Abstract
Eight duikers, representing 3 different species cohoused in a single zoological collection, died in a 10-month period. Black, red-flanked, and yellow-backed duikers were affected, appearing clinically with a combination of anorexia, diarrhea, ataxia, tremors, and/or stupor, followed by death within 72 hours of onset of clinical signs. Consistent gross findings were pulmonary ecchymoses (8/8), generalized lymphadenomegaly (6/8), ascites (5/8), and pleural effusion (4/8). Dense lymphocyte infiltrates and arteritis affected numerous tissues in most animals. Ibex-associated malignant catarrhal fever (MCF) viral DNA was detected in all cases by polymerase chain reaction and in situ hybridization. Identical ibex-MCF virus sequence was detected in spleen of a clinically healthy ibex (Capra ibex) housed in a separate enclosure 35 meters away from the duikers.
Keywords
Malignant catarrhal fever (MCF) is a worldwide, often fatal, lymphoproliferative disease predominantly affecting domestic and wild ruminants, caused by a group of MCF viruses (MCFV) from the genus Macavirus, sub-family Gammaherpesvirinae, family Herpesviridae. 13,17 MCFV infections are persistent, and usually not clinically significant in their adapted host species, but can cause fatal disease if infection occurs in a nonadapted species. The MCFV group currently comprises 10 members, 7 of which have been associated with disease. 6,17,19 Common histologic lesions of MCF include lymphoproliferation, vasculitis, and mucosal ulceration. 17,19
MCF poses a serious risk to zoological collections, and while zoos make every attempt to avoid housing MCF-susceptible animals together with, or close to, known carrier species, chronic infections in adapted host species are generally nonclinical and only detected by the presence of MCFV-specific antibodies in these species. The problem is compounded because susceptibility to MCF disease is not known for all animal species, so, despite careful husbandry, fatal transmission of MCFV does occur in zoological collections. 8,14,16
Duikers are small to medium-sized antelopes, including 22 species that belong to the genus Cephalophus within the Cephalophinae subfamily of the Bovidae family. Duikers are native to sub-Saharan Africa where some species are threatened by extinction due to habitat loss and intensive hunting. 3 Duikers are common in zoological collections, and known susceptibility to MCF disease is limited to a single report of a red-flanked duiker (Cephalophus rufilatus), infected with alcelaphine herpesvirus-1, the carrier species of which is the wildebeest. In that case, a newly purchased wildebeest bull (Connochaetes taurinus) that shared a common feeding and service shed was considered the likely source of infection. 14
The ibex-MCFV was first recognized in Nubian ibex, which were asymptomatic carriers of the virus. 11 The virus has been previously associated with disease in bongo antelopes that were in contact with healthy ibex. 8,16 We have identified a second species susceptible to infection with ibex-associated MCFV. We present here ibex-MCFV-associated malignant catarrhal fever in 8 duikers housed close to a group of healthy Ibex (Capra ibex), which were infected with an identical strain of ibex-MCFV.
Eight duikers, including 4 red-flanked duikers (Cephalophus rufilatus), 2 black duikers (Cephalophus niger), and 2 yellow-backed duikers (Cephalophus silvicultor), died at a zoological collection in Southern California. Duiker cases 1 and 2 were received for necropsy in January and cases 3 to 8 between September and November of 2017. They included 5 males and 3 females, and ages ranged from 4 months to 8 years. Clinical disease in these animals lasted between 1 and 3 days, and the most frequently observed clinical signs included anorexia, tremors, ataxia, diarrhea, anemia, and stupor. Sudden death without premonitory signs was reported in one animal (Table 1).
Signalment, Epidemiologic, and Clinical Data of 8 Duikers With Ibex-Malignant Catarrhal Fever.
