Abstract
Collagenofibrotic glomerulonephropathy (CFGN) is characterized by the deposition of type III collagen within the mesangial matrix and the absence of mesangial cell proliferation. A case of CFGN in a 2.7-year-old female cynomolgus macaque was investigated in the present study. Clinically, the animal was shown to have severe systemic edema along with hypoproteinemia. At necropsy, the kidneys were swollen and pale. The glomerular lesions were characterized by massive diffuse and global accumulation of fibrous materials in the mesangial areas. Neither mesangial cell proliferation nor changes in other organs were found. The fibrous materials were confirmed by the results of immunohistochemical and electron microscopic findings to consist mainly of randomly arranged, curve-shaped, twisted, and entwined type III collagen. This is the first case report of CFGN in nonhuman primates to date.
Keywords
Collagenofibrotic glomerulonephropathy (CFGN) in humans was first described by Arakawa et al. 1 in 1979 as glomerulonephropathy with a characteristic deposition of fibrillar materials within the mesangial and subendothelial spaces. Since 1988, when the fibrillar materials were identified as type III collagen, this disease has been termed “primary glomerular fibrosis,” 7 “collagen type III glomerulopathy,” 8 or “collageno-fibrotic glomerulonephropathy.” 2 In 1995, the disease was classified as CFGN by WHO, 17 and thereafter this disease has been well documented in humans. 5, 15
Case reports of CFGN in animals, such as glomerulosclerosis and glomerular collagen formation in the Newfoundland dog by Koeman et al. in 1994, 10 renal glomerular fibrosis in the pig by Shirota et al. in 1995 16 and in the cat by Nakamura et al. in 1996, 14 can be found. There have been no published reports concerning CFGN among nonhuman primates to date. Here we present the first case study of the morphologic and immunohistochemical characteristics of CFGN in a cynomolgus macaque.
A 2.7-year-old female cynomolgus macaque (Macaca fascicularis) imported from China was shown to have severe systemic edema and abnormal blood biochemistry indicating nephrotic syndrome (Table 1). The animal was sacrificed by exsanguination under anesthesia by ketamine because of poor prognosis resulting from the nephrotic syndrome. At necropsy, the kidneys were swollen and pale. Systemic edema in the cutaneous tissue and in the serosa of the digestive tract, as well as increased fluid content in the pericardial cavity, was noted. Other organs and tissues appeared to be normal. The animal carried antibodies for the measles and simian varicella viruses but was negative for B virus, malaria, tuberculosis, shigella, salmonella, nematode infection, and Hepatitus B virus surface antigen (HBs). The animal had no history of treatment prior to the study. The animal used in this study was treated in accordance with the ethical guidelines on animal care, handling, and termination followed at Chugai Pharmaceutical Co. Ltd. These guidelines meet the generally accepted international criteria for humane treatment, sparing the animal needless pain and suffering and ensuring that the experiments conducted are of actual scientific benefit to mankind.
Peripheral blood and serum chemistry values.∗.

∗ WBC = peripheral white blood cell counts; RBC = peripheral red blood cell counts.
† House data of the female Cynomolgus Macaque at Chugai Pharmaceutical Co., Ltd. (n = 67).
The kidneys were fixed in 20% neutral buffered formalin and embedded in paraffin for histopathologic examination. Hematoxylin and eosin (HE) stain, periodic acid-Schiff (PAS) reaction, Masson's trichrome stain, periodic acid-methenamine silver (PAM) stain, and Congo red stain were performed for all sections.
For immunohistochemical analysis, antibodies against type III collagen (rabbit antibovine type III collagen, whole serum, COSMOBIO, Tokyo, Japan, dilution 1:1000), type IV collagen (mouse antihuman collagen IV, clone CIV 22, mouse IgG1, kappa, Dako, Carpenteria, CA, USA, ready-to-use solution) and α-smooth muscle actin (α-SMA, clone ASM-1, mouse IgG2a, Progen Biotechnik GmbH, ready-to-use solution) were applied as the primary antibodies. Immunohistochemical staining was performed according to the labeled streptavidin-biotin (LSAB) method with the Dako LSAB2 kit (Dako, Carpenteria, CA, USA). The immunoreaction was visualized by peroxidase-diaminobenzidin reaction. The sections were finally counterstained with hematoxylin.
For electron microscopic examination, specimens of the renal cortex fixed in 20% neutral buffered formalin were additionally fixed in 2% glutaraldehyde, post-fixed in 1% osmium tetroxide, and embedded in epoxy resin (Epok 812, Oken Company, Tokyo, Japan). Ultrathin sections were double-stained with uranyl acetate and lead citrate and observed under an electron microscope (JEM-1010, JEOL, Tokyo, Japan).
Histopathologic changes in the glomerulus were characterized by deposition of eosinophilic substances in the expanded mesangial area and marked diffuse and global enlargement of glomeruli (Figs. 1,2). A majority of the eosinophilic substances had a homogenous appearance, but some were of a fine fibrillary and/or fine granular appearance (Fig. 3). A portion of the lesions contained eosinophilic, rough granular or amorphous plasmalike substances with red blood cells. Narrowing of the capillary lumen in glomeruli and decrease in the number of nuclei of the mesangium and/or endothelium were noted, as were thickened Bowman's capsules. The urinary tubules in the cortex were focally atrophic and dilated, with some containing proteinaceous casts. Furthermore, peritubular fibrosis, along with infiltration of lymphocytes and plasma cells in the interstitium, was observed.
