Abstract
The schistosome Heterobilharzia americana infects several mammalian species in the southeastern United States, including horses, but infections have not been reported in camelids. This is a report of H. americana infection in a 6-year-old llama with extensive cardiac pathology and congestive heart failure. Parasite-induced granulomas were widely disseminated and included overwhelming involvement of the lungs and liver. Microscopic lesions in the heart included myofiber degeneration and necrosis, with extensive replacement fibrosis. Polymerase chain reaction amplification and sequencing confirmed the presence of H. americana in the lungs.
The American schistosome Heterobilharzia americana is the cause of canine schistosomiasis. Infection is dependent on the presence of specific freshwater snails that serve as the intermediate host, among which Bakerilymnaea cubensis and Pseudosuccinea columella are known to be susceptible. 10 The most important natural definitive host is the raccoon, 1 although other species can be infected. The life cycle has been studied in mice, 10 and infections in other mammals are presumed to follow a similar course. Infection begins following contact with free-standing, snail-infested water containing free-swimming cercariae, the larval form of the parasite. Cercariae penetrate the skin and migrate to the lungs, eventually travelling via the circulatory system to the liver, where growth and development of the worms occurs. Adult worms travel via the portal vein to the mesenteric veins, where males and females live in copula. Eggs shed into mesenteric venules migrate to the intestinal tract and escape the circulatory system with the help of proteolytic enzymes. Eggs that manage to cross the intestinal mucosa and reach the lumen are shed in the feces. Antigens released from the eggs stimulate an intense immune response, and aberrant migration leads to characteristic foci of granulomatous and eosinophilic inflammation in the intestine and other organs, most notably the liver. Adult worms are difficult to locate in the mesenteric veins of large animals and are only rarely found.
Recently, equine infections resulting in characteristic small, disseminated, fibrotic, and eosinophilic hepatic granulomas with occasional involvement of the lungs and intestine have been documented. 4,5 While innumerable granulomas may be in the liver of horses, clinically significant hepatic dysfunction is usually not evident, and equine heterobilharziasis is most often reported as an incidental finding. This is in contrast to cases of canine schistosomiasis, which are often associated with hypercalcemia and significant gastrointestinal disease. 3,6 Infections in camelids have not been reported.
Published accounts of H. americana infections in horses are currently limited to Texas, 5 although a report of similar hepatic lesions in horses from central and northern Florida indicates that equine infections are present there as well. 2 A more widespread distribution throughout the entire Gulf Coast and South Atlantic region of the United States, the area corresponding to the geographic range of canine schistosomiasis, is likely. The potential for infection of camelids may similarly extend throughout this region.
History
A 6-year-old female short-haired llama from Harris County, Texas, a location within the greater Houston metropolitan area, was admitted to the Texas A&M University Veterinary Medical Teaching Hospital (College Station, TX) in November 2011 for clinical signs related to heart disease. The llama had a history of chronic weight loss and dyspnea. Clinical and diagnostic findings included heart enlargement and pericardial, pleural, and peritoneal effusions indicative of congestive heart failure. The clinical impressions were suggestive of dilated cardiomyopathy and bronchopneumonia. The llama was euthanized and submitted for necropsy because of its worsening condition.
Necropsy
At necropsy, the llama weighed 95 kg. There was approximately 20 L of hazy, yellow fluid containing coagulae of fibrin in the peritoneal cavity and about 600 ml of clear, light yellow fluid in the pleural cavity. Thousands of firm, discrete, raised, 2 to 4 mm in diameter, pale yellow to white nodules, which were interpreted as granulomas, were over the pleural surface of the cranioventral lung field, affecting up to 80% of the lung surface (Fig. 1, Suppl. Fig. S1). Thin, coalescing plaques of fibrin occasionally covered some of the granulomas and pleural surfaces. On cut section, the lung parenchyma contained hundreds of granulomas in all affected lung lobes. The granulomas had the appearance and consistency of small fibrous beads, and 3 to 4 of them fell from the pleural and cut surfaces each time the lung was handled.
