Abstract
Spontaneous progressive nephropathy dominated by glomerular lesions in common marmosets has been reported. However, the histopathologic characteristics, including the relationship between glomerular and tubulointerstitial lesions, have not been described in detail. In the present study, the authors examined the histopathologic characteristics of the background renal lesions in common marmosets (3 males and 9 females, 3 to 8 years old). The severity of glomerular lesions was graded into 3 classes: grade I, no alteration; grade II, hilar/focal increase of mesangial matrix; grade III, global/diffuse increase of mesangial matrix. Tubulointerstitial lesions (tubular regeneration and hyperplasia and interstitial inflammation and fibrosis) were scored according to the area of each lesion. The renal lesions were characterized by enlargement of glomeruli, expanded mesangial area with increase of periodic acid–Schiff reaction-positive matrix, tubular regeneration and hyperplasia, and interstitial inflammation and fibrosis. Glomerular lesions progressed with increasing mesangial matrix and aging. Additionally, the tubulointerstitial lesions became exacerbated with progressing glomerular lesions. Tubular hyperplasia was divided into 4 types according to the structure of the cell layer (simple or stratified-like), the area of increased lining cells (partial or entire), cytoplasmic staining (eosinophilic or basophilic), brush border and thickness of basement membrane, and the activity of cell proliferation. In conclusion, the background renal lesions in common marmosets were characterized by glomerular lesions with increase of mesangial matrix, which progressed with aging, and secondary tubulointerstitial lesions, including tubular hyperplasia. Those lesions were thus diagnosed as progressive glomerulonephropathy in common marmosets.
Spontaneous progressive nephropathy dominated by glomerular lesions in Callitrichidae, including common marmosets (Callithrix jacchus), has been reported. 2,4 The nephropathy was characterized by mesangial proliferative glomerulonephritis (increase of mesangial cells and matrix) and tubulointerstial nephritis. The pathogenesis of the disease is yet unknown, although participation of IgM and IgA has been reported. 3 The disorder begins early in life, at the infant stage, and in one study, already at 1 year of age, 70% of the callitrichids examined had been affected. 1 However, the histopathologic characteristics, including the relationship between glomerular and tubulointerstitial lesions, have not been described in detail.
Common marmosets are useful as a nonhuman primate animal model in various fields, including physiology, drug metabolism, general toxicology, and reproductive biology, because of their small size, availability, and relatively rapid generational turnover. 16 Recently, transgenic marmosets were successfully created, meaning that marmosets will draw increasing attention in biomedical research. 14 Therefore, it is important to have detailed information on background lesions in this species.
In the present study, we therefore examined the histopathologic characteristics of background renal lesions in common marmosets.
Materials and Methods
Animals
Twelve common marmosets (3 males and 9 females, 3 to 8 years old) obtained from Clea Japan, Inc. (Tokyo, Japan) or bred in-house were used in this study. The animals were housed individually in stainless steel cages placed in an animal room on a 12-hour light/dark cycle (lighting, 7
Histopathology
The kidneys were fixed in 10% neutral buffered formalin and embedded in paraffin for histopathologic examination. Hematoxylin and eosin stain, periodic acid–Schiff (PAS) reaction, Masson’s trichrome stain, and periodic acid–methenamine silver (PAM) stain were performed. For immunohistochemical (IHC) analysis, antibodies against proliferating cell nuclear antigen (PCNA, clone PC10), IgM (rabbit polyclonal, anti-human), IgA (rabbit polyclonal, anti-human), and IgG (rabbit polyclonal, anti-human) were applied as the primary antibodies (Dako, Glostrup, Denmark). IHC staining was performed according to the labeled streptavidin–biotin method with the Dako LSAB2 kit (Dako, Glostrup, Denmark). The immunoreaction was visualized by peroxidase–diaminobenzidin reaction. The sections for PCNA were finally double stained with PAS reaction to identify the tubular segments. The other sections for IgM, IgA, and IgG were finally counterstained with hematoxylin. Masson’s trichrome stain, PAM stain, Congo red stain, and IHC staining for PCNA, IgM, IgA, and IgG were performed on representative animals.
Histopathologic Evaluation of Glomerular and Tubulointerstitial Lesions
The severity of glomerular lesions was graded into 3 classes (Table 1 and Fig. 1).
