Abstract
Thirty-seven adult female moose (Alces alces) from 2 distinct but adjacent populations in Elk Island National Park (EINP), Alberta, Canada (19 in north EINP and 18 in south EINP), were fitted with mortality-sensing VHF radio-collars, and radio signals were acquired daily to ascertain mortality status. At capture, serum, whole blood, and feces were collected; pregnancy was determined; teeth were aged by visual inspection; and a portion of liver was assessed by ultrasound examination. Postmortem examination was conducted on 20 suitable carcasses. Clinical pathological abnormalities, including eosinophilia, polycythemia, elevated levels of liver enzymes in serum, hemoglobin, hematocrit, and red blood cell distribution, and liver damage as seen in ultrasound images occurred only in moose from north EINP. Infected moose had 4.7 ± 4.8 Fascioloides magna flukes per liver (mean ± SD). The proportion of moose pregnant at capture was similar in both populations (74% in north EINP, 61% in south EINP). Proportional mortality was significantly higher in moose from the north (68%) than the south (32%). Fascioloides magna was associated as a cause of death in 7 of 14 (50%) moose in the north where cause of death was determined, while predation (n = 1), acute toxemic syndrome (n = 3), dystocia (n = 1), and roadkill and undetermined causes (n = 3) were additional causes of mortality. F. magna was associated with poor body condition and was a major cause of mortality in north EINP but not south EINP, despite very similar habitat and proximity, suggesting a significant role for these flukes in affecting health and viability of naturally infected moose populations.
Giant liver fluke (Fascioloides magna) is a digenetic trematode occurring in wild and domestic ungulates across North America and Europe. It is enzootic in 5 broad geographic areas within the United States and Canada and results in highly variable tissue damage depending on the host species. 22,30 Within North America, white-tailed deer (Odocoileus virginianus), elk (or wapiti, Cervus canadensis), and caribou (Rangifer tarandus) are considered normal definitive hosts while other ungulates such as moose (Alces alces), bison (Bison bison), feral pigs (Sus scrofa), domestic cattle (Bos taurus), and horses (Equus caballus) are considered dead-end hosts. In the latter, adult flukes migrate to the liver and occasionally produce eggs, but these do not reach the small intestine. These dead-end hosts are not epidemiologically significant in maintaining giant liver fluke populations. Other wild and domestic ungulates such as domestic sheep (Ovis aries) and wild bighorn sheep (Ovis canadensis) are considered aberrant hosts in which the flukes fail to reach maturity and are associated with significant morbidity and mortality during systemic migration. 22 Aquatic snails in the genus Lymnaea act as intermediate hosts in North America.
F. magna is associated with relatively benign infections in normal definitive host species, with rare exceptions in some individuals. 22 In contrast, the flukes often cause severe liver damage and systemic lesions in aberrant and dead-end hosts, with wide variation among individuals, making it difficult to determine whether or not F. magna significantly affects wild ungulate populations. 17,21,34
Moose populations in several areas of North America are experiencing rapid population declines, and there is continued concern among wildlife managers that F. magna may be contributing. However, reports and opinions as to the cause of the declines are controversial. 15,19 Clinical evidence of a significant effect was absent in 3 experimentally infected moose, 16 although exposure dose and duration of infection were limited. Prevalence in infected moose populations is highly variable, ranging from 20% to 89%, 17,19,23,34 and severe infection has been reported in some free-ranging individuals. 7 However, the role of flukes in declining moose populations remains unclear, and other factors such as predation, lower productivity, loss of condition, other diseases, parasites, and environmental stress also may contribute. 17,19,21
Lankester 15 correctly pointed to an “absence of published evidence linking the death of moose to giant liver fluke infection” and subsequently concluded that liver flukes do not play a significant role in moose mortality. However, evaluation of giant liver fluke infection in free-ranging moose populations as part of longitudinal studies has not been previously undertaken. Despite opportunistic surveillance, there was no evidence of F. magna in Elk Island National Park (EINP) prior to 1987, 2 and the occurrence has steadily increased in elk, moose, and plains bison since then (M. J. Pybus, personal communication, 2017). In 2009, we initiated a study of F. magna in naturally infected free-ranging moose within EINP.
The park is physically bisected into separate regions (north and south EINP), each bounded by a high-perimeter page wire fence. Annual aerial surveys previously indicated a sharply declining moose population density in the north after 2000 but not in the south (Suppl. Fig. S1). While other ungulate densities in EINP typically have similar changes in the north and south over time, this is not the case for moose where densities between the 2 areas are currently very disparate. This provided the opportunity to determine cause-specific mortality of moose and assess the potential role of F. magna in the 2 isolated populations. Our objectives were to assess differences in (1) clinical parameters at capture through hematology, serology, fecal parasitology, and ultrasound examination of moose from the 2 regions with a focus on F. magna and (2) cause(s) of death and pathologic changes associated with moose mortality in the 2 populations. Survival analysis and population demographics for the 2 moose populations will be provided in a separate publication (Rettie et al, unpublished data).
Materials and Methods
Elk Island National Park is located in central Alberta, Canada (53°30′N, 112°30′W). The park (194 km2) comprises 2 regions bounded by a 2.2-m-high wire fence. A 4-lane divided highway functionally divides the park into a Main Park Area north of the highway (134 km2, north) and an Isolation Area to the south (60 km2, south) (Fig. 1). The fencing was originally created to contain an extant elk population in the early 20th century, but eventually, the 2 regions were designed to contain and separate 2 different subspecies of bison, which are managed separately by the park. The 2 regions contain virtually identical habitat and ungulate assemblages of elk, bison, moose, white-tailed deer, and mule deer (Odocoileus hemionus), except that the bison population in the north is Bison bison bison (plains bison) and in the south is Bison bison athabascae (wood bison). Fencing effectively contains large mammal species, including elk, moose, and bison, but permits limited passage of smaller ungulates species such as deer in certain areas. Large carnivores in EINP include transient black bears (Ursus americanus) and resident coyotes (Canis latrans) with occasional cougars (Puma concolor) and wolves (Canis lupus). Opportunistic sampling from dead ungulates within EINP from 1987 to 2015 indicates the absence of F. magna in the south (n = 42: 13 elk, 24 moose, 5 white-tailed deer; all negative), whereas in the north, the parasite is well established (n = 134: 77 elk, 48 moose, 9 white-tailed deer; 49% cumulative prevalence) (M. J. Pybus, personal communication, 2015).