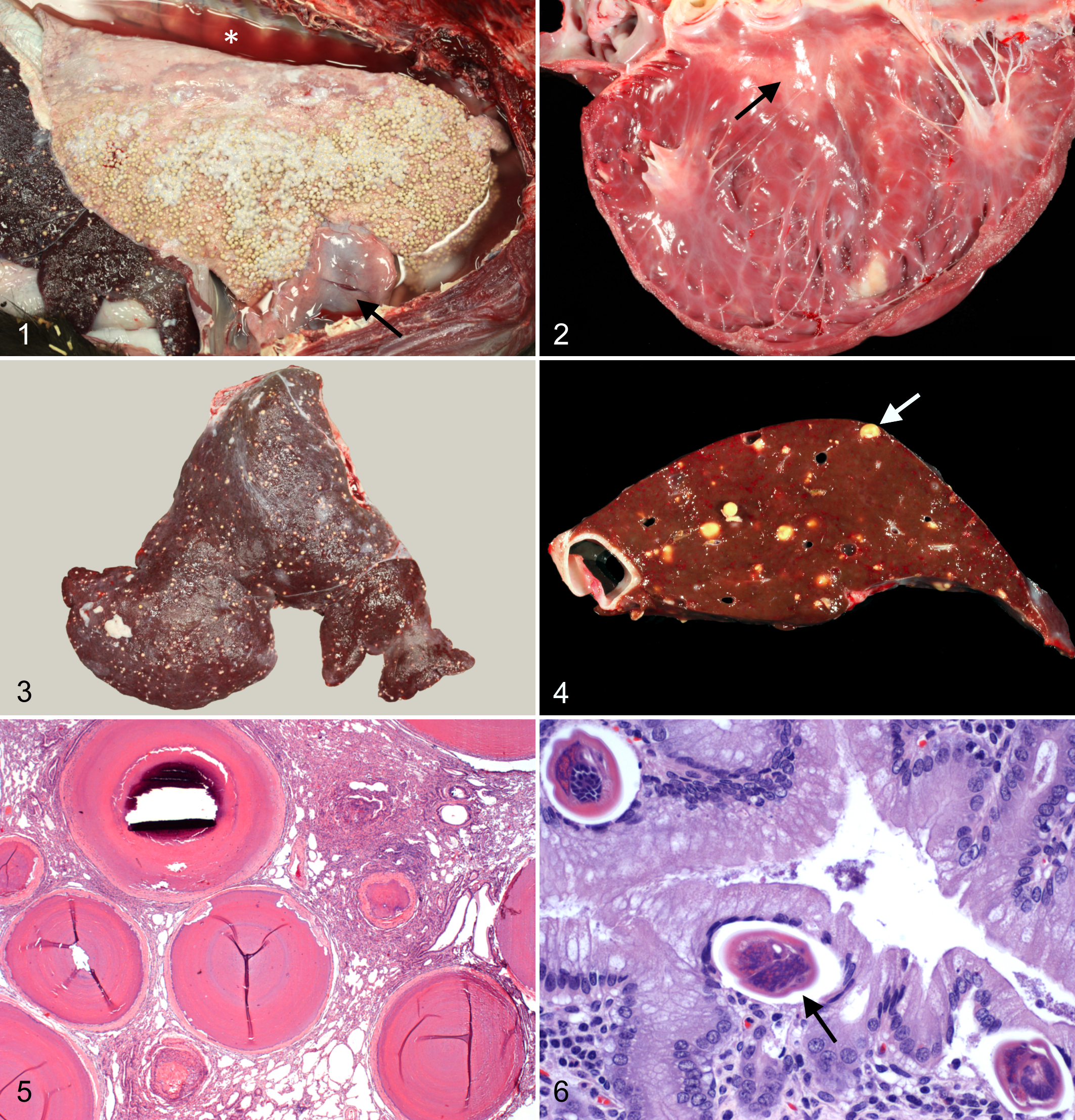
Heterobilharziasis, pleural effusion, and pericardial edema; thoracic cavity, llama. Granulomas and patches of fibrin cover the pleural surface of the lung. The pericardium is thickened and edematous (arrow), and the thoracic cavity contains abundant clear fluid (*).