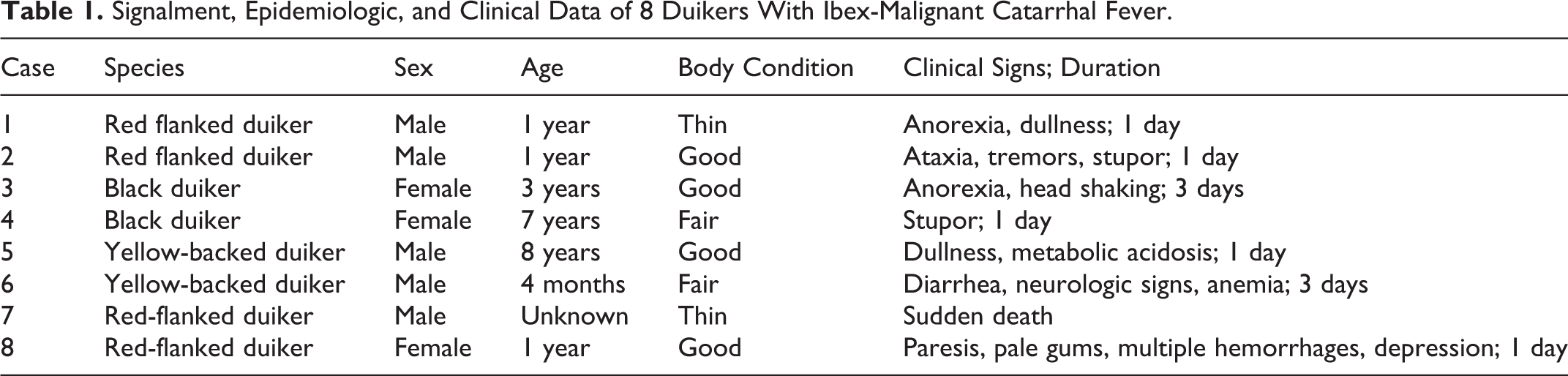
Each duiker species was housed in a separate enclosure, and the ibex were housed approximately 35 meters away in a separate exhibit. A single caretaker attended the yellow-backed duikers, while independent caretakers looked after the ibex and red-flanked duikers. At the time of the outbreak, bleach footbaths were used to access the exhibits.
The 8 affected duikers were submitted for necropsy and diagnostic workup to the San Bernardino branch of the California Animal Health and Food Safety (CAHFS) Laboratory. Subsequent to detection of ibex-associated MCF in duikers, all 7 clinically healthy but infected ibex (Capra ibex) from the same facility were culled for management purposes and submitted for necropsy. Full necropsies were performed on all affected duikers and ibexes and surveillance samples of tissues were collected for histopathology. All tissues were fixed in 10% neutral buffered formalin, pH 7.2, for between 1 and 3 days, and processed by routine histological methods to obtain 5-μm-thick, hematoxylin and eosin–stained sections. At necropsy, all 8 duikers had multifocal ecchymoses in lungs (Fig. 1). Other findings (Supplemental Table S1) included generalized lymphadenomegaly (n = 6), bilateral renomegaly with mottled white to red renal cortices (n = 6; Fig. 2), urinary bladder mucosal hemorrhages (n = 5; Fig. 3), ascites (n = 5), hydrothorax (n = 4), hepatomegaly (n = 4), and mucosal hemorrhages in the cecum and colon (n = 2). No significant gross lesions were identified in the 7 Ibex.
Malignant catarrhal fever, red flanked duiker.
Microscopic findings in the duikers are summarized in Supplemental Table S2. A consistent finding was a dense lymphocyte infiltration and thick perivascular cuffing in multiple organs including kidneys, testicles, lungs, liver, rumen, abomasum, small intestine, pancreas, heart, small and medium-sized blood vessels, brain, spleen, and lymph nodes (Figs. 4, 5, Supplemental Figures S1–S6). Vascular and perivascular inflammation was composed predominantly of lymphocytes, a subset of which had round to irregular, bizarrely shaped, hyperchromatic to vesicular nuclei, with an occasional single, prominent nucleolus. Among the lymphocyte population, there were 1 to 3 mitotic figures per high power field, and an abundance of extracellular pyknotic and karyorrhectic cellular debris (Fig. 6). Vascular fibrinoid necrosis was a consistent finding in cases 2 to 8. No significant findings were identified in the adrenal or thyroid glands. No significant histologic lesions were identified in the ibex.
