Abstract
A retrospective study was performed to characterize 52 new cases of feline epulides between 1995 and 2001, with clinical and pathological results classified according to Head's histopathologic criteria for canine epulides. The incidence of the fibromatous, acanthomatous, ossifying, and giant cell epulis were respectively 57.7% (30/52), 7.7% (4/52), 5.8% (3/52), and 28.8% (15/52). Giant cell epulides presented significant differences in clinical behavior compared with the fibromatous type, including rapid growth (P < .0001), presence of ulcerative changes (P < .01), and rapid recurrence after surgery (P < .01) from which euthanasia was judged necessary in 4 cases. Fifteen giant cell epulides were additionally examined in order to characterize the lesion both histochemically and immunohistochemically and to investigate the origin of the multinucleated giant cells (MGCs). Van Gieson staining showed osteoid and woven bone formation in 11 cases. Both the MGCs and a fraction of the mononuclear cells were positive for vimentin, tartrate-resistant acid phosphatase (TRAP), a commonly accepted marker for osteoclasts, and the polyclonal antibody receptor activator of nuclear factor κβ (RANK), a cytokine leading to the differentiation of osteoclast progenitors into mature osteoclasts in presence of its ligand. MGCs were negative for smooth muscle actin, MIB-1, and factor VIII. The giant cell epulis may be a variant of the fibromatous and ossifying epulis in which extensive ulceration and inflammation results in increased osteoclastic activity. The osteoclast-like giant cells are most likely formed from a monocyte/ macrophage-like osteoclast precursor that differentiates into osteoclasts under the influence of mononuclear osteoblast-like stromal cells.
Epulis is a nonspecific, clinical descriptive term referring to a benign local exophytic growth of the oral mucosa. Epulides are microscopically characterized by a dense, well vascularized stroma populated by stellate cells with abundant fibrillar collagen resembling the periodontal ligament. In the dog several forms can histologically be distinguished: 1) fibromatous, 2) acanthomatous, 3) ossifying, and 4) giant cell epulis,2,5,7–9,18,26,27 while in cats fibromatous, ossifying, and rarely giant cell epulides are reported.4,22 Fibromatous epulides in the cat tend to have ossifying and acanthomatous components.4,22
Giant cell epulides are microscopically characterized by presence of numerous multinucleated giant cells (MGCs), scattered in fibrous, well-vascularized, and highly cellular stroma containing numerous round and spindle-shaped mononuclear cells. Focal areas with osteoid-like material can be noticed.8 Although clinical behavior and histopathologic features are well established in dogs, feline epulides are infrequent, representing up to 7.8% for the fibromatous type and up to 0.5% for the giant cell epulis of all feline oral neoplasms.22 Most feline epulides are single, but multiple occurrence is reported in the fibromatous type and tend to have a higher recurrence rate after surgery.4 Giant cell epulides are both clinically and pathologically less well characterized but exhibit an aggressive clinical course and are, unlike in dogs, reported to recur after surgery.19,28
Because of the resemblance of the MGCs to foreign body–type giant cells, the giant cell epulis sometimes is considered to represent a reactive giant cell-rich granulomatous lesion, but the real origin remains unknown.19,21
In human oral cavity proliferations containing MGCs, like the central and peripheral giant cell granuloma, MGCs have been considered as phagocytes,12 foreign body cells,24 or osteoclasts.1,6,20 The MGC has characteristics of the osteoclast phenotype, including expression of tartrate-resistant acid phasphatase (TRAP), and is able to perform lacunar bone resorption. Recently, the major cellular and humoral factors that induce osteoclast formation have been determined. Osteoclast formation involves an interaction between stromal cells, which express the receptor activator of nuclear factor κβ ligand (RANKL), and receptor activator of nuclear factor κβ (RANK)-expressing mononuclear phagocyte osteoclast precursors that are observed in the circulation and tissues.10,17 Mononuclear stromal cells in these jaw lesions are thought to be composed of a heterogeneous population of macrophage-like and fibroblast-like cells.6,25
The aim of this retrospective study was to present the clinical and pathological characteristics of 52 cases of feline epulides and to clarify the origin of the MGCs. Therefore, all giant cell epulides were analyzed both histochemically and immunohistochemically.
Materials and Methods
The subjects of this study were all cats (n = 52) with proliferating oral-cavity lesions that after surgical excision were histologically diagnosed as epulides at the Veterinary Pathology Department of Utrecht University, Faculty of Veterinary Medicine between 1995 and 2001.The specimens were fixed in 10% neutral buffered formalin and bisected along their axis. Specimens that were hard to cut or included bone were decalcified after fixation in formic acid. Tissues were embedded in paraffin, sectioned at 5 μm, and stained with haematoxylin and eosin (HE). All cases were histopathologically reexamined by 2 pathologists (TSGAM van den Ingh and IJS Neyens) and classified based upon light microscopy according to Head's histopathologic criteria for canine epulides.8,9 Histopathologic features made and modified from Head's histopathologic criteria of canine epulides are shown in Table 1.
Histological features of fibromatous, ossifying, acanthomatous, and giant cell canine epulides.
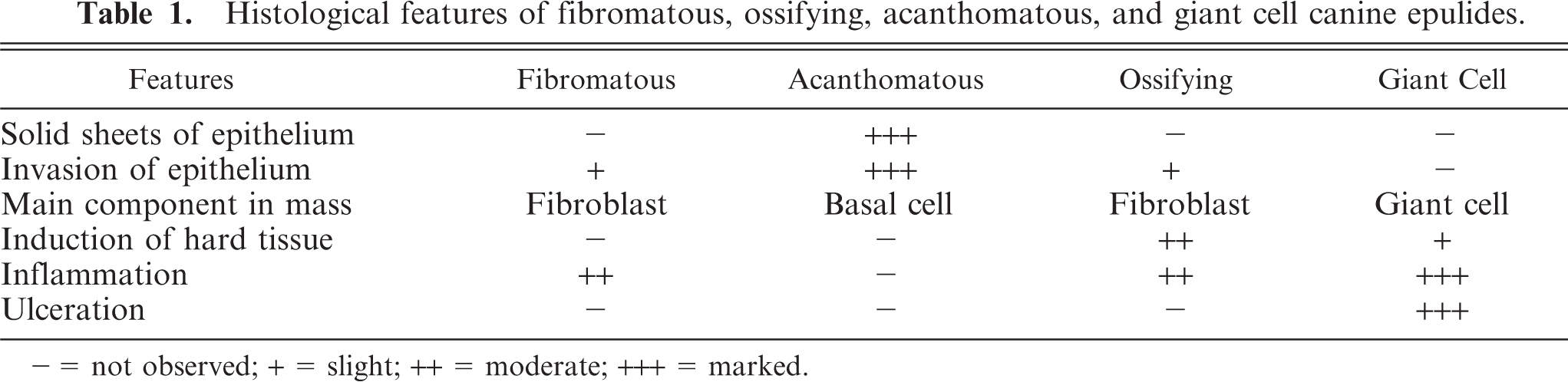
− = not observed; + = slight; ++ = moderate; +++ = marked.
Medical records were reviewed for age, sex, breed, site, size, number of lesions, prior surgery, and treatment. Follow-up data were conducted by telephone to obtain information about the postsurgical course of the disease in the following 2 years. The overall survival time (ST) was defined from the day of diagnosis until the day of death or last follow-up. The recurrence free interval (RFI) was considered from the day of diagnosis until the day recurrence was noticed. Statistical analysis was carried out with the SPSS (Chicago, IL) statistical package. The Kaplan-Meier product limit method was used to estimate the ST and RFI.
Fifteen cases diagnosed as giant cell epulis were stained according to Van Gieson (VG). Histochemical staining to identify the origin of MGCs was performed for TRAP, an enzyme unique to osteoclasts,15 using the acid phosphatase commercial Test kit no. 386-A (SIGMA Chemicals, St. Louis, MO). Sections were deparaffinized and rehydrated with xylol for 10 minutes, rinsed in TRIS-buffer for 5 minutes (replaced 3 times), and rinsed in distilled water for 3 minutes (replaced twice). Without prior fixation sections were immediately incubated with the SLP-solution (acid leukocyte phosphatase) and the substrate (1 capsule Fast Garnet GBX; SIGMA Chemicals, St. Louis, MO) at 37°C for 1 hour. After rinsing in distilled water for 3 minutes, sections were counterstained with Mayer's hematoxylin (Merck KgaA, Darmstadt, Germany), washed with running water, dehydrated, and mounted with Eukitt (O. Kindler GmBH, Freiburg, Germany).
The following immunohistochemical markers were selected for further identification of the origin of the MGCs: 1) vimentin, 2) alpha smooth muscle actin, 3) MIB-1, 4) factor VIII, and 5) RANK (Table 2). Positive controls were feline fibrosarcoma for vimentin; smooth muscle for smooth muscle actin; intestinal crypts for MIB-1, and feline skin for factor VIII. For negative control purposes, the primary antibody was replaced by a nonspecific antibody of the same isotype. Polyclonal antibodies were incubated with a biotinylated secondary goat antirabbit immunoglobulin, and monoclonal antibodies were incubated with a biotinylated horse antimouse secondary immunoglobulin (Table 2).
