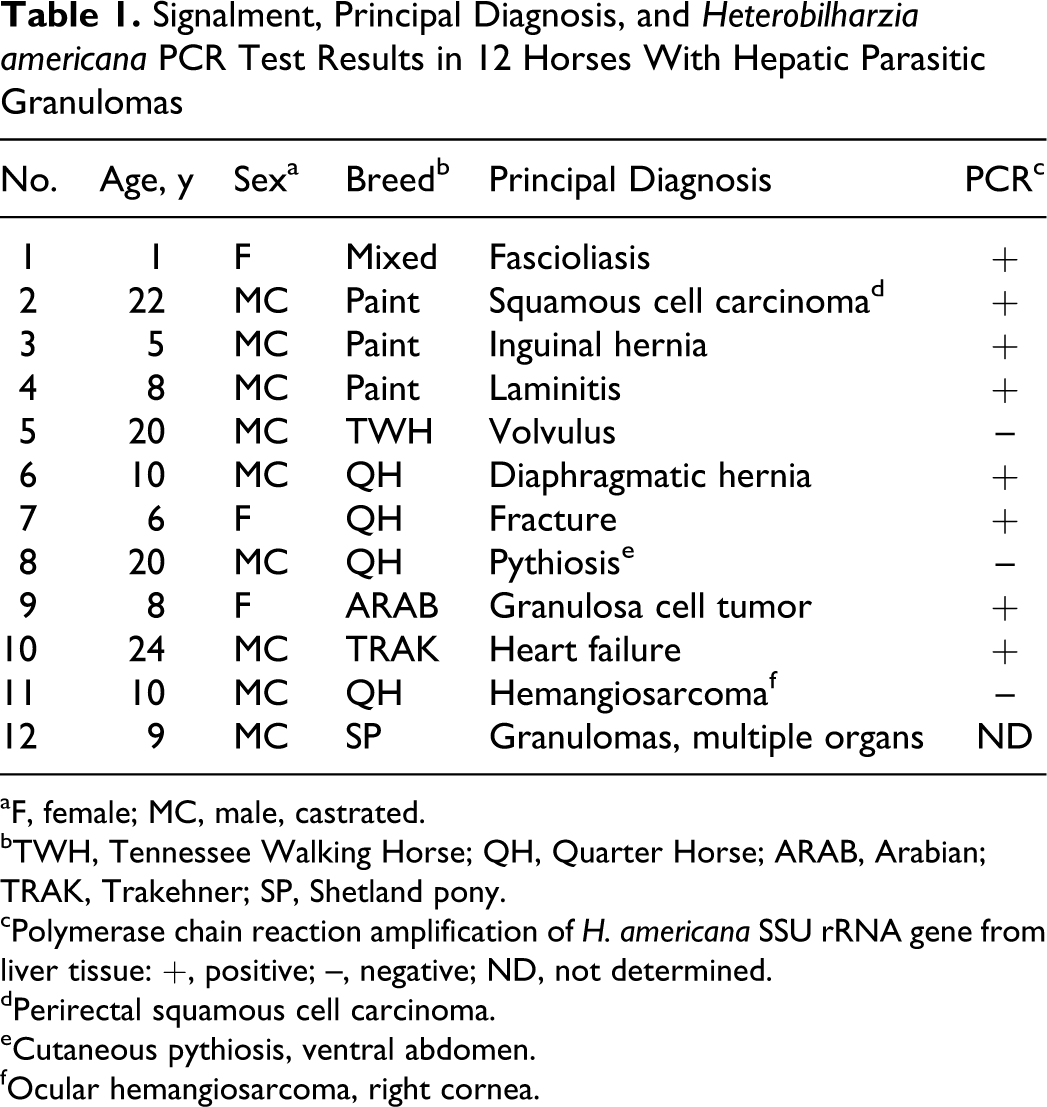
Abstract
The schistosome Heterobilharzia americana infects dogs, raccoons, and other mammals in the southeastern United States. Migration of eggs into the liver results in parasitic granulomas with varying degrees of fibrosis and inflammation. Recently, hepatic parasitic granulomas in horses were shown to be caused by H. americana infection. In the present study, samples of liver from 11 of 12 horses with hepatic granulomas identified at necropsy (n = 11) or surgical biopsy (n = 1) were used for DNA extraction, polymerase chain reaction amplification and sequencing using primers specific for a portion of the H. americana small subunit ribosomal RNA gene. A polymerase chain reaction amplicon of the correct size was produced from the extracted DNA in 8 of the 11 horses. Amplicons from 5 of the 8 positive horses were sequenced and had 100% identity with H. americana. In all but 2 of the 12 horses, Heterobilharzia was not responsible for the primary clinical disease, and the hepatic granulomas were considered an incidental finding.