Criteria for Grading Glomerular Lesions

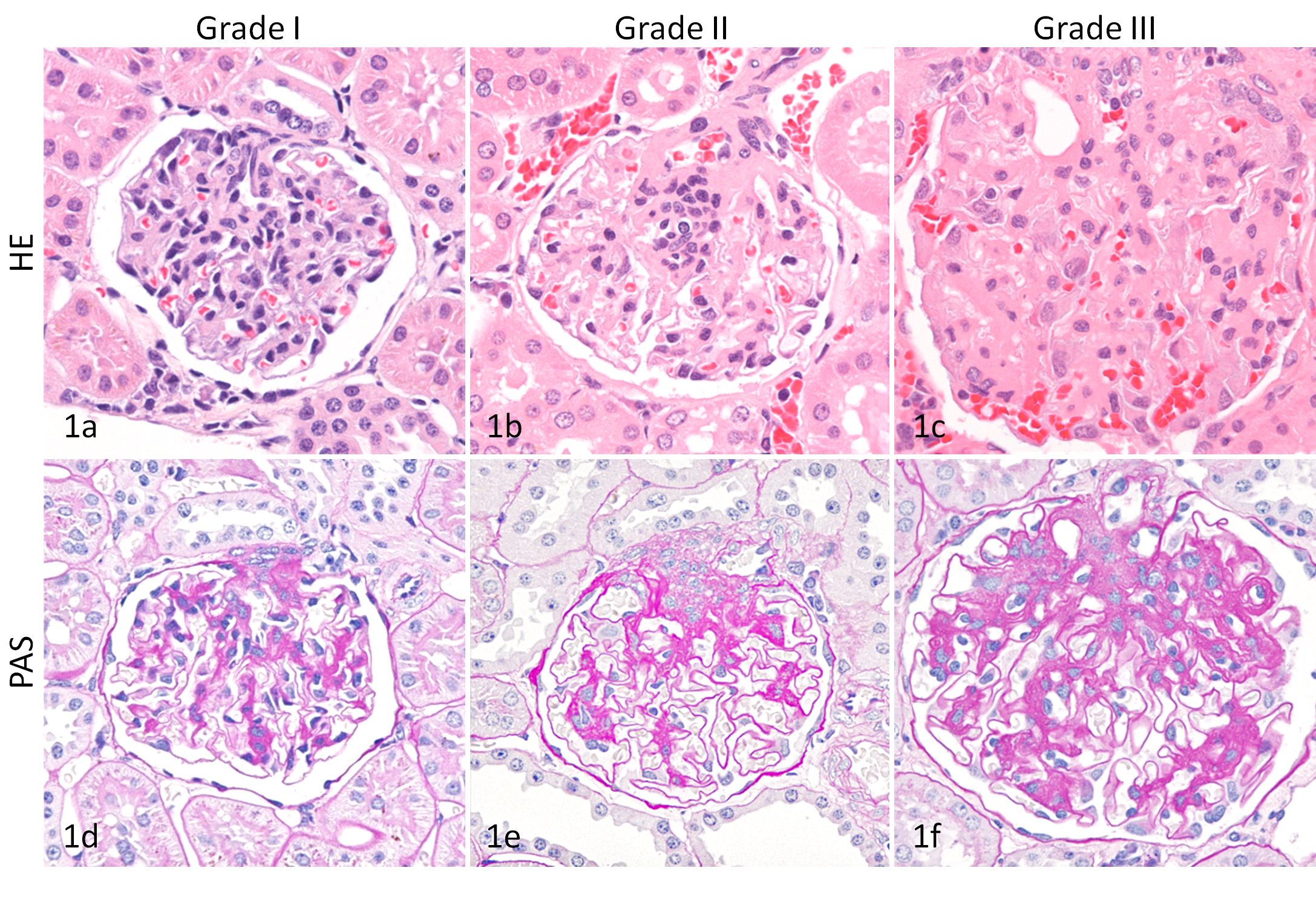
Kidney; common marmoset. (A) Grade I, no alteration in the glomerulus. HE. (B) Grade II, glomerular lesion with increase of mesangial area, predominantly in the hilar area in the glomerulus. HE. (C) Grade III, glomerular lesion with global increase of mesangial area in the glomerulus. HE. (D) Grade I, no alteration in the glomerulus. PAS reaction. (E) Grade II, glomerular lesion with increase of mesangial matrix, predominantly in the hilar area in the glomerulus. PAS reaction. (F) Grade III, glomerular lesion with global increase of mesangial matrix in the glomerulus. PAS reaction.
