Abstract
A private zoological facility experienced an outbreak of malignant catarrhal fever (MCF) in 3 bongo antelope (Tragelaphus eurycerus). All cases were periparturient bongos that presented acutely anorectic beginning ~6 weeks after being housed with a Nubian ibex. Disease quickly progressed to respiratory distress and death within 24–72 hours of onset of clinical signs. Consistent gross findings in affected bongos were pulmonary edema and small pale tan foci in the livers. Histological lesions included a nonsuppurative vasculitis in multiple tissues, which is well recognized in MCF, but additionally included necrotizing cholangiohepatitis and neutrophilic, necrotizing myocarditis. Ibex-associated viral DNA was detected by polymerase chain reaction and was identical in sequence whether derived from bongos or ibex. The sequence closely matched an MCF viral DNA fragment that had been amplified from an ibex and bongo in a previous case report.
Keywords
Malignant catarrhal fever (MCF) is a herpesviral disease of ungulates with a high case-fatality rate in susceptible species. 13,14 Disease is often sporadic in domestic animals; however, significant epidemics occur. 8,9,11,14,16 The causative agents are a group of gammaherpesviruses in the genus Macavirus. 2 Disease results when virus is transmitted from an endemic, asymptomatic reservoir species to a clinically susceptible species. Outbreaks of MCF in domestic cattle caused by infection with the macaviruses Ovine herpesvirus-2 (OvHV-2) and Alcelaphine herpesvirus-1 (AlHV-1) have been well-described following exposure to the reservoir species, sheep, and wildebeest (Connochaetes sp), respectively. 13,14 Infection may affect virtually any organ and thus may present with a wide variety of clinical manifestations; however, in domestic ruminants the most common clinical presentations are gastrointestinal, central nervous system, and ocular disease. 14 Recognition of these patterns of clinical disease facilitates a differential diagnosis of MCF, identification of affected animals, and the initiation of appropriate intervention.
In zoological and similar facilities, timely clinical diagnosis of MCF is more challenging. Many outbreaks involve macaviruses other than OvHV-2 and AlHV-1; they occur in various combinations of reservoir and susceptible species, and the clinical presentations may vary from those classically described for domestic ruminants. 3,7,12 MCF or MCF-like disease has been identified in a wide variety of hoofstock within zoological collections, and subclinical carrier states have been confirmed or suspected in, suggesting that these populations are at risk for unidentified or poorly characterized MCF viruses. 3,4,7 One such novel virus (MCFV-ibex) has been previously reported to cause fatal disease in a bongo, and the likely reservoir sources were unaffected Nubian ibex that were housed nearby. 7,12 This article further characterizes the susceptible host–reservoir (bongo–ibex) combination, including the unique clinical presentation and gross and histological lesions of MCFV-ibex disease. This information strengthens the epidemiological link that establishes ibex as the source of MCFV that is potentially fatal in bongos. Importantly, the clinical presentation of infected bongos differs markedly from the common presentations in MCF-infected domestic ruminants.
In the fall of 2009, a private zoological facility pastured 3 separate groups of bongo antelope (Tragelaphus eurycerus). One group, composed as a breeding program, consisted of 3 pregnant adult female bongos and 1 adult male bongo. The second group was a “bachelor group” of 2 subadult male bongos that were kept in a mixed-species enclosure. On November 19, one of the female bongos calved, at which time all 3 female bongos and the calf were moved to an isolation barn. A newly acquired 3-year-old male Nubian ibex (Capra nubiana) that had been placed in quarantine in this same isolation barn 5 days earlier was present in the barn when the bongos arrived. This male ibex (ibex 1) was released from quarantine after a 5-day period during which the bongos and ibex were cohoused. All bongos and ibex 1 appeared healthy during this time. No other animals were moved into or out of this barn. The bongos remained in the barn for the next 6 weeks. Within a 3-week period all 3 adult females successively exhibited an acute onset of anorexia rapidly progressing to respiratory distress and death within 24–72 hours. Of note, the 3 male bongos, housed in a mixed-species enclosure that included periodic exposure to resident female ibex, remained healthy prior to and during the outbreak.