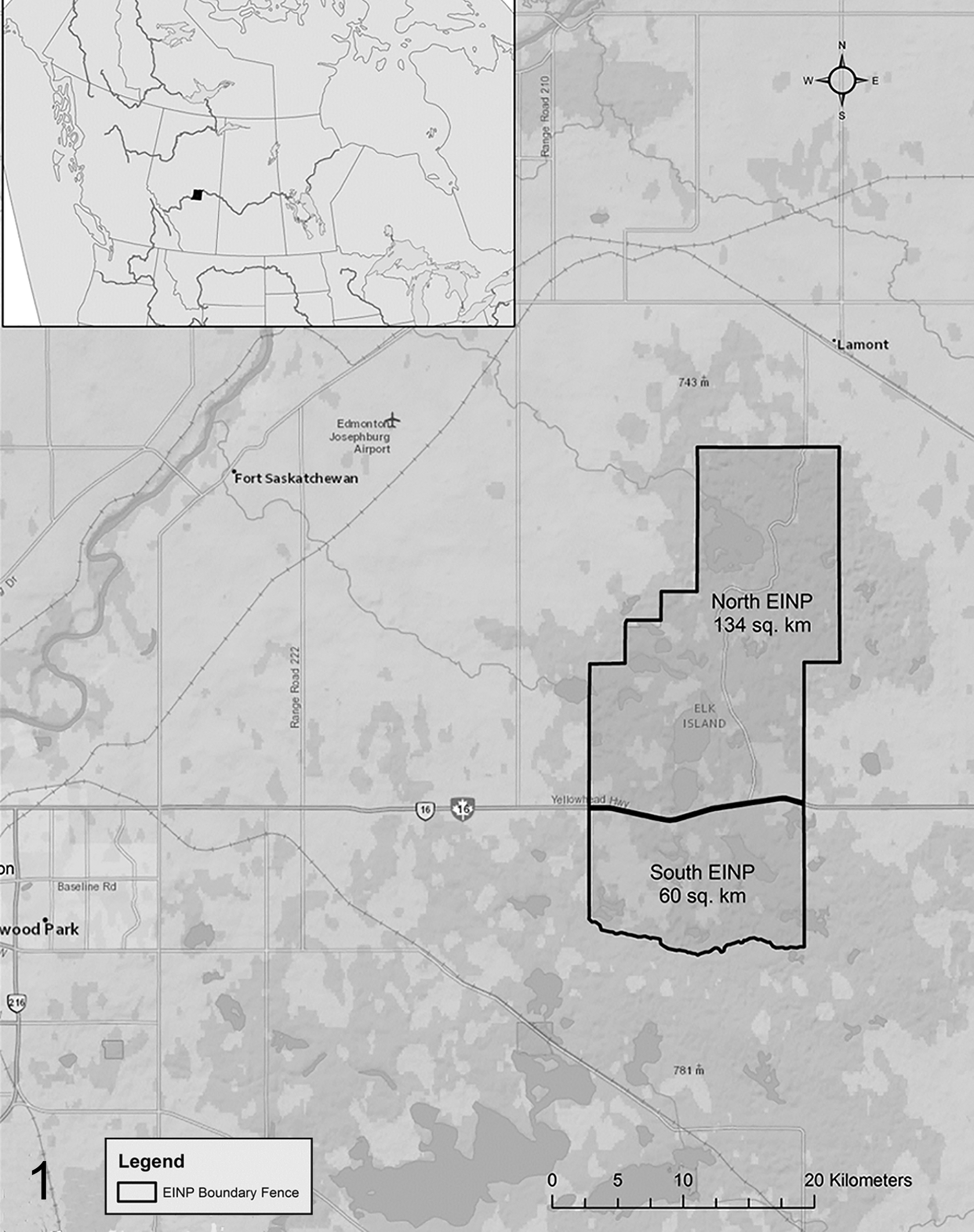
Location of Elk Island National Park, including proximity of the Main Park (north) and Isolation Area (south).
Adult female moose were captured in January 2010 and 2011 using carfentanil (3–3.3 mg per moose), etorphine (7.5–9 mg per moose), or etorphine-xylazine (9 mg etorphine and 25 mg xylazine per moose) delivered via 3-ml Pneu-Darts from a Dan-Inject Model JM Special rifle shot from a Bell 206B helicopter (Bighorn Helicopters, Cranbrook, British Columbia). Reversal was accomplished with an intramuscular injection of naltrexone (30:1 ratio for etorphine, 100:1 ratio for carfentanil). Blood was collected via jugular venipuncture into sterile tubes with and without anticoagulant (Becton-Dickinson, Mississauga, Ontario, Canada 6 ml EDTA and 10 ml Serum Separator Tube (SST), respectively). Serum was harvested following centrifugation at 3000 rpm for 10 minutes and held at 4°C. Fecal samples were collected per rectum and held at 4°C for up to 96 hours. Livers from immobilized moose were examined by transcutaneous ultrasonography using a 5- to 8-MHz curved array transducer and a portable ultrasound machine (Sonosite Titan, Bothell, WA). Hair was plucked from the skin on an area approximately 5 by 5 cm to allow better visualization of a portion of the dorsal right lobe of the liver to a depth of approximately 8 cm. As moose are a dead-end host for F. magna and do not typically shed eggs, ultrasound visualization of a portion of the liver was used to infer the presence or absence of hepatic lesions at time of capture. This was a novel technique not previously used for detection of F. magna in wild or domestic species but was based on clinical ultrasonographic techniques developed for domestic animals. 3,27 The age was determined by visual inspection of incisors using eruption and wear of lower incisiform teeth into broad age classes (calf, yearling, adult). 20 Body condition of moose was assessed by palpation of subcutaneous fat deposits and muscle definition using a 6-point scale (1 to 6, emaciated, poor, fair, moderate, very good, excellent) based on Franzmann. 12
VHF radio-collars (Lotek, Newmarket, Ontario, Canada) with mortality sensors were attached and mortality status was monitored daily from fixed stations. When a mortality signal was detected, the carcass was located and, when possible, transported to the Alberta Agriculture Diagnostic Laboratory in Edmonton for postmortem examination by a board-certified veterinary pathologist (N.N.). When carcasses were not accessible or were partially or completely scavenged, Parks Canada staff conducted field necropsies. A first incisor was collected for cementum annuli analysis by Matson’s Laboratory (Milltown, MT). Carcasses of uncollared moose found opportunistically by park staff were also collected. All capture procedures and study design were approved by the Parks Canada Animal Care Committee (permit 3800).