Heterobilharzia americana, a trematode in the family Schistosomatidae, is the cause of canine schistosomiasis. Infections in dogs and raccoons have been well characterized and result in granulomatous inflammation in various organs, including the liver. 1,8,9 Eggs released from the female, which lives in copula with the male in the mesenteric veins, migrate to the liver through the portal circulation where a characteristic inflammatory response develops around individual or small clusters of eggs. Severe infections may result in liver dysfunction, fibrosis, and nodular regeneration. 6 The geographic range of the infection depends on the presence of the intermediate host, the lymnaeid freshwater snail (Bakerilymnaea cubensis or Pseudosuccinea columella). Reported cases of canine schistosomiasis have been limited mainly to the Gulf Coast and south Atlantic regions of the United States, an area that includes part of the known range of the intermediate host. The natural definitive host is the raccoon. In some areas of Texas, the prevalence of adult H. americana infection is as high as 47% in raccoons of all ages, and up to 85% in older raccoons. 7
Although H. americana infections in horses in this same geographic region have long been suspected, 2,3 definitive evidence has only come recently with the successful amplification and sequencing of a portion of the 18 S small subunit ribosomal RNA (SSU rRNA) gene of H. americana from the liver of an infected horse. 5 Horses with presumptive heterobilharziasis typically had small randomly disseminated granulomas in the liver. On antemortem abdominal ultrasonography, severely affected livers had hyperechoic foci that gave the liver a “starry sky” appearance, enhanced by areas of dystrophic mineralization within the granulomas. 3 Most infections, however, do not result in clinically evident liver dysfunction, and hepatic infection is not suspected until postmortem examination after death from an unrelated cause. Granulomas may also be found in the small intestine, diaphragm, and lungs. 2,5 Although the granulomas may be numerous, eggs or eggshell fragments are only rarely observed. The association of these hepatic granulomas with the presence of H. americana has been documented, based on histopathology and DNA sequencing, in a single horse. 5 The purpose of the current study is to describe the gross and histologic lesions in 12 horses with hepatic granulomas and to report the molecular evidence of an association of the granulomas with H. americana infection in 8 of 11 horses tested.
Materials and Methods
The 12 horses in this study were from the Gulf Coast and southeastern regions of Texas (Table 1). All horses were admitted to the Texas A&M University Veterinary Medical Teaching Hospital (College Station, Texas) for unrelated reasons and were either euthanized and submitted for necropsy (n = 11) or examined by abdominal ultrasonography followed by percutaneous liver biopsy and histopathology (n = 1). One horse (No. 1) was euthanized in 2005 and another (No. 12) in 2008; the remaining 10 horses were euthanized (Nos. 2–8, 10, 11) or biopsied (No. 9) over a 16-month period in 2010–2011. All horses in the study had multiple small granulomas in the liver. One horse (No. 1) also had evidence of hepatic fibrosis secondary to fascioliasis. The horses included 9 geldings and 3 mares of multiple breeds and ranged in age from 1 to 24 years. The clinical disease varied among cases, but only 1 horse had obvious clinical evidence of hepatic disease (No. 12). Four horses (Nos. 3, 5–7) were euthanized due to an acute traumatic injury or colic, and 7 horses (Nos. 1, 2, 4, 8, 10–12) were euthanized because of a chronic disease condition. One horse (No. 9) was treated by ovariectomy and recovered. In all cases, the clinical findings were compatible with the final diagnosis. For optimization of the polymerase chain reaction (PCR) analyses, only horses with fresh, unfixed liver tissue available for DNA extraction were included in the molecular testing.
