Abstract
Chlamydia pecorum is an obligate intracellular pathogen with a wide host range including livestock such as sheep, cattle, goats, and pigs as well as wildlife species such as koalas. Chlamydial polyarthritis is an economically important disease resulting in swollen joints, lameness, stiffness, and weight loss in young sheep. In the present study, tissues from sheep experimentally or naturally infected with Chlamydia pecorum were assessed by histopathology and immunohistochemistry. Carpal, hock, and stifle joints as well as spleen, liver, kidney, lymph nodes, lung, and brain of 35 sheep from different inoculation groups were available. Two different C. pecorum strains (IPA and E58), different routes of administration (intraarticular or intravenous), UVA-irradiated IPA strain, and corresponding noninfected control groups were investigated. Similar investigations on tissues from 5 naturally infected sheep were performed. The most obvious inflammatory lesions were observed in synovial tissues and, notably, in the renal pelvis from the experimentally infected group and naturally infected animals. This resulted in chronic or chronic-active arthritis and pyelitis. Intralesional chlamydial inclusions could be demonstrated by immunohistochemistry in both tissues. Immunohistochemical evaluation of the presence and distribution of macrophages, T and B cells in synovial tissues revealed macrophages as the most prevalent inflammatory cell population. Previous observations indicated that C. pecorum isolates can infect circulating monocytes. Together with the finding of the histological lesions in synovial tissues and internal organs alongside the presence of C. pecorum DNA, these observations suggest chlamydial arthritis in lambs is the result of hematogeneous spread of C. pecorum.
Chlamydia pecorum is a gram-negative, obligate intracellular bacterium with a broad host range including livestock such as sheep, cattle, goats, and pigs as well as wildlife species like koalas. 6,17,52 Infection with C. pecorum can present with clinical manifestations such as arthritis, conjunctivitis, infertility, enteritis, endometritis, vaginitis, pneumonia, and mastitis, as well as sporadic bovine encephalomyelitis in cattle. 24,25,37,43,50,51 The most important economic manifestation of C. pecorum, mainly prevalent in Australia and New Zealand, is polyarthritis in sheep, which causes swollen joints, lameness, stiffness, and weight loss in lambs. 50 Moreover, it is hypothesized that the agent also infects the gastrointestinal tract of lambs asymptomatically. 13 In Australian sheep flocks, prevalence of gastrointestinal shedding of >30% has been reported. 53
Recent studies have used comparative genomics and molecular typing to identify differences in strain virulence. 1,23,25,41 As such, a C. pecorum–specific multilocus sequence analysis scheme utilizing 7 concatenated housekeeping gene fragments has been developed to examine the genetic diversity of and relationships between Australian sheep, cattle, and koala isolates. 25 The MLVA sequence type (ST) 23 could be found in association with ovine conjunctivitis, ovine polyarthritis, and bovine encephalomyelitis in Australian livestock. 25 It was also shown that sheep with polyarthritis had unique sequence types (eg, ST23) when compared to STs of C. pecorum strains shed in feces (eg, ST62, ST63, ST81). 1
Polyarthritis in feeder lambs due to a PLGV (psittacosis-lymphogranuloma venerum) agent was described in the United States in the 1960s. 48,49 The agent induced inflammatory changes in synovial membranes and had the ability to pass the synovia-blood barrier followed by entry into the blood stream and causing mild inflammatory reactions in internal organs. Experimentally infected sheep showed identical pathological changes to those observed in sheep recovering from polyarthritis associated with natural infection. 48 These early infection studies were recently reproduced by Australian researchers. 21 While these experimental studies 21,48,49 focused on clinical signs and gross findings due to C. pecorum infection in lambs, limited information is available on the histopathomorphological changes in infected animals. Therefore, the aim of our study was to describe histologic lesions and localization of C. pecorum antigen in joints and visceral organs, and to develop quantitative and semiquantitative scoring systems. The use of such scoring systems might standardize and better characterize the pathogenesis of C. pecorum–induced arthritis in sheep and other arthritic diseases.