Livers were either frozen or examined fresh. All livers were examined in detail as described. 23 In general, livers were sliced thinly (5 mm) and intact flukes were gently extracted and rinsed in tap water. Partial and complete flukes were staged as immature or gravid (eggs present). Severity of infection was categorized on a scale of 0 to 4: no evidence of flukes (0), ∼10% damaged liver (1), ∼25% (2), ∼50% (3), and >75% (4). Selected lesions from livers and other organs were examined using standard histological methods. Samples for microbiology were forwarded to IDEXX Laboratories in Edmonton, Alberta, at the discretion of the pathologist and were only submitted for 3 moose that died with similar lesions of subcutaneous hemorrhage and edema (described later). Lesions associated with giant fluke infection were considered incidental if <25% of the liver was affected or other causes of death were apparent. 18
Sera, whole blood, and feces were evaluated for health assessment. Sera and whole blood from captured moose were submitted to Prairie Diagnostic Services in Saskatoon, Saskatchewan, for a complete blood count and serum chemistry analyses. Pregnancy status was assessed through serum progesterone determination using an in-house enzyme immunoassay from the Toronto Zoo Reproductive Physiology Laboratory. 31 Moose were considered pregnant if serum progesterone was ≥3 ng/ml. 26 Fecal pellets were assessed by a modified Wisconsin flotation technique for occurrence of gastrointestinal parasite eggs. 6
Hematological, serum chemistry, and parasitological (intensity) parameters were compared between north and south and whether lesions were present on ultrasound using a Mann-Whitney U test. Reference intervals for serum biochemical and hematologic parameters were compared to data from free-ranging moose captured in Norway using similar drugs (etorphine and ketamine/medetomidine) but darted from the ground. 1 Preliminary association between ultrasound lesions at capture and severity of lesions at necropsy was explored using a scattergram (Suppl. Fig. S2) and Spearman’s rank correlation. Differences in parasite prevalence between north and south were tested using the prtest command in STATA. Significance level for all tests was P < .05. All statistical tests were completed using STATA 13.1 (StataCorp, College Station, TX).
Results
A total of 37 adult (7.3 ± 3 years old, mean ± SD) female moose (19 north, 18 south) were captured and radio-collared in the park. One calf was captured (∼9 months of age) but excluded from further analysis. Overall mean age at time of death was 10 ± 4.3 years, and no difference was observed in age at death between moose examined in the north and south (t = 1.1046, P = .306). Fifteen of 19 (79%) moose captured in the north and 6 of 18 (33%) in the south were found dead between September 2010 and April 2015 (Table 1). In addition, 1 uncollared adult female and 1 adult male moose found dead in the north in 2013 and 2015, respectively, were available for necropsy.
Causes of Mortality and Flukes Found in 23 Adult Female Moose Found Dead in Elk Island National Park, 2010 to 2015.
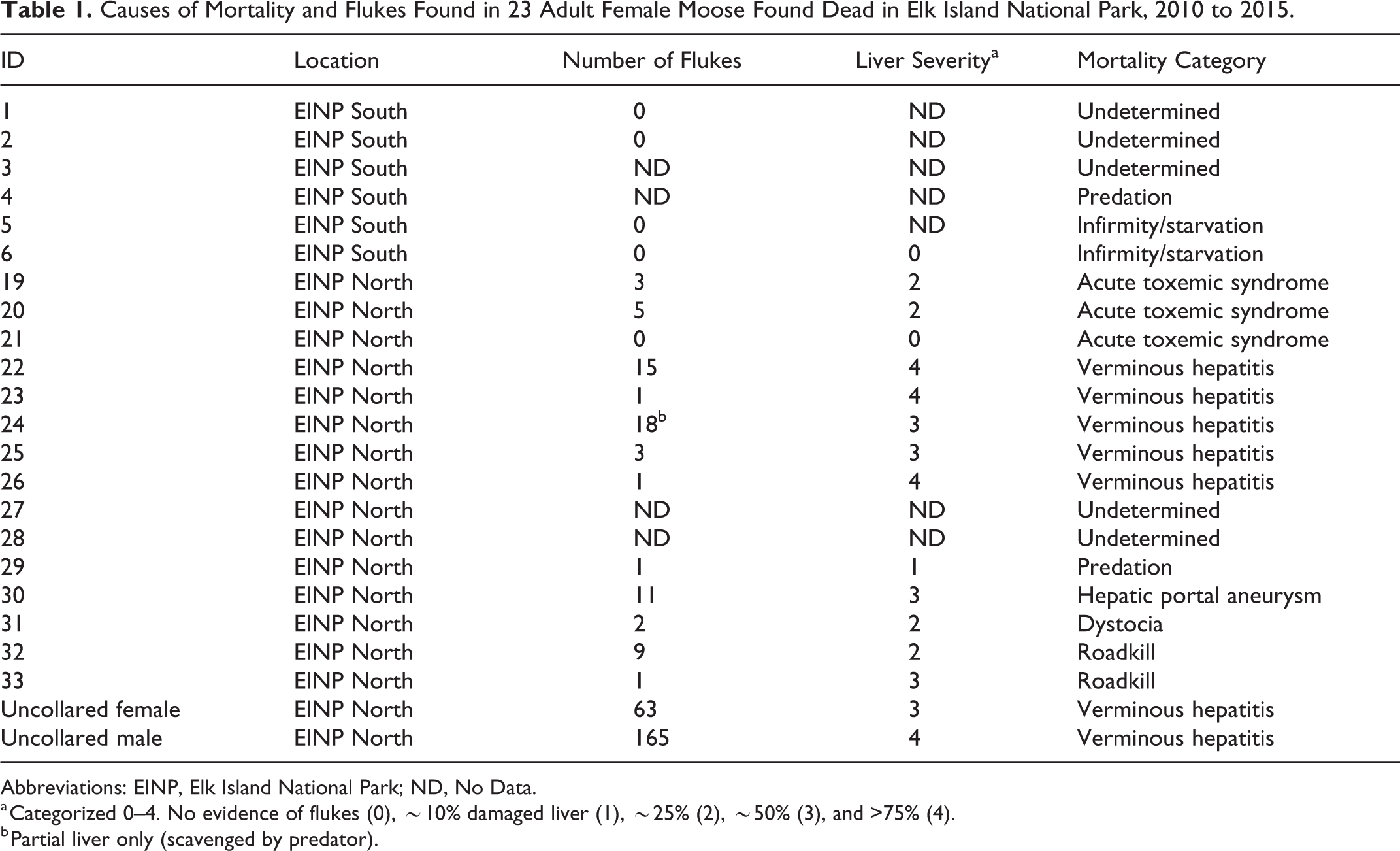
Abbreviations: EINP, Elk Island National Park; ND, No Data.
a Categorized 0–4. No evidence of flukes (0), ∼10% damaged liver (1), ∼25% (2), ∼50% (3), and >75% (4).
b Partial liver only (scavenged by predator).
Clinical Pathology Findings at Capture
Ultrasound lesions were observed in 8 of 19 (42%) moose from the north and 1 of 18 (6%) from the south, and this difference was statistically significant (χ2 = 4.97, P = .026). Lesions included both linear and circular hyperechoic regions within the liver parenchyma. Mean body condition did not vary significantly between north and south EINP (P = .357), but moose with ultrasound lesions had significantly lower mean body condition score (4.5, n = 9) than moose without liver lesions (5.5) (n = 18, P = .014).
Six of 9 moose with liver lesions detected at capture later died; all were from the north, and all 6 had F. magna infection with associated liver damage. Fourteen of 24 moose without apparent liver lesions at capture later died, 4 of which were infected with liver flukes, all of which were from the north. There was no apparent relationship between hepatic lesions observed by ultrasound at capture and severity of lesions observed at necropsy (Spearman’s ρ = 0.3050, P = .289; Suppl. Fig. S2), although the time between capture and necropsy differed tremendously (range, 77–1524 days).