The area of mesangial matrix was measured as the PAS-positive area in the mesangial area. Each area of mesangial matrix was measured quantitatively by using Aperio ScanScope System with image analysis software included (Aperio, Vista, CA). The whole glomerular area was measured by tracking out the glomerular tuft. In each section, 50 glomeruli were selected at random and then evaluated. The area of mesangial matrix (%) was calculated by the following formula: (area of mesangial matrix/whole glomerular area) × 100. The individual area of mesangial matrix (%) was obtained as the average score of these glomeruli in each section.
Tubulointerstitial lesions (tubular regeneration and hyperplasia, interstitial inflammation and fibrosis) in the cortex and outer stripe of the medulla were scored according to the area of each lesion: 0, 0%; 1, 1–20%; 2, 21–40%; 3, ≥ 41%).
Statistical Analysis
The correlation between age and area of mesangial matrix was analyzed by simple regression analysis. The significance of the correlation value was tested with Pearson correlation coefficient test via Mini StatMate software (ATMS Co., Ltd., Tokyo, Japan). Difference with P values less than .05 was considered to be significant.
Results
Histopathology
Glomerular lesions
Two animals (3-year-olds) grouped into grade I showed no alteration in the glomerulus. In 4 animals (3- to 6-year-olds) grouped into grade II, increase of mesangial area predominantly in the hilar area was focally observed. In 6 animals (3- to 8-year-olds) classified as grade III, enlargement of glomeruli and global (partly segmental) and diffuse increase of mesangial area were noted (Fig. 1).
