Abstract
Cutaneous lymphoma is a common skin neoplasm of pet rabbits in Europe but is rarely reported in pet rabbits in North America. These neoplasms have not been previously characterized, nor has the cause for the apparent predilection for cutaneous lymphoma in European pet rabbits compared with North American pet rabbits been investigated. In this retrospective study, the authors morphologically and immunohistochemically characterized 25 cutaneous lymphomas in European pet rabbits according to the World Health Organization classification. Tumors were classified as diffuse large B cell lymphomas, with 14 lymphomas exhibiting a centroblastic/centrocytic subtype and 11 tumors exhibiting a T cell–rich B cell subtype. To investigate a potential viral etiology of these lymphomas, 3 diffuse large B cell and 3 T cell–rich B cell lymphomas were evaluated by polymerase chain reaction for retroviral and herpesviral genes. Neither virus was detected. In contrast to other domestic animals, cutaneous lymphomas in European pet rabbits were highly pleomorphic and frequently contained multinucleated giant cells. Unexpectedly, the second most common subtype was T cell–rich B cell lymphoma, a subtype that is rare in species other than horses. Based on a limited number of samples, there was no support for a viral etiology that would explain the higher incidence of lymphoma in European pet rabbits compared with American pet rabbits. Further investigation into genetic and extrinsic factors associated with the development of these tumors is warranted.
Over the past decade, rabbits have become increasingly popular as companion animals; however, most publications on neoplasia in this species have been derived from spontaneous or induced tumors in animals kept under laboratory conditions. It is unclear whether data derived from such studies are applicable to pet rabbits.
Most pet and laboratory rabbit breeds are derived from the European rabbit, Oryctolagus cuniculus, which is distinct from the wild rabbit in the Americas, the cottontail rabbit, Sylvilagus sp. Uterine adenocarcinoma is the most common tumor reported in older rabbits, and by age 5 to 6 years, 80% of females will develop this neoplasm. 18 Although uterine tumors can occur in younger rabbits, lymphoma is the most common tumor in juvenile rabbits. It typically presents as a multicentric disease affecting one or more organs, with the stomach, kidney, liver, spleen, and lymph nodes frequently involved. 18 Recent reports of lymphoma in rabbits also involved the cecum, spinal cord, and Harderian gland, where tumors were all B cell lymphomas. 14,19,26
Unlike multicentric lymphoma, cutaneous lymphoma is rarely diagnosed in rabbits in the United States. A recent 16-year study of pet rabbits in the United States found that trichoblastomas were by far the most common cutaneous neoplasm, with collagenous hamartomas and leporipoxvirus-associated Shope fibromas being the next most frequent cutaneous tumors in pet rabbits. Just 1 of 179 rabbits (0.55%) in this study had cutaneous lymphoma. 28 On the other hand, in Europe, lymphoma appears to be a commonly diagnosed malignant cutaneous neoplasm in pet rabbits. A recent survey of a private lab in Germany found that 45 of 280 rabbits (14%) with cutaneous neoplasms had lymphoma. 27
The difference in incidence between these 2 populations raises the question of factors predisposing the European pet rabbit population to this tumor. Various viral infections are associated with lymphoproliferative diseases in other species and in wild rabbits (Sylvilagus sp.) 11 –13,18,23 and therefore may also play a role in cutaneous lymphoma in pet rabbits.
The purpose of this retrospective study was to morphologically and immunophenotypically characterize cutaneous lymphomas in European pet rabbits and to investigate in a subset of cases the possibility of a viral infection as a potential cause.
Materials and Methods
Cases
A review of the records at the Fachpraxis für Tierpathologie, a private diagnostic laboratory in Munich, Germany, revealed 25 pet rabbits with a histologic diagnosis of cutaneous lymphoma, for which formalin-fixed paraffin-embedded tissues were available.