Details of immunohistochemical reagents and methods.
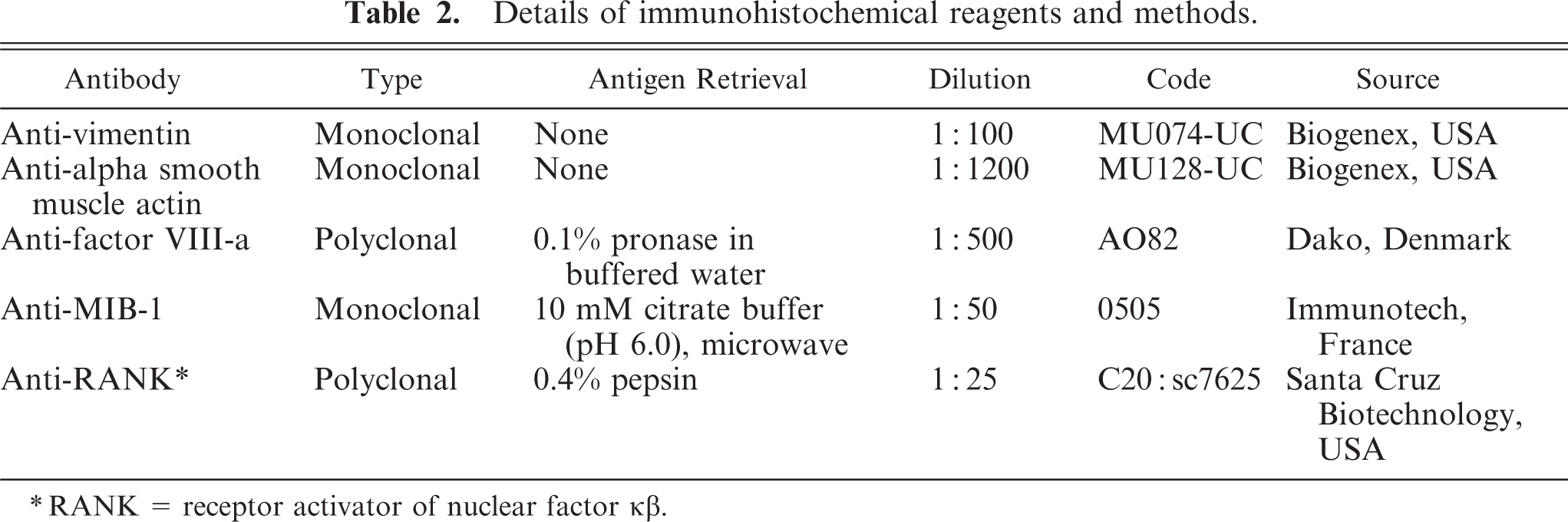
RANK = receptor activator of nuclear factor κβ.
Immunohistochemistry for the RANK was performed with the ABC kit (Vector Laboratories, Burlingame, CA) at room temperature. Sections were mounted on poly-L-lysine (Sigma Diagnostics) coated slides, deparaffinized and rehydrated, and then incubated in 1% H2O2 in methanol for 30 minutes to block endogenous peroxidase activity. After rinsing in phosphate-buffered saline (PBS), sections were digested with 0.4% pepsin for 5 minutes in order to unmask the antigen. Subsequently, sections were rinsed with PBS/Tween and incubated with normal horse serum 0.1% for 15 minutes to avoid unspecific background staining. Thereafter, sections were incubated with a polyclonal primary antibody of goat origin raised against a peptide mapping at the carboxy terminus of the protein (anti-RANK9C-20)°sc-7625 at 4°C overnight in a humidified chamber. The slides were rinsed again in PBS/Tween and incubated for 30 minutes with a biotinylated secondary horse antigoat immunoglobulin (1:125) and peroxidase-labeled streptavidin. Incubation was stopped in PBS/Tween, before the ABC complex (Vector Laboratories) was administrated for 30 minutes. After rinsing in PBS, the sections were stained in a 3,3′-diaminobenzidine tetrahydrochloride (Sigma) 0.5%, diluted in Tris/HCL buffer, for 10 minutes. The slides were then rinsed in distilled water, counterstained with Mayer's hematoxylin (Merck KgaA), washed with running water, dehydrated, and mounted with Eukitt (O. Kindler GmBH). Positive controls were human dental tissue and human bone tissue both decalcified in EDTA-solution, of proven reactivity with RANK. The specificity of anti-RANK antibodies for feline osteoclasts was investigated by examining their reactivity against other MGC-containing lesions of inflammatory origin (feline granulomatous inflammation foreign body type, feline leprosy).
Results
The major breed of the cats presented was the European domestic shorthair. The male°female ratio was 50%, and the age varied from 8 months to 20 years. The majority of the cases presented as a slowly growing solitary epulis, although few grew fast and expansively and were associated with dental disruption. Epulides were located around premolars and molars in either mandible or maxilla (Table 3).
Clinical presentation of 52 cats with epulides.
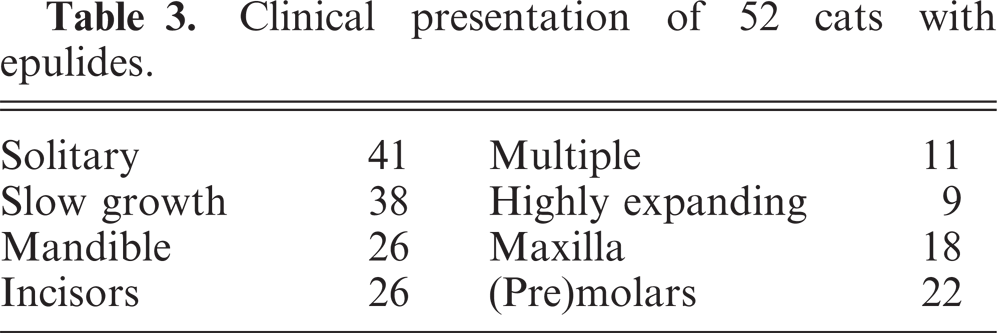
Most feline epulides could not be diagnosed as 1 of the 4 canine classical types but showed a combination of lesions.
The most frequently diagnosed (n = 30) epulis was the fibromatous epulis with a combination of acanthomatous and ossifying components, similar to those previously reported in cats.22 Epulides were in general up to 1 cm in diameter, and the overlying gingival epithelium was intact in the majority (n = 18) of the cases. Microscopically, the overlying gingival epithelium was normal to hyperplastic, often with rete ridge formation. The stroma was densely cellular composed of stellate cells and tightly packed fibrillar collagen resembling that of the periodontal ligament (Figs. 1, 2). Nearly all epulides also contained variably sized islands of osteoid like material (Fig. 2, ∗). Male°female ratio was 16:14, and 90% was castrated/spayed. The breed of cats was predominantly European domestic shorthair (n = 25), but Persian (cases 1 and 13), Abyssinian (cases 9 and 16), and Siamese (case 11) were also represented. In 4 cases the reason for surgery was recurrence or renewed occurrence of an epulis. Multiple epulides were reported in 9 cases. Four out of the total 6 recurrences occurred in multiple epulides within the 2-year follow-up period after marginal excision (Table 4A).
Case summaries of feline fibromatous epulides.