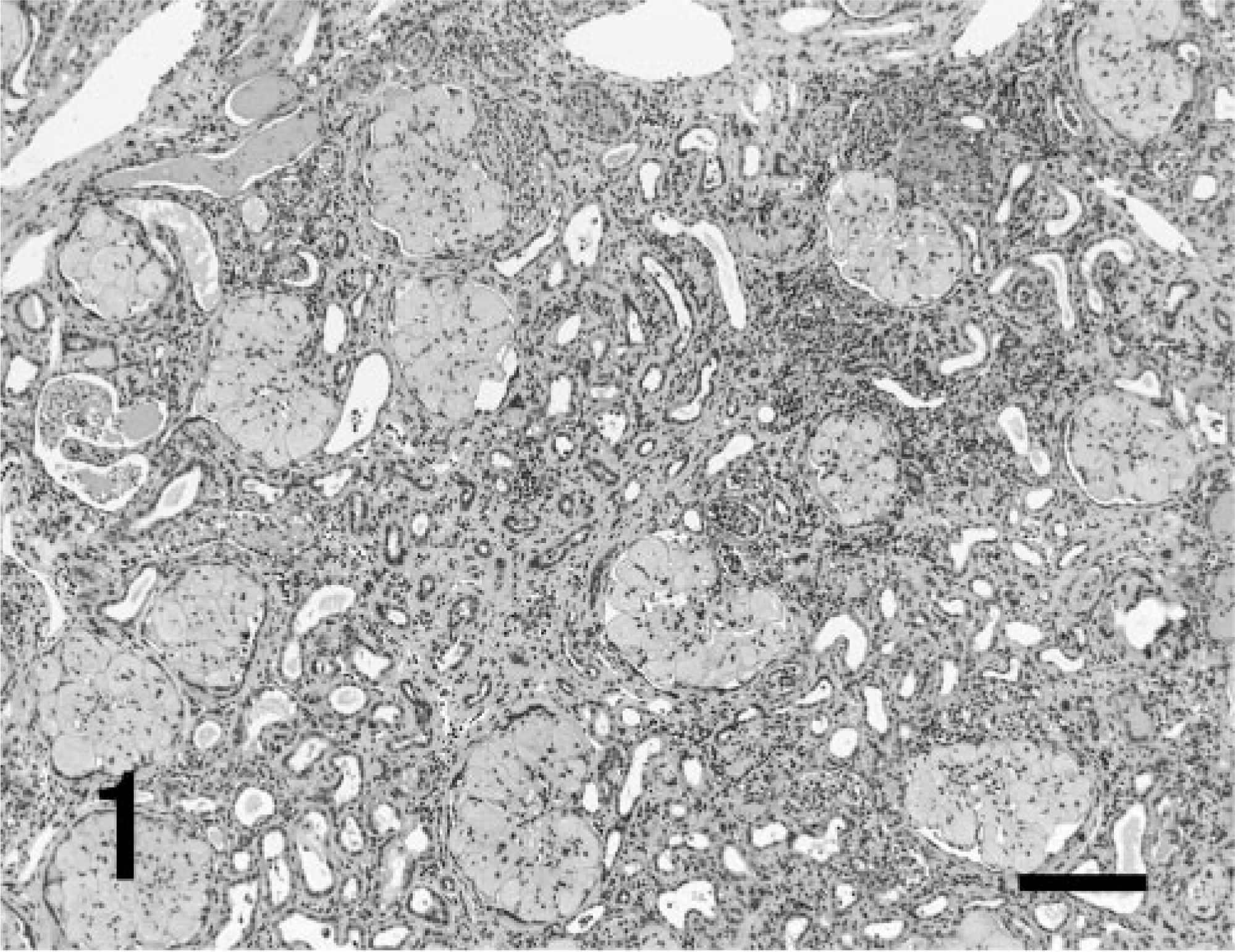
Renal cortex; cynomolgus macaque. Marked diffuse and globular enlargement of glomeruli is noted. The urinary tubules in the cortex are focally atrophic and dilated. Some of the tubules contain proteinaceous or cellular casts. Interstitial infiltration of lymphocytes and plasma cells as well as peritubular fibrosis were also observed. HE. Bar = 200 μm.
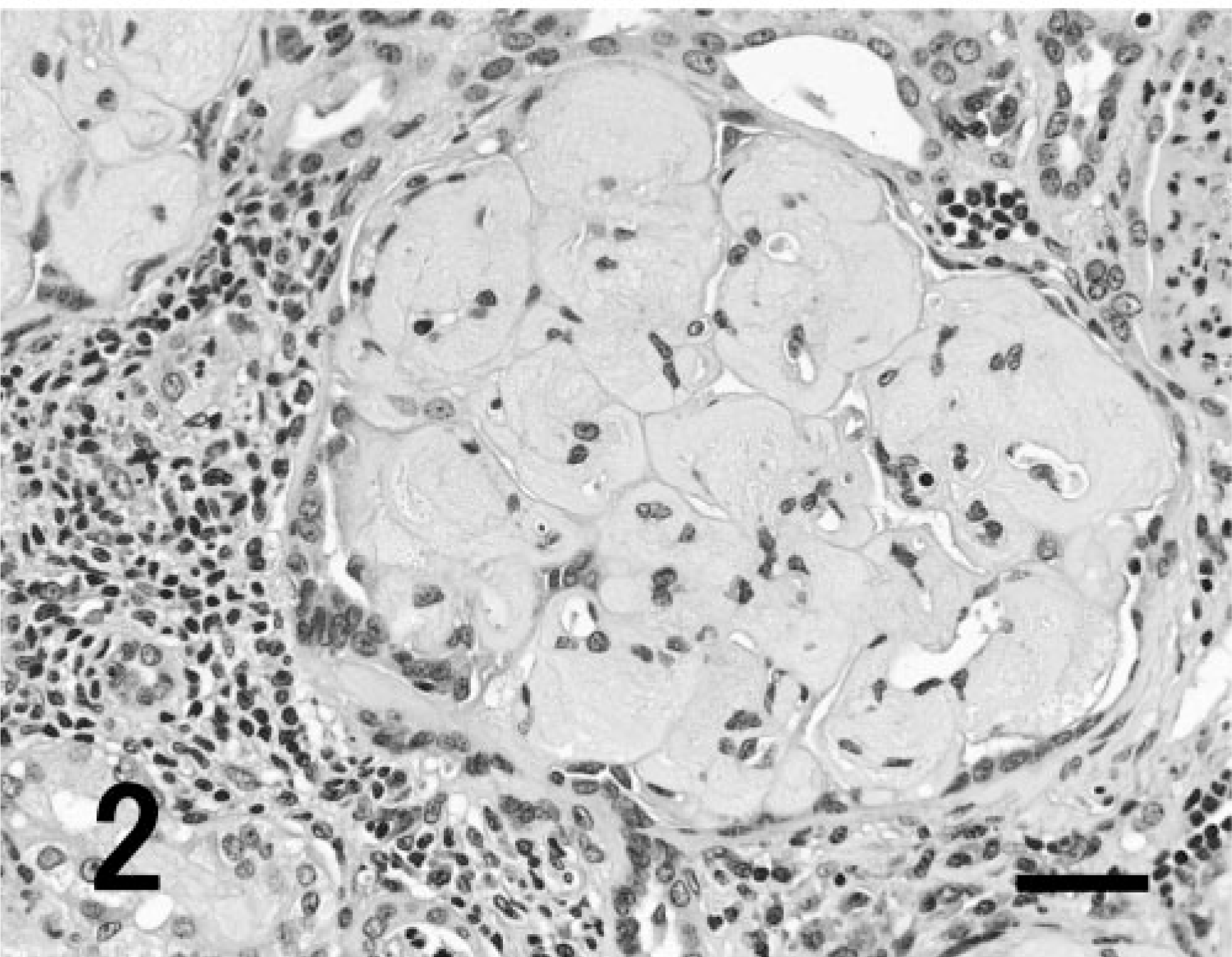
Renal glomerulus; cynomolgus macaque. Globular deposition of eosinophilic substances in the expanded mesangial area is observed. Narrowing of