Abstract
Canine schistosomiasis due to Heterobilharzia americana is a clinically underdiagnosed disease in dogs, which is found primarily in the Gulf Coast and south Atlantic region of the United States. A 3-year-old dog from Texas with a clinical diagnosis of systemic mineralization of unknown origin in the absence of evidence of hypercalcemia was found at necropsy to have severe disseminated H. americana infection involving the liver, pancreas, small and large intestine, lungs, and kidneys. Calcification of many of the large number of H. americana eggs gave the false impression of soft-tissue mineralization on radiographic and ultrasonographic images. Polymerase chain reaction amplification and sequencing of DNA derived from formalin-fixed sections of small intestine and liver, using primers specific for a 487–base pair segment of the H. americana small subunit ribosomal RNA gene, confirmed the presence of H. americana.
Heterobilharzia americana, the etiologic agent of canine schistosomiasis, is a uniquely North American trematode in the family Schistosomatidae. 10 Its intermediate host is a lymnaeid freshwater snail (Bakerilymnaea cubensis or Pseudosuccinea columella), and its natural definitive host is the raccoon. The life cycle has been well documented, 9 and infections in dogs occur when they come in contact with the free-swimming cercariae, the multicellular larval stage of the schistosome, which swim near the surface of snail-infested fresh water following their release from infected snails. The fork-tailed cercariae attach to the skin of the dog and penetrate intact epidermis. Studies of Schistosoma japonicum, one of the major human schistosomes and phylogenetically related to H. americana, have shown that cercariae attach to the skin with their ventral suckers and release proteolytic enzymes that facilitate entry into the skin and penetration of the basement membrane and blood vessel wall. 7 Heterobilharzia americana is believed to use a similar mechanism. Losing their tail, the immature worms, known as schistosomula, migrate via the blood to the lungs and then the liver, where they mature into adults. The adult worms live in mesenteric and intrahepatic portal veins where the male and female worms live in copula. Eggs, with internal ciliated larval miracidia, are deposited by the female in mesenteric venules. Proteolytic enzymes secreted through pores in the eggshell allow the eggs to escape from the capillary lumen and migrate to the intestinal tract, while aberrant migration leads to infection of other tissues. Granulomatous and eosinophilic inflammation occurs in response to the presence of the eggs lodged in tissues. Eggs that manage to migrate through the intestinal wall enter the intestinal lumen and are released in the feces. Miracidia are rapidly released from the eggs following contact with fresh water. The miracidia actively seek out and infect the appropriate snail intermediate host by penetrating its soft mantle. Larval stages multiply in the snail with the eventual release of the cercariae in fresh water. Raccoons are similarly infected by skin penetration of the cercariae and develop lesions comparable to those in dogs, although the degree of periportal fibrosis in the liver can be more severe. 1 Other mammalian species known to be naturally infected include the bobcat, armadillo, Brazilian tapir, beaver, coyote, mountain lion, mink, nutria, opossum, red wolf, swamp rabbit, and white-tailed deer. 12 Suspected and confirmed cases of natural infections in horses have also been reported. 2,3
While H. americana is found primarily in the Gulf Coast and south Atlantic states from Texas to North Carolina, the majority of the reported infections in dogs have been from Texas and Louisiana. An expanding incidence in certain geographic regions of Texas has been noted (Snowden K, White S, Lewis, B, Craig T: 2009, Distribution and characterization of Heterobilharzia americana infections in the dog in Texas. In: Proceedings of the 22nd World Association for the Advancement of Veterinary Parasitology Conference, August 8–13, Calgary, Canada). A lack of awareness of the existence of this blood fluke and its associated pathology, particularly among veterinarians from nonendemic areas of the United States, as well as the often nonspecific signs and laboratory data associated with infection, may lead to a misdiagnosis.