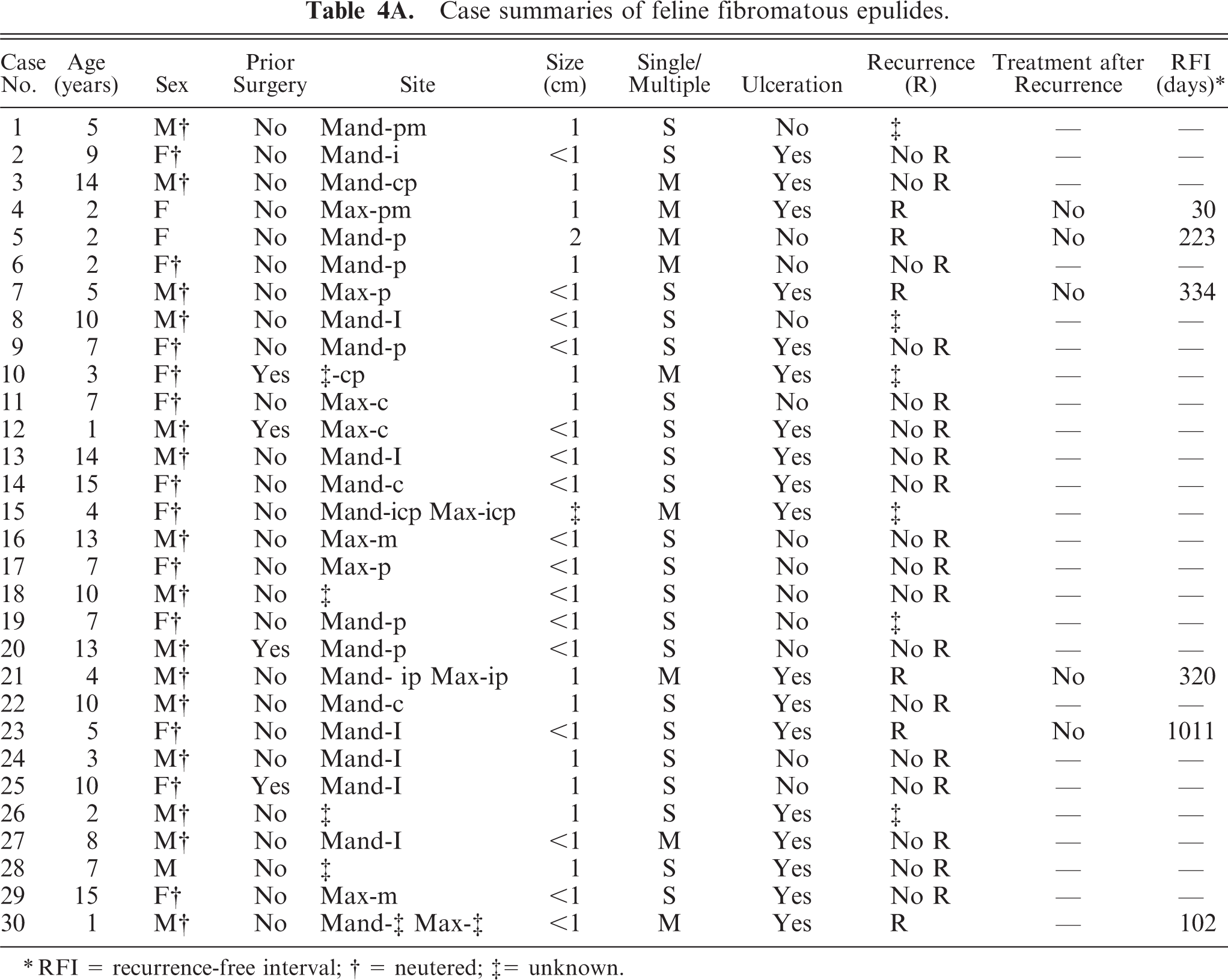
RFI = recurrence-free interval
= neutered
= unknown.
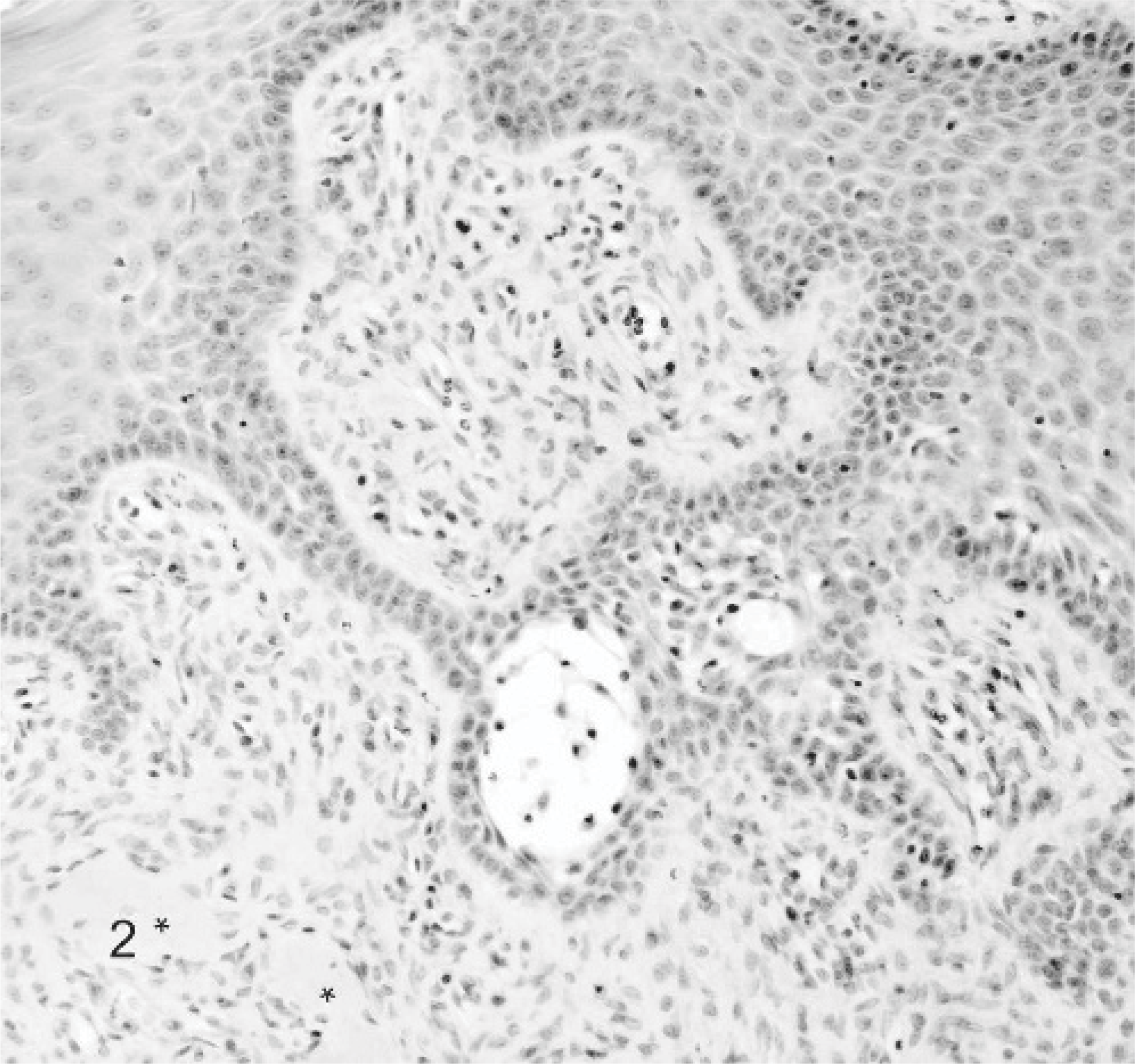
Fibromatous epulis; cat. Case 14. Nearly all fibromatous epulides also have areas with ossifying (∗) and acanthomatous components with rete ridge formation. HE stain.
Fibromatous epulis; cat. Case 14. The fibromatous epulis is characterized by normal to hyperplastic gingival epithelium, densely cellular stroma composed of stellate cells and tightly packed fibrillar collagen resembling that of the periodontal ligament. HE stain.
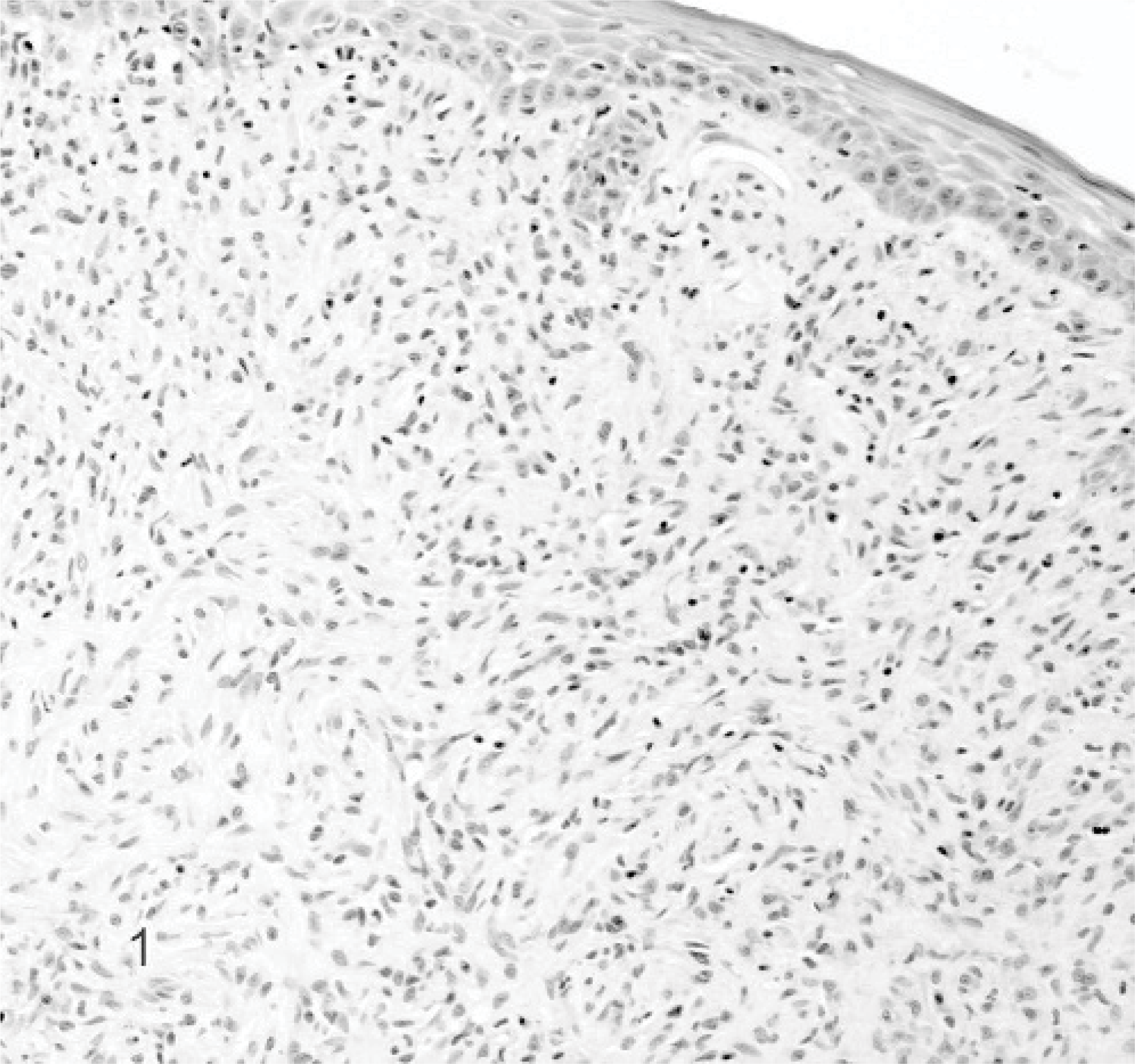
In 4 cats, an acanthomatous epulis was diagnosed. All cats were female European domestic shorthair with a solitary 1-cm-diameter proliferating epulis. Microscopic characteristics of these epulides were infiltrative growth of deep in the submucosa invading cords or solid clusters of proliferating columnar epithelial basal cells with a palisade arrangement (Fig. 3). Recurred multiple growth at the start of the survey was found in case 3, without recurrence after marginal excision in the 2-year follow-up period after marginal excision (Table 4B).