capillary lumen in glomerulus and a decrease in nuclearity of the mesangial area and endothelium are noted. Bowman's capsules are thickened. Interstitial infiltration of lymphocytes and plasma cells and cellular casts are also observed. HE. Bar = 50 μm.
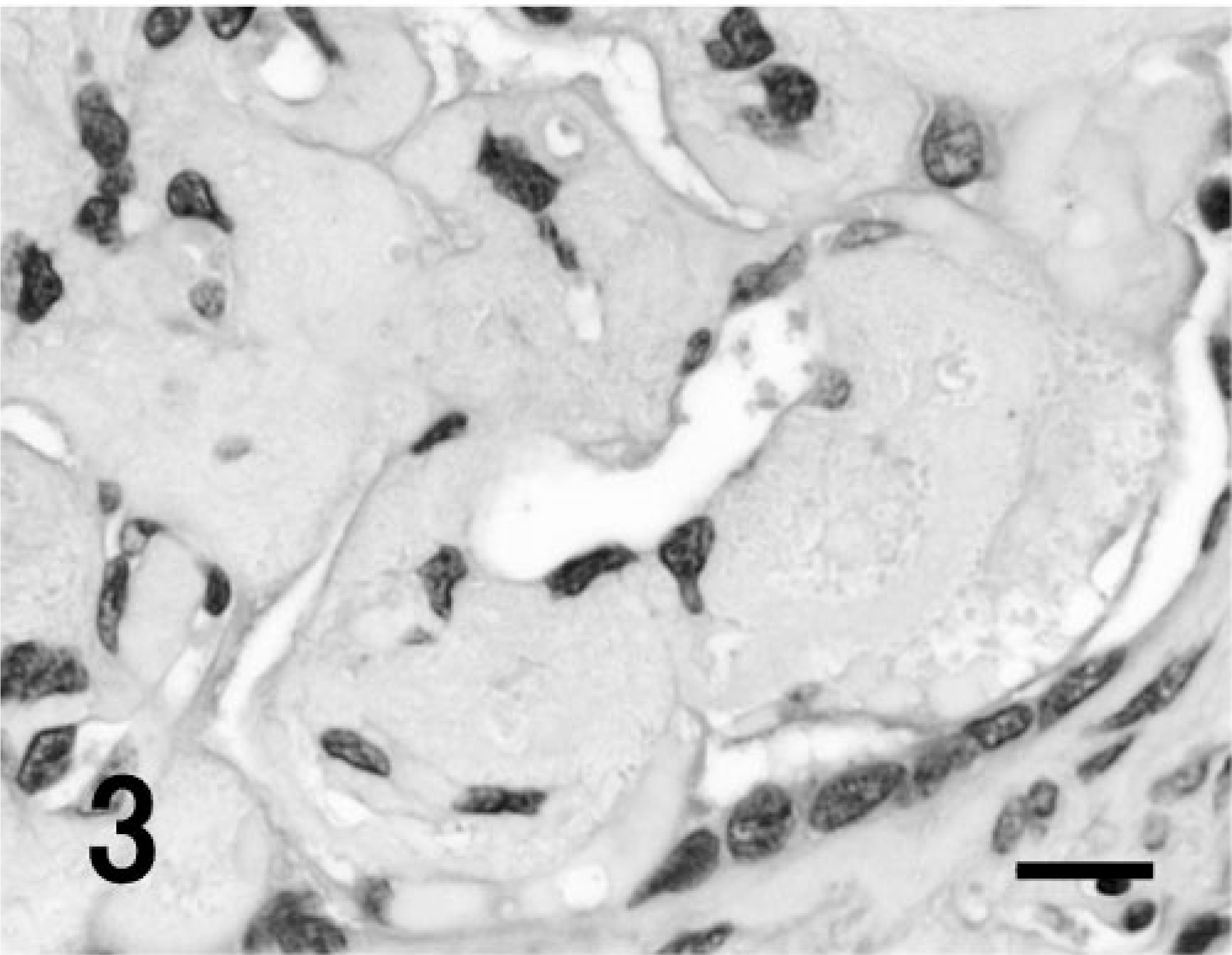
Renal glomerulus; cynomolgus macaque. A majority of the eosinophilic substances have a homogenous appearance, but some of them are fine fibrillary and/or fine or rough granular. Some parts of the lesions contain amorphous plasmalike substances with red blood cells. HE. Bar = 15 μm.