Signalment, Principal Diagnosis, and Heterobilharzia americana PCR Test Results in 12 Horses With Hepatic Parasitic Granulomas
aF, female; MC, male, castrated.
bTWH, Tennessee Walking Horse; QH, Quarter Horse; ARAB, Arabian; TRAK, Trakehner; SP, Shetland pony.
cPolymerase chain reaction amplification of H. americana SSU rRNA gene from liver tissue: +, positive; –, negative; ND, not determined.
dPerirectal squamous cell carcinoma.
eCutaneous pythiosis, ventral abdomen.
fOcular hemangiosarcoma, right cornea.
Tissue samples from all major organs of the necropsied horses were immersed in 10% buffered formalin, routinely processed for histologic examination, sectioned at 4 μm, and stained with hematoxylin and eosin. A minimum of 6–10 granulomas were dissected from the liver of 10 horses (Nos. 1–8, 10, 11) without formalin fixation and frozen at –80°C for subsequent PCR amplification and sequencing. Granulomas from the lungs of 1 horse (No. 6) and the small intestine of another (No. 7) were processed in the same manner. Hepatic samples obtained surgically via percutaneous liver biopsy (No. 9) were processed in toto. For the PCR assay, DNA extraction and amplification were performed using a commercial kit and primers specific for the H. americana SSU rRNA gene, as described. 4 Positive samples were sequenced and compared with the H. americana SSU rRNA gene sequence in GenBank (accession no. AY157220).
Results
The number of macroscopic hepatic granulomas ranged from a few dozen to several thousand. The most unifying characteristic of the granulomas was their spherical shape, the random and disseminated pattern of their distribution in the liver, and the presence of fibrosis (Fig. 1). The granulomas ranged from 1 to 4 mm in diameter, often coalescing in more severely affected areas. The degree of mineralization varied; those livers with numerous mineralized granulomas were firm and difficult to section. In the most severely affected livers, the appearance of numerous small, pale gray to white granulomas scattered through the darker hepatic parenchyma corresponded to the starry-sky pattern seen with abdominal ultrasound. 3,5 In 4 horses (Nos. 2, 6, 10, 12), granulomas were found in other tissues, including the serosa of the small intestine and colon (Fig. 2) and within the mediastinum, lung, and heart. Very rarely, serosal inflammation extended into the mesentery, and in No. 12, which was not included in the DNA analyses because no unfixed hepatic tissue was available, linear areas of granulomatous inflammation extended along the mesenteric vessels (Fig. 3). In 2 horses (Nos. 10 and 12), the severity of the hepatic lesions and involvement of the heart were considered to be responsible for the primary clinical disease.