Diagnostic tests on all animals were performed at the California Animal Health and Food Safety Laboratory system and included lung and liver aerobic culture, Salmonella PCR on liver, fecal flotation, and heavy metal and selenium levels on liver. Spleen from each animal was tested for epizootic hemorrhagic disease virus, bluetongue virus, and bovine viral diarrhea virus (quantitative real-time polymerase chain reaction (qRT-PCR), and for ovine herpesvirus-2 and bovine herpesvirus-1 by conventional PCR. Kidney was tested for Leptospira spp. by qRT-PCR. No bacterial or viral pathogens were detected by these methods. No parasite eggs were observed on fecal flotation. Copper was deficient in duiker cases 2, 3, 5, 6, 7, and 8. All other liver minerals were within normal limits.
A multiplex PCR for detection and identification of 6 MCFV (OvHV-2, Ibex-MCFV, AlHV-1, AlHV-2, CpHV-2, and CpHV-3) was performed on blood (6/8) and/or fresh, frozen splenic tissue (8/8) from clinically affected duikers. 4 Only DNA from ibex-MCFV was amplified in all analyzed samples. To confirm the virus identity, blood and/or spleen samples from duikers and spleen from all 7 Ibex were evaluated by herpesvirus consensus PCR targeting a region of the herpesviral DNA polymerase gene as previously described. 20 Amplification of an expected 232 base-pair fragment was obtained from the spleen of all 7 ibex and 8 duikers, and blood from 6 duikers. PCR amplification products were subsequently cloned using a TOPO TA Cloning Kit (Invitrogen) and sequenced (Eurofins Genomics LLC). Alignment of the viral DNA segments (177 base-pair non-primer DNA sequence) from the duikers and ibexes was carried out (AlignX, Vector NTI Software, Invitrogen) and showed that all sequences were identical. The consensus viral sequence obtained in this study shared 97.7% to 100% identity to the sequences previously obtained from asymptomatic ibex (GenBank: AY212112) 11 and from ibex-associated MCF-affected bongos, 8,16 confirming ibex-MCFV as the virus causing the MCF outbreak in the duikers.
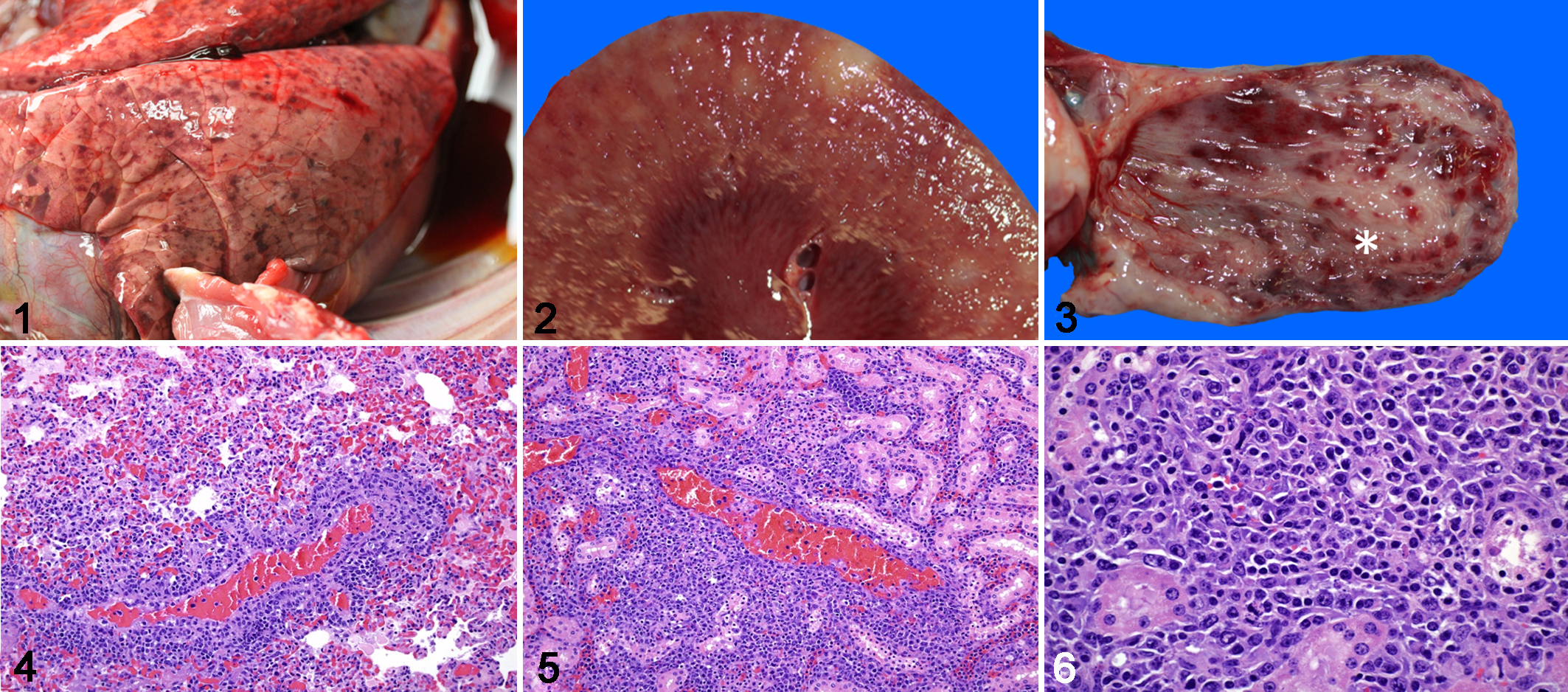