In some animals grouped into grades II and III, mesangial cell proliferation with approximately 4 to 6 cells in the peripheral mesangial area was observed with increase of mesangial matrix, while up to 3 mesangial cells in the peripheral mesangial area were noted in animals of grade I. In some animals classified as grade III, enlarged and/or polynucleated podocytes, some of which contained hyaline droplets, were also observed.
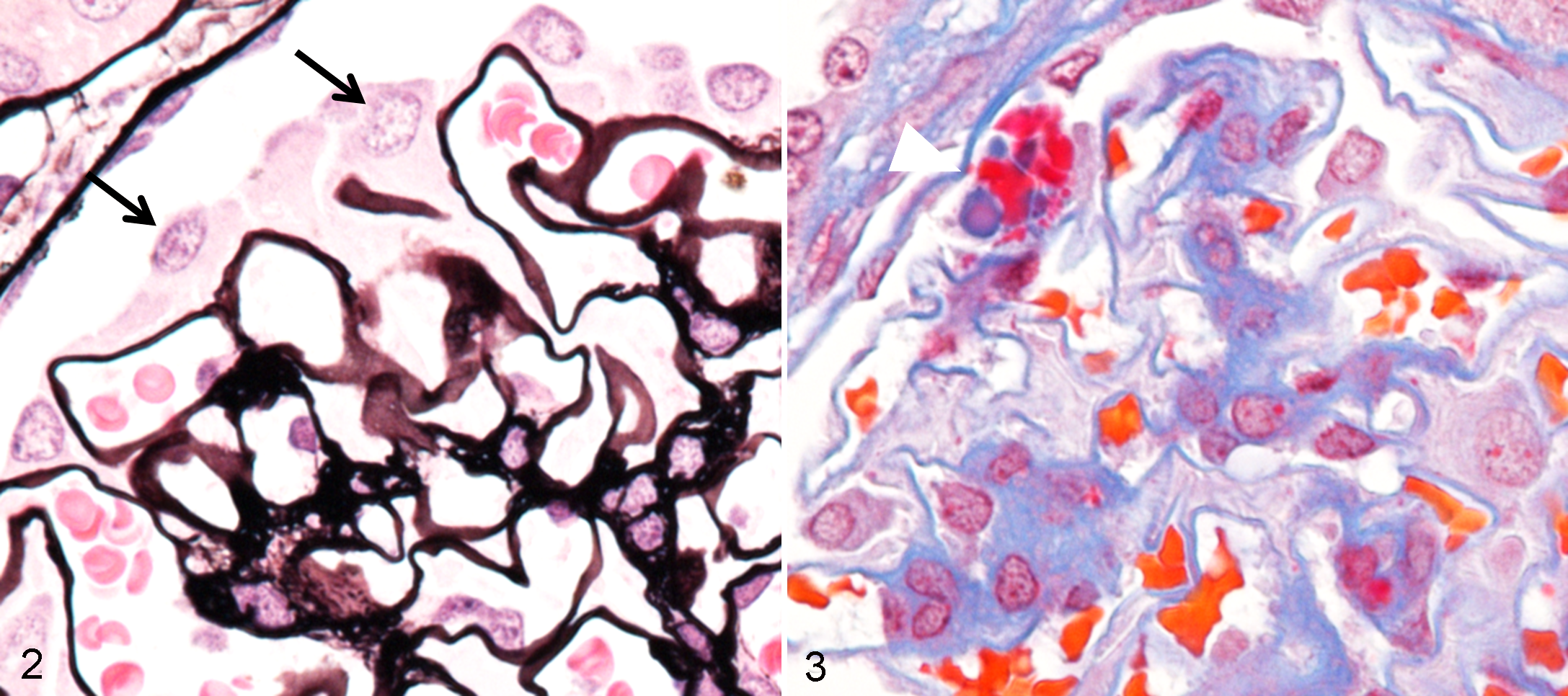
The mesangial area was stained positive for PAS reaction, negative for Congo red stain, and deep to pale blue with Masson’s trichrome stain. However, no abnormalities in the thickness of basement membrane were observed in PAS- and PAM-stained sections (Fig. 2). Although droplets of podocytes were stained red with Masson’s trichrome stain (Fig. 3), no other depositions either stained red with Masson’s trichrome stain or stained positive for IHC of immunoglobulins, including IgM, IgA, and IgG, were detected.
Tubulointerstitial lesions
Tubular degeneration (swelling and vacuolation) or necrosis, regeneration, and hyperplasia in the cortex and outer stripe of the medulla were noted according to nephron unit. Those lesions were mainly observed with accompanying interstitial inflammation and fibrosis. Hyaline casts were frequently seen in distended tubules. Inflammatory infiltrates consisted predominantly of lymphocytes, plasma cells, and some neutrophils.
Relationship Between the Glomerular Lesions and Age
A correlation between the area of mesangial matrix and age was noted (Fig. 4). In addition, the severity of areas of mesangial matrix corresponded to the glomerular grades.
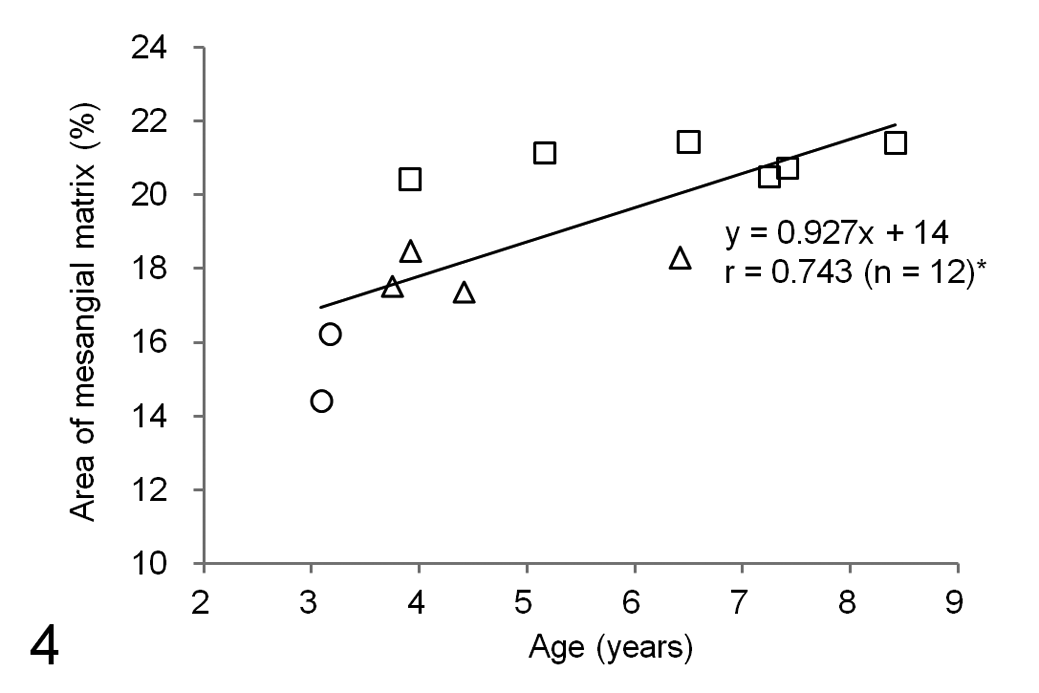
Correlation between age and the area of mesangial matrix. Each point represents the value for a single animal. The functional equation is a regression equation. r = correlation coefficient. The area of mesangial matrix corresponds to 3 glomerular grades: grade I; open circles, grade II; open triangles, grade III; open squares.