Case summaries of feline acanthomatous epulides.

RFI = recurrence-free interval
= neutered
= unknown.
Acanthomatous epulis; cat. Case 1. The acanthomatous epulis is characterized by infiltrative growth of deep in the submucosa invading cords or solid clusters of proliferating columnar epithelial basal cells with a palisade arrangement. HE stain.
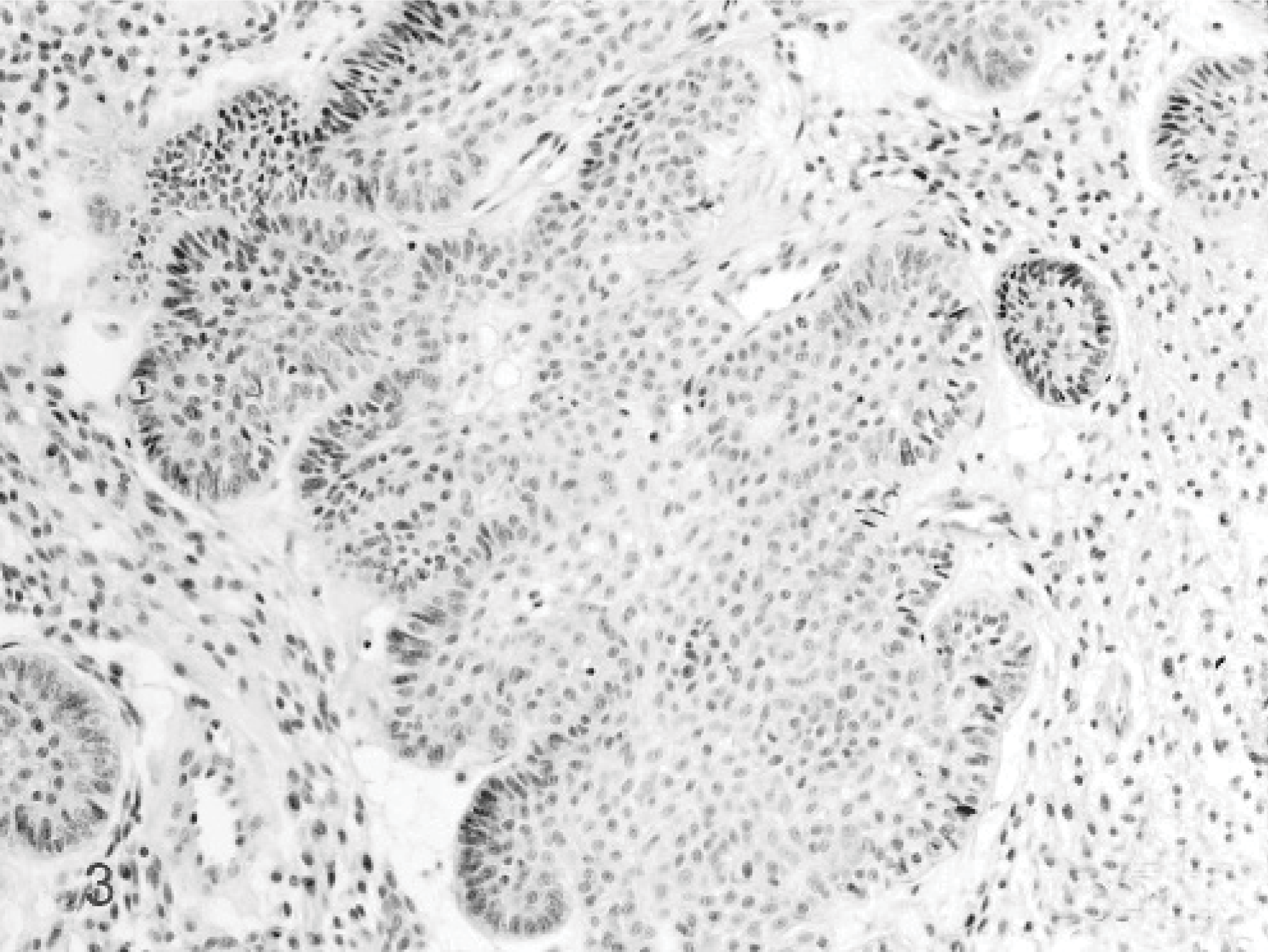
Only in 3 cats a solitary, 1-cm-diameter ossifying epulis was diagnosed, microscopically characterized by hyperplastic gingival epithelium, well-vascularized collagenous stroma populated by stellate cells and large areas of osteoid, cementin-like, or dentin-like material (Fig. 4). No recurrence was reported (Table 4C).
Case summaries of feline ossifying epulides.
RFI = recurrence-free interval
= neutered
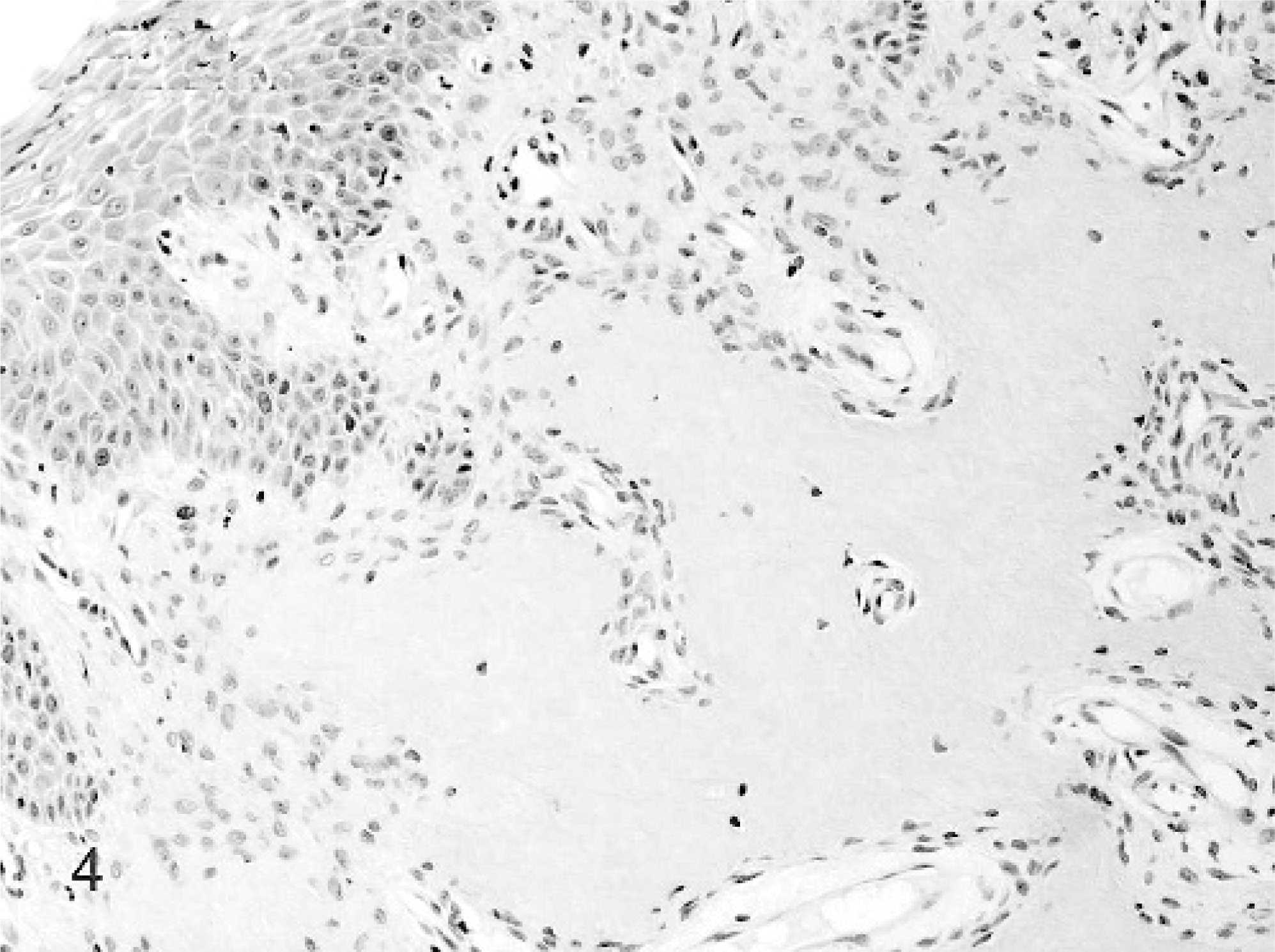
Ossifying epulis; cat. Case 2. The ossifying epulis is characterized by hyperplastic epithelium, well-vascularized collagenous stroma populated by stellate cells and large areas of osteoid-like, cementin-like, or dentin-like material. HE stain.