Clinical signs associated with H. americana infection are related to gastrointestinal disease and include mucoid to bloody diarrhea, lethargy, weight loss, vomiting, and hyporexia or anorexia. 4,5,8 More than 50% of cases are also associated with hypercalcemia, 4,6,13 which is likely due to H. americana–induced granulomatous disease. The presence of the granulomas, formed in response to the dissemination of eggs to various organs, is believed to result in unregulated calcitriol synthesis by macrophages. Serum parathyroid hormone-related protein is elevated in some cases. 6 Abdominal ultrasound results have either been within normal limits 6 or shown evidence of nonspecific hepatic disease, peritoneal effusion, thickening of the intestinal wall, enlarged mesenteric lymph nodes, splenomegaly, or renal disease. 4,5,13 While affected tissues may contain areas of dystrophic mineralization, 4 radiographic or ultrasonographic evidence suggesting widespread soft-tissue mineralization has not been reported as a feature of this disease.
A 3-year-old, intact female, mixed-breed dog from the Gulf Coast region of Texas was examined at the Texas A&M University Veterinary Medical Teaching Hospital (College Station, Texas) because of vomiting, lethargy, and anorexia. The dog, which had been rescued by the current owners at 1 year of age, had lived mostly indoors for the past 2 years with access to 3 acres of land.
Plasma biochemical abnormalities included hypoglycemia (55 mg/dl; reference [ref.] interval: 60–135 mg/dl), hypocholesterolemia (72 mg/dl; ref. interval: 120–247 mg/dl), hypoalbuminemia (1.1 g/dl; ref. interval: 2.4–3.6 g/dl), and hyperammonemia (170 µg/dl; ref. interval: 0–50 µg/dl), which were supportive of hepatic insufficiency. Although alkaline phosphatase was elevated (292 U/l; ref. interval: 24–147 U/l), alanine aminotransferase and gamma glutamyltransferase were within normal limits. Additional biochemical abnormalities included mild hypocalcemia (7.3 mg/dl; ref. interval: 9.3–11.8) and mild hyperphosphatemia (8.2 mg/dl; ref. interval: 2.9–6.2). Complete blood cell count abnormalities included anemia (packed cell volume 26.2%; ref. interval: 31–56%), reticulocytosis (reticulocyte 4.4%; ref. interval: 0–1.5%), and the presence of nucleated erythrocytes, indicative of a regenerative anemia. Increased prothrombin time, partial thromboplastin time, and d-dimers were suggestive of thromboembolic disease or subclinical disseminated intravascular coagulation, although a platelet count was within normal limits. Abdominal fluid analysis suggested bile peritonitis with abdominal sepsis. A urinalysis was not performed.
Abdominal radiographs demonstrated multiple, thin linear areas in the wall of the small intestine (Fig. 1). Abdominal ultrasound revealed a large amount of echogenic peritoneal effusion and numerous hyperechoic pinpoint foci throughout a patchy heterogeneous and hyperechoic liver. In a large part of the small intestine and colon, the submucosa was diffusely hyperechoic (Fig. 2), with the affected areas corresponding to the linear areas of mineralization observed on the abdominal radiographs. The tunica muscularis was also affected but to a lesser extent. Areas of mineralization were also found in the pancreas. The findings were consistent with widespread soft-tissue mineralization. Calculi were present in the gall bladder, kidneys, and urinary bladder. Possible causes considered for the presumed widespread and unusual mineral deposition included atypical manifestation of hyperadrenocorticism, vitamin D intoxication, hypothyroidism, and imbalance of calcium-to-phosphorus ratio. Canine schistosomiasis was not considered. Due to its poor condition, the dog was euthanized and submitted for necropsy.
