Medicine between January 2000 and October 2010. The submission forms were reviewed for clinical data, including age, breed, sex, and location of the tumor. Additional clinical information, including presence of periodontal disease and recurrence after excision, was obtained by mail and oral communication with the referring veterinarian. The specimens had been submitted in 10% neutral buffered formalin and were decalcified in 15% formic acid, if needed, before trimming. Tissues were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. All cases were reevaluated by 2 anatomic pathologists (AVD, MDS) and classified histologically as “classic” or “collision” peripheral giant cell granulomas. The mitotic index was determined for 10 consecutive high power fields, when possible. Histochemistry for tartrate-resistant acid phosphatase (TRAP), an enzyme unique to osteoclasts, was performed on 2 collision and 6 classic peripheral giant cell granulomas. The acid phosphatase commercial test (Kit No. 386-A; Sigma Chemicals, St. Louis, MO) was performed as described. 5 Limitations of this study include those inherent to retrospective studies—namely, incomplete medical records and loss of cases to follow-up.
Results
A total of 2609 canine fibromatous epulides of periodontal ligament origin (including the formerly classified fibromatous and ossifying epulis), acanthomatous ameloblastomas (formerly acanthomatous epulis), and peripheral giant cell granulomas (formerly giant cell epulis) were diagnosed from January 1, 2000, to October 1, 2010, by the Biopsy Service of the Laboratory of Pathology and Toxicology. The prevalence of peripheral giant cell granulomas in the studied population was 0.99% (26/2609). If acanthomatous ameloblastomas were excluded, the prevalence of peripheral giant cell granulomas was 1.2% (26/2198).
Clinical information for the 26 cases of canine peripheral giant cell granuloma is summarized in Table 1. The age at diagnosis ranged from 2 to 12 years (mean, 7.4 years; median, 8.25 years). Approximately half the animals (11/26) were 8 to 10 years of age; however, 8 dogs were younger than 6 years. There were 5 Golden Retrievers, 2 Pomeranians, and 7 dogs of mixed breeding (including 1 Golden Retriever and 1 Pomeranian mix). The male: female ratio was 1.08, with 12 females (1 intact) and 13 males (5 intact). Sex was not recorded on the submission form for 1 dog. Eighteen tumors were in maxillary gingiva adjacent to incisor or premolar teeth in 6 and 10 cases, respectively, with site not further specified in 2 cases. Four tumors were in mandibular gingiva adjacent to incisor, canine, or premolar teeth in 1, 1, and 2 cases, respectively. Location was not reported for 4 dogs. Of the 10 dogs with information in the medical record about periodontal disease at diagnosis, 4 had no periodontal disease and 6 had evidence of periodontal disease. Follow-up information about recurrence was available for 16 cases (follow-up period ranged from 1.5 months to 9 years). In 2 cases, recurrence was reported within 2 months of initial surgical excision.