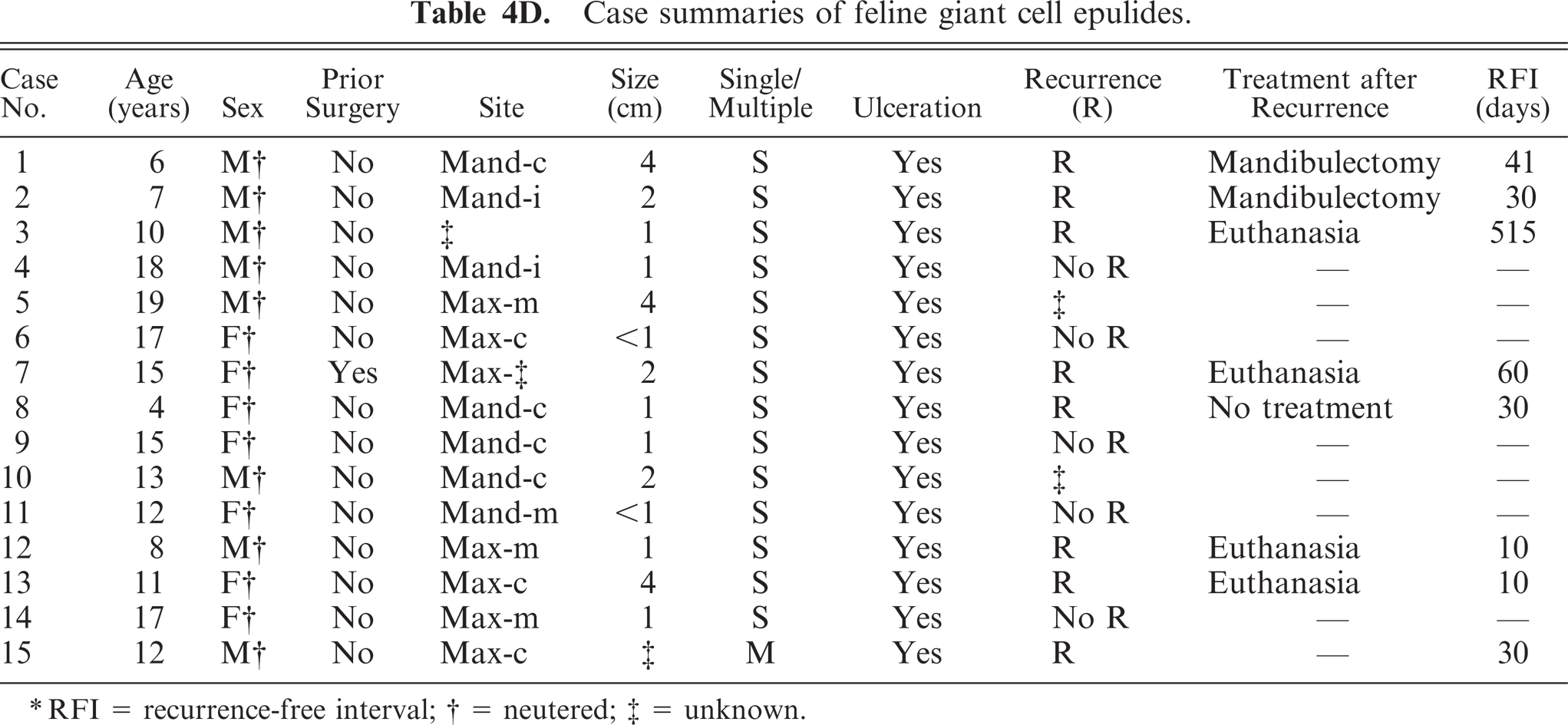
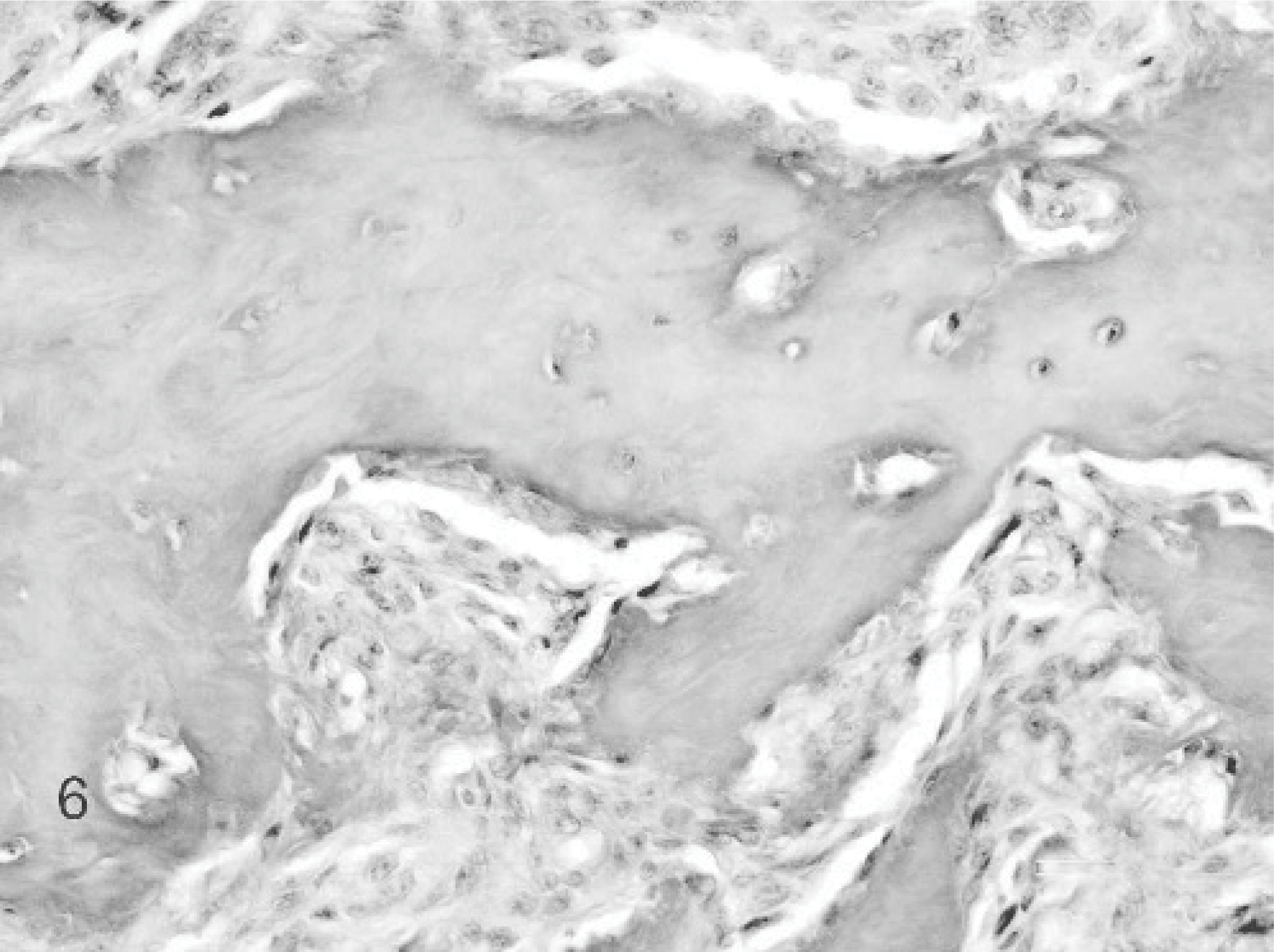
The giant cell epulis was the second most important type containing 15 cases. All animals showed severely ulcerated variably sized mostly single proliferative epulides. Microscopically, all giant cell epulides were nonencapsulated, poorly demarcated, and covered by hyperplastic gingival epithelium with large ulcerated areas. The most important feature was presence of numerous MGCs, scattered in a background of fibrous well-vascularized and highly cellular stroma containing numerous round and spindle-shaped mononuclear cells. No evident cellular pleomorphism, anaplasia, or mitotic figures were found. Inflammation, hemorrhages, and islands of osteoid were frequently present. The MGCs were often associated with osteoid forming Howship's lacunae (Fig. 5). All cats were neutered European domestic shorthairs between 4 and 19 years of age with a male°female ratio of 8:7. Seven out of 13 cases available for follow-up presented a recurrence within 2 months after marginal excision. One cat had a recurrence after 515 days. In 4 cases, euthanasia was performed in relation to recurred growth of the epulis (Table 4D).