Histopathology
Serial sections 3 to 5 μm in thickness were prepared from formalin-fixed paraffin-embedded tissue samples from each case. Sections from each tissue were routinely stained with hematoxylin-eosin (HE) and evaluated according to the current World Health Organization (WHO) classification of malignant lymphomas. 7,24
Immunohistochemistry
Immunohistochemistry for CD3 and CD79a was performed on tissue from all animals, to classify neoplasms as T or B cell origin, following previously published methods. 16,17 Briefly, deparaffinization, antigen retrieval, labeling, and counterstaining were performed with the BOND MAX Automated Staining System (Vision BioSystems/Leica, Bannockburn, Illinois) using secondary anti-rat and anti-mouse antibodies, purified against rabbit, in combination with the BOND Compact Polymer Detection System (Vision BioSystems/Leica). Antigen retrieval was achieved using the BOND Epitope Retrieval Solution 1 (Vision BioSystems/Leica) for 20 minutes. Sections were incubated with a rat-monoclonal anti-CD3 (Peter Moore, University of California at Davis, Davis, California) at a concentration of 1 in 10 and a mouse-monoclonal anti-CD79a (Dako Cytomation, Carpenteria, California) at a concentration of 1 in 100. Labeling was “visualized” with 3, 30-diaminobenzidine substrate (Vision BioSystems/Leica), and sections were counterstained with hematoxylin. Antibodies were validated in normal rabbit lymph node, where perimembrane labeling for CD79a was present in lymphocytes in follicular centers, whereas perimembrane labeling for CD3 was present in paracortical lymphocytes. For negative controls, the primary antibodies were replaced with homologous nonimmune sera or buffer. Only perimembrane labeling within neoplastic cells was interpreted as positive.
Polymerase Chain Reaction
Tissue from tumors of 6 cases (3 diffuse large B cell lymphomas and 3 T cell–rich B cell lymphomas) were evaluated by polymerase chain reaction (PCR) for retroviral and herpesviral genes. Formalin-fixed paraffin-embedded tissue sections were deparaffinized with Citrisolv (Fisher Scientific, Waltham, Massachusetts) and washed in ethanol prior to nucleic acid extraction, following the guidelines in the DNeasy Tissue Kit Handbook (QIAGEN, Valencia, California). Nucleic acids (DNA and RNA) were extracted from the samples using the DNeasy Tissue Kit. RT (reverse transcriptase)–PCR and PCR reactions were carried out with the One-Step RT-PCR Kit (QIAGEN) and Taq PCR Master Mix Kit (QIAGEN), respectively.
For retrovirus RT-PCR, initial testing was performed using degenerate primers that were designed based on conserved regions of the polymerase genes of murine, feline, and ape gammaretroviral sequences. The assay targets a 300-bp fragment of the polymerase gene with the following primer sequences: (forward primer) 5′-GAYTTCACHGARATMAAGCC-3′ and (reverse primer) 5′-TCTRTTCATTCTTTCTACCTG-3′. Cycling parameters were 50°C for 30 minutes and 95°C for 15 minutes, followed by 40 cycles of 94°C for 30 seconds, 52°C for 30 seconds, and 72°C for 30 seconds, followed by a final elongation of 72°C for 7 minutes. Further retrovirus RT-PCR testing was performed using 2 pairs of degenerate primers from the MOP-2 primer set of Seifarth et al 20 for the detection of a portion of the gammaretroviral reverse transcriptase gene sequence (125–150 bp). The primers were modified by the removal of the 6–base clamp extension at the 5′ end. Each of the primer pairs—set A, (fwd) 5′TGGAAAGTGYTRCCMCARGG3′ and (rev) 5′CAGCAGSAKGTCATCCAYGTA-3′ and set B, (fwd) 5′-TKKACMSKMYTRCYYCARGGG-3′ and (rev)5′-KMWKRYCATCMAYRTAMTG-3′—was used in a separate uniplex reaction. Cycling conditions started with reverse transcription at 45°C for 60 minutes, then an incubation at 95°C for 15 minutes, followed by 3 cycles of 94°C for 30 seconds, 45°C for 3 minutes, and 72°C for 1 minute, then 35 cycles of 94°C for 30 seconds, 50°C for 2 minutes, and 72°C for 1 minute, with a final extension step at 72°C for 7 minutes. Feline leukemia provirus–positive blood was used as the positive control for all retrovirus RT-PCR testing. Nuclease-free water was used as the negative control template for all RT-PCR and PCR testing.