Relationship Between the Glomerular Lesions and Tubulointerstitial Lesions
A correlation between the glomerular lesions and tubulointerstitial lesions, such as tubular regeneration and hyperplasia and interstitial inflammation and fibrosis, was noted (Fig. 5). Tubular regeneration and hyperplasia and interstitial inflammation observed in animals of grade I and grade II were graded under score 2. Meanwhile, only animals of grade III showed those lesions graded as score 3. In addition, interstitial fibrosis was noted only in animals of grade III. Consequently, the tubulointerstitial lesions became conspicuous with progressing glomerular lesions.
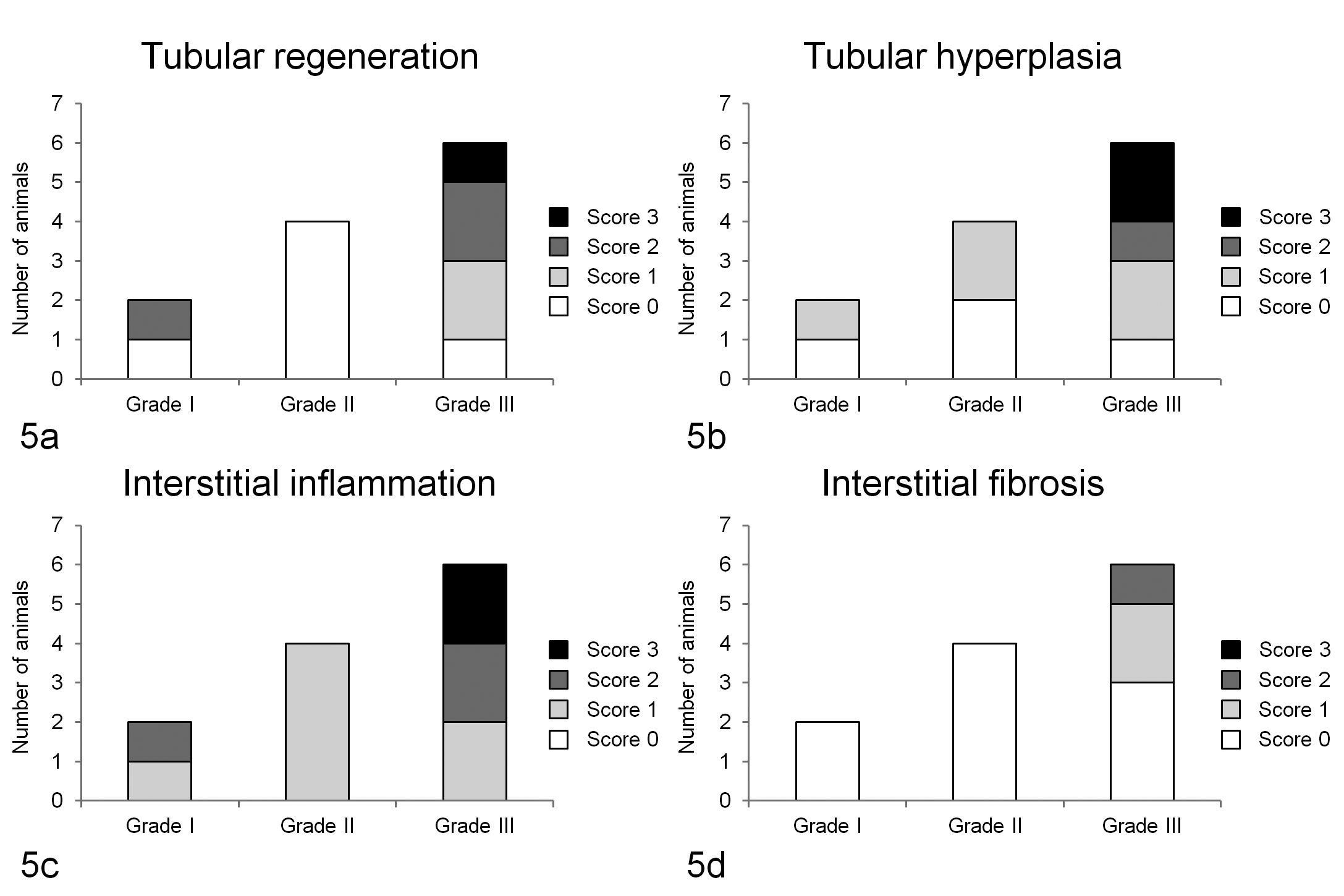
Comparison of glomerular lesions and tubulointerstitial lesions: Comparison of the severity of glomerular lesions with tubular regeneration (A), tubular hyperplasia (B), interstitial inflammation (C), and interstitial fibrosis (D).