Routine field gross necropsies were performed by 1 of 3 attending clinical veterinarians on all 3 affected female bongos. From bongo No. 1, a section of lung was the only tissue submitted to a private laboratory for histopathological examination. Fresh tissues from bongo Nos. 2 and 3 as well as tubes of clotted blood from bongo Nos. 1 and 2 and the live calf born from bongo No. 3 were submitted to the California Animal Health and Food Safety Laboratory at Davis, California (CAHFS–Davis) for diagnostic testing and histopathological examination. In addition, serum and EDTA blood were later submitted to CAHFS–Davis from ibex No. 1 and 4 resident female ibex. Tissues received from bongo No. 2 included lung, heart, bronchiolar lymph node, esophagus, and liver. Tissues received from bongo No. 3 included heart, lung, and liver. In addition, for bongo No. 2, the liver was tested for heavy metals, selenium, and vitamin E levels. Lung, bronchial lymph node, liver, and a pericardial swab were cultured on blood, MacConkey’s, chocolate, and Noble agars and D broth, and fluorescent antibody tests were run on frozen lung for infectious bovine rhinotracheitis (IBR), bovine viral diarrhea (BVD), and parainfluenza-3 (PI3) viruses. In addition, both pooled and individual tissues from bongo No. 2 were submitted in viral transport media to the Foreign Animal Disease Diagnostic Laboratory (FADDL), USDA, APHIS for virus isolation.
Serum from the cohoused male ibex, 4 pastured ibex, bongo No. 2, and the live calf born from bongo No. 3 were submitted to Agricultural Research Service (ARS), USDA, Pullman, Washington, or to FADDL for cELISA testing (ARS) or immunoperoxidase (FADDL) for antibodies to MCFV. Polymerase chain reactions (PCR) for detection of several MCF viruses were performed on blood and tissue samples at multiple institutions including the following: fresh sections of lung, liver, and bronchial lymph node (bongo No. 2) were submitted to the FADDL for nested PCR testing of OvHV-2, AIHV1, and AIHV2. 1 Lung, liver, and bronchial lymph node from bongo No. 2 and liver and kidney from bongo No. 3 were submitted to ARS for multiplex PCR for several MCF viruses known to cause disease (see Table 1). Samples of blood preserved in EDTA from the 5 ibex, which included ibex No. 1 and 4 resident female ibex, and bongo No. 1 were also submitted to ARS for PCR analysis for MCF viral DNA (see Table 1). To confirm the identity of the ibex-associated MCFV in the affected bongo and ibex, tissue samples (lung, lymph node, liver, and kidney from bongo Nos. 2 and 3) and blood samples (from ibex) were also evaluated by PCR using degenerate primers that target a region of the herpesviral DNA polymerase gene. 15 The PCR amplification conditions were described in a previous report. 6 PCR amplification products were purified and cloned into the pCR2.1 vector using TOPO TA Cloning Kit (Invitrogen, Grand Island, NY, USA). Plasmid DNA was extracted and sequenced, which was carried out by Amplicon Express (Pullman, Washington). Alignment of the amplified segments (177 base pair nonprimer DNA sequence) from the bongo and ibex was carried out by Vector NTI Software (Invitrogen) (Figure 1a). A phylogenetic tree including the ibex-associated MCFV from this study and known disease-associated macaviruses in the MCF virus group was generated based on nucleotide sequences of DNA polymerase gene fragments constructed by using the DNASTAR MegAlign program (Figure 1b). Routine gross necropsies were also performed at CAHFS–Davis on ibex No. 1 and 3 female ibex that were culled for management purposes following the outbreak.