Some hematology and serum biochemistry values differed between moose from the north and south and for moose with and without liver lesions. Liver-specific enzymes (γ-glutamyl transferase [GGT], glutamate dehydrogenase [GLDH] and, sorbitol dehydrogenase [SDH]) were not significantly different between moose from the north and south, but mean levels in moose from the north were above reference intervals for moose 1 (Table 2). Moose captured in the north had eosinophilia, slight polycythemia, hypochloremia, and larger red blood cell distribution width compared to moose captured in the south, with values exceeding the reference range for moose captured under similar conditions (Table 3). Serum urea values were also significantly higher in moose from the north compared to the south but were below reference intervals. Fibrinogen was significantly higher in moose captured in the north than those in the south, but mean values were within the established reference range for domestic cattle 13 (Table 2).
Serum Chemistry Values of Adult Female Moose Captured in Elk Island National Park by Location and Liver Ultrasound Result.
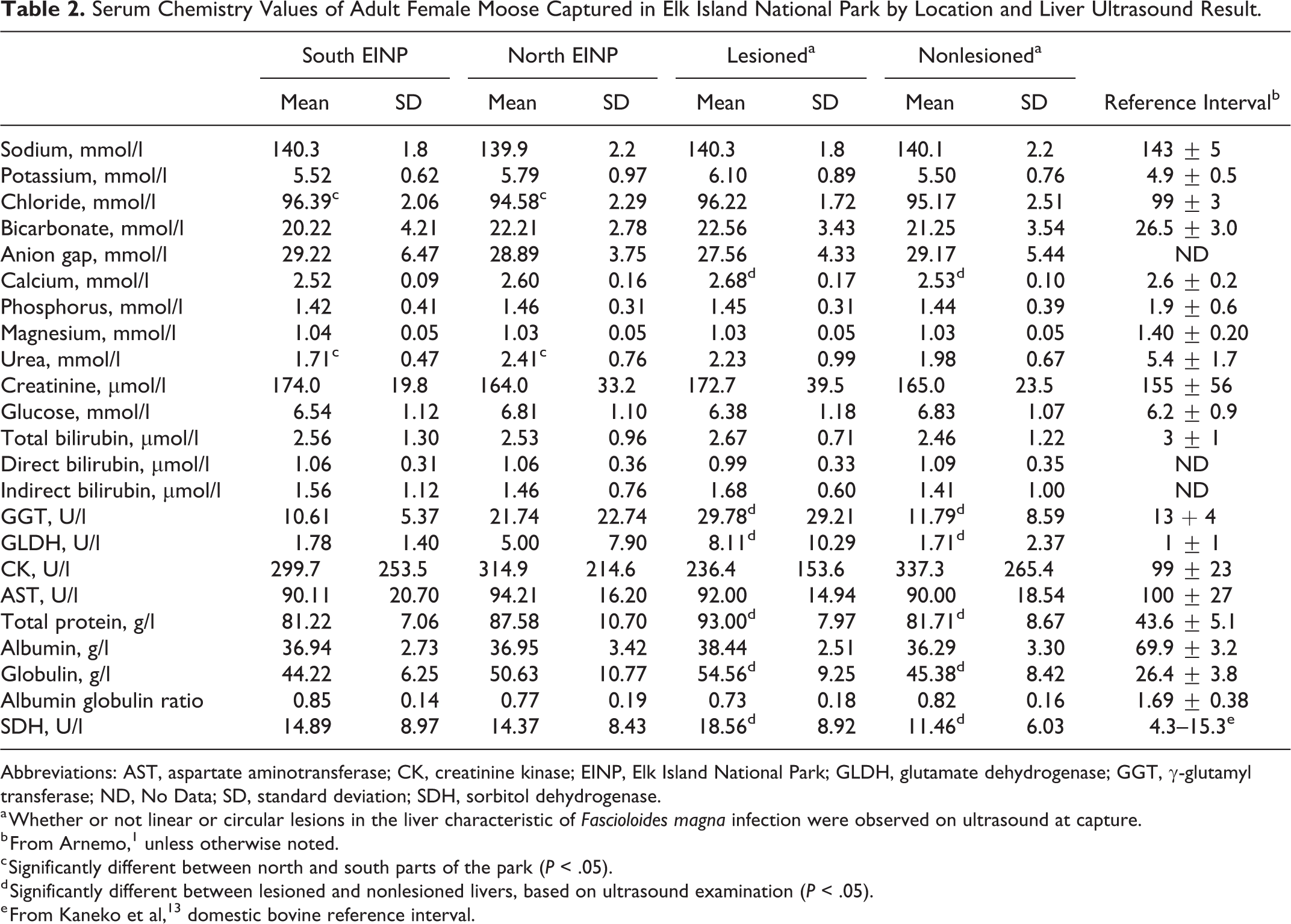
Abbreviations: AST, aspartate aminotransferase; CK, creatinine kinase; EINP, Elk Island National Park; GLDH, glutamate dehydrogenase; GGT, γ-glutamyl transferase; ND, No Data; SD, standard deviation; SDH, sorbitol dehydrogenase.
a Whether or not linear or circular lesions in the liver characteristic of Fascioloides magna infection were observed on ultrasound at capture.
b From Arnemo, 1 unless otherwise noted.
c Significantly different between north and south parts of the park (P < .05).
d Significantly different between lesioned and nonlesioned livers, based on ultrasound examination (P < .05).
e From Kaneko et al, 13 domestic bovine reference interval.
Hematological Values of Adult Female Moose Captured in Elk Island National Park by Location and Liver Ultrasound Result.