Case summaries of feline giant cell epulides.
RFI = recurrence-free interval
= neutered
= unknown.
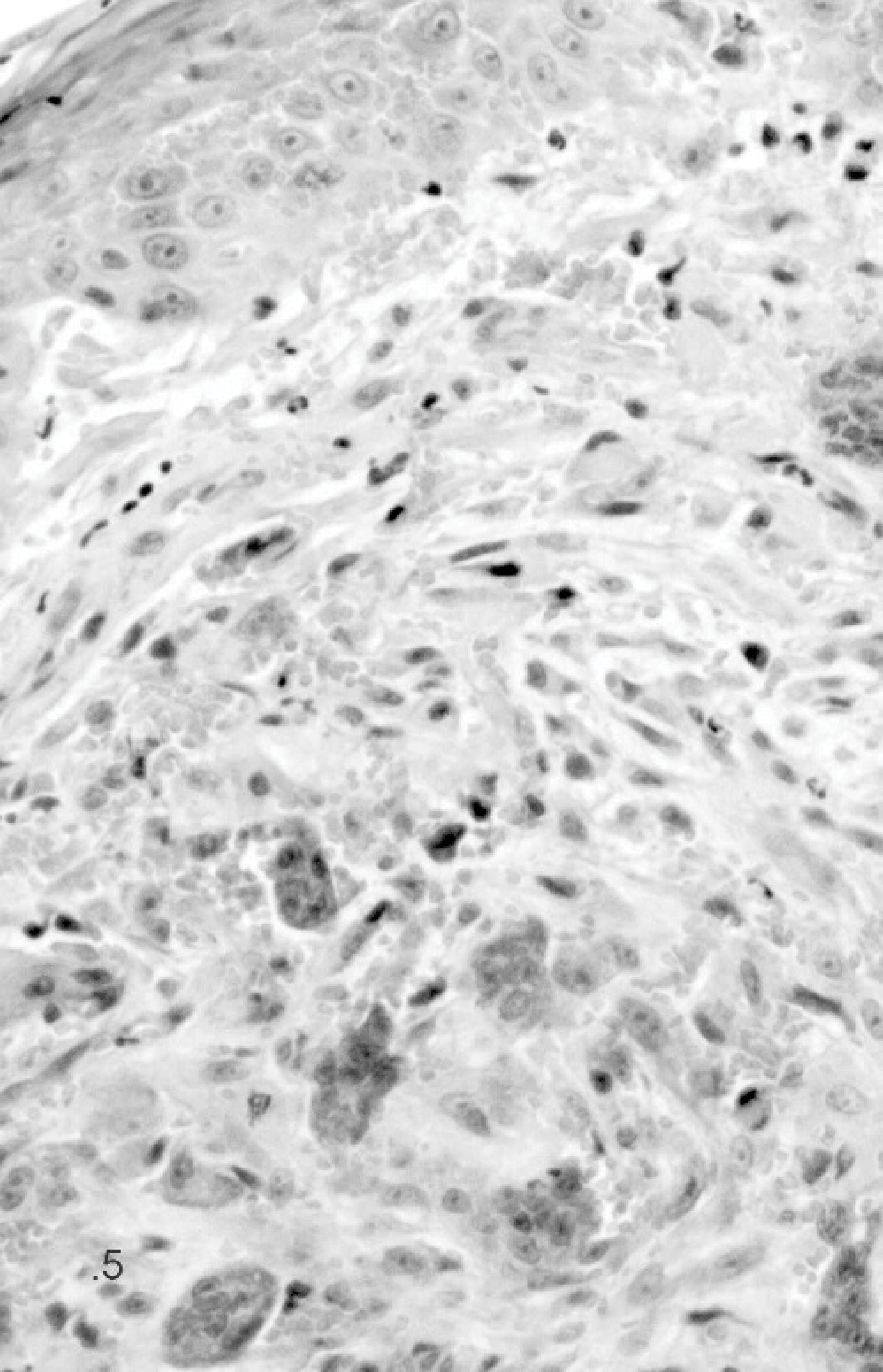
Giant cell epulis; cat. Case 12. The giant cell epulis is characterized by large numbers of MGCs scattered in fibrous, well-vascularized stroma containing round and spindle-shaped mononuclear cells. HE stain.
A chi-square test concluded that giant cell epulides presented significant difference in clinical behavior compared to the fibromatous type, because of their rapid growth character (P < .0001) and the presence of ulcerative changes (P < .01) in all giant cell epulides. The follow-up data were also significantly different, and recurrence after surgery (P < .01) was more common in giant cell epulides compared to fibromatous epulides (Fig. 10). Fibromatous epulides showed few recurrences (5 / 30), while the giant cell epulides had a high percentage of recurrences (8:13) within 515 days after marginal excision, while 5 / 13 cases showed no recurrence within the following 2 years. Death (euthanasia) related to the epulis was significant higher (P < .01) in giant cell epulides compared to the other types.
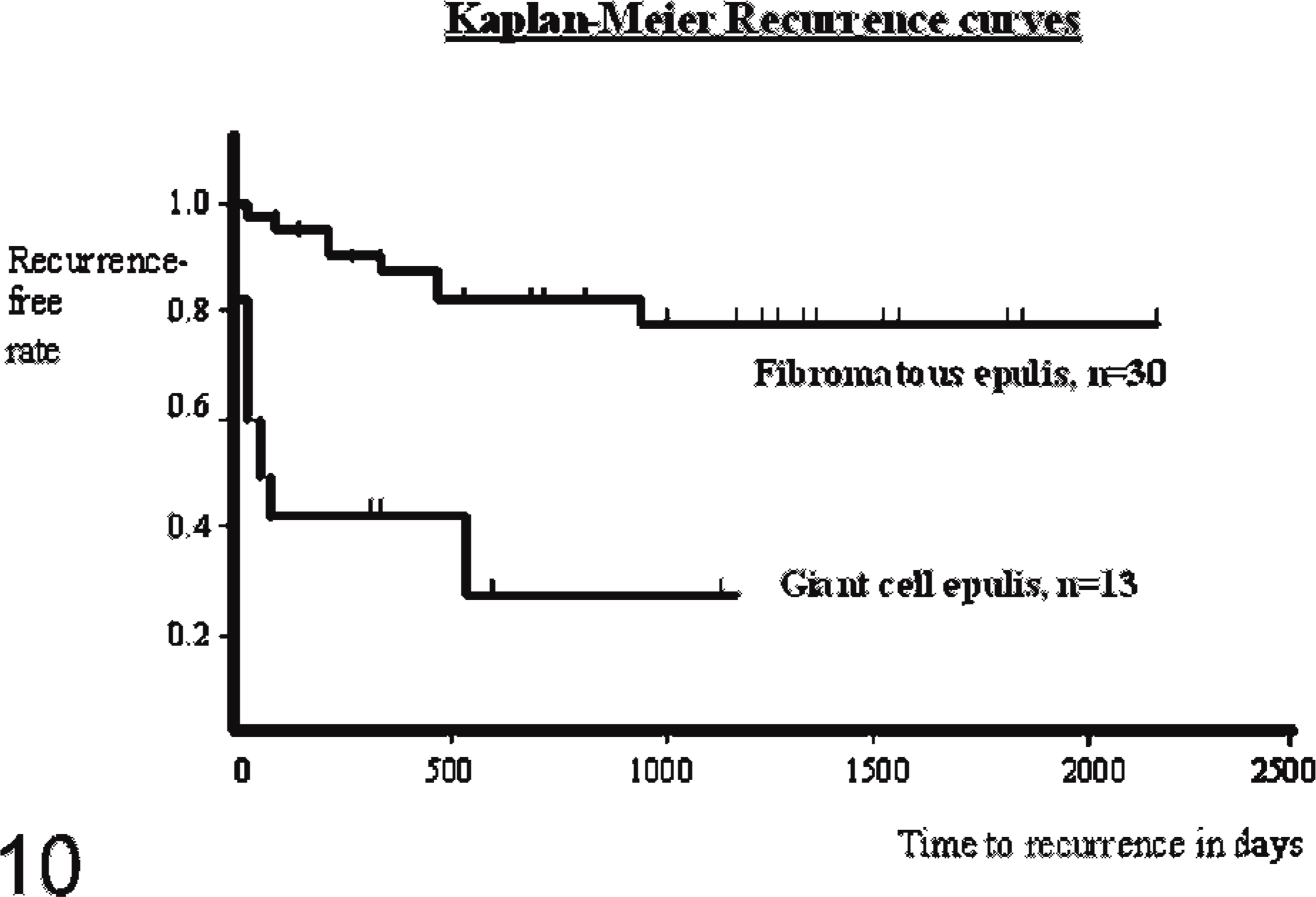
Kaplan-Meier recurrence curves for the feline fibromatous epulis and the feline giant cell epulis.