The eosinophilic substances, which were of homogenous, fine fibrillary, or fine granular appearance, were weakly positive for PAS reaction (Fig. 4), stained deep to pale blue with Masson's trichrome stain (Fig. 5), deep to pale brown with PAM stain (Fig. 6), and were negative for Congored stain (Fig. 7). The substance was positive for immunohistochemical staining of type III collagen (Fig. 8), and negative for both type IV collagen and α-SMA (Fig. 9).
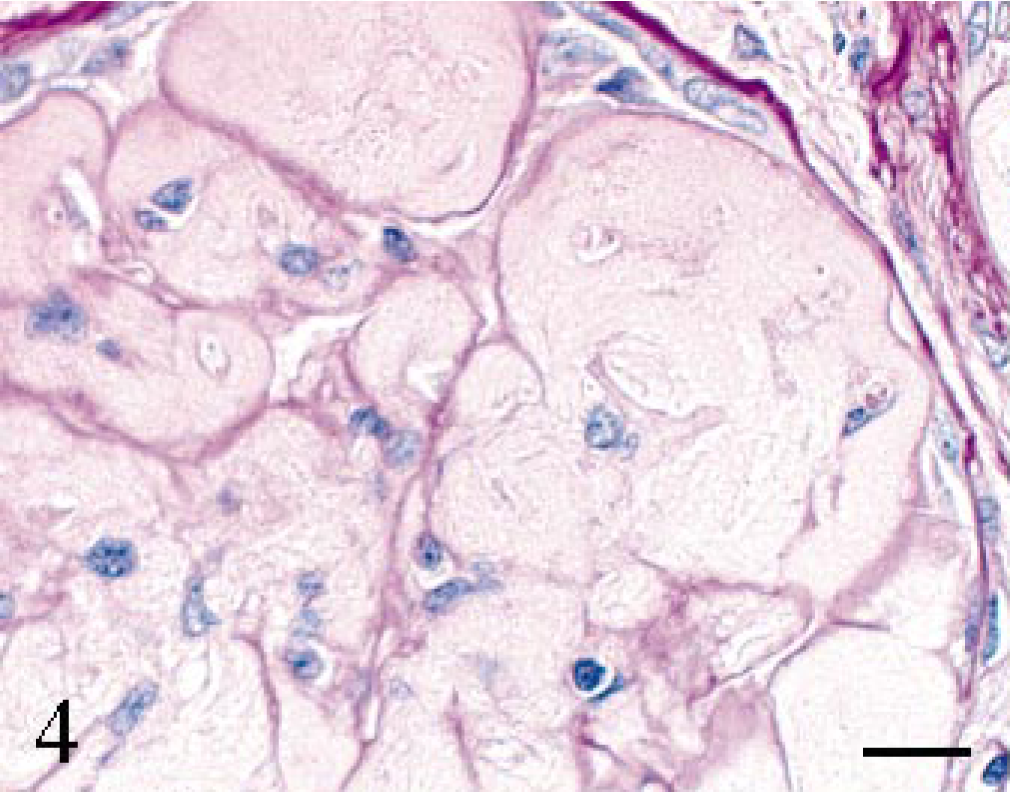
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is weak positive. PAS. Bar = 15 μm.
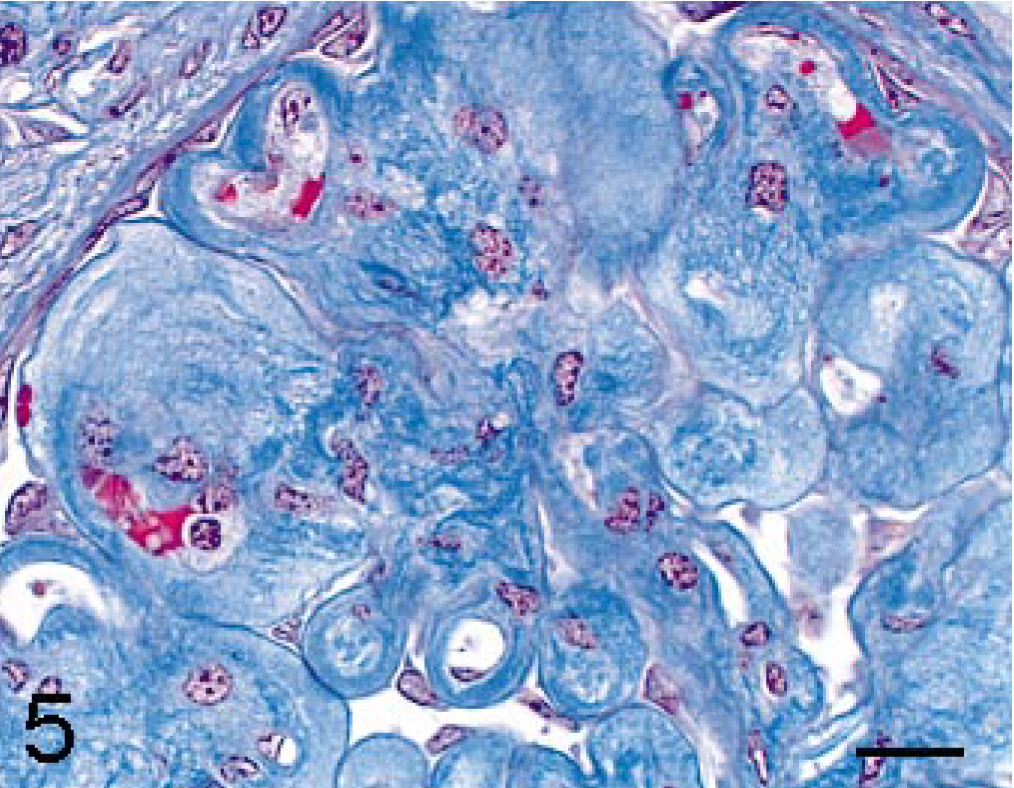
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is stained deep to pale blue. Masson's trichrome. Bar = 15 μm.