The pericardial sac was thick and edematous (Fig. 1), and the right dorsocaudal aspect was adhered to the right caudal lung lobe by 2 opaque white bands of fibrous tissue. The wall of the pericardial sac in this area contained 5 granulomas similar to those in the lung. The pericardial sac contained 50 to 60 ml of clear, light yellow fluid. The heart weighed 500 g or 0.53% of body weight (reference range, 265–502 g or 0.47 ± 0.07% of body weight) 9 and was diffusely dilated, thin walled, and flaccid. Large, locally extensive areas of epicardial pallor extended over both ventricles, especially over the dorsal and lateral aspect of the left ventricle (Suppl. Fig. S2). Affected areas extended into the myocardium and reached the endocardium in multiple locations (Fig. 2). All cardiac valves were grossly normal.
The liver was firm, covered with thin strands and plaques of fibrin, and contained hundreds of hard, 3 to 4 mm in diameter granulomas randomly scattered over the capsular surface and throughout all liver lobes (Figs. 3, 4), giving it an appearance comparable to that seen with H. americana infection in horses. 4,5 Variable numbers of granulomas, similar in size and morphology to those in the liver, were found in the mesentery (Suppl. Fig. S3), jejunum (Suppl. Fig. S4), stomach, left kidney, uterus (Suppl. Fig. S5), and lymph nodes. Dissection of mesenteric, portal, and intrahepatic veins did not reveal any adult trematodes.
Histopathology
Tissue samples from all major organs were fixed in 10% buffered formalin, routinely processed for light microscopy, and stained with hematoxylin and eosin. Histologically, the heart had extensive fibrosis in multiple areas with myofiber degeneration and necrosis, sarcoplasmic vacuolization, and occasional myofiber mineralization (Suppl. Fig. S6). There was no evidence of attenuated wavy fibers or extensive fatty infiltration. Lung sections contained numerous granulomas composed of acellular, densely packed, finely mineralized collagen arranged in tight, concentric layers and surrounded by a distinct outer capsule of fibrous connective tissue (Fig. 5). The intervening alveolar septae were mildly thickened due to increased amounts of fibrous tissue. Rare, degenerate, mineralized eggs and empty eggshells were in the center of the granulomas in the lung and free in the intervening lung parenchyma. Fibrosing granulomas were also present in the liver, mesenteric lymph nodes, kidney, mesentery, endometrium and myometrium of the uterus, and small intestine.
A section of gastric mucosa from the glandular region of the proximal compartment of the stomach contained multiple schistosome eggs within the superficial lamina propria, which was mildly inflamed (Fig. 6). The eggs had an average size of 53 × 37 μm and consisted of a 2- to 5-μm-thick eggshell with an internal miracidium. The size and morphology of the eggs was similar to that seen with H. americana infection in the dog, 7,11,13 raccoon, 1,12 and horse. 4,5 Multiple granulomas with mineralized centers surrounded by a fibrous tissue capsule were in the submucosa. Schistosome eggs were not seen in organs other than the gastric mucosa and lungs. Adult worms were not identified in any sections. Acid-fast stains of sections of lung and lymph node, including those with granulomas, were negative for acid-fast bacteria.
Polymerase Chain Reaction
Polymerase chain reaction (PCR) analysis was performed on fresh, unfixed liver and lung tissue. Samples containing granulomas were initially frozen at –80°C. Granulomas were subsequently dissected from surrounding tissue and used for DNA extraction. DNA extraction and PCR amplification of a sequence of 487 base pairs were performed using a commercial kit (PurGene DNA Purification System, Gentra Systems Inc., Minneapolis, MN) and primers for the H. americana SSU ribosomal RNA (rRNA) gene, as previously described. 3 Positive and negative controls were included in all assays.