Materials and Methods
Ethics Statement
This study was carried out in accordance with the New South Wales (NSW) Animal Research Act (1985) and Animal Research Regulation (2010); Australian Code for the Care and Use of Animals for Scientific Purposes. The protocol was considered and approved by the University of the Sunshine Coast Animal Ethics Committee (AN/S/18/56) and the Elizabeth Macarthur Agricultural Institute (EMAI) Animal Ethics Committee (M17/09). Animals were housed in Quarantine Containment Level 2 facilities at the Elizabeth Macarthur Agricultural Institute (EMAI) and veterinary procedures were carried out by qualified veterinarians. Naturally infected sheep were part of a project approved and monitored by Murdoch University Animal Ethics Committee (R3004/17).
Experimental Design and Sampling
The experimental design was implemented during a previous study 21 and consisted of 35 Australian Merino sheep, 6 to 7 months of age and originating from a flock without any history of arthritis or Chlamydia-related health problems. These 35 sheep were selected following 2 rounds of prescreening for Chlamydia in over 100 sheep, collecting rectal swabs for testing by C. pecorum–specific qPCR (qualitative polymerase chain reaction) as well as blood samples for complement fixation test. 4 Both tests confirmed Chlamydia-negative status in the 35 animals selected for experimental studies. The objectives of the previous study 21 were to (1) establish an infection model of chlamydial arthritis, (2) determine the optimal delivery route, (3) identify potential differences between C. pecorum isolates IPA and E58 in vivo, and (4) observe both infection and recovery phases following infection. The C. pecorum IPA isolate used in this study originated from the joint of a sheep with polyarthritis in the United States, while the E58 is also a US isolate but was retrieved from the brain of a calf with sporadic bovine encephalomyelitis. 33
All 35 sheep were weighed and randomly assigned to 4 groups: (1) IPA-group (n = 10), (2) E58-group (n = 10), (3) UV-irradiated IPA-group (n = 10), and (4) control group (n = 5). The infection groups received 107 inclusion-forming units (IFU) of C. pecorum E58 or IPA strain either into the right carpal joint via the intraarticular route (n = 5 per group) or intravenously (n = 5 per group). The control group (n = 5) received sucrose phosphate glutamate by the intraarticular route (n = 5).
After inoculation, the sheep were observed daily and a gait score 21 was evaluated to guide the appropriate time of euthanasia due to animal welfare reasons. Therefore, euthanasia, necropsy, and tissue sampling were performed at various time points (Supplemental Table S1) after the administration of the inoculum as described previously. 21 Tissues of carpal, hock, and stiftle joints and from internal organs (kidney, liver, spleen, lung, lymph node, and brain) from experimentally infected animals were collected in 10% formalin and processed as previously described. 21
In an additional study, 5 sheep, approximately 6 months of age, from a commercial farm approximately 165 km south-east of Perth, Western Australia, were assessed as having swollen joints and chronic lameness. One sheep also had evidence of conjunctivitis at the time of examination. Sheep were euthanized via intravenous barbituate overdose. There were no gross lesions in the abdominal and thoracic organs sampled during necropsy. Samples of left and right carpal joint tissue, liver, kidney, mesenteric lymph node, and lung were collected from these naturally infected animals (from the same anatomic location for each animal), fixed in 10% formalin, and processed as previously described. 21 Samples of spleen were collected for all sheep except number 4. In addition, tissues of the right stifle joint, the left conjunctiva, and third eyelid from animal 1 were available.
In total, 312 formalin-fixed and paraffin-embedded (FFPE) tissues from 35 experimentally inoculated sheep and 5 naturally infected sheep were included in this study. Hematoxylin and eosin–stained slides were prepared from all tissues according to standard procedures.