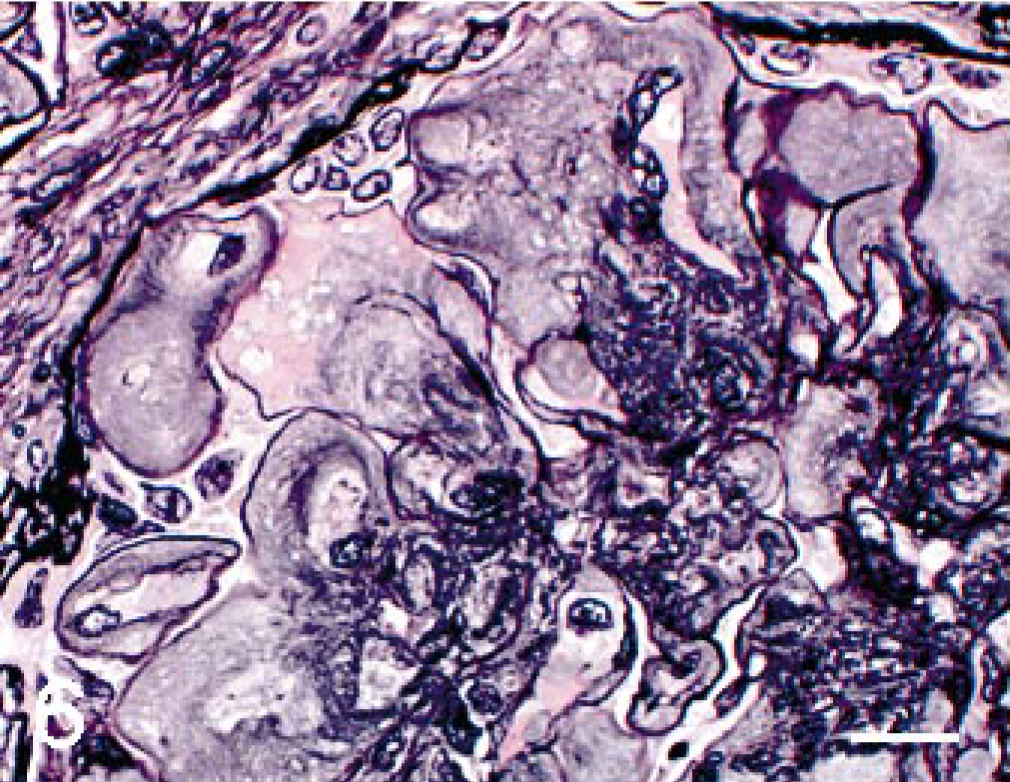
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is either stained deep to pale brown or negative. PAM. Bar = 15 μm.
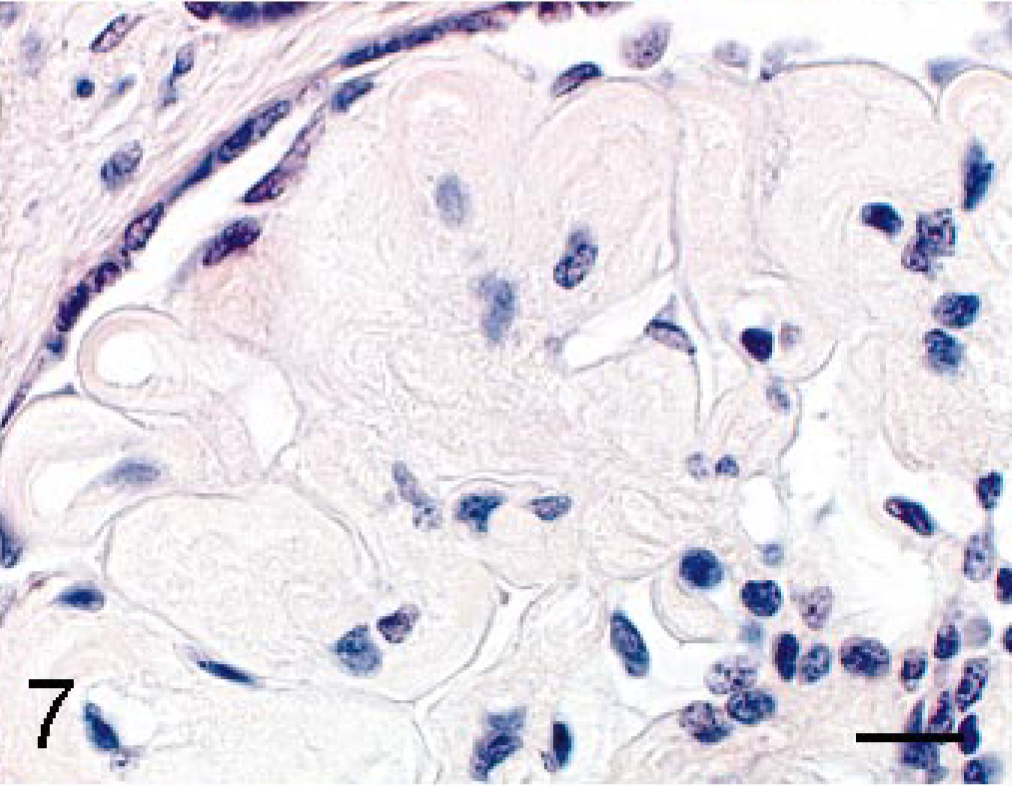
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is negative. Congo red. Bar = 15 μm.