For in situ hybridization (ISH), we designed (Advanced Cell Diagnostics Cat # 519391) a 35 paired probe set targeting 693 bp of the DPOL gene and 443 bp of the orf25 gene, which encodes the herpes virus major capsid protein in the ibex-MCFV. As a reference, these correspond to regions 22440-23112 (DPOL) and 50677-51119 (orf25) of the ovine herpesvirus 2 genome (DQ198083). The probe set was designed to recognize both viral genome and transcripts. Colorimetric ISH was performed manually on 5-µm sections of formalin-fixed paraffin-embedded tissue on Superfrost Plus slides (Fisher Scientific) using the RNAscope 2.5 Red assay kit (Cat #322360, ACD) per the manufacturers’ recommendations, and as previously reported. 18 Tissues tested by in situ hybridization included the testicle and bone marrow of case 1 (Figs. 7–11). In all tissues the nuclei of a subset of lymphocytes were labelled by the ibex-MCFV probe (Figs. 8, 11). ISH-positive cells were a subset of the inflammatory cells that surrounded and/or were present within the walls of vessels, most of which were small to medium caliber arteries. Scattered ISH-positive cells were also present within the bone marrow (Fig. 11). Sequential sections of all ibex-MCFV-positive tissues were negative when probed with the OvHV-2 probe or an unrelated probe.
Malignant catarrhal fever, red flanked duiker, case 1.
A presumptive diagnosis of MCF was made for this group of duikers based on clinical, gross, and microscopic findings. The final diagnosis and evidence of causality was established by PCR followed by sequencing of amplicons, coupled with detection by ISH of viral nucleic acid present within the nuclei of intralesional lymphocytes. Sequence identity also allowed us to confirm the source of the virus as separately housed ibex.
Duiker cases 1 and 2 occurred in January and cases 3 to 8 between September and November in the same year. The reason for the temporal separation is not clear, but the disease in both groups was similar in time course so independent exposures are likely and correlated with the birth of ibex calves located approximately 35 meters away from the duiker enclosures. A detailed epidemiological study of this outbreak will be published elsewhere.