To detect chlamydial DNA, qPCR 26 was performed on the joint tissues of both experimentally and naturally infected animals (sterile dry swabs from the synovia of the carpus) and the organs of only the experimentally infected animals. Positive qPCR results were listed per group, organ, or joint and were compared against each other considering copies per microliter.
Joints
From experimentally infected animals, a total of 32 histologic sections of carpal, hock, and stifle joint synovium were examined (Supplemental Table S2). From naturally infected animals (n = 5), a total of 12 slides from carpal, hock, and stifle joints (10/12 carpal joints and 2/12 stifle joints) were investigated (Supplemental Table S3). The assessment was performed by 2 pathologists in a blinded manner (at a magnification of 100× and 400×) by using a light microscope and pathological diagnoses were elaborated.
Five main microscopic findings, focusing on inflammation, were identified and assessed qualitatively: the presence of inflammation, the type of inflammation, the degree and distribution of inflammation, and whether the surrounding tissue was affected or not (Fig. 1). Then, inflammatory synovial lesions were evaluated according to semiquantitative parameters resulting in a newly developed scoring system with a maximum possible score of 48 for each animal (Table 1). In addition to parameters described in Figure 1, the scoring system also included important findings such as the presence of fibrin and granulation tissue and positive immunolabeling for Chlamydiaceae. Furthermore, the synovia was evaluated according to criteria previously described 10,15,30 and total scores per experimental group were evaluated.
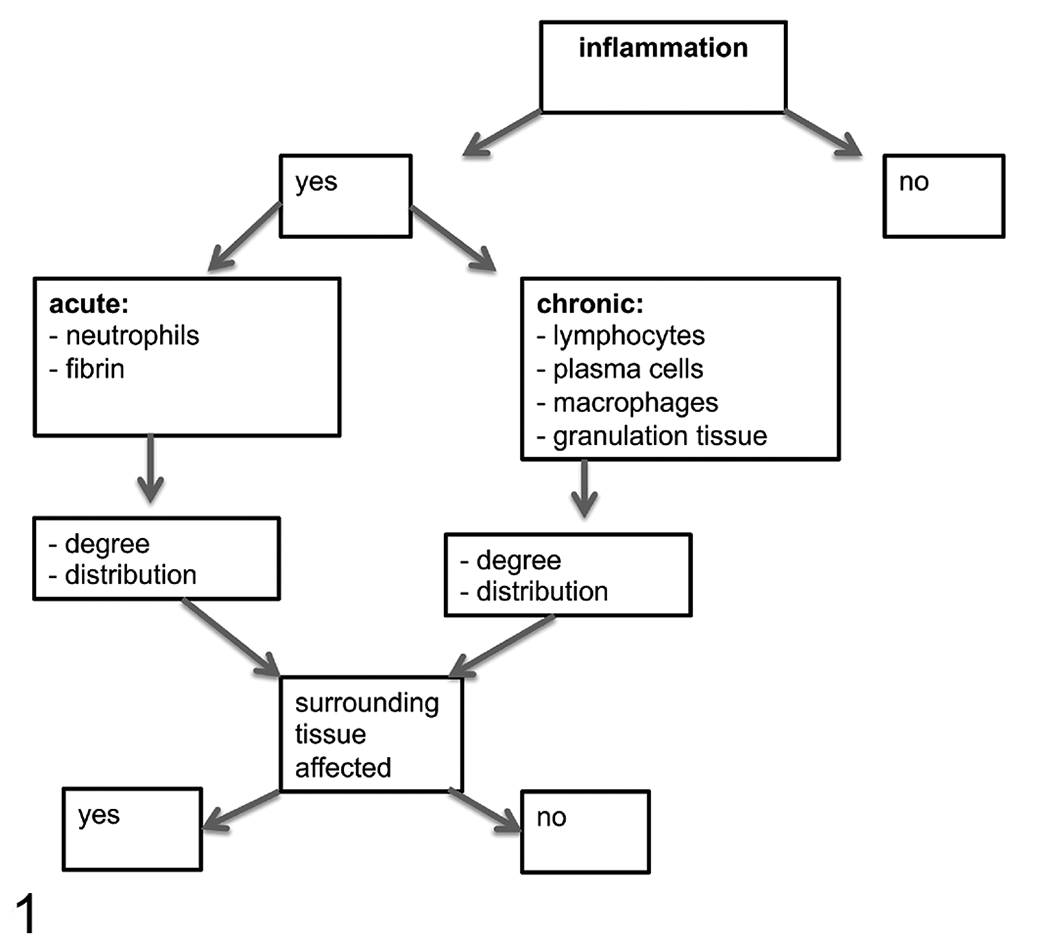
Flow chart for assessment of joint inflammation.