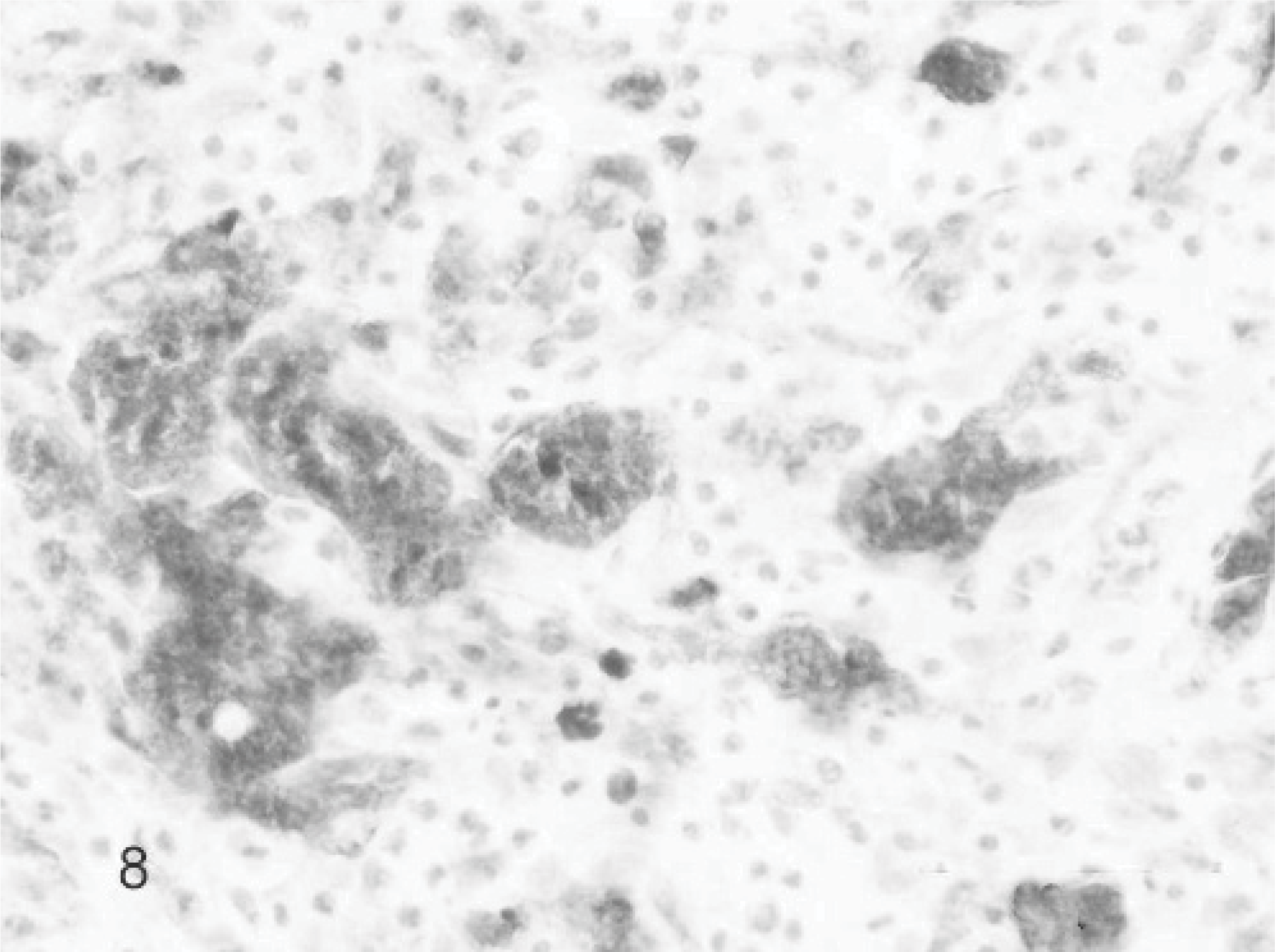
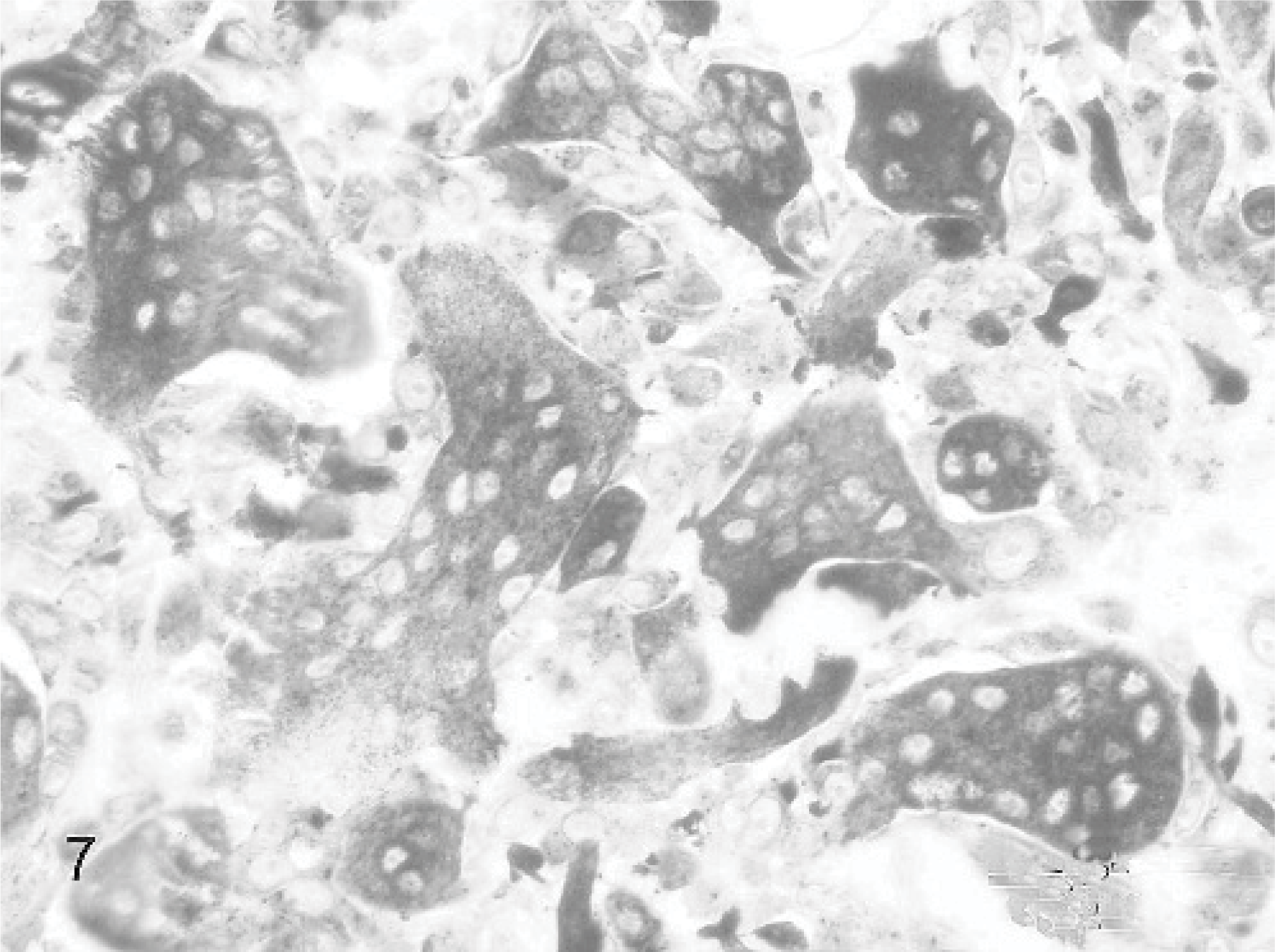
Additional staining techniques to clarify the origin of the MGCs showed in the Van Gieson staining 11 cases focal areas of osteoid and woven bone formation with lacunar resorption by osteoclastic activity (Fig. 6). All MGCs were strongly positive for the osteoclast marker TRAP. Also a substantial part of the mononuclear stromal cells showed positive staining; these positive cells tended to surround TRAP-positive MGCs and the foci containing osteoid-like material (Fig. 7). All MGCs reacted positively with monoclonal antibodies to vimentin, as did the mononuclear stromal cells (Fig. 8). The alpha smooth muscle actin staining was negative in all MGCs. Immunoreactivity for MIB-1, the proliferation marker, showed randomly scattered positivity in the nuclei of a small amount of mononuclear stromal cells, while the MGCs were negative. The factor VIII staining showed positivity of the well-differentiated endothelial cells of the vascular proliferation at the periphery of the lesion. MGCs were negative for factor VIII. Finally, the polyclonal antibody RANK, a cytokine leading to the differentiation of osteoclast progenitors into mature osteoclasts in presence of its ligand, was positive in the cytoplasm of the MGCs and also in mononuclear stromal cells (Fig. 9). In both the foreign body–type feline granulomatous inflammation and feline leprosy multinucleated cells were negative in the RANK staining, suggesting a different origin of the MGCs.