PCR amplification of nucleic acid was initially unsuccessful. Because of the extensive fibrosis of the granulomas, it was hypothesized that the lack of amplification might be due to inadequate DNA extraction using standard physical and chemical treatments. Therefore, additional granulomas were cut into thin slices with a scalpel blade and then ground into fine powder with a mortar and pestle prior to DNA extraction. Using this method, amplicons of the correct size were produced from granulomas taken from the lung but not the liver. Positive samples were sequenced and compared with the H. americana SSU rRNA gene sequence in GenBank (accession no. AY157220). Sequences derived from the amplified DNA had a 99% identity with H. americana with a few base pairs of sequence ambiguity. The final diagnosis was congestive heart failure and disseminated heterobilharziasis.
Discussion
H. americana infection is an increasing problem in Texas, with dogs and horses being the most common domestic species affected. Of the 146 horses necropsied over a 10-month period at Texas A&M University between September 8, 2011, and July 5, 2012, a total of 19 horses (13%) had hepatic parasitic granulomas at necropsy. This represents a 5% increase in infections compared with the previous 20 months, when 7.9% were infected. 5 During the same 10-month period, 2 of a total of 5 llamas necropsied had similar granulomas, including the llama in this report. Infections in llamas had never been recognized prior to this time. We believe the increased incidence and severity of infections may be related to recent drought conditions in Texas that continued through much of 2012. With decreased amounts of fresh, free-flowing water, horses and llamas may be coming in contact with pools of stagnant, snail-infested water containing high concentrations of H. americana cercariae more frequently than in the past.
In horses, hepatic fibrosing granulomas resulting from infection with H. americana have generally been regarded as an incidental finding. 2,4,5 In most cases, little evidence of overt hepatic dysfunction is found, and extrahepatic lesions are usually limited to small numbers of granulomas in the intestines or lungs. The massive numbers of granulomas in the llama in the present case are presumed to be due to a heavier than usual parasite load and a correspondingly large numbers of eggs produced. Failure to locate the adult schistosomes is not unusual, since they are only rarely found in natural infections during a routine necropsy.
The gross and histologic appearance of the hepatic granulomas in the llama were similar to those found in cases of equine heterobilharziasis. 4,5 Other causes of hepatic fibrosing and mineralizing granulomas in camelids include liver fluke infection, larval nematode migration, and resolving embolic bacterial infections. The uniform nature and disseminated pattern of the granulomas along with the absence of cirrhosis and parasitic ova were different from the hepatic lesions associated with liver flukes. 8 This also distinguished them from subcapsular nodules and fibrous tracts associated with migration of nematode larvae and irregular areas of fibrosis and inflammation secondary to resolving bacterial emboli.
Unlike the response in the horse, there was extensive granuloma formation in other organs of the llama, particularly the lungs, which were severely compromised. The resulting dyspnea initially led to an erroneous clinical suspicion of bronchopneumonia. Myocardial damage was severe and consisted of myofiber degeneration and necrosis with extensive fibrosis, presumably due to vascular compromise by the circulating eggs, leading to myocardial ischemia. While granulomas were not found in the myocardium, they were present in the pericardium, and it is conceivable that eggs may have become lodged in coronary vessels. Alternatively, pulmonary hypertension resulting from the massive infection of the lungs may have led to right-sided heart failure, although chronic cardiac overload is usually associated with right ventricular hypertrophy, which was not seen in this case.
The infection in this llama had a combination of unique features that set it apart from other cases of H. americana infection in domestic animals, which up to this point have been limited to the dog 6,7,11,13 and horse. 4,5 These include unusually large numbers of granulomas, widespread dissemination to multiple organs, extensive fibrosis of granulomas in the lungs and liver, and myocardial damage that led to congestive heart failure. While H. americana infection in horses has generally been considered an incidental finding, 4,5 in rare cases, the granulomatous response may be so vigorous and widespread that it leads to cardiac involvement and congestive heart failure. 5 The same appears to be true for llamas, which, based on the lesions in this case, may be more susceptible to pulmonary involvement and a more vigorous fibroblastic response.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