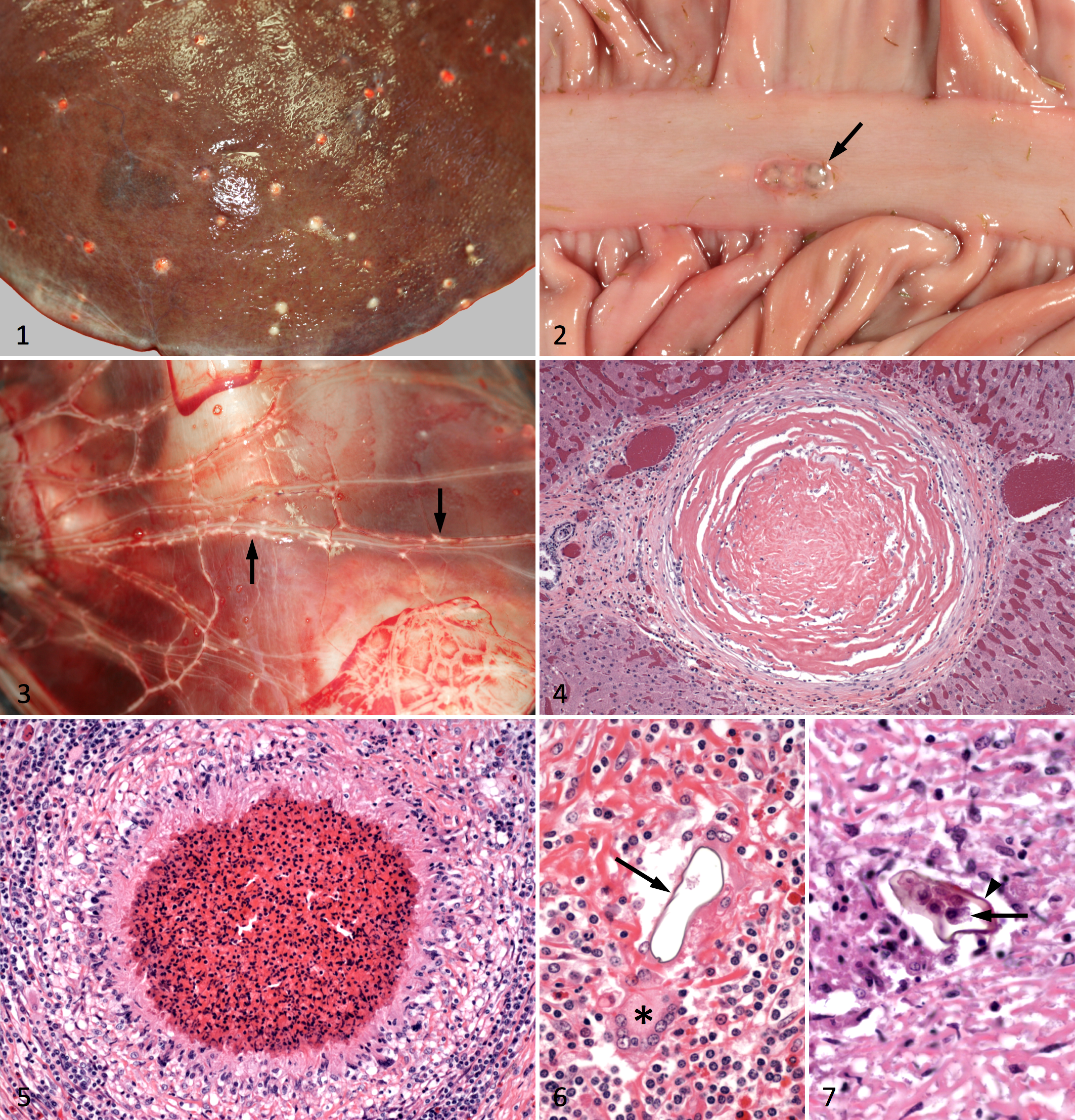
Liver; horse No. 6. Disseminated parasitic granulomas (PCR positive for Heterobilharzia americana) beneath the hepatic capsule.
Histologically, the granulomas usually had sclerotic cores surrounded by thick, loosely arranged collagen fibers and few leukocytes (Fig. 4). The central cores were often mineralized; thus, tissue sectioning without decalcification of those samples frequently resulted in loss of the central part of the granulomas. The area immediately surrounding the dense fibrous tissue generally contained at least mild inflammation consisting of variable numbers of lymphocytes, plasma cells, macrophages, and eosinophils. In contrast to the resolved, sclerotic granulomas, some livers contained granulomas with a dense center of degenerate eosinophils surrounded by a well-defined layer of epithelioid macrophages and occasional multinucleated giant cells (Fig. 5). Granulomas with less fibrous tissue and a greater number of inflammatory cells were more likely to contain fragments of empty eggshells (Fig. 6), although eggshells were rare, even in the most severely affected livers. Intact eggshells were thin, yellow-brown, up to 68 μm in length, 1-2 μm thick, and occasionally surrounded one or more round degenerate cells, each of which was approximately 10 μm in diameter (Fig. 7).