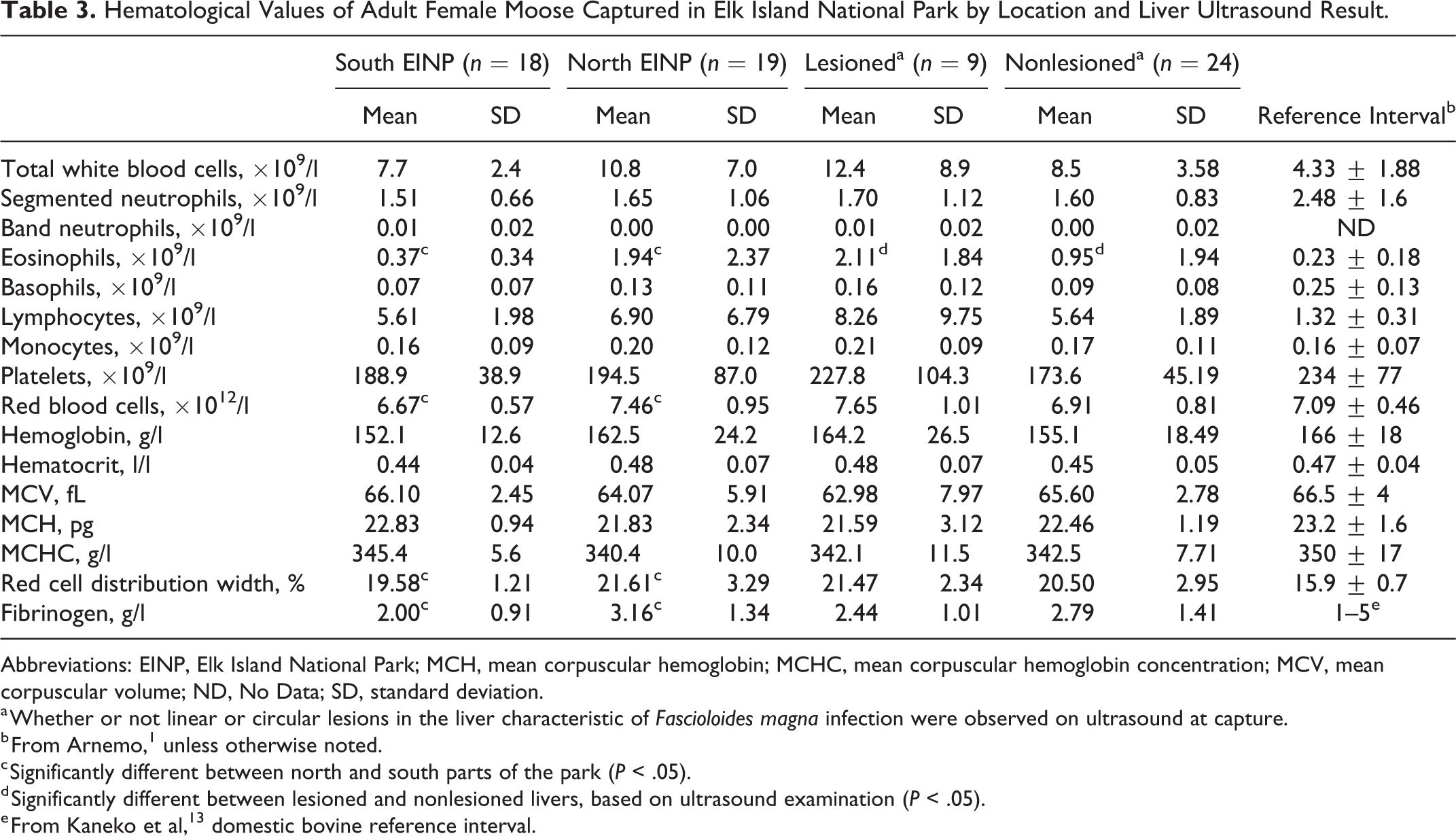
Abbreviations: EINP, Elk Island National Park; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; ND, No Data; SD, standard deviation.
a Whether or not linear or circular lesions in the liver characteristic of Fascioloides magna infection were observed on ultrasound at capture.
b From Arnemo, 1 unless otherwise noted.
c Significantly different between north and south parts of the park (P < .05).
d Significantly different between lesioned and nonlesioned livers, based on ultrasound examination (P < .05).
e From Kaneko et al, 13 domestic bovine reference interval.
In contrast, values of SDH, GGT, and GLDH; serum globulins; total protein; calcium; and eosinophils were significantly higher in moose with liver lesions on ultrasound compared to those without (Tables 2 and 3), and all were above reference ranges except for calcium. Although serum albumin level was not associated with capture location or liver lesions on ultrasound, levels in all moose were significantly elevated compared to reference intervals.
There was no significant difference (χ2 = 0.669, P = .413) in the proportion of moose pregnant at time of capture in the north (14 of 19; 73.7%) vs the south (11 of 18; 61.1%) or in moose with liver ultrasound lesions (7 of 9; 77.8%) vs those without lesions (16 of 24; 66.7%, χ2 = 0.383, P = .536). There was no significant difference in intensity of gastrointestinal parasites from the north and south, but prevalence of Capillaria spp was significantly higher (P = .023) in the south (Table 4).
Quantitative Fecal Flotation Results From Adult Female Moose Captured in Elk Island National Park.
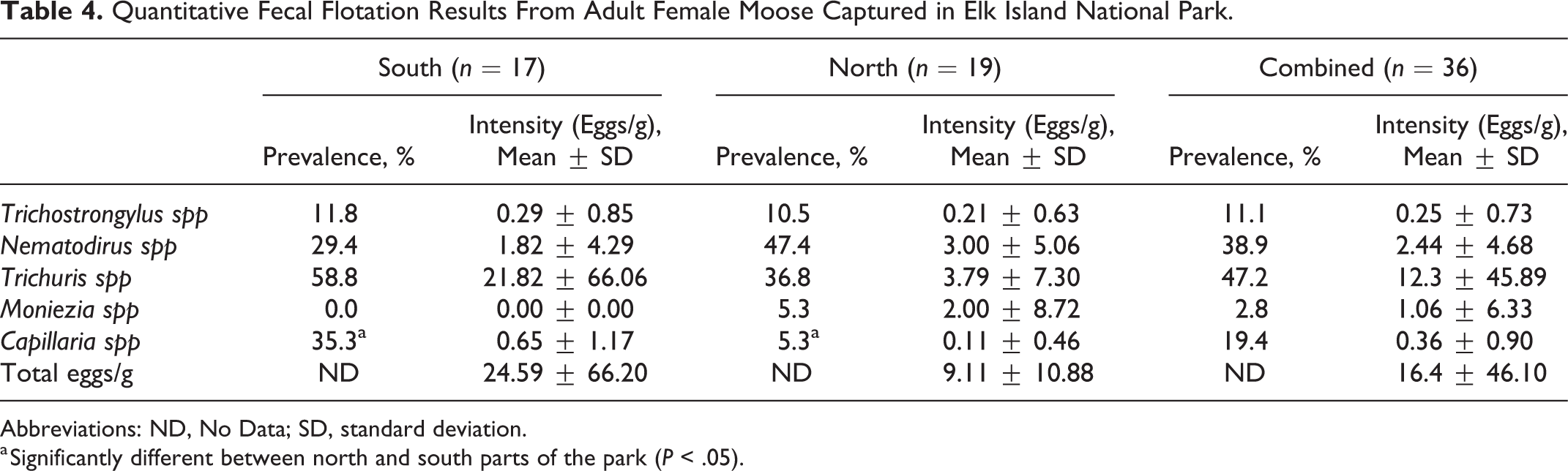
Abbreviations: ND, No Data; SD, standard deviation.
a Significantly different between north and south parts of the park (P < .05).
Gross Pathology
Fourteen of 20 (70%) moose necropsied were infected with giant liver fluke (14 of 15; 95% north, 0 of 5 south) at time of death. Of the 20 animals necropsied, 6 did not have lesions or evidence of giant liver fluke, and 5 of these were from the south.