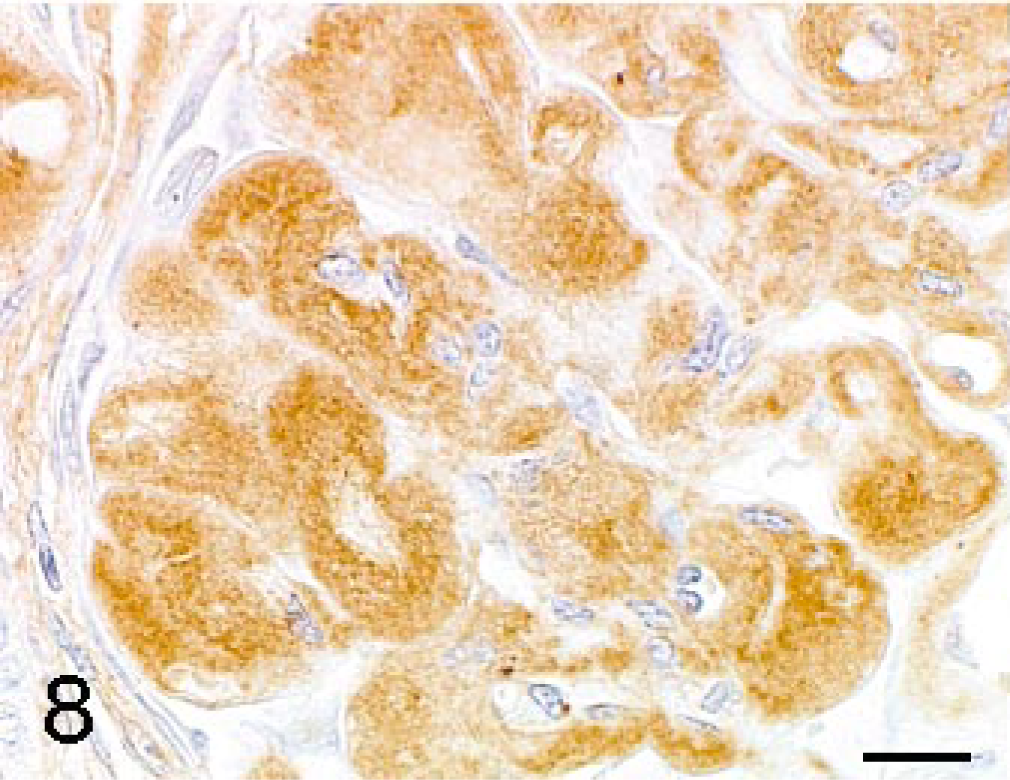
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is positive for type III collagen. Labeled streptavidin-biotin method, counterstain by hematoxylin. Bar = 15 μm.
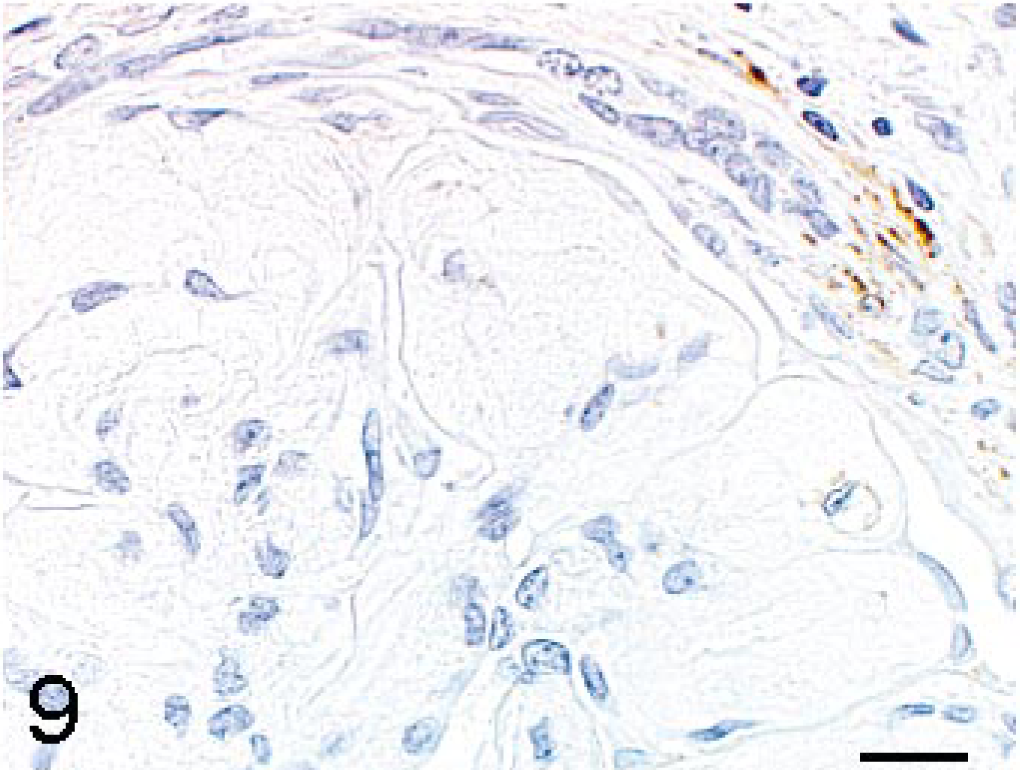
Renal glomerulus; cynomolgus macaque. The expanded mesangial area is stained negative for α-SMA. Labeled streptavidin-biotin method, counterstain by hematoxylin. Bar = 15 μm.
In addition, the rough granular or amorphous plasmalike substances were positive for PAS reaction and were stained orange or pale blue with Masson's trichrome stain. These substances were negative for PAM stain, Congored stain, type III collagen, type IV collagen and α-SMA. Type III collagen was detected in tissues surrounding regenerating urinary tubules. Positive reactions for type IV collagen or α-SMA were noted along the capillary walls of glomeruli or smooth muscle cells of the periglomerular arterioles, consistent with the control tissue.
The mesangial areas and subendothelial spaces were expanded through electron microscopy and found to have a clear or lytic appearance (Fig. 10). Within these lesions, there were various amounts of randomly arranged, curve-shaped, twisted, and entwined collagenous fibrils (Fig. 11). No low electron density lesions on the lamina densa or electron dense deposits in the mesangium or in the capillary walls were observed; however, the capillary lumens were sometimes compressed or occluded by massive deposition of fibrils in the mesangium. Epithelial cells were flattened in some parts.