Summary of Diagnostic Findings in 3 Bongo and 5 Ibex From the Wildlife Park
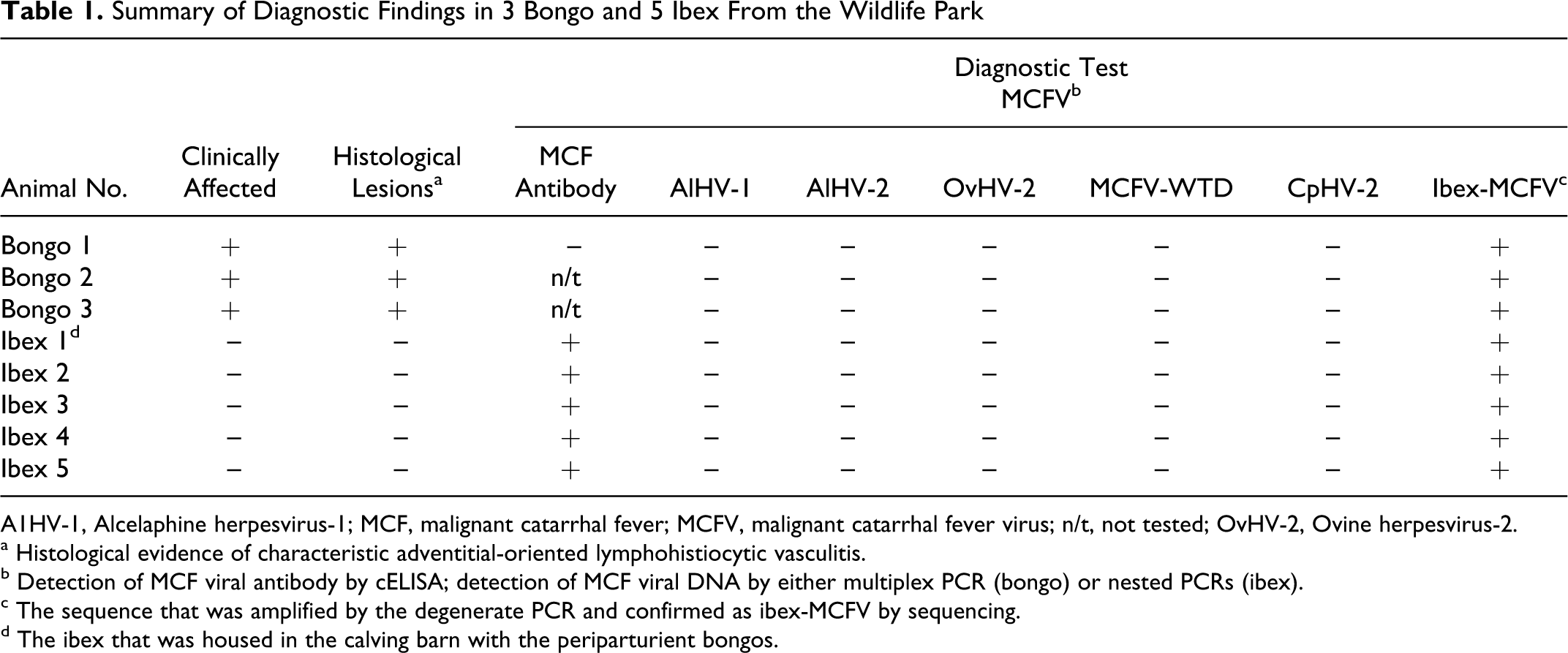
A1HV-1, Alcelaphine herpesvirus-1; MCF, malignant catarrhal fever; MCFV, malignant catarrhal fever virus; n/t, not tested; OvHV-2, Ovine herpesvirus-2.
a Histological evidence of characteristic adventitial-oriented lymphohistiocytic vasculitis.
b Detection of MCF viral antibody by cELISA; detection of MCF viral DNA by either multiplex PCR (bongo) or nested PCRs (ibex).
c The sequence that was amplified by the degenerate PCR and confirmed as ibex-MCFV by sequencing.
d The ibex that was housed in the calving barn with the periparturient bongos.