Cause of death was determined for 17 of the 23 moose (Table 1; 3 were not submitted due to extensive scavenging and lack of tissues). Causes of death included severe verminous hepatitis, predation, roadkill, infirmity/starvation, dystocia, and hepatic portal aneurysm. Giant liver fluke was directly associated with mortality in 7 of 14 moose from the north for which cause of death was determined (Table 1, 6 collared moose and 1 uncollared). Six of these moose had severe verminous hepatitis with systemic complications, and 1 died as a result of hepatic portal aneurysm. In the south, none of the 6 mortalities were attributed to liver fluke. Two of these moose died of cachexia: 1 was aged and had poor and missing teeth, generalized lymphadenitis, and concurrent infestation with Dermacentor albipictus (winter tick), and 1 died in conjunction with severe winter tick infestation and had poor body condition, major loss of hair over much of the body, and an abundance of ticks. One moose was predated, and 3 were scavenged and cause of death was undetermined.
Livers affected by liver flukes were notably enlarged and lesions generally affected >75% of the tissue. The wet weight of infected livers (6652 ± 2198 g, mean ± SD, n = 12) was higher than that of uninfected livers (3391 ± 99 g, mean SD, n = 2). Black pigmentation, considered pathognomonic for giant liver fluke infection, was identified in all affected livers, and occasionally in hepatic lymph nodes, adjacent abdominal tissues, and the pleural cavity. Lesions generally were similar to those reported previously. 14,22 The primary lesions were multiple firm, large, often confluent, fibrous abscess-like structures (Fig. 2) ranging in size from 5 to 15 cm in diameter (Fig. 3). Large organized thrombi of fibrin and degenerate inflammatory cells encapsulated by fibrous tissue were present in large hepatic blood vessels, including the vena cava and portal veins. Moose with chronic liver lesions (n = 6) also had diffuse fibrosis of the pleura and epicardium, as well as of the hepatic capsule and abdominal surface of the diaphragm. Portions of some lungs were firmly adherent to the pleural wall, pericardium, and/or diaphragm. Local areas of thickened alveolar septa containing extracellular protein deposits, likely as a result of inflammation, were present in many infected moose.
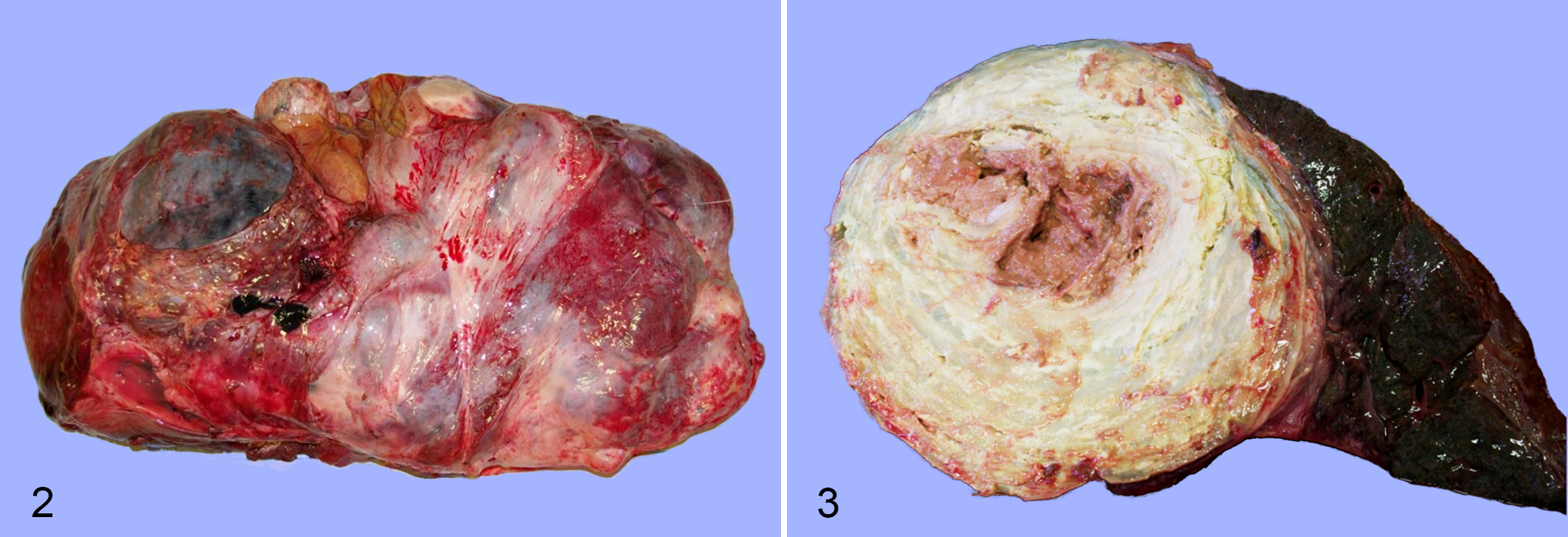
Severe chronic Fascioloides magna infection, liver, moose. Figure 2. The liver is enlarged and there are multiple large, nearly confluent masses, fibrous capsular adhesions, and minimal normal parenchyma. Figure 3. On cut section, the liver mass has a central core of brown caseous necrosis and yellow lamellated concentric rings of connective tissue. The adjacent parenchyma is atrophied.
Viable or discernable remnants of F. magna were found in the liver within fresh tracts or fibrous structures. In 11 livers that could be assessed for fluke numbers (1 liver was partially scavenged), the mean intensity was 4.7 ± 4.8 (SD) flukes (range, 1–18). Two moose had higher numbers of flukes: 63 and 165 (Table 1). Gravid adult flukes were found in these 2 moose (Figs. 4, 5), whereas flukes in the other moose were immature. Live and dead flukes often occurred within the same liver.