Clinical Characteristics of 26 Canine Peripheral Giant Cell Granulomas
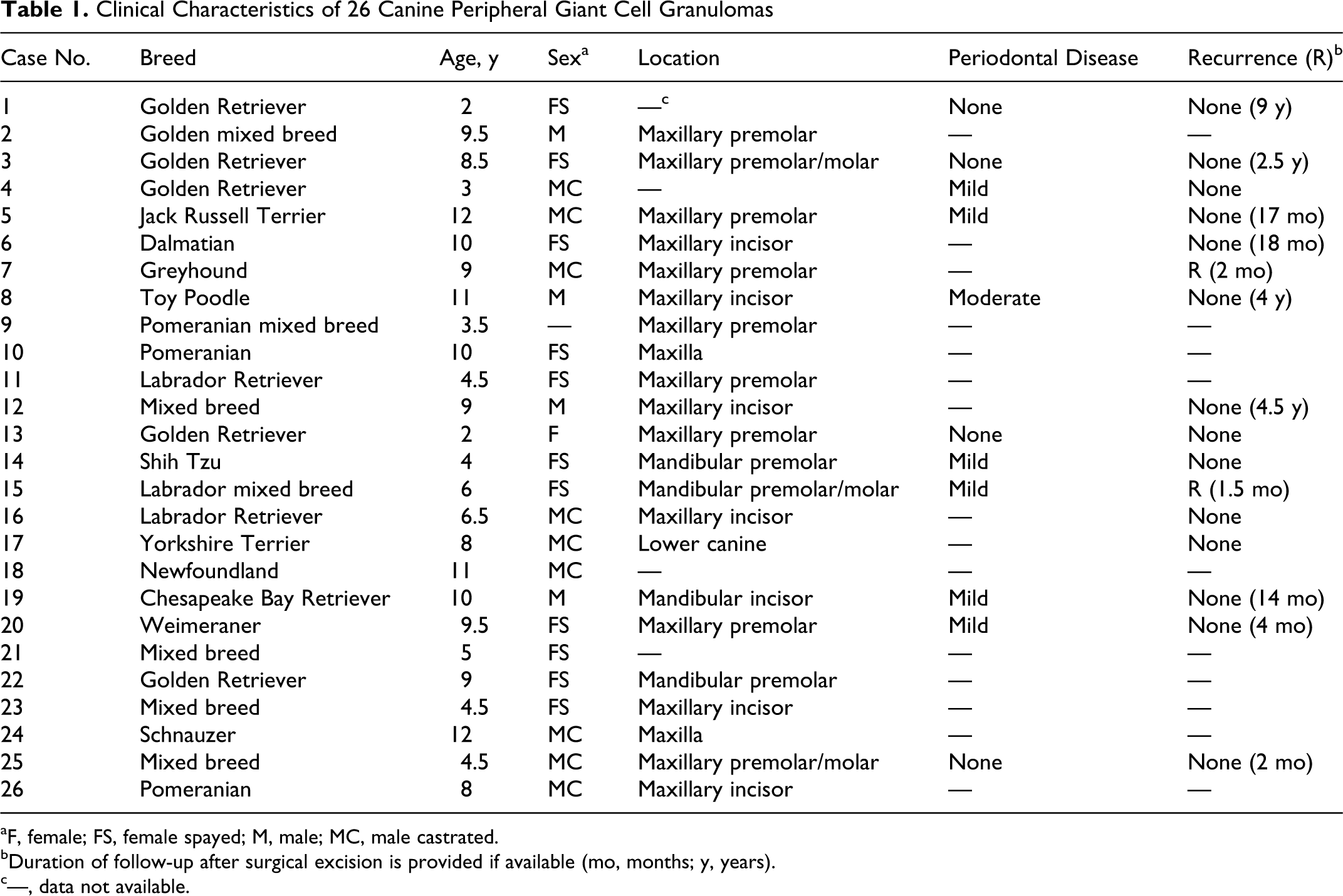
aF, female; FS, female spayed; M, male; MC, male castrated.
bDuration of follow-up after surgical excision is provided if available (mo, months; y, years).
c—, data not available.
Histologic features of the peripheral giant cell granulomas are summarized in Table 2. The “classic” peripheral giant cell granuloma (21/26) was characterized by variable numbers of MNGCs in a dense population of mononuclear spindle-shaped cells with variable amounts of collagenous matrix (Figs. 1, 2). The mononuclear spindle cell population was mildly pleomorphic, haphazardly arranged, or in short fascicles, with moderate amounts of eosinophilic cytoplasm, indistinct cell borders, and oval nuclei with indistinct nucleoli.