PCR assays were performed on hepatic granulomas from 11 of 12 horses and on granulomas from the lung or small intestine from 2 cases (Nos. 6 and 7, respectively). Assays of hepatic granulomas from 8 of the 11 horses (72.7%) produced amplicons of the correct size for H. americana (Table 1). Assays of tissue from the lung and small intestine of Nos. 6 and 7 also produced amplicons of the correct size. Amplicons from 5 of the 8 positive horses (Nos. 1, 2, 7, 9, 10) were sequenced directly from genomic DNA; all 5 resulting sequences had 100% identity with the H. americana SSU rRNA gene.
Discussion
Hepatic granulomas associated with H. americana infection are relatively common in horses in southeastern Texas, and new cases are being recognized with increasing frequency at our institution. Of the 328 horses necropsied over a 20-month period at Texas A&M University between January 1 2010, and August 31 2011, a total of 26 (7.9%) were found to have hepatic parasitic granulomas at necropsy. Nine of those 26 horses were included in the present study along with 2 horses euthanized in 2005–2008 and 1 horse examined by percutaneous liver biopsy in 2011. In addition to the presence of typical parasitic granulomas in the liver, a major criterion for inclusion in molecular analyses was the availability of fresh liver tissue for DNA extraction, amplification, and sequencing. Formalin-fixed tissue was not used for PCR amplification because of the failure to produce H. americana-specific PCR amplicons from formalin-fixed tissue in previous attempts. 5 Of the 11 horses for which granulomas from fresh or frozen liver tissue were available, a portion of the H. americana SSU rRNA gene was successfully demonstrated in 8. Sequencing of amplicons generated from 5 of the 8 PCR-positive horses demonstrated that all 5 cases contained DNA with 100% identity with H. americana.
In all but 2 of the 12 horses in this study, the presence of hepatic granulomas or documented infection with H. americana was considered an incidental finding unrelated to the primary disease of each horse. The direct, long-term impact on hepatic function in cases of severe heterobilharziasis is still unknown. In some cases, granulomatous inflammation resulting from the presence of aberrant H. americana eggs in extrahepatic tissues such as the heart may result in debilitating disease, including heart failure (No. 10). The possibility of a similar correlation between H. americana infection and clinically relevant hepatic dysfunction, however, has not been extensively examined, in large part because heterobilharziasis in horses is rarely discovered prior to necropsy. A recognizable ultrasonographic pattern has been demonstrated in horses with fibrosing hepatic granulomas identical to those described in this report. 3 Some horses had elevated liver enzymes, most notably γ-glutamyltransferase and aspartate aminotransferase, but this occurred in only 50% of affected horses and was sometimes associated with primary hepatic disease unrelated to heterobilharziasis.
Percutaneous liver biopsy may be useful to detect fibrosing granulomas in the liver of horses, and PCR assay of obtained liver samples may facilitate antemortem identification of H. americana, as demonstrated in this study (No. 9). Infected horses had undoubtedly been exposed at some point to snail-infested water, resulting in contact with the free-swimming and skin-penetrating cercariae, the larval stage of the schistosome. No. 10 had had frequent access to wet pasture directly adjacent to a pond. Although pasture conditions varied, all horses in this study were from the Gulf Coast and southeastern counties of Texas. Whether or not infected horses contribute to the spread of H. americana by shedding of eggs is unknown, but we consider this unlikely and suggest that the horse is most likely a dead-end host, given the chronic nature of the granulomas, the scarcity of eggs in tissue sections, and the degenerated state of the rare eggs present.
The small, fibrosing granulomas in the liver of horses caused by H. americana infection have a characteristic, widely disseminated pattern of distribution that is noticeably different from the occasional subcapsular nodules and fibrous tracts caused by larval migration of Parascaris equorum or Strongylus spp. Although H. americana was suggested as the cause of the granulomas as early as 1995, 2 positive identification has only come more recently with the successful PCR amplification and sequencing of the H. americana SSU rRNA gene from the liver of an affected horse along with the identification of a pair of adult schistosomes. 5 In this case series, H. americana DNA was documented in the granulomas of 8 of 11 affected horses.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