Herpesvirus PCR testing was carried out with the universal herpesvirus PCR protocol of VanDevanter et al. 25 Equine herpesvirus-1 DNA was used as the positive control.
PCR products were analyzed by electrophoresis in 2% agarose gels prestained with ethidium bromide and visualized on a UV transilluminator.
Results
Clinical History and Follow-up
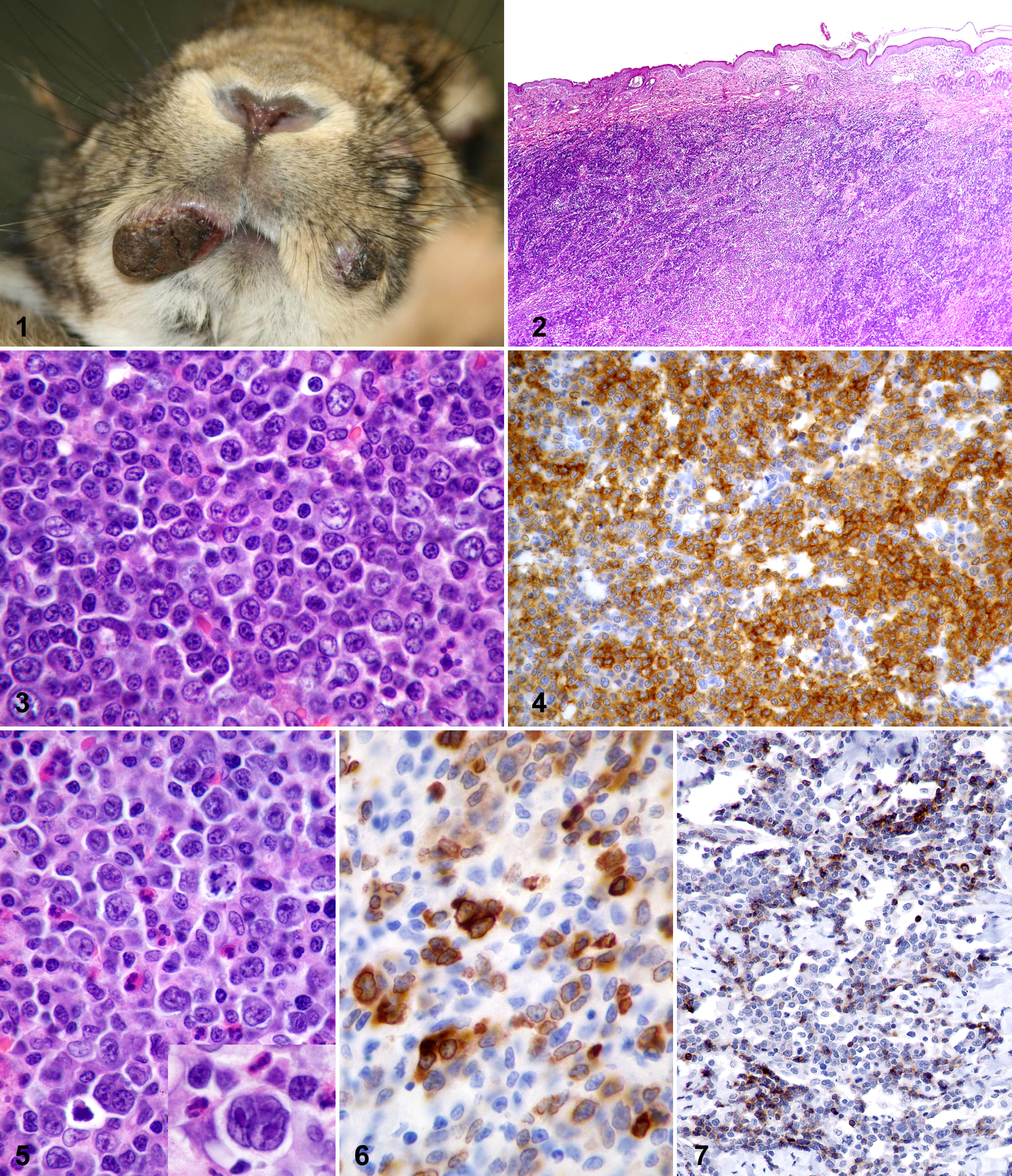
Twenty-five rabbits were included in the study. All specimens had been submitted by veterinarians located throughout Germany. Limited clinical history and/or follow-up information were available for 15 animals. Among those for which information was available, various breeds were represented, and the age range was 1.5 to 6 years (mean = 4 years). No sex predilection was identified. All animals had a previous diagnosis of cutaneous or subcutaneous lymphoma, which presented as one or more nodules at various locations on the body (Fig. 1). Six animals had solitary cutaneous nodules, 3 had 1 to 3 nodules, and 6 had 4 or more nodules. One rabbit (case No. 15) also had splenic malignant lymphoma at the time of diagnosis. All animals were treated only surgically; chemotherapy was not reported. Seven animals developed postsurgical extracutaneous spread of disease (lymph node, intestine, eye) or developed multiple-organ involvement without specification of the other organs. When survival time postdiagnosis was reported, it was not indicated whether the animal was euthanized or died naturally. Table 1 summarizes the tumor distribution, results of lymphoma classification according to WHO guidelines, and survival time postdiagnosis, when available, for each animal.