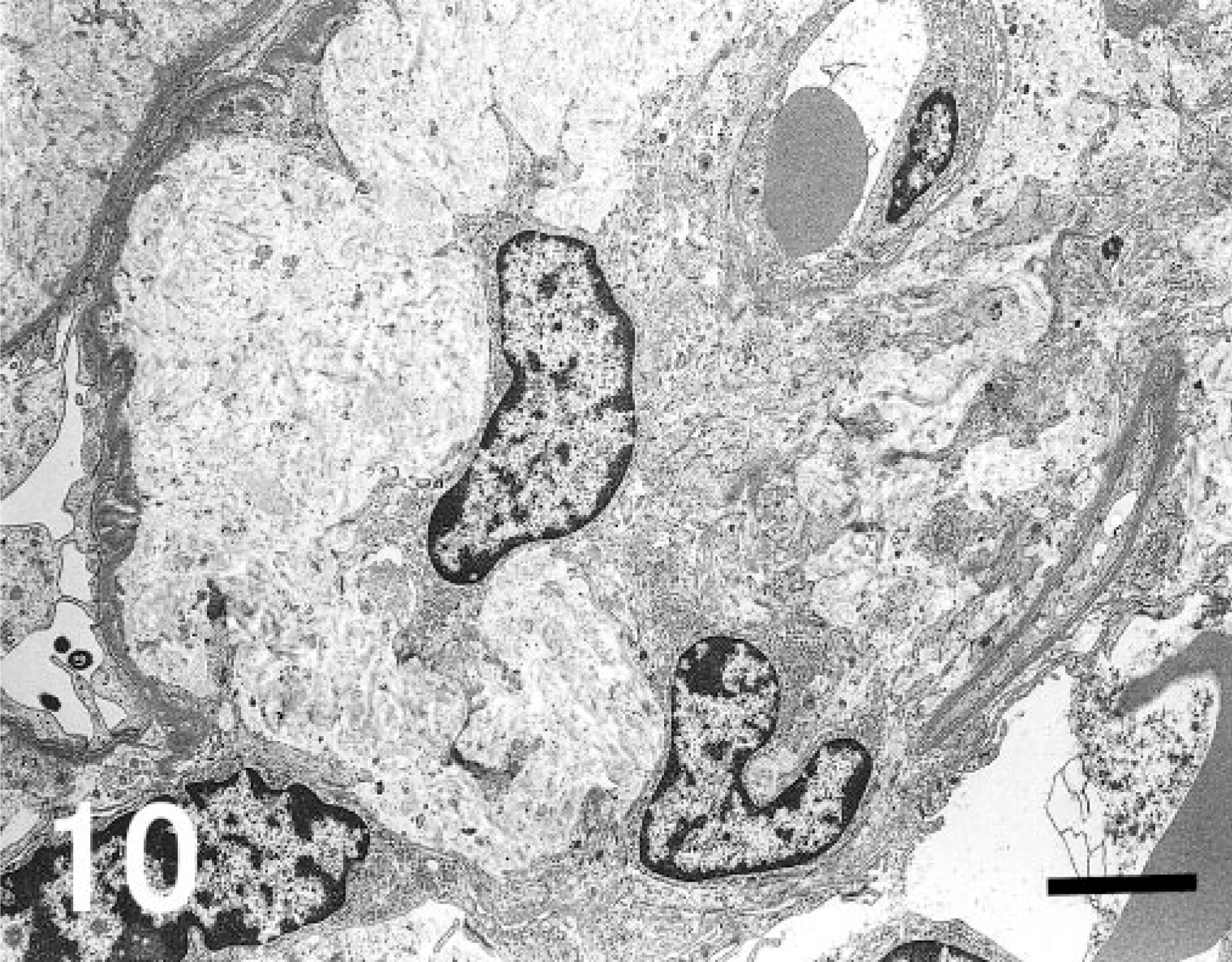
Electron micrograph. Kidney; cynomolgus macaque. The mesangial areas and subendothelial spaces are expanded and have a clear or lytic appearance. Within these lesions, there are various amounts of collagenous fibrils. The capillary lumen is compressed by massive deposition of fibrils in the mesangium. Uranyl acetate and lead citrate. Bar = 2.7 μm.
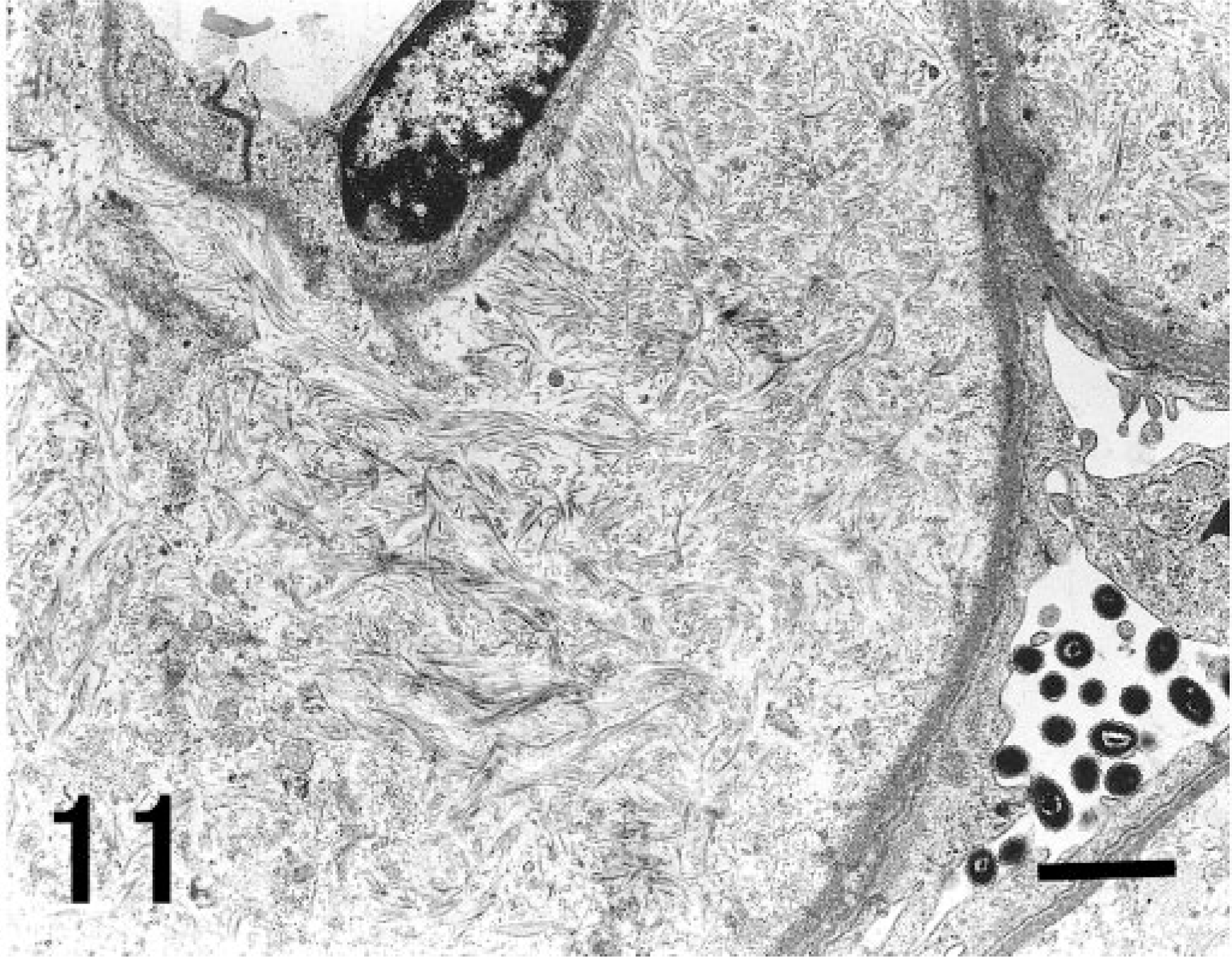
Electron micrograph. Kidney; cynomolgus macaque. Randomly arranged, curve-shaped, twisted and entwined collagenous fibrils are noted in the expanded mesangial area. Uranyl acetate and lead citrate. Bar = 1 μm.
The clinical, histopathologic, histochemical, immunohistochemical, and electron microscopic findings of the glomerular lesions in the present case closely resemble those of CFGN in humans 2, 3, 5, 15 and other species. 10, 14, 16 There are no known case reports of CFGN in nonhuman primates other than the present report.
The criteria for diagnosis of fibrillary glomerulopathy in humans is defined by ultrastructural changes that demonstrate glomerular deposits of microfibrils or microtubules. 12 Fibrillary glomerulopathy can be classified into amyloid and nonamyloid glomerulopathies by Congo red staining.
In the present case, homogenous or fine fibrillary to granular, eosinophilic substances in the glomeruli were observed in HE-stained sections under light microscopy. These findings were thought to concur with the randomly arranged, curve-shaped, twisted, and entwined type III collagen fibrils identified by immunohistochemistry and electron microscopy. In contrast, typical changes seen in amyloidosis consist of deposits in the mesangial area with a homogenous appearance in HE-stained sections and are of a fine, straight fibrillary structure with a width of 7 to 15 nm under electron microscopy. 9, 13 In addition, many cases show the deposition of amyloid in both the glomeruli and the blood vessels of the kidney and among other organs. 9, 13 Detailed light microscopic examination of the HE-stained section enabled differentiation between the changes in the present case and those in amyloidosis.