MCF epizootics have been identified in numerous collections when a susceptible species is housed in close proximity to a carrier species. Close contact is not always required for infection as indirect transmission can occur through utensils, feedstuff, personnel, and/or by synanthropic animals such as birds, unavoidably present in such settings. 1,7,12 Ibex-associated MCF has been described in a group of 3 female pregnant or postparturient bongo antelopes (Tragelaphus euryceros) cohoused with a male adult Nubian ibex (Capra nubiana) for a short period of time (5 days) in an isolation barn. 8 The proximity to a carrier animal plus stress due to late-term pregnancy and low liver copper levels were considered as potential predisposing factors in those cases. Copper deficiency is associated with increased susceptibility to infectious diseases because of a decline in super oxide dismutase and cytochrome oxidase, which provoke decreased numbers and functions of both neutrophils and lymphocytes. 15 In this study, 6 duikers also had low hepatic copper levels, which may have contributed to susceptibility, the development of the disease, or both. Ibex-associated MCF has been previously described in a bongo antelope housed across (16 m) from an exhibit containing Nubian ibex, 16 similar to the situation observed here. Direct contact between the duikers and ibex never occurred.
The clinical presentation of ibex-MCFV infection in bongos (Tragelaphus euryceros) after a short period of exposure to a Nubian ibex (Capra nubiana) bore some resemblance to that of the infected duikers in this case. 8,16 In the bongo cases, anorexia and respiratory distress was observed, with a rapid clinical course and death in 1 to 6 days, as was observed in the majority of duikers included in this report. One duiker (case 7) was found dead without premonitory signs; sudden death was not a feature of the bongo cases. Gross lesions in the bongos included mottled red to dark lungs, fibrinous pericarditis, hyperemia of small intestine, and a friable liver. In the duikers, gross lesions were more striking and included fluid in cavities, hepatomegaly, renomegaly, and vascular changes in several organs. Histologically, adventitial perivascular mononuclear infiltrates with occasional transmural inflammation and vascular necrosis were frequently observed in the lung, heart, liver, kidney, and digestive system of the bongos, 8,16 with necrotizing cholangiohepatitis and myocarditis reported as unique lesions. 8 Similar lesions were commonly seen in this series of cases in duikers (Supplemental Table S2).
Although the clinical presentation and the gross and histologic lesions of bongos and duikers were overlapping, the histologic findings in the duikers were more florid. The atypical lymphocyte population shared in all the duiker cases of MCF were so dense that lymphosarcoma was initially considered likely until multiple other animals were affected and the virus was detected. Lymphocyte infiltration can be widely variable among MCF cases, often being quite sparse (OvHV-2-induced disease, eg, in bison) or quite heavy. 17 This feature was also described in one series of AlHV-1-associated MCF cases in captive wild ruminants, in which marked lymph node enlargement with lymphoma-like features was also recognized in 10/25 animals. 2 This, and other studies, raise the hypothesis of virus-induced lymphocytic transformation. 2,10 Virus-induced lymphocyte transformation is a rare occurrence as a sequela to infection with other members of the gammaherpesvirus family, such as Epstein-Barr virus and Kaposi’s sarcoma-associated herpesvirus. 5,9 MCF affects artiodactyls, and no characterization of clonality or partial clonality in this population is currently possible.
The detection of identical viral sequences in both the duikers and ibex in this study provides strong evidence that ibex-MCFV was the cause of outbreak of MCF in the duikers.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Carvallo_et_al - Ibex-Associated Malignant Catarrhal Fever in Duikers (Cephalophus Spp)
Supplemental Material, Combined_supplemental_materials-Carvallo_et_al for Ibex-Associated Malignant Catarrhal Fever in Duikers (Cephalophus Spp) by Francisco R. Carvallo, Francisco A. Uzal, Janet D. Moore, Kenneth Jackson, Akinyi C. Nyaoke, Lisa Naples, Jordan Davis-Powell, Cynthia K. Stadler, Brandon A. Boren, Cristina Cunha, Hong Li and Patricia A. Pesavento in Veterinary Pathology
Footnotes
Acknowledgements
We thank CAHFS San Bernardino histopathology and microbiology laboratories for their technical expertise. We also thank Shirley Elias and Xiaoya Chen for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