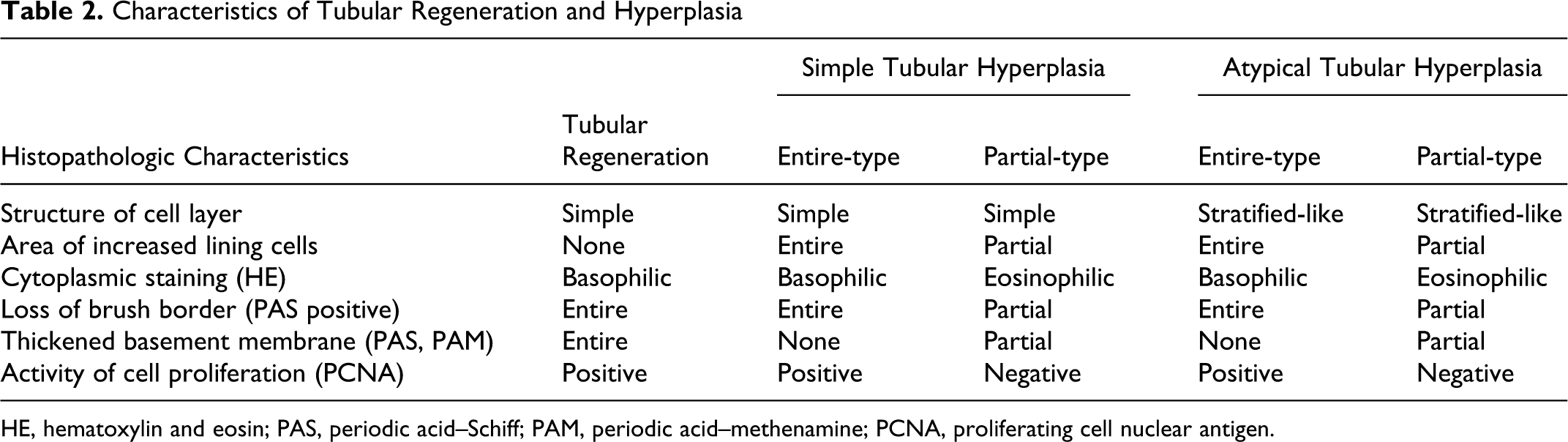
Characteristics of Tubular Regeneration and Hyperplasia
Regenerating tubules were characterized by simple epithelium, no area of increased lining cells, basophilic cytoplasm, loss of brush border, thickened basement membrane, and relatively high activity of cell proliferation (PCNA positive) (Table 2 and Fig. 6).
Characteristics of Tubular Regeneration and Hyperplasia
HE, hematoxylin and eosin; PAS, periodic acid–Schiff; PAM, periodic acid–methenamine; PCNA, proliferating cell nuclear antigen.
Tubular hyperplasia was divided into 4 types according to the structure of the cell layer (simple or stratified-like), the area of increased lining cells (partial or entire), cytoplasmic staining (eosinophilic or basophilic) with or without brush border and thickened basement membrane, and the activity of cell proliferation (PCNA positive or negative) (Table 2 and Fig. 6).