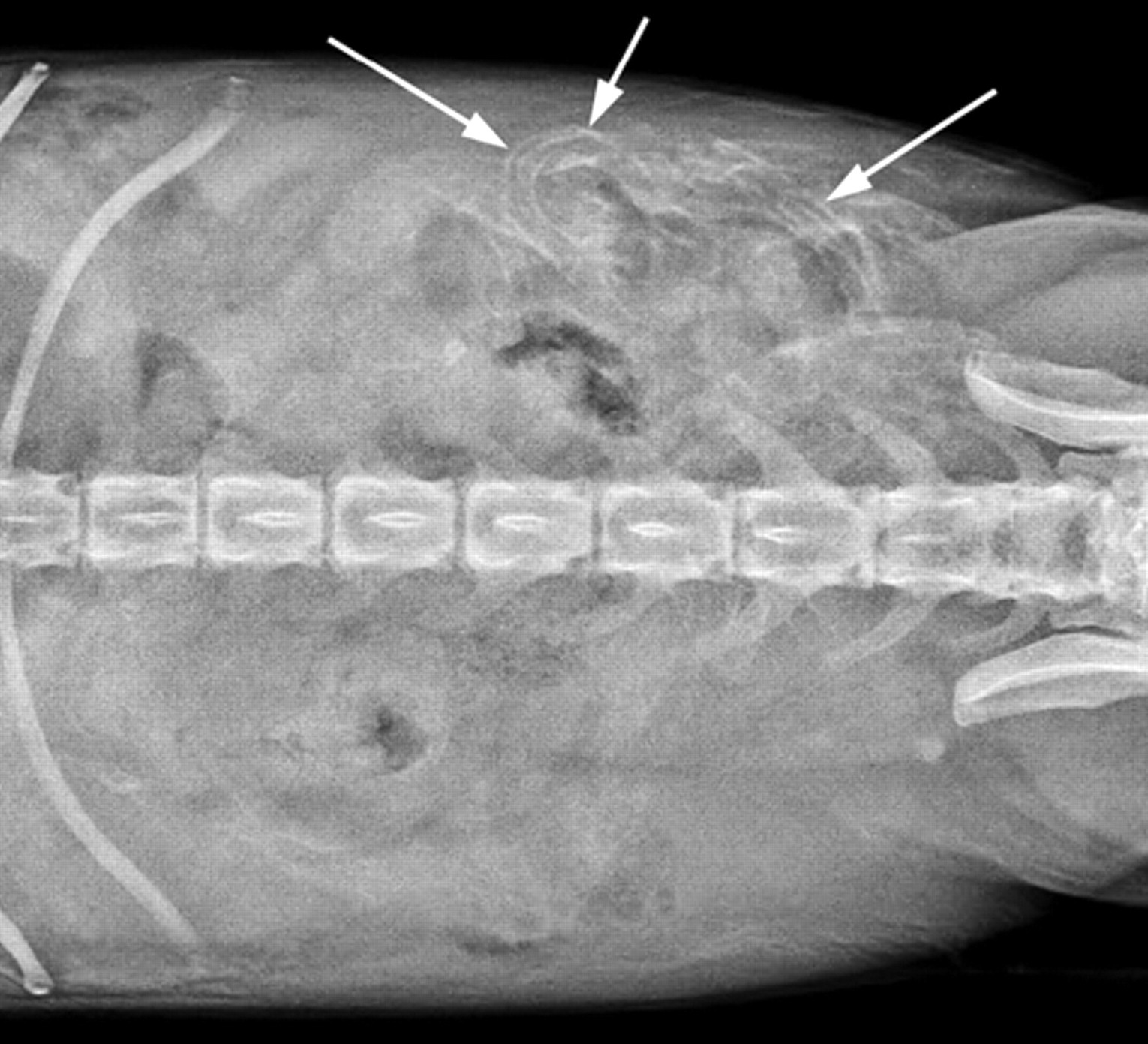
Ventrodorsal radiographic image of the abdomen; dog. Thin, linear areas of mineralization are in the wall of the small intestine (arrows) in the left mid abdomen.
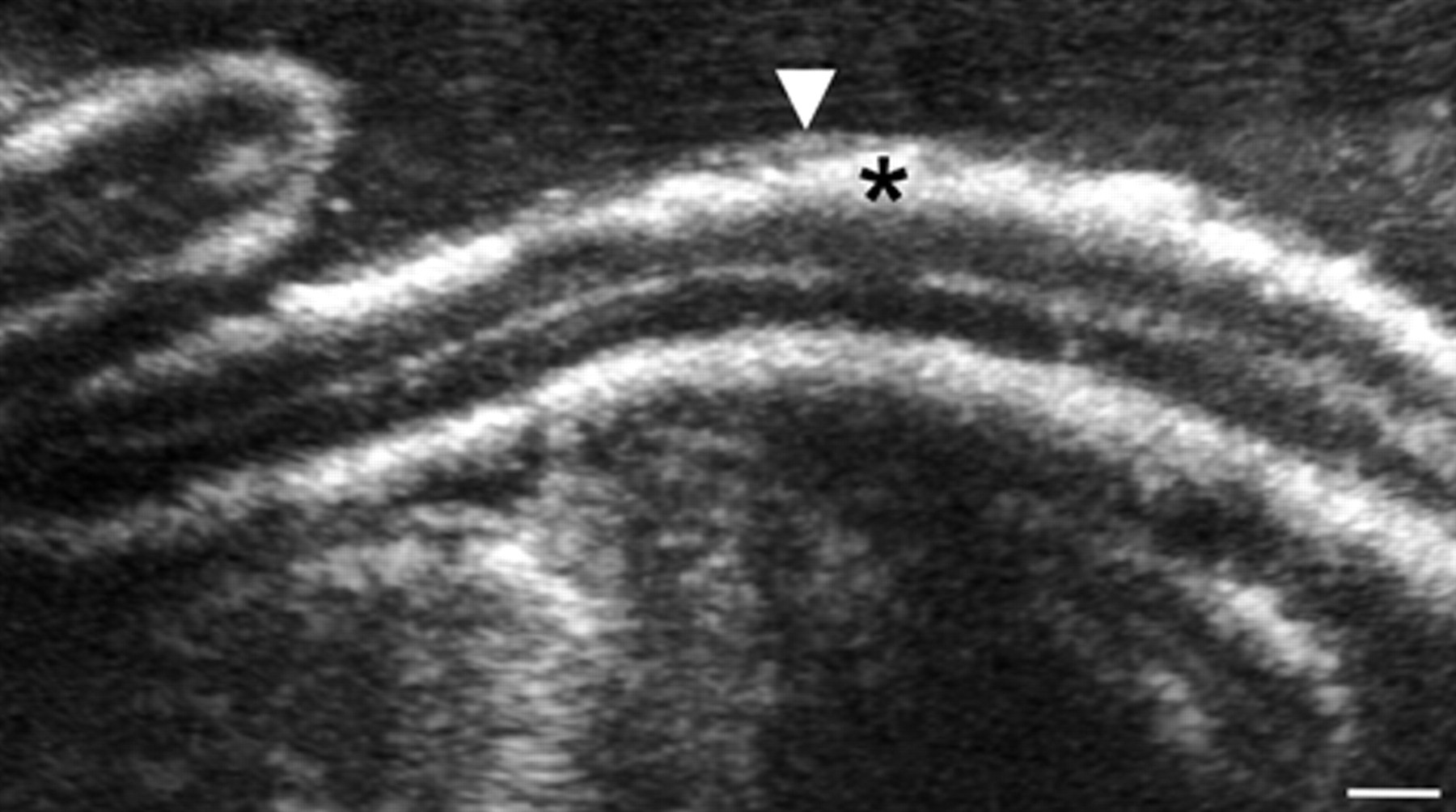
Ultrasonographic image of the abdomen; dog. Thick, linear, hyperechoic areas of mineralization extend circumferentially throughout the submucosa (*) and portions of the tunica muscularis (arrowhead) of a segment of the small intestine. Bar = 0.25 cm.