Semiquantitative System for Histological Scoring of Joint Lesions.
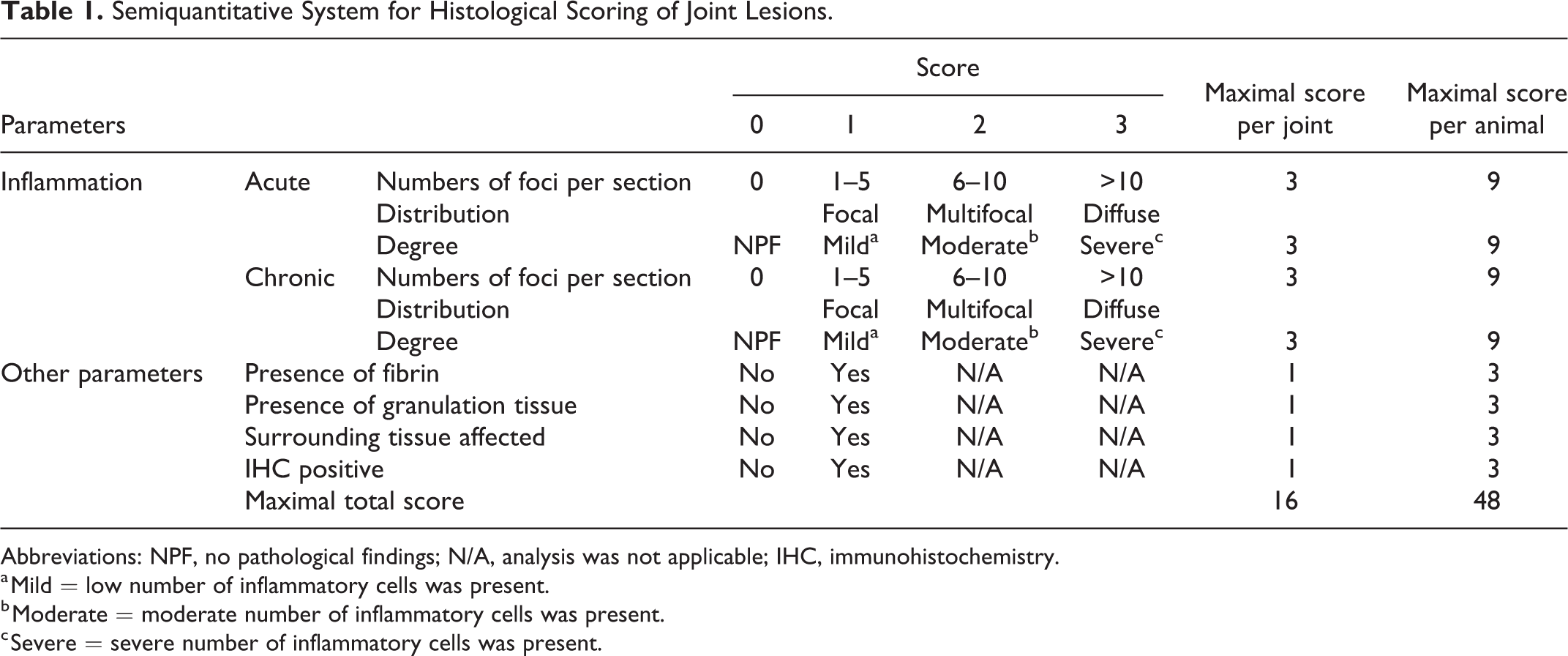
Abbreviations: NPF, no pathological findings; N/A, analysis was not applicable; IHC, immunohistochemistry.
a Mild = low number of inflammatory cells was present.
b Moderate = moderate number of inflammatory cells was present.
c Severe = severe number of inflammatory cells was present.