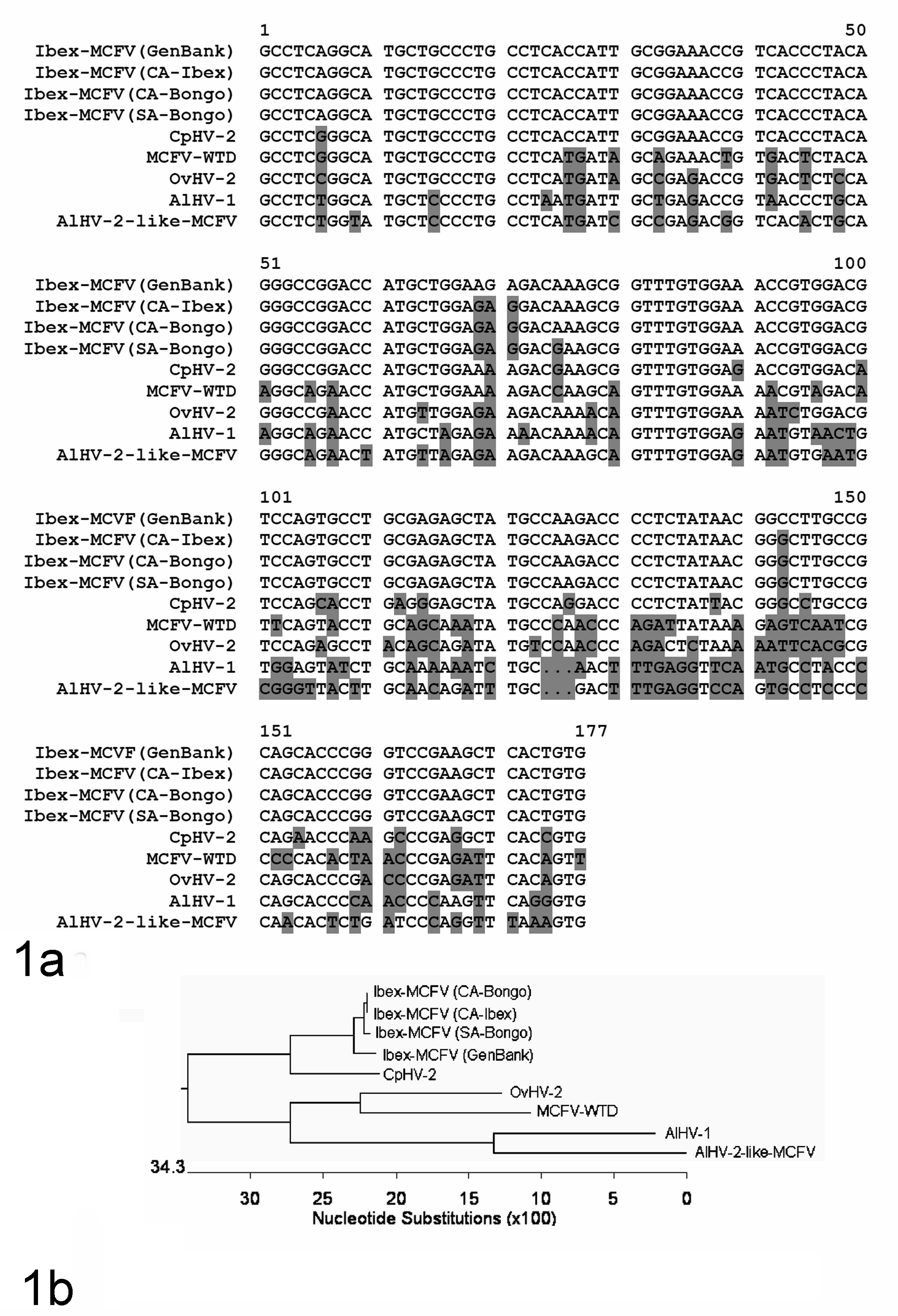
a. Partial DNA sequence alignment of the MCFV DNA polymerase gene derived from bongo (CA-Bongo, this outbreak) and ibex (CA-Ibex, this outbreak) with previously reported MCFV. The sequences include 3 ibex-MCFV (Genbank, accession AY212112), MCFV from a bongo with clinical MCF from San Antonio Zoo (SA-Bongo), caprine-associated (CpHV-2 (AF275941)), ovine-associated (OvHV-2 (AF031812)), deer associated (MCFV-WTD (AF181468)), AlHV-1(NC_002531), and AlHV-2-like MCFV (AY092762). The sequence alignment was carried out by the Vector NTI Software (Invitrogen). b. Phylogenetic tree of the known disease-associated MCFVs based on nucleotide sequences of DNA polymerase gene fragments (above). The phylogenetic tree was constructed by using the DNASTAR MegAlign program (Lasergene, Madison, WI).