Distribution, Classification, and Outcome of Cutaneous Lymphomas in Pet Rabbits
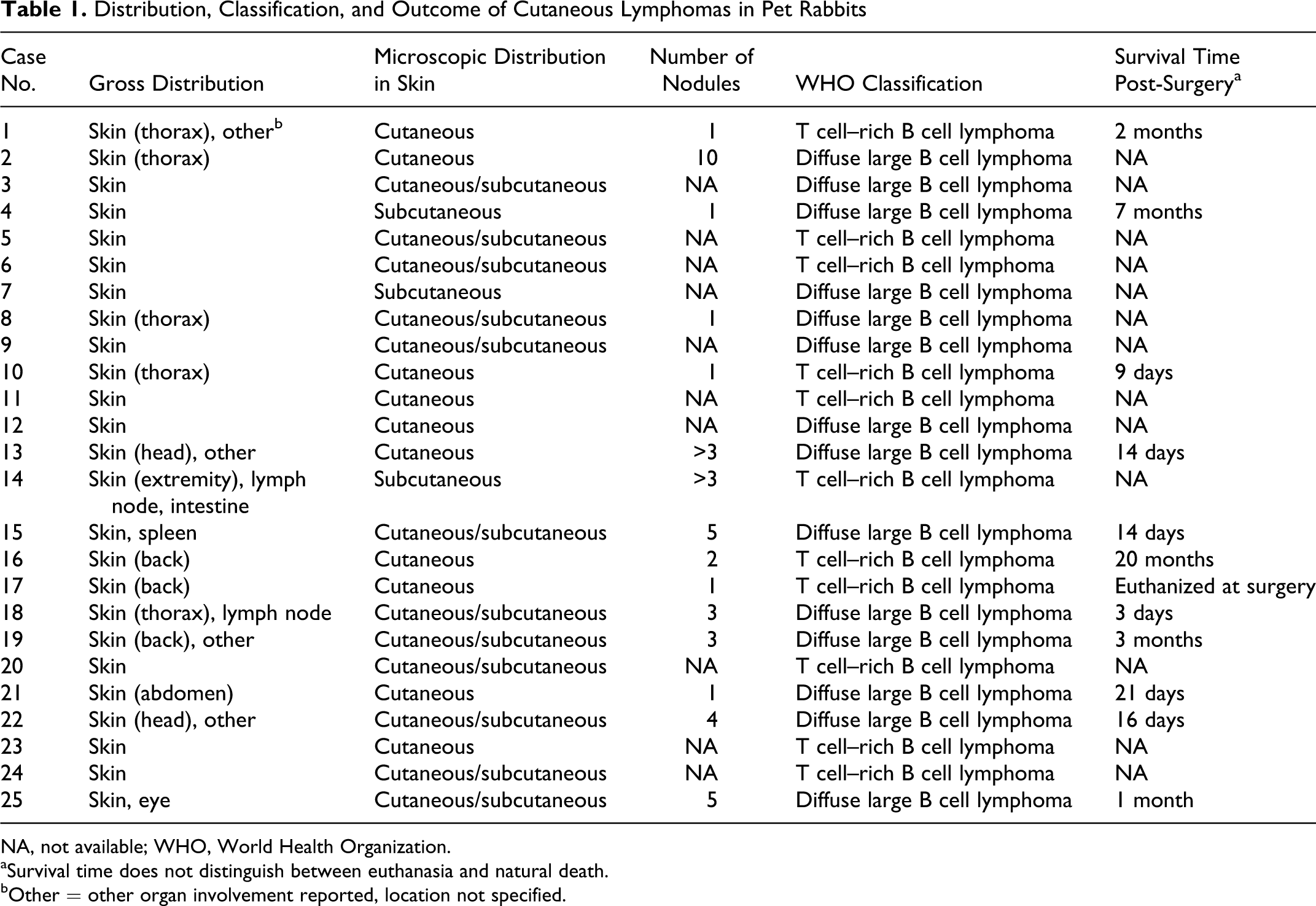
NA, not available; WHO, World Health Organization.
aSurvival time does not distinguish between euthanasia and natural death.
bOther = other organ involvement reported, location not specified.
Histopathology and Immunohistochemistry
Histologically, all cutaneous neoplasms were composed of dense sheets of lymphocytes within the dermis and/or subcutis (Fig. 2). Neoplastic cells frequently extended close to the dermoepidermal junction; however, epitheliotropism was not identified. Lymphomas were classified into 2 subtypes of diffuse large B cell lymphoma. One subtype (n = 14) was composed of a population of predominantly large lymphocytes with nuclei typically greater than twice the size of an erythrocyte. These cells had large euchromatic, round to rarely indented nuclei with multiple prominent large, distinct membrane-associated nucleoli typical of centroblasts (Fig. 3). Mitotic figures were observed in most high-power fields. Large lymphoid cells were admixed with variable numbers of intermediate-size lymphoid cells more characteristic of centrocytes, with indented nuclei, densely stippled chromatin, and smaller indistinct nucleoli (Fig. 3). Immunohistochemistry revealed that the vast majority of the lymphoid cells exhibited positive labeling for CD79a (Fig. 4) and were negative for CD3, consistent with a diagnosis