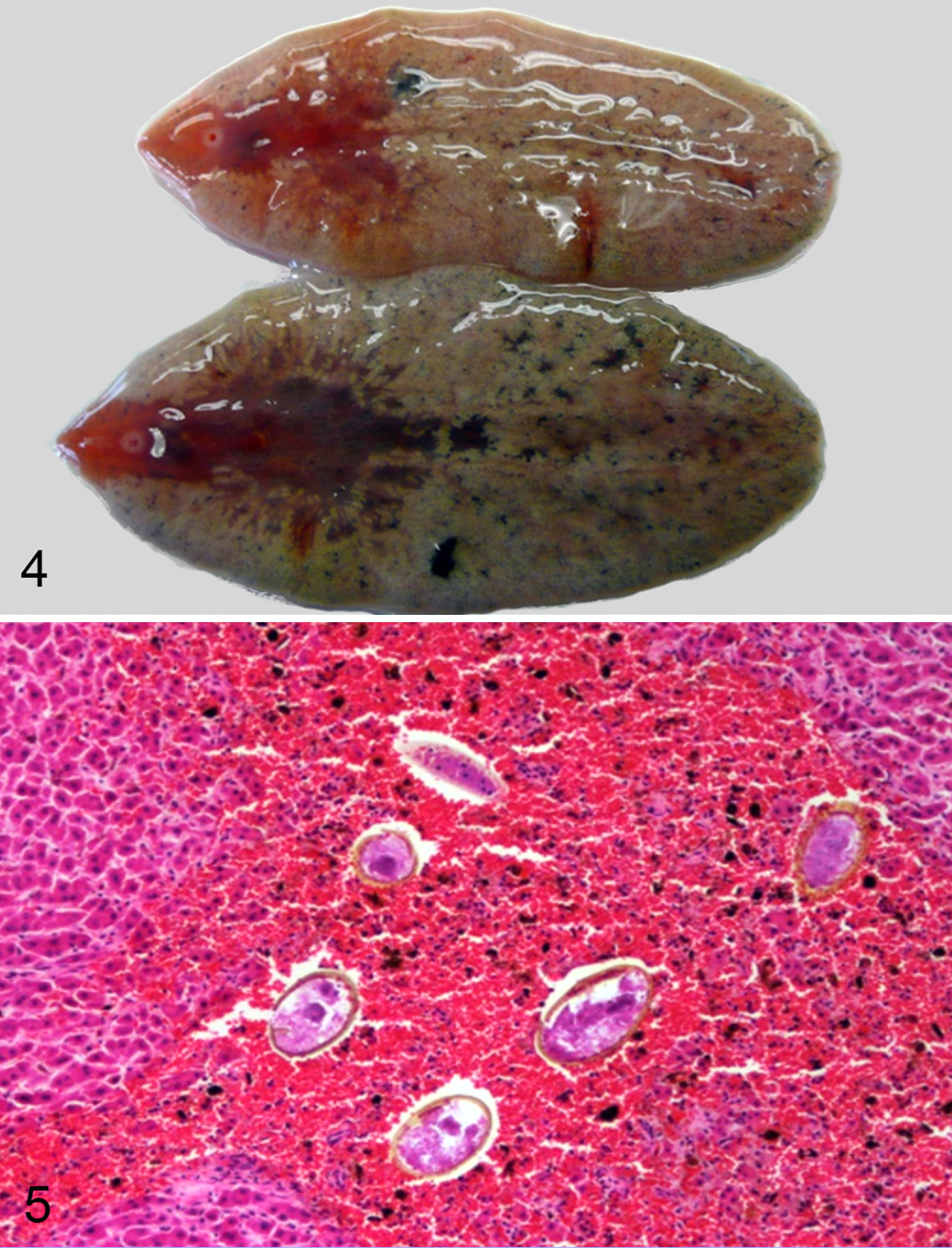
Gravid adult Fascioloides magna trematodes.
Three collared moose from the north had a very different, yet consistent pathological presentation. All were in good body condition and appeared to die suddenly within a 3-month period (early November 2011 to early February 2012). There was general bruising and edema in all subcutaneous areas of the body, including connective tissues, fat, and superficial musculature, and protected areas such as the ventral abdomen and inner thighs. Selected tissues were examined microscopically but were too autolysed for meaningful evaluation. Affected subcutaneous tissues were cultured aerobically from 2 moose and anaerobically from 1. Cultures from 1 moose yielded Streptococcus bovis, Pseudomonas fluorescens, and Escherichia coli. Cultures were negative for Pasteurella spp and Clostridium spp. Two of the 3 moose had generalized chronic hepatic damage in approximately 25% of the liver, including liquefactive areas and/or tracts in the hepatic parenchyma, organized thrombi, and vascular occlusion in association with 3 and 5 immature F. magna flukes, respectively. The third moose had no discernable liver lesions.
In addition, 1 uncollared adult male moose was found opportunistically in distress in the north in April 2015; it was euthanized and a necropsy was conducted. The liver was grossly enlarged (wet weight 8.75 kg) with characteristics of severe verminous hepatitis associated with giant liver fluke infection. The liver contained 21 immature flukes, 120 nongravid adults, and 24 gravid adults. Blood flow within the liver appeared restricted by a major organized thrombus attached to the intimal surface of the hepatic vein near the hilus and by smaller thrombi in the distal branches of the hepatic vein.
Discussion
This article describes a mortality study of radiocollared moose in 2 adjacent but separate populations in EINP. Clinical observations in live moose and postmortem examination of 20 moose revealed that F. magna infection caused morbidity and mortality in the north EINP moose population but not in the south population. This suggests that F. magna has an important role in the viability of naturally infected moose populations.
Clinical findings at the commencement of this study indicate the presence of lesions and both a hepatic and a systemic response to the presence of flukes in the liver. This is demonstrated by ultrasound results, total serum protein levels, increased liver-specific enzyme activity in serum, and hematologic changes all consistent with chronic hepatic inflammation. In moose with ultrasound lesions, serum levels of total protein, globulin, GGT, and GLDH were significantly elevated above reference values, consistent with both parenchymal and biliary damage. GGT levels are widely used as an index of biliary injury and can be an independent predictor of increased risk of death in humans. 32 Our clinical observations in these moose are consistent with F. magna disease in other species: eosinophilia has been reported in experimentally infected calves, 4 and elevations in total protein and serum globulins have been reported in experimentally infected white-tailed deer. 11 Eosinophilia is associated with migrating immature F. magna in naturally infected red deer, Cervus elaphus, in a population characterized by moderately poor body condition. 28 High GGT values may also play a role in appetite suppression in sheep infected with the closely related Fasciola hepatica 8 and are common in chronic debilitating fasciolosis. 24
In this study, hepatic lesions associated with F. magna infection cover a spectrum from acute damage associated with migrating flukes to chronic damage associated with established or deteriorating stationary flukes. Severity of liver damage generally increases with chronicity and is accompanied by a progressive reduction in the amount of normal hepatic tissue. Immature migrating flukes occur even in moose with long-term damage, indicating ongoing and suggesting cumulative infection over time. Hepatic enlargement as reflected in the increased liver weight of infected individuals may be an attempt to maintain functional liver tissue by hyperplasia but is more likely a reflection of the large mass of fibrotic tissue and accumulated inflammatory and necrotic material. The added mass and functional inefficiency of such livers may contribute to the deteriorating body condition observed in chronically infected moose.
In addition to hepatic lesions, most fluke-infected moose had significant lesions in other tissues. Chronic serosal inflammation, large fibrous masses similar to those in the liver, and extensive fibrous adhesions between serosal surfaces within the thorax and abdomen and especially between the diaphragm, liver, and lungs were common and might have compromised pulmonary and gastrointestinal movement. Organized thrombi were occasionally found in the caudal vena cava and larger hepatic veins, suggesting that circulation through hepatic tissues might be compromised and predispose infected moose to aneurysm and restricted vena caval blood flow. Such aneurysm and sudden death have been seen in fluke-infected black-tailed deer (Odocoileus hemionus columbianus), 5 elk, 23 and cattle. 33
The anatomic hepatic lesions described herein are consistent with those reported by previous authors, but the direct link to mortality has not previously been documented. Lesions associated with verminous hepatitis in the current study appeared more common, more severe, and more generalized than those described in other studies. 34 The lack of established populations of large carnivores within our study area may have contributed to longer life span of individual moose and opportunity to develop more extensive liver lesions. In addition, relatively high densities of a normal host for F. magna (elk) within this closed ecosystem (Suppl. Fig. S1) in conjunction with abundant high-quality semiaquatic snail habitat (intermediate host) may also have contributed to the severe lesions observed.