Necropsy Results
All bongos were in good body condition with good hair coats and formed feces in their colons. Lungs in all cases were wet, heavy, and mottled red to dark red. In bongo Nos. 2 and 3, capsular and cut surfaces of liver had infrequent, soft, scattered 0.1- to 0.3-cm-diameter irregular pale tan foci. In bongo No. 2, additional gross findings were a thickened pericardial sac with cloudy, fibrinous pericardial fluid that adhered to the epicardium, and ecchymotic and paint-brush hemorrhages scattered over the atrial epicardia. Bronchiolar lymph nodes in bongo No. 2 were light brown, soft, and mildly enlarged.
Histological examination was performed on lung from all 3 bongos; heart and liver from bongo Nos. 2 and 3; trachea, esophagus, and bronchial lymph node from bongo No. 2; and kidney from bongo No. 3. Histological examination of the lung from bongo No. 1 was performed by a private laboratory, and reported findings included congestion, edema, patchy perivascular adventitial accumulations of mononuclear cells, and flooding of scattered alveoli with mixed bacteria. Bongo Nos. 2 and 3 were sent to CAHFS–Davis. In both cases, the lungs were congested and edematous with diffuse and often prominent adventitial perivascular inflammatory cell infiltrates (Fig. 2). The infiltrates were composed primarily of medium to large lymphocytes with lesser macrophages, rare plasma cells, and neutrophils. Random mitoses and individual cell necrosis (karyorrhexis) of mononuclear cells were also noted. Mononuclear cells focally transmigrated through vascular media and intima with associated focal mural necrosis (Fig. 3). Proteinaceous fluid and fibrin were present in alveolar and airway lumen.
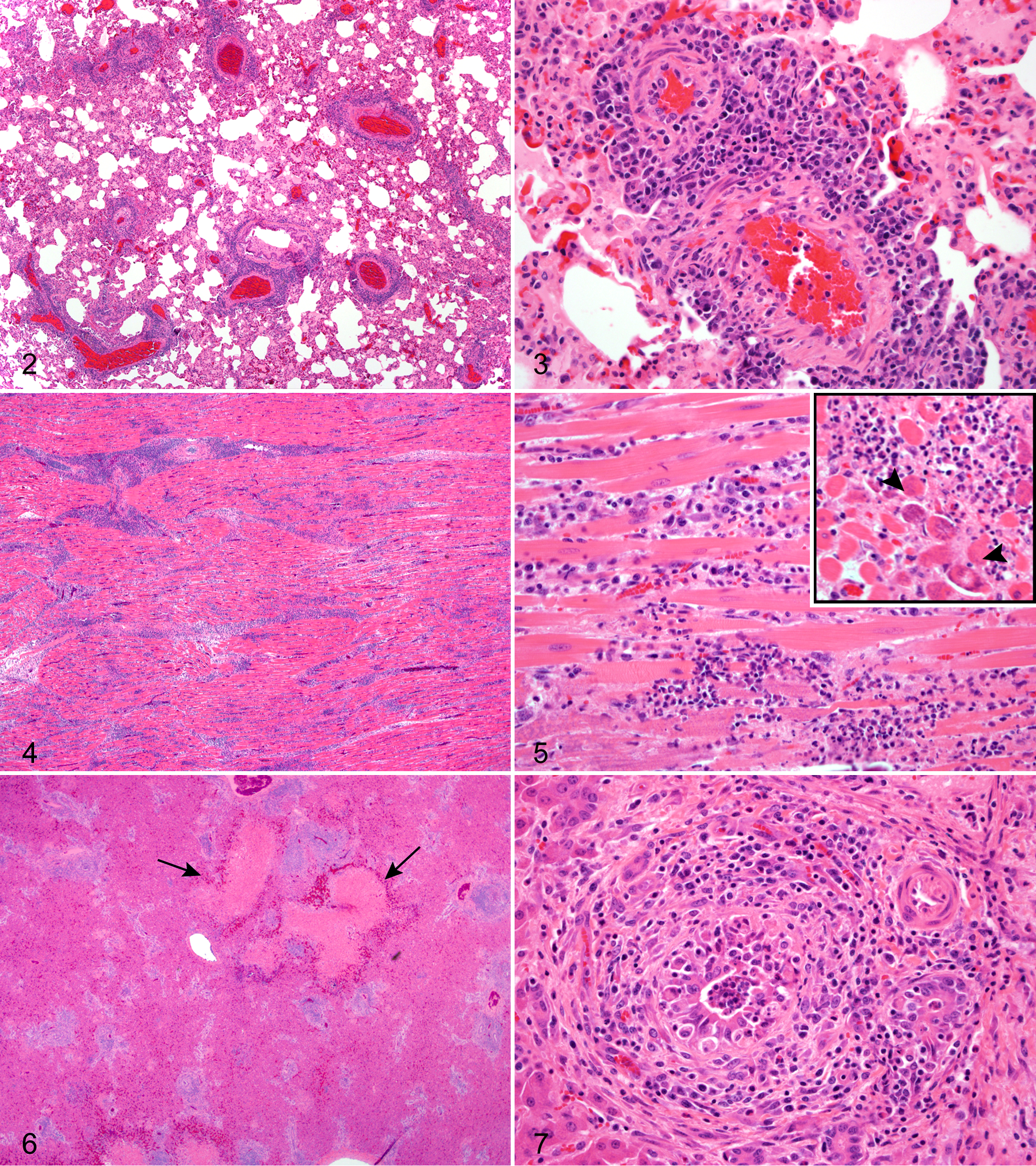
Lung; bongo No. 2. There are dense perivascular infiltrates, some of which partially extend around adjacent bronchioles. HE.