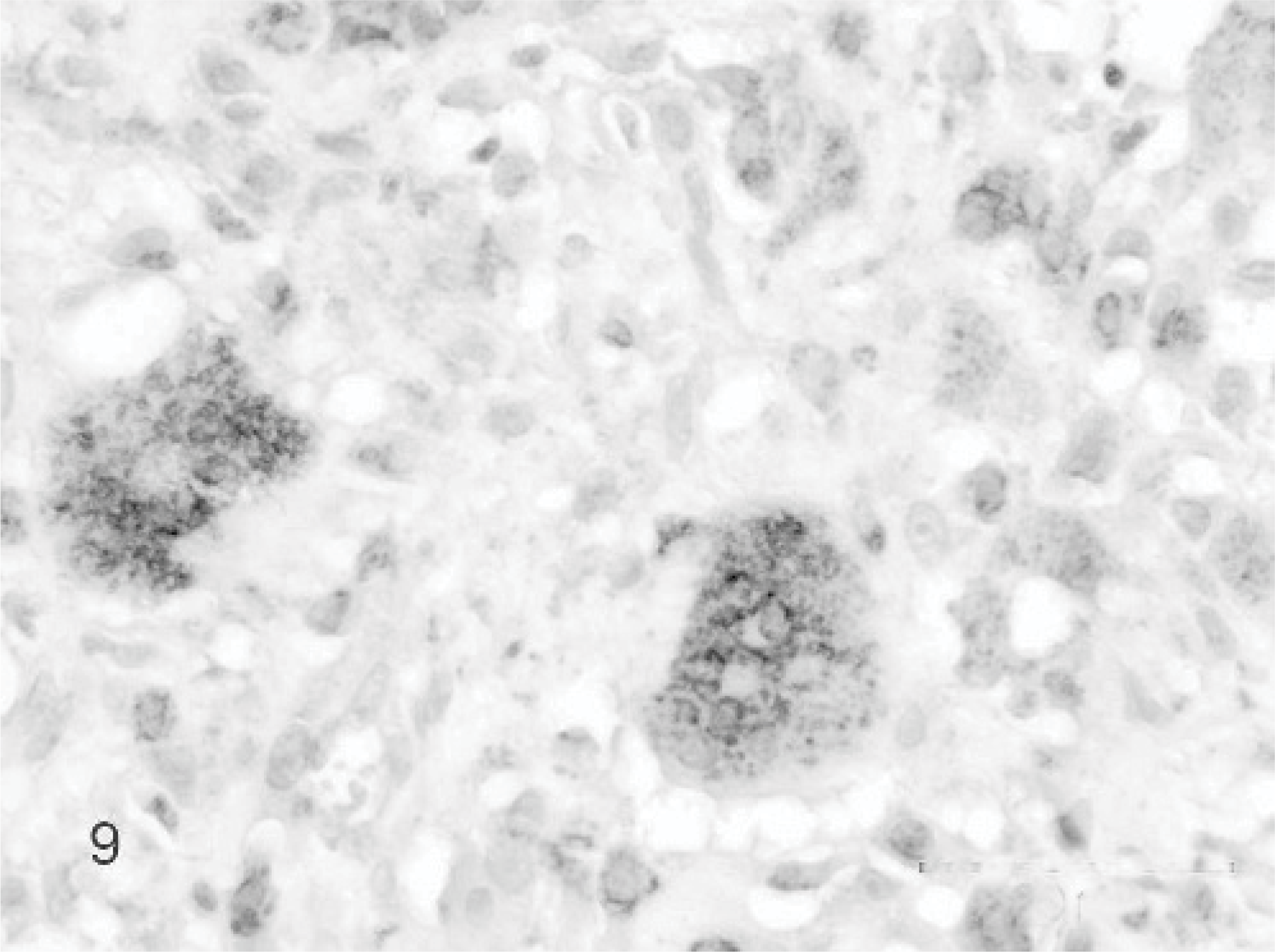
Giant cell epulis; cat. Case 13. Cytoplasm of MGCs and many mononuclear stromal cells are positive for RANK immunolabeling. Mayer's hematoxylin counterstain.
Giant cell epulis; cat. Case 9. MGCs and mononuclear stromal cells are positive for vimentin immunolabeling. Mayer's hematoxylin counterstain.
Giant cell epulis; cat. Case 7. All MGCs and many mononuclear stromal cells are positive for TRAP histochemical labeling. Mayer's hematoxylin counterstain.
Giant cell epulis; cat. Case 1. MGCs are closely associated with osseous material, creating typical resorption lacunae of Howship. Van Gieson staining.
Discussion
The most common type of epulis in the cat is the fibromatous epulis, usually with both acanthomatous and ossifying components. It is a benign solitary or multiple occurring hyperplastic lesion, slowly growing and nonulcerating. Marginal excision is usually successful. Although recurrences are rare, multiple epulides tend to recur more often after marginal excision, consistent with previous findings.4 Acanthomatous and ossifying epulides were rare in this survey, and their clinical course was similar to the fibromatous epulis. In contrast, the second most important type of epulis is the giant cell type, which represents an epulis with a potentially more aggressive clinical course. Nearly all investigated feline giant cell epulides were poorly demarcated solitary lesions with severe ulceration. Microscopically, giant cell epulides are composed of numerous MGCs scattered in a heterogeneous background population of polygonal to elongate stromal cells, with hemorrhage and islands of osteoid material or woven bone. MGCs are also found in association with osteoid forming Howship's lacunae. Reactive inflammation and vascular proliferation is common in ulcerated areas, representing a reactive rather than a neoplastic component of the lesion. A substantial amount of the moderately proliferative active mononuclear stromal cells expressed osteoclast-precursor characteristics, evidenced by the positive TRAP staining and by the identification of the RANK. The MGCs exhibit features of nonproliferating terminally differentiated osteoclasts, including TRAP positivity and lacunar bone resorption. MGCs may be formed by the fusion of mononuclear stromal cells and are under control of a RANK/RANKL regulatory axis.3 They are probably upregulated by persistent inflammatory cytokines or other local environmental factors associated with the extensive ulceration of the giant cell epulis, resulting in increased formation of osteoclasts and subsequent osteolysis.
The osteoclast-like MGCs may be derived from a monocyte/macrophage-like osteoclast precursor, which differentiates into osteoclasts under the influence of mononuclear osteoblast-like stromal cells.11,13
Although the fibromatous and ossifying epulis in the cat shows no evidence at all for osteoclastic activity, and recurrences are very rare, the giant cell epulis most likely is a variant of the fibromatous and ossifying epulis in which rapid growth and extensive ulceration and inflammation results in strongly increased osteoclastic activity. The morphological and immunohistochemical findings show that the MGCs in the feline giant cell epulis express the characteristic phenotypes of osteoclasts and that the RANK/RANKL may play an important role in the formation of the giant cells in these lesions. For most feline epulides, because of their localization, only marginal excision is possible. In combination with a low recurrence rate the prognosis in general is good. But this is not true for the giant cell epulis. The high recurrence rate and poor prognosis of the giant cell epulis in the cat after marginal excision alone may be related to the rapid growth and poor demarcation of the lesion associated with the persistent inflammatory component.
Other therapy modalities, such as external beam radiation therapy, may result in better survival statistics of feline giant cell epulides, as described in both the canine fibromatous, acanthomatous and ossifying epulis,14,23 and the feline fibromatous and ossifying epulis.16 In conclusion, a concise clinicohistopathologic differentiation of feline epulides is important in order to give a valid prognosis and to prevent possible recurrences.
Footnotes
Acknowledgements
We would like to thank Dr. Van De Waele and Dr. M. Heimann for providing positive control tissues.