of a centroblastic/centrocytic lymphoma. The second subtype (n = 11) was composed of a more pleomorphic neoplastic cell population comprising small numbers of large lymphocytes in a background of small mature lymphocytes (Fig. 5). The large lymphocytes resembled centroblasts but exhibited great variability in nuclear shape, with frequent bi- and multinucleated cells (Fig. 5 inset). Mitotic figures were common, including bizarre forms. Moderate numbers of heterophils and/or eosinophils were scattered throughout these lymphomas (Fig. 5). Immunohistochemistry confirmed that the population of large cells, including the multinucleated cells, were positive for CD79a (Fig. 6), whereas the smaller cells were positive for CD3 (Fig. 7), consistent with a diagnosis of a T cell–rich B cell lymphoma.
Polymerase Chain Reaction
Neither retroviral nor herpesviral nucleic acid was detected in tissues from any of the 6 cases tested.
Discussion
Lymphoma classification systems in veterinary species have evolved in parallel with human classification systems. 7,24 Although older classification systems were based on specific biological or morphological characteristics, the current WHO system of classification of hematopoietic neoplasms attempts to define lymphomas as specific disease entities, taking into account cellular morphology and lineage, topography, and general biology. 7,24 This classification system has been applied to the classification of lymphoma in dogs 24 and lions, 8 and we found that it was also readily applied to pet rabbit lymphomas.