In both cases in which the heart was examined (bongo Nos. 2 and 3), there was a vasculitis that was focused primarily within the adventitia, with rare mural vascular infiltrates and necrosis. In both cases there was also a more diffuse and striking pattern of myocardial inflammation (Fig. 4) with acute, scattered, monophasic myofiber necrosis and accompanying interstitial neutrophilic infiltrates localized around necrotic myofiber segments (Fig. 5). Myofibers were occasionally mineralized (Fig. 5, inset). There were scattered, mild, mononuclear infiltrates present beneath the endocardium and epicardium sometimes associated with accumulations of fibrin. The livers from bongo Nos. 2 and 3 exhibited a similar pattern of inflammatory changes. Randomly scattered irregular foci of acute hepatic necrosis (Fig. 6) were present throughout the liver. In addition, portal tracts were expanded with dense infiltrates of lymphocytes, histiocytes, and plasma cells. In random portal tracts there also were prominent neutrophilic infiltrates that surrounded, disrupted, and occluded bile ducts (Fig. 7). Portal fibrosis was mild in bongo No. 3, but in bongo No. 2 there was focal bridging portal fibrosis and biliary hyperplasia. Kidney was available for examination in bongo No. 3 only. There were scattered infiltrates of lymphocytes and histiocytes that expanded the interstitium in multiple segments and coalesced around small to medium-sized arterioles and/or glomeruli. Histopathological changes in the esophagus from bongo No. 2 were very limited and consisted of minimal focal superficial mucosal, perivascular lymphocytic infiltrates with accompanying mild epidermal exocytosis and rare single apoptotic epithelial cells. The remainder of the gastrointestinal system was not submitted for examination in any of the affected bongos. The bronchial lymph node from bongo No. 2 was markedly congested, edematous, and subcapsular, and medullary sinuses were filled with mixed inflammatory cell infiltrates. At necropsy, all ibex that had been culled for depopulation (subsequent to the outbreak) were unremarkable.
Other Diagnostic Tests
Liver vitamin E, selenium, and all heavy metals from bongo No. 2 were within the normal range for domestic livestock excepting copper, which was low (1 ppm). No bacteria or viruses were isolated (by culture) from fresh tissues from bongo No. 2. Fluorescent antibody test on lung for IBR, BVD, and PI3 viruses at CAHFS–Davis were negative. All ibex samples were positive, and the bongo No. 1 sample was negative for the presence of MCF-specific antibody (see Table 1). Lung, liver, and bronchial lymph node from bongo No. 2 and liver and kidney from bongo No. 3 were uniquely positive for ibex-associated MCFV (Table 1). EDTA blood from all 5 ibex tested and bongo No. 1 as well as the surviving calf from bongo No. 2 was submitted to ARS for PCR analysis for MCF viral DNA and were all uniquely positive for ibex-associated MCFV (Table 1), excepting the surviving calf from bongo No. 2.
The clinical, histological, molecular, and epidemiological data from this case provide strong evidence of point-source transmission of MCFV from a single male Nubian ibex to periparturient female bongos. The features of disease in bongos that are unique compared with other MCFV-related diseases include a rapid clinical course, a necrotizing cholangiohepatitis, and neutrophilic, necrotizing myocarditis. Serofibrinous pericarditis and myocarditis, to our knowledge, have been previously reported only in Indonesian swamp buffaloes with MCF-like disease. 5 Erosive or ulcerative oronasal lesions and enteritis, which are common in cattle, bison, and/or deer affected with MCF, were not detected grossly in the affected bongos, and there was no clinical evidence of gastrointestinal disease. However, we are unable to rule out involvement of the gastrointestinal system since, aside from esophagus from bongo No. 2, no gastrointestinal tissues were available for histological examination. The sequence analysis of the MCFV amplified DNA detected in all of the bongos was identical to the MCFV detected in all ibex tested, which included the in-contact male ibex 1 and the resident herd of female ibex (Table 1, Fig. 1).
Fatal disease occurred in the pregnant or postparturient female bongos after cohabitation with the single male ibex 1, following a postexposure incubation period of approximately 2–6 weeks, consistent with a variable 1- to 16-week incubation period reported with various outbreaks of MCFV. 3,4,10,13,14 The pastured male bongos, presumably exposed by periodic cohabitation with resident female ibex, were unaffected, suggesting that other factors could contribute to viral transmission or disease progression, including proximity to infected animals and stress (affecting host viral shedding or susceptibility of the female bongos). In particular, late-term pregnancy and/or low levels of liver copper may have altered the immune response or disease susceptibility of the bongos, and stress (transport, housing) in the ibex might have increased viral shedding. It is notable that the exposed pregnant/postparturient female bongos acquired the MCFV infection, whereas the neonatal calf of bongo No. 2 (also housed in the isolation barn with the male ibex) was seronegative for MCFV by cELISA and remains healthy.
Footnotes
Acknowledgements
We thank the CAHFS–Davis and UC Davis VMTH Histopathology Laboratories for their technical expertise.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