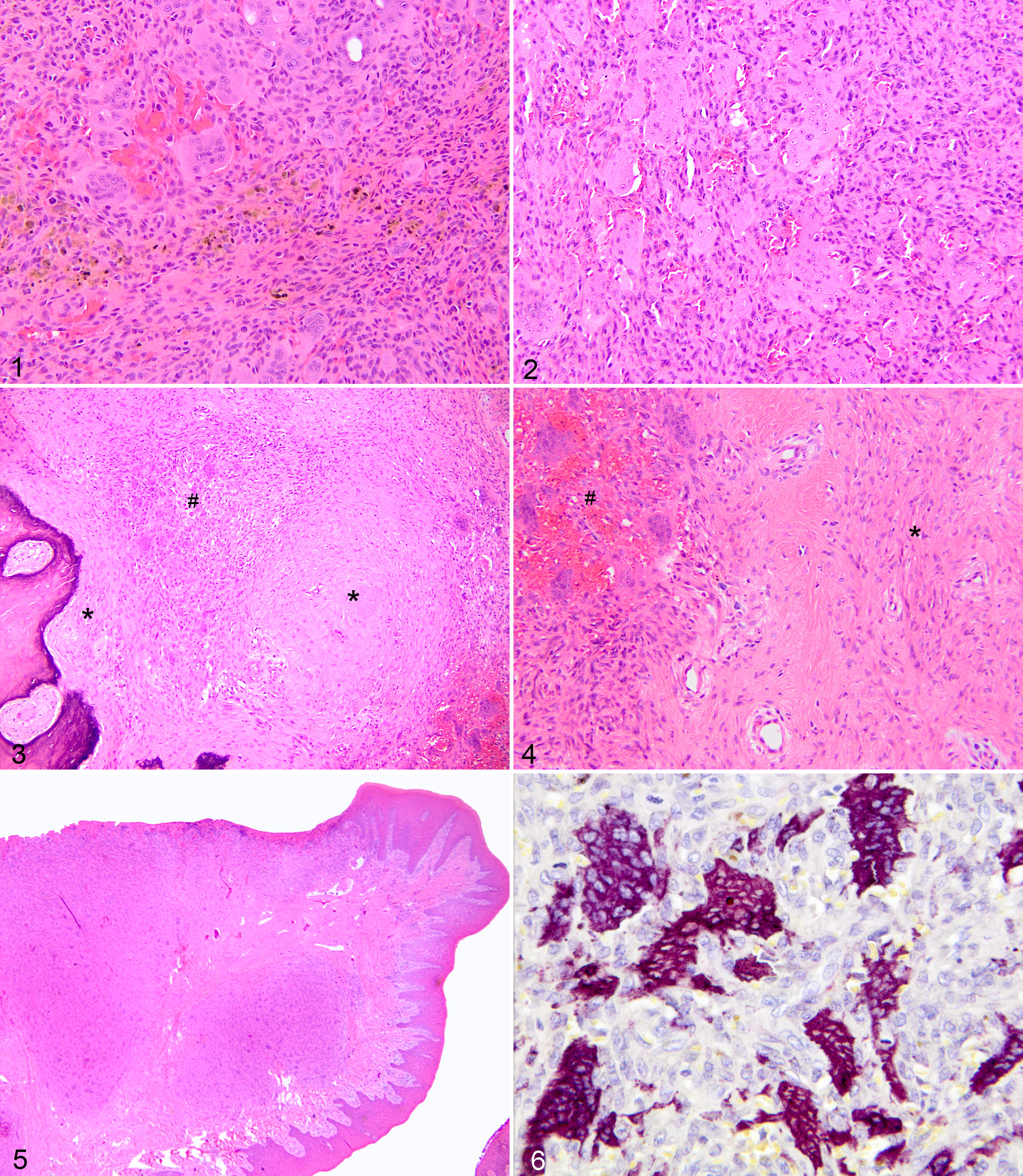
Gingiva; dog No. 14. “Classic” peripheral giant cell granuloma with multinucleated giant cells and spindle cells in collagenous matrix. Note focal hemorrhage and hemosiderin deposition. Hematoxylin and eosin (HE).