The tumors in our study were classified as diffuse large B cell lymphomas of either a centroblastic/centrocytic subtype (14/25) or a T cell–rich B-cell subtype (11/25). Although diffuse large B cell lymphoma is the most common malignant lymphoma found in the lymph nodes and viscera of the dog, cow, cat, horse, and pig, T cell lymphoma is the most common cutaneous lymphoma in these species. 23,24 Cutaneous lymphoma in other species is most commonly of T cell origin and often exhibits epitheliotropism (mycosis fungoides). 4 Epitheliotropic T cell lymphoma has previously been reported in rabbits, 29 but epitheliotropism was not observed in rabbits in this study. T cell–rich B cell lymphomas are only commonly described in horses, where they typically present as multiple subcutaneous tumors. 15 They are rare in other species, where they usually involve the lymph nodes. 1,21 A case of disseminated T cell–rich B cell lymphoma involving the skin and multiple viscera was previously reported in a rabbit. 5
Seven of 12 (58%) animals for which follow-up information was available had reported involvement of other organs, and 10 of 12 (83%) died or were euthanized by 3 months postdiagnosis. Although these data suggest an aggressive disease with a rapid progression, the true course of disease is undetermined because many of the animals were euthanized due to poor long-term prognosis. Regardless, the high percentage of early death/euthanasia differs from cutaneous lymphomas in other species, which typically have a slowly progressive course of disease. 2,22
The tremendous difference in incidence of lymphoma between the European and American pet rabbit populations raises the question of possible factors leading to a higher incidence of lymphoma in European pet rabbits. Housing conditions and feeding practices most likely resemble those of pet rabbits in North America; therefore, genetic factors and/or infectious agents endemic to Europe may be the most likely explanation for this difference. Because numerous viruses are associated with lymphoproliferative disease in other animal species, 9 –13,23,30,31 we hypothesized that a viral etiology might underlie cutaneous lymphomas in rabbits. In humans, T lymphotropic virus (HTLV-1), a retrovirus, causes adult T cell leukemia/lymphoma and other lymphoproliferative disorders. Epstein-Barr virus (EBV), a gamma herpesvirus, has been associated with Hodgkin and Burkitt lymphoma in humans. 10,31 Some of the most widely known virally induced lymphoproliferative diseases in veterinary medicine include retrovirus-induced leukemias and lymphomas in cats and cattle caused by feline leukemia virus and bovine leukemia virus, respectively. 23 An association has also been made between Herpesvirus sylvilagus, a gamma herpesvirus similar to EBV, and lymphoproliferative disease in rabbits. 11 –13,18 To the authors’ knowledge, this association has only been found in cottontail rabbits but not domestic rabbits. Furthermore, human retrovirus type 5 (HRV-5), a retrovirus associated with chronic inflammatory conditions and lymphoma in humans, has recently been confirmed to be an endogenous retrovirus of rabbits. 3,6 There are also experimental models of viral lymphomagenesis in rabbits. 9,30 An EBV-like virus from cynomolgus macaques (Cyno-EBV) can induce lymphoproliferation and T cell lymphomas when administered orally in laboratory rabbits. 10 HTLV-1 has been shown to infect rabbits; however, the infection does not consistently induce lymphoma. 30 Neither retroviral nor herpesviral nucleic acid was detected in the tissues tested on a selected number of cases in this study.
In conclusion, adult European pet rabbits have an apparent predisposition to develop nonepitheliotropic diffuse large B cell lymphomas. This is in contrast to cutaneous lymphomas commonly encountered in the veterinary diagnostic pathology setting, which are most often epitheliotropic T cell lymphomas in the skin of dogs and cats. These rabbit tumors are highly heterogeneous, and centroblastic/centrocytic as well as T cell–rich B cell subtypes are common. Local spread and involvement of distant organs are not uncommon. Neither retroviral nor herpesviral nucleic acid was identified in these cases, and further investigation into the factors associated with the higher incidence of lymphoma in the European pet rabbit population is warranted.
Footnotes
Acknowledgements
We thank Tom Wood of the Immunohistochemistry Laboratory at the DCPAH, Michigan State University for technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