Simple tubular hyperplasia was characterized by simple epithelium and atypical tubular hyperplasia by stratified-like epithelium. The cytoplasm of entire-type tubules had basophilic cytoplasm, and affected cells entirely lined the tubular circumference with loss of brush border and normal basement membrane. Additionally, the affected cells were positive for PCNA. Meanwhile, partial-type tubules were mainly eosinophilic. Characteristically, the affected cells only partially lined the tubular circumference with thickened basement membrane and loss of brush border and were uniformly negative for PCNA.
Interestingly, degenerative changes of tubules and surrounding interstitial inflammation were more frequently observed with entire-type tubules than partial-type tubules.
Discussion
Spontaneous progressive nephropathy dominated by glomerular lesions with increase of mesangial cells and matrix in Callitrichidae has been reported. 1,2,4 In the present study, we therefore examined details of the histopathologic characteristics of the background renal lesions in common marmosets.
The glomerular lesions were characterized by increase of mesangial matrix with slight mesangial cell proliferation, which coincided with data from a previous report. 2 However, we could not detect the presence of immunoglobulins in the mesangial area by IHC, although involvement of IgM and IgA has been mentioned as a possible pathogenesis. 3 In the preliminary study, some animals were contributed to electron microscopy. As a result, thickening and hump formation in the basement membrane were noted in animals with severe glomerular lesions. Further examinations, including electron microscopy to clarify the pathogenesis of this nephropathy, are needed.
The tubulointerstitial lesions were characterized by tubular regeneration and hyperplasia and interstitial inflammation and fibrosis. Although interstitial inflammation has been previously mentioned, other findings, such as tubular hyperplasia, are reported for the first time in the present study, as far as we know. Among them, partial-type tubules were most unique in that there was no appreciable increase of PCNA positivity, and in particular, the foci of altered tubular cells were uniformly negative for this marker.
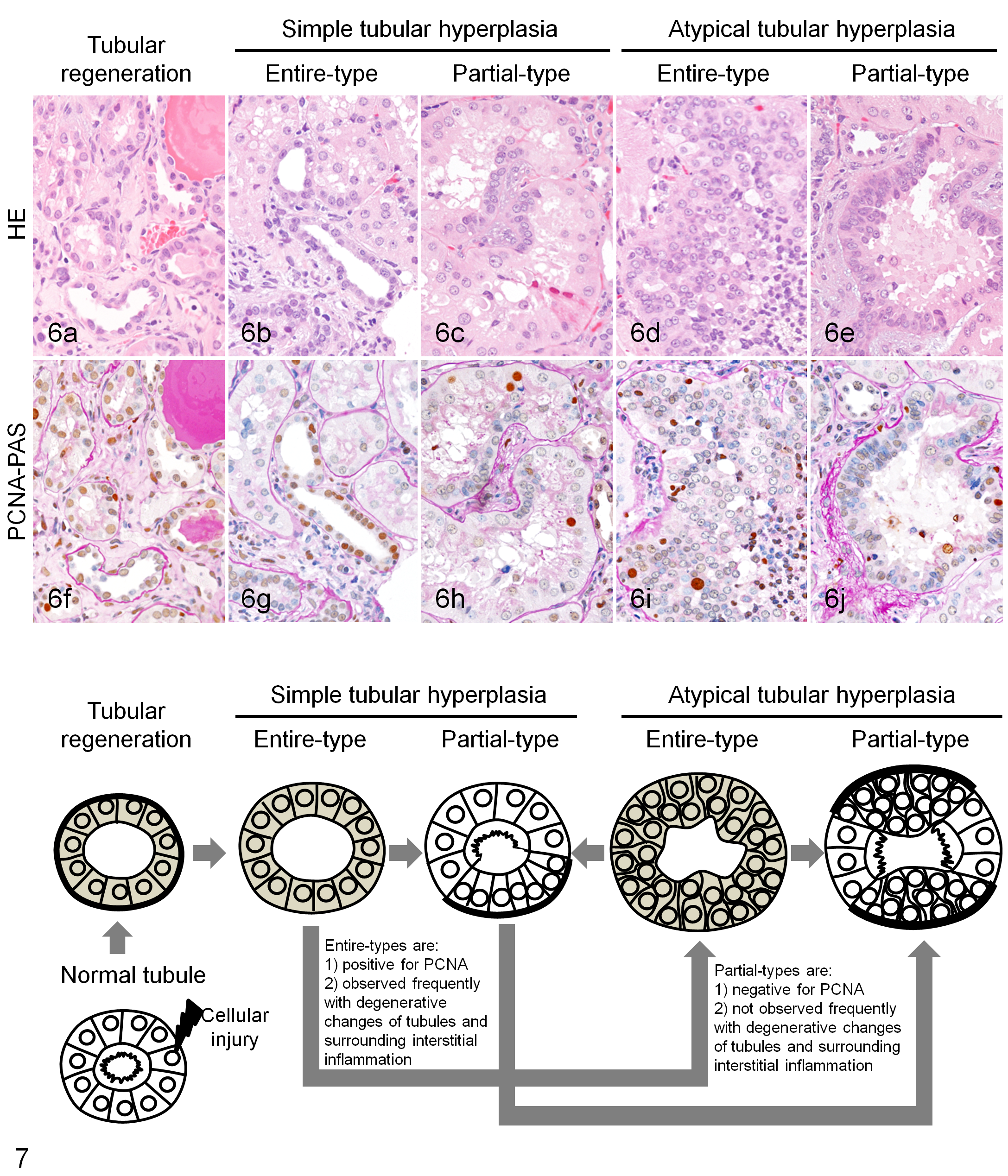
Regenerating and entire-type tubules were positive for PCNA and were observed more frequently with degenerative changes and surrounding interstitial inflammation than partial-type tubules were. These 4 types of tubules were observed mixing within nephron units and were readily discerned from the typical tubular regeneration, since the number of cells and size of regenerating tubules are not usually increased. 