Histologic Features of “Classic” and “Collision” Peripheral Giant Cell Granulomas
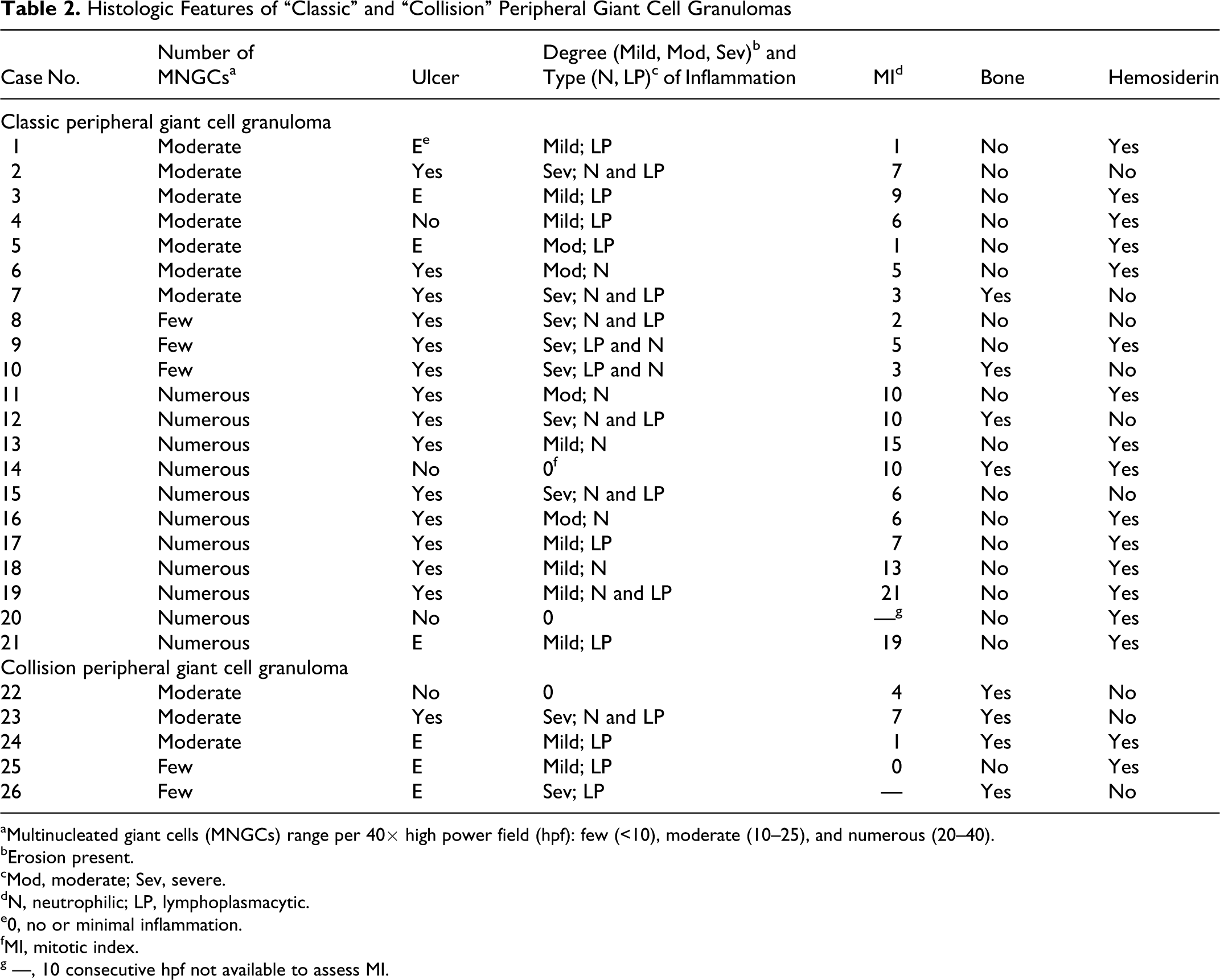
aMultinucleated giant cells (MNGCs) range per 40× high power field (hpf): few (<10), moderate (10–25), and numerous (20–40).
bErosion present.
cMod, moderate; Sev, severe.
dN, neutrophilic; LP, lymphoplasmacytic.
e0, no or minimal inflammation.
fMI, mitotic index.
g —, 10 consecutive hpf not available to assess MI.