At necropsy, the abdominal cavity contained approximately 200 ml of reddish brown, blood-tinged fluid with rare fibrin strands. The liver was small (2.65% of body weight), pale, and had an uneven capsular surface with many scattered, 1–2 mm, minimally raised and rounded, pale gray to white areas (Fig. 3). These areas extended into the parenchyma and were faintly visible and randomly scattered throughout the liver on cut section. A localized area of hemorrhage with clotted blood was on the visceral surface of the right medial and lateral liver lobes in an area surrounding the neck of the gall bladder and adjacent to the duodenum. This area was considered the source of the abdominal fluid. A well-circumscribed hepatocellular adenoma was in the right medial lobe, and the gall bladder contained a small amount of inspissated bile along with a small number of 2–3 mm in diameter, dark green to black choleliths. The wall of the small intestine was mildly thickened but was considered to be of normal consistency and without evidence of mineralization. The thickening was more noticeable following fixation and was mostly confined to the mucosa and submucosa. The kidneys contained multiple, 1–2 mm in diameter, firm, well-circumscribed white areas within the outer cortex (Fig. 3). A 7.0 mm × 4 mm, hard, green and yellow nephrolith was in the pelvis of the right kidney. Numerous, small uroliths, up to 2 mm in diameter, were in the urinary bladder.
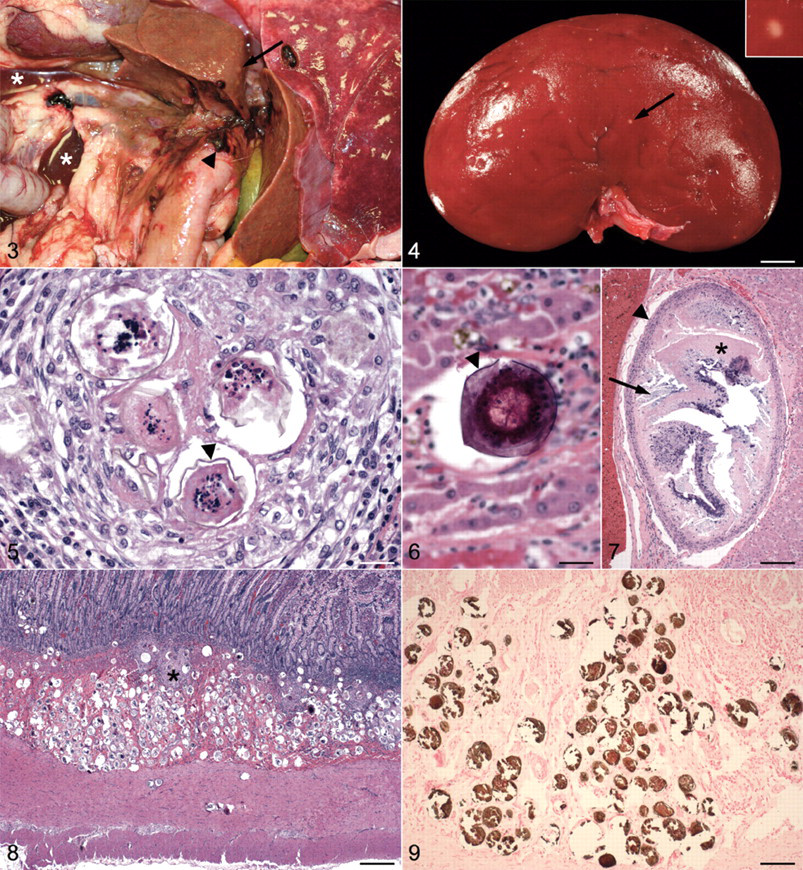
Abdominal cavity; dog. The dog is in left lateral recumbency with the right caudal and middle lung lobes to the right. The liver is small, pale, and contains multiple small, elevated, pale gray to white nodules (arrow). Areas of hemorrhage are around the base of the right medial and lateral liver lobes, around the common bile duct, and on the duodenal serosa (arrowhead). Two small pools of reddish brown, blood-tinged fluid are free within the abdominal cavity (*).
Samples of tissue from all major organs were immersed in 10% formalin, processed in a standard manner, and 4-µm sections were stained with hematoxylin and eosin or von Kossa stain. Due to the presence of a large number of mineralized eggs in some tissues, a 15-min surface decalcification of tissue blocks with a 50:50 mixture of 50% formic acid and 20% sodium citrate was also used to prevent tears in the tissue during sectioning and artifactual loss of eggs. Measurements of eggs were made using digital photomicroscopy and a calibrated imaging software system. a
Histologically, the liver contained multifocal areas of random, lymphoplasmacytic, histiocytic, and eosinophilic to granulomatous hepatitis with intralesional schistosome eggs consistent with H. americana (Fig. 3). The eggs were 55–75 µm in diameter and consisted of a thin, 1–2 µm, yellow–brown, round to collapsed and often mildly undulated outer eggshell surrounding a multicellular miracidium. Eggs and egg fragments were often partially or completely mineralized (Fig. 3). Some of the more heavily mineralized eggs were inadvertently pulled from the tissue during sectioning, leaving behind fragments of eggs or only round empty spaces surrounded by a thin layer of fibrous tissue. A cross section of one expanded portal arteriole contained 18 closely packed eggs within its lumen. Additional changes in the liver included periportal to bridging fibrosis, lobular collapse, nodular regeneration, bile duct hyperplasia, mild cholestasis, and scattered individual histiocytes and lipogranulomas with abundant, intracytoplasmic, yellow–brown, granular pigment. Rare portal vessels contained a single, large, up to 1 mm × 0.63 mm, ovoid structure containing a central area of irregular, necrotic, eosinophilic material that was partially to heavily mineralized (Fig. 3). In some sections, the central eosinophilic material was more organized and lined by a thin cuticle, suggesting that it was a degenerate adult schistosome. The presumed schistosome was surrounded by additional areas of necrosis and mineralization and lined by an outer layer of macrophages and fibroblasts. A hepatocellular adenoma in the right medial lobe lacked the chronic inflammatory changes found in the remaining sections of liver and did not contain any eggs, in spite of the fact that these changes were easily visible in areas immediately surrounding the adenoma.
The small intestine contained the largest number of H. americana eggs (Fig. 3). Eggs were most heavily and diffusely concentrated in the submucosa where they were frequently mineralized and sometimes surrounded by areas of granulomatous inflammation. The large number of mineralized eggs in this area often resulted in empty spaces and tears in the tissue due to artifactual removal of eggs during sectioning. A short period of decalcification of tissue blocks resulted in better preservation of sections, with many more eggs remaining in the tissue than could be appreciated otherwise. Eggs extended into the muscularis mucosa where they were often arranged in linear chains of up to 10 closely aligned eggs. Dense clusters of up to 25 eggs were sometimes found in deep cryptal areas of the mucosa. In a few areas, a linear chain of eggs had migrated up to the base of the villi. Similar, although less severe, changes were found in the colon. A von Kossa stain of the small intestine demonstrated a large amount of calcium salt in the mineralized eggs within the submucosa (Fig. 3).
The abundance of mineralized eggs in the submucosa of the small intestine correlated well with the markedly hyperechoic areas observed in the abdominal ultrasonographic images of the small intestine (Fig. 2). In the histologic sections, soft-tissue mineralization, apart from the eggs themselves, was not found in any of the tissues examined.
Similar dense clusters of H. americana eggs and egg fragments, undergoing varying degrees of mineralization and often surrounded by areas of granulomatous inflammation, were present in the pancreas. Single eggs or discrete, focal areas of granulomatous inflammation containing up to 15 eggs were scattered throughout the lungs. Within the cortex of the kidneys there were multiple, 0.7–1 mm in diameter granulomas with necrotic, partially mineralized centers. These granulomas corresponded to the white foci observed grossly (Fig. 3). While their histologic appearance was similar in size and shape to the few larger granulomas in the liver (Fig. 3), those in the kidney contained only necrotic centers that lacked any organized structure. The inflammatory cell population surrounding the granulomas in the kidneys contained numerous eosinophils, but eggs were not found. The ultrasound images of the kidneys did not contain hyperechoic, mineralized foci in the renal parenchyma, and radiographic images of the kidneys and lungs did not detect any abnormalities at all. Significant histologic changes were not found in the heart, brain, stomach, cecum, rectum, or adrenal glands.
Three, 20-µm thick curls of tissue taken from blocks of formalin-fixed, paraffin-embedded samples of liver and small intestine were used for DNA extraction using a commercial kit, b following the manufacturer’s instructions for paraffin-embedded tissue. Using genomic DNA, a portion of the 18S small subunit ribosomal RNA gene was amplified by polymerase chain reaction (PCR) and sequenced, utilizing primers designed to be specific for H. americana (Het 1298F 5′-ATGGCCGTTCTTAGTTGGTG-3′ and Het 1824R 5′-CGTCTTCTCAGCATCAGTCG-3′). Identical unambiguous sequences of 487 base pairs were obtained from both hepatic and intestinal tissues. The sequences showed 100% identity with H. americana sequences in GenBank (accession nos. DQ230918 and AY157220). The new sequence was assigned the GenBank accession number HQ339878.
Heterobilharzia americana infection in dogs is an increasing yet underdiagnosed problem in the southeastern United States. A reported higher incidence in clusters of urban areas (Snowden K, et al.: 2009, Distribution and characterization of Heterobilharzia americana infections in the dog in Texas) is likely related to a combination of the presence of raccoons, which serve as the definitive host, standing fresh water containing the appropriate snail intermediate host, and a population of dogs with enough exposure to the cercariae-infested water to allow for penetration of the cercariae through the dog’s skin. The 2007 report of canine H. americana infections in Kansas (Wilson N, Johnson EM, Brunker J, Ritchey J: 2007, Heterobilharziasis in a dog. In: Proceedings of the 52nd Annual Meeting of the American Association of Veterinary Parasitologists, July 14–17, Washington, DC), which presumably resulted from the earlier importation of infected raccoons from Texas and Florida, 12 suggests that spread of the disease to new areas is likely to continue.
While previous reports have documented the life cycle of H. americana 9 and the pathologic changes associated with infections in dogs, 5,11,14 the severity of the infection in the present case, the number of organs involved, including the liver, small intestine, colon, pancreas, lung, and kidneys, and the lack of hypercalcemia in the face of clinically diagnosed soft-tissue mineralization, is somewhat unusual. Polymerase chain reaction amplification of DNA from formalin-fixed sections of small intestine and liver confirmed the presence of H americana.
One of the more striking features about the current case was the number of mineralized eggs in multiple tissues that gave the false impression of soft-tissue mineralization on the abdominal radiographs and ultrasound images. To the authors’ knowledge, ultrasonographic findings suggesting widespread systemic mineralization in dogs with H. americana infection have not been previously reported. Although mineralization of H. americana eggs is not uncommon and is considered to be part of the host response to chronic infection, the large number of eggs, particularly in the intestinal tract, liver, and pancreas, combined with evidence of calculi in the kidney, urinary bladder, and gall bladder, led to an antemortem diagnosis of atypical systemic mineralization of unknown origin, most likely associated with metabolic disease. The severity and distribution of the mineral was uncharacteristic for most common causes of dystrophic or metastatic mineralization.
An antemortem diagnosis of H. americana infection can often be made with the aid of a fecal sedimentation test. A PCR-based assay for the detection of eggs in feces or from hepatic or intestinal biopsies is also available on a research basis. Postmortem diagnosis is readily accomplished when the characteristic eggshells and miracidia, many of which are associated with granulomatous inflammation, are found in the intestine, liver, or other tissues. The level of suspicion of H. americana infection should be increased when there is a history of diarrhea, lethargy, weight loss, vomiting, and anorexia in dogs living in an endemic area. Since the adult male and female schistosomes live in copula in the mesenteric veins, eggs may reach the liver through the portal system and result in hepatic dysfunction and related biochemical abnormalities, as noted in the present case. Radiographic and ultrasonographic findings mimicking soft-tissue mineralization, particularly in the small intestine and liver, should be considered as possible further evidence of infection. A greater awareness of the prevalence of H. americana in endemic areas of the United States and the clinical, pathologic, and imaging changes associated with canine infections will aid in more accurate diagnosis and treatment.
Footnotes
a.
SPOT software, Diagnostic Instruments Inc., Sterling Heights, MI.
b.
PurGene DNA Purification System, Gentra Systems Inc., Minneapolis, MN.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