Nonamyloid glomerulopathy is classified into 2 groups by the presence of immunoglobulin-derived or nonimmunoglobulin-derived fibrils in the glomeruli. 12 The former group includes cryoglobulinemia, 3 immunotactoid glomerulopathy, 11 and fibrillary glomerulonephritis, 4 and the latter includes diabetic fibrillosis, nail-patella syndrome, 6 and CFGN. 1, 2, 5, 15 CFGN differs from the other diseases in the ultrastructural features of the fibrillar materials, such as appearance, size, and periodicity in the cross-striations. Diffuse and globular distribution of the eosinophilic deposits, as well as the absence of thickening in the glomerular basement membrane, the absence of mesangial cell proliferation, and the presence of immunoreactivity for type III collagen are also characteristic features. 5 Changes, such as uniform microtubular structures seen in immunotactoid glomerulopathy and fine, straight, randomly arranged fibrils seen in amyloidosis, were not observed. The lamina densa was of normal thickness and did not show the “moth-eaten” appearance seen in nail-patella syndrome. There was neither the electron-dense deposition characteristic of cryoglobulinemias nor the mesangial cell proliferation seen in fibrillary glomerulonephritis.
Two hypotheses for the pathogenesis of CFGN in human and other species have been proposed. 5, 15, 16 According to one, systemic alterations in collagen synthesis and/or degradation occur, releasing precursors of type III collagen into the systemic circulation. Subsequently, the serum level of procollagen III peptides is elevated and increased collagen precursors become entrapped in the glomerular basement membrane. The other hypothesis suggests that activated and transformed mesangial cells produce type III collagen in the mesangial matrix.
In the present case, eosinophilic substances in the expanded mesangial area contained not only type III collagen but also proteinaceous or plasmalike substances. Furthermore, neither mesangial cell proliferation nor activation of mesangial cells, which can be assessed by immunohistochemical staining of α-SMA, were detected. These findings indicate that the various substances observed in the expanded mesangial area were derived from blood components. However, as the serum levels of type III procollagen peptide or other blood components were not measured, the pathogenesis in this case was not clarified. Further studies of the pathologic mechanisms of fibril formation may allow a better understanding of CFGN.
Footnotes
Acknowledgements
We would like to thank Ms. Yayoi Karasawa at Chugai Pharmaceutical Co., Ltd. and Mr. Susumu Tominaga at National Defense Medical College for their expert technical assistance.