The collision peripheral giant cell granuloma (5/26) had histologic features of classic peripheral giant cell granuloma with distinct areas that resembled fibromatous epulis of periodontal ligament origin (Figs. 3, 4). The fibromatous epulis–like areas were characterized by stellate mesenchymal cells in fibrous stroma that resembled the periodontal ligament with regularly spaced blood vessels. Areas of fibromatous epulis and peripheral giant cell granuloma, although distinct, often merged focally.
Peripheral giant cell granulomas were unencapsulated, nodular, or multinodular masses separated by normal gingival collagen (Fig. 5). Lesional tissue was focally contiguous with the overlying mucosal epithelium or separated from it by a band of collagen fibers; often, both patterns were present in the same biopsy sample. The presence of ulceration and granulation tissue and the severity of inflammation varied greatly among the specimens. Ulcers and erosions were present in 15 and 7 cases, respectively. Inflammatory cells consisted predominantly of lymphocytes and plasma cells, except in ulcerated areas, where neutrophils were usually the predominant leukocyte. The mitotic index ranged from 1 to 21 per 10 high power fields (hpf). The mitotic index in the 2 epulides that recurred was 3 and 6. Mitotic figures were in the mononuclear spindle cell population. Most lesions had variability in the number of mitotic figures among fields. Bizarre mitotic figures or other nuclear or cellular features of anaplasia were not found. Hemosiderin was present in 16 of 26 cases; bone was present in 8 cases (4/5 of the collision peripheral giant cell granulomas and 4/21 of the classic peripheral giant cell granulomas). The lesions extended to the surgical margins in all but 1 case.
The striking histologic feature of peripheral giant cell granuloma was the presence of multinucleated giant cells. In “classic” peripheral giant cell granulomas, the number of MNGCs per 40× hpf was quite variable. Three had less than 10, 7 had 10 to 25, and 11 had 25 to 45. In collision peripheral giant cell granulomas, the number of giant cells varied from less than 10 to 30 per 40× hpf. The MNGCs were often adjacent to ulcers; however, MNGCs were also deeper in the subepithelial tissue and, in some cases, were more numerous in areas without inflammation or ulceration (Fig. 5). Two of the 11 cases with numerous MNGCs had no or minimal inflammation, 5 of 11 had mild inflammation, and 3 of 11 did not have ulcers. All 3 peripheral giant cell granulomas with very few MNGCs were accompanied by severe ulceration, neutrophilic inflammation, and granulation tissue. As described in humans 13 and dogs, 22 2 types of MNGCs were present in 12 of 26 cases. The type I MNGCs comprised cells with many large to oval vesicular nuclei, prominent nucleoli, and slightly basophilic cytoplasm (metabolically active cells). 20,22 The type II MNGCs were less common and smaller cells with brightly eosinophilic cytoplasm and small, condensed hyperchromatic and irregular nuclei (degenerating cells). 20,22 In the classic peripheral giant cell granulomas with moderate numbers to numerous MNGCs, the MNGCs and fewer mononuclear spindle cells were strongly positive with TRAP histochemistry; however, in the classic peripheral giant cell granulomas with few MNGCs and in the collision peripheral giant cell granulomas (with few MNGCs), the cells were negative (Fig. 6).