5,6,12 We therefore presumed that the partial-type tubules could be the result of once-proliferative tubules. In the long-standing, persistent tubule regeneration caused by chronic glomerular lesions, overproduction of cells in the form of entire-type tubules might have happened. Partial-type tubules could be derived from once-proliferative entire-type tubules. Accordingly, we considered that all 4 type tubules might be regenerative phase of simple/atypical tubular hyperplasia (Fig. 7). Although this study has not confirmed it entirely, partial-type simple tubular hyperplasia might originate from partial regeneration of tubules, caused by cellular injury to a restricted area in the circumference of the tubules. Although all 4 types of tubular hyperplasia could be diagnosed as simple or atypical tubular hyperplasia, 6,12 the results obtained in this study suggest that we should consider the possibility that all tubular hyperplasia is not “currently” proliferating when we observe renal lesions in common marmosets.
In the present study, we found that glomerular lesions with increased mesangial matrix progress with age and that the tubulointerstitial lesions become exacerbated with progressive glomerular lesions. The nephropathy thus resembles chronic progressive nephropathy (CPN) of rodents, 5,7–10 which is characterized histopathologically by glomerular lesions, including adhesion to Bowman capsule, glomerular atrophy and sclerosis, protein casts, degenerative or regenerative renal tubular lesions, and interstitial fibrosis with inflammation, although mesangial matrix proliferation and partial-type tubular hyperplasia were not previously reported, as far as we know. 8 The nephropathy in common marmosets is similar to CPN of rodents in that protein casts, degenerative or regenerative renal tubular lesions, and interstitial fibrosis with inflammation were conspicuous and some of glomerular lesions mentioned above were also present. We speculated that the pathogenesis of the secondary tubulointerstitial lesions in common marmosets was related to protein leakage caused by increased permeability of the basement membrane in glomeruli, as in CPN of rodents. 7,8 We considered that this pathogenesis is also supported by the presence of podocyte lesions, such as hyaline droplets with enlargement of the cytoplasm.
In conclusion, the background renal lesions in common marmosets were characterized by glomerular lesions with increase of mesangial matrix, which progressed with aging, and secondary tubulointerstitial lesions, including tubular hyperplasia. Those lesions were thus diagnosed as progressive glomerulonephropathy in common marmosets.
Footnotes
Acknowledgements
We would like to thank Ms Yayoi Takai at Chugai Research Institute for Medical Science, Inc. and Dr Takeshi Watanabe at Chugai Pharmaceutical Co., Ltd. for technical assistance. We would also like to thank Dr Naoaki Yamada and Dr Junko Sato at Kashima Laboratory, Mitsubishi Chemical Medience Corporation, for helpful comments on diagnosis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