In this study, there was a clear difference in the mortality, clinical observations, and postmortem findings between the north EINP moose population and the south EINP population. While adjacent, these 2 populations are not in contact, being separated by a major highway and fencing. Mortality among moose from north EINP of 68% was significantly higher than south EINP (32%). At the time of radio-collaring (commencement of the study), there was a significant difference in the ultrasound findings between north and south populations. The measured serum enzymes and hematologic values exceeded reference values for moose in north EINP at commencement of the study but were not statistically different from south EINP. However, these parameters were statistically elevated for moose with ultrasound lesions when compared to those without. The clinical pathology results support the suggestion that from the commencement of the study, moose in the north EINP population were infected with F. magna while those in the south EINP population were not. Postmortem examination demonstrated that F magna was associated with death in fully half the moose in the north where cause of death was determined, while it was not found in any of the moose examined from south EINP. Therefore, the various clinical parameters obtained and the gross pathology all support the assertion that F. magna is an important cause of morbidity and mortality in affected moose populations. Mortality in free-ranging moose with chronic systemic lesions is similar to prior reports. However, in other F. magna enzootic areas in North America, the exact role of F. magna in such mortality has been uncertain. 7,14,19,23 Because of a lack of other coincidental mortality factors, this study more clearly demonstrates a direct connection of F. magna with moose mortality and population decline.
The severity and chronicity of F. magna lesions in moose are indications of a poorly adapted host-parasite relationship. 22 While gravid flukes and ova were found in 2 moose in this study, these are the exception. Most flukes recovered from moose livers were immature and associated with fresh migratory tracts in the liver and occasionally in the thoracic and abdominal cavities. Contact between immature flukes leads to maturity, mating, and cessation of migration of F. magna in white-tailed deer and, to a lesser extent, in elk and caribou. 10 Immature flukes in moose were rarely found together, suggesting that migration within the parenchyma is continuous and ongoing, providing opportunity for secondary bacterial infection and/or intrahepatic vascular thrombosis and aneurysm.
Considering the magnitude of the differential mortality between north and south EINP in the radio-collared animals, it is probable that the declining moose population in the north is associated with the negative effects of giant liver fluke in a naive population. There was no evidence of significant alternate disease or parasite infection affecting the health of individual moose in either population in EINP, which have been confounding factors in other studies. 17,19,34 Meningeal worm (Parelaphostrongylus tenuis) may be a confounding factor in mortality studies of some moose populations, but this parasite does not occur in Alberta. 15,19,34 Similarly, severe weight loss and alopecia associated with winter tick (D. albipictus) infections 15,34 were apparent only in 1 moose from south EINP (approximately 70% hair loss). Winter ticks occur throughout EINP but during our study were present on only a few moose and in light infestations, except the moose mentioned above, as assessed using previously described methods. 25 Chronic wasting disease (CWD) had not been detected in EINP or within 100 km of the park in susceptible wild ungulates at the time of this study. Despite these findings, the number of moose examined in this study represents a fairly small sample size with low numbers of mortalities, somewhat restricting external inference to other moose populations.
The 3 moose that died between November 2011 and February 2012 in the north were of interest. All 3 had a similar and distinct pattern of diffuse subcutaneous bruising and edema and were in good body condition. The lesions were suggestive of an acute systemic toxemia similar to some clostridial diseases, 29 but Clostridia and Pasteurella spp were not detected. The role of F. magna in these cases is unknown. Extensive damage to tissues and disruption of normal hepatic architecture provides opportunity for breach of vascular tissues leading to subsequent bacterial septicemia, as may have occurred in 2 moose in this study. A generalized correlation between liver cirrhosis and sepsis is reported in the medical literature. 9
This study provided an opportunity to employ ultrasound examination of the liver of moose in a field situation. Although not designed to fully evaluate field application of ultrasound, the technique did detect fluke-associated liver damage, which was associated with elevated serum hepatic enzymes, elevated serum globulins, and concurrent eosinophilia. We found that approximately 50% of nonlesioned moose at capture (based on ultrasound) had lesions at necropsy. There are a number of possible explanations for this variance. In our study, postmortem examination occurred only after natural mortality of individual moose. The depth of ultrasonography was limited and it was not possible to visualize the entire liver, so only the upper third of the liver closest to the right body wall was examined. Restricted depth of penetration of ultrasound in the abdomen of moose would result in deep lesions being missed. There was a significant time lag (up to 4 years) between ultrasound at capture and time of death, during which time small lesions could enlarge and uninfected moose could become infected and develop chronic illness. Despite such limitations, field use of hepatic ultrasonography appears to warrant further evaluation for antemortem diagnosis of F. magna in dead-end hosts, particularly in conjunction with clinical chemistry and hematology assessments.
Giant liver fluke appeared in north EINP sometime prior to 1987 but was not detected during elk slaughters conducted in the 1960s and 1970s. Although the 2 fenced areas and a major highway effectively partition the large ungulate assemblages between north and south EINP, it is likely that white-tailed deer, a suitable definitive host for F. magna, occasionally move between these fenced areas. Why giant liver fluke only occurs in the north is unknown, considering that both areas are ecologically identical and 30 years have passed with no evidence of spread to the south. It seems likely that the flukes will eventually “cross the road” and infect deer, elk, and moose populations in south EINP. Periodic monitoring would help determine when this happens and if similar population impacts occur in moose populations in south EINP.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818823776 - Fascioloides magna in Moose (Alces alces) From Elk Island National Park, Alberta
Supplemental Material, DS1_VET_10.1177_0300985818823776 for Fascioloides magna in Moose (Alces alces) From Elk Island National Park, Alberta by Todd K. Shury, Margo J. Pybus, Nick Nation, Normand L. Cool and W. James Rettie in Veterinary Pathology
Footnotes
Acknowledgements
We readily acknowledge park staff at EINP for support in various aspects of this project, particularly Clayton Szafron, Archie Handel, and Brett Sarchuk for retrieving moose for necropsy and all other aspects of fieldwork. Christine Wilson, Geoff Skinner, and Bighorn Helicopters assisted with moose captures. Brent Wagner conducted the fecal floatations and Gabriella Mastromonaco assisted with the reproductive hormone assays. Dr. Robert McCorkell from the University of Calgary kindly performed all ultrasound examinations during field capture. We also recognize Alberta Agriculture for use of the Edmonton diagnostic facility and technical assistance during postmortems.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This study was funded by Parks Canada Agency.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