Discussion
Peripheral giant cell granuloma is a rare oral lesion in dogs, 9,10,16,22,25 with a prevalence (among fibromatous epulis of periodontal ligament origin and acanthomatous ameloblastoma) of 0.99% in the present study. Peripheral giant cell granulomas are reportedly more prevalent in cats, comprising 28.8% of the group of gingival tumors that includes fibromatous epulis and acanthomatous ameloblastoma. 5
The age of dogs with peripheral giant cell granuloma ranged widely, with almost 1 of 3 of the cases in animals younger than 6 years of age. In previous reports, the age of 4 dogs with peripheral giant cell granuloma ranged from 6.3 to 11 years; 25 in 15 cats, the age ranged from 4 to 19 years. 5 Although no sex predisposition for peripheral giant cell granulomas has been reported for dogs or cats, there is a slight female predominance in human cases. 13,15 Peripheral giant cell granulomas are most often in maxillary gingiva, similar to fibromatous epulis, and in contrast to acanthomatous ameloblastoma, for which 58.8% arise around mandibular canine teeth. 25 In humans, peripheral giant cell granulomas are located predominantly in mandibular gingiva. 13,15
Marginal excision was usually curative in this study, with recurrence reported in only 2 cases, each within 2 months after excision. No histologic features distinguished the peripheral giant cell granulomas that recurred from those that did not. Previously reported canine peripheral giant cell granulomas did not recur after surgical excision. 10,22,25 Similarly, low recurrence (up to 9%) of canine fibromatous epulis of periodontal ligament origin has been reported. 25 In contrast, feline peripheral giant cell granulomas tend to recur soon after surgery. 5 Cellular pleomorphism or nuclear atypia were not present in the cases evaluated in the present study; the mitotic index varied but did not correlate with biologic behavior.
Five cases in this study had histologic features of both peripheral giant cell granuloma and fibromatous epulis of periodontal ligament origin and were classified as “collision” peripheral giant cell granulomas. This combination of histologic features could represent a transition between 2 lesions of periodontal ligament origin. The “collision” peripheral giant cell granuloma may be a variant of fibromatous epulis or a collision between 2 different and separate oral lesions. None of the “collision” peripheral giant cell granulomas in this report recurred after excision.
The pathogenesis of peripheral giant cell granuloma is poorly understood. Many consider peripheral giant cell granuloma a reactive and non-neoplastic lesion. 1,2,13,22 An association with trauma and hemorrhage has been suggested in humans. 1,2,13 Several peripheral giant cell granulomas in this study had intralesional hemosiderin, consistent with previous hemorrhage. An association between periodontal disease and the development of peripheral giant cell granuloma has also been suggested in humans. 13,15 In dogs, fibromatous epulides of periodontal ligament origin are associated with more severe dental plaque deposition than are acanthomatous ameloblastomas and may develop in association with chronic inflammation. 25 The presence of periodontal disease in dogs with peripheral giant cell granuloma has not been previously reported, but no correlation was evident in this study.
The MNGCs in peripheral giant cell granulomas resemble osteoclasts, 3,5,7,20 and the histochemical reactivity for TRAP, an enzyme unique to osteoclasts, in the MNGCs and a fraction of the mononuclear cell population is consistent with osteoclastic origin. 5,14 The variable TRAP reactivity among lesions with numerous, moderate, or few MNGCs is interesting, although the significance of this finding is unknown. We cannot rule out 2 different cells of origin for the MNGCs in these lesions. The formation of MNGCs in peripheral giant cell granulomas has been attributed to increased osteoclastic activity secondary to ulceration and inflammation. 5 In the present study, most peripheral giant cell granulomas had gingival ulceration or erosion and inflammation with MNGCs present in the areas of ulceration. However, in several cases, numerous MNGCs were also in areas without ulceration or inflammation, and some peripheral giant cell granulomas with few MNGCs had severe ulceration, neutrophil infiltration, and granulation tissue formation. It has also been suggested that the MNGCs are formed by the fusion of macrophages in granulomatous inflammation. 25 Granulomatous inflammation was not present in any case in the current study.
In summary, canine peripheral giant cell granuloma, with the exception of the “collision” variant, is histologically distinct from fibromatous epulis of periodontal ligament origin. However, its biologic behavior (in contrast to feline peripheral giant cell granuloma) is similar, with rare recurrence after even marginal surgical excision. It is much less aggressive than canine acanthomatous ameloblastoma, which is invasive and not cured by marginal excision. There is no correlation between the histologic characteristics (category, number of giant cells, mitotic index) of the peripheral giant cell granulomas and their biological behavior.
Footnotes
Acknowledgement
The authors thank Mrs Juli Burns for histochemical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