Internal Organs
From experimentally infected animals, a total of 72 slides from internal organs (liver, kidney, brain, spleen, lymph node, and lung) were examined. From naturally infected animals (n = 5), a total of 5 slides were examined from internal organs while conjunctival tissues were investigated from only one animal. The assessment was performed by 2 pathologists in a blinded manner as described above (Supplemental Table S4). Four main microscopic findings of inflammation were identified and assessed qualitatively: the presence of inflammation, and the type, degree, and the distribution of inflammation (Fig. 2). Inflammatory lesions of internal organs were also evaluated according to semiquantitative parameters, resulting in a newly developed scoring system with a maximum possible score of 39 for each animal (Table 2). In addition to the parameters described in Figure 2, the scoring system also included the presence of positive immunolabeling for Chlamydiaceae.
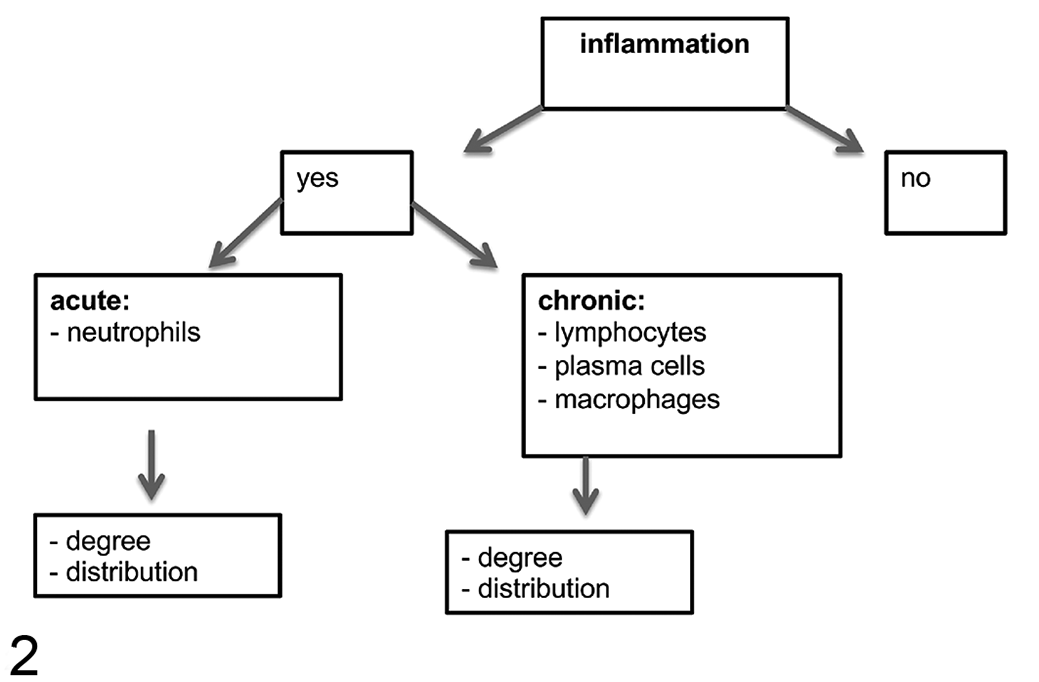
Flow chart for assessment of inflammation in internal organs.
Semiquantitative System for Histological Scoring of Internal Organ Lesions (Brain, Kidney, Liver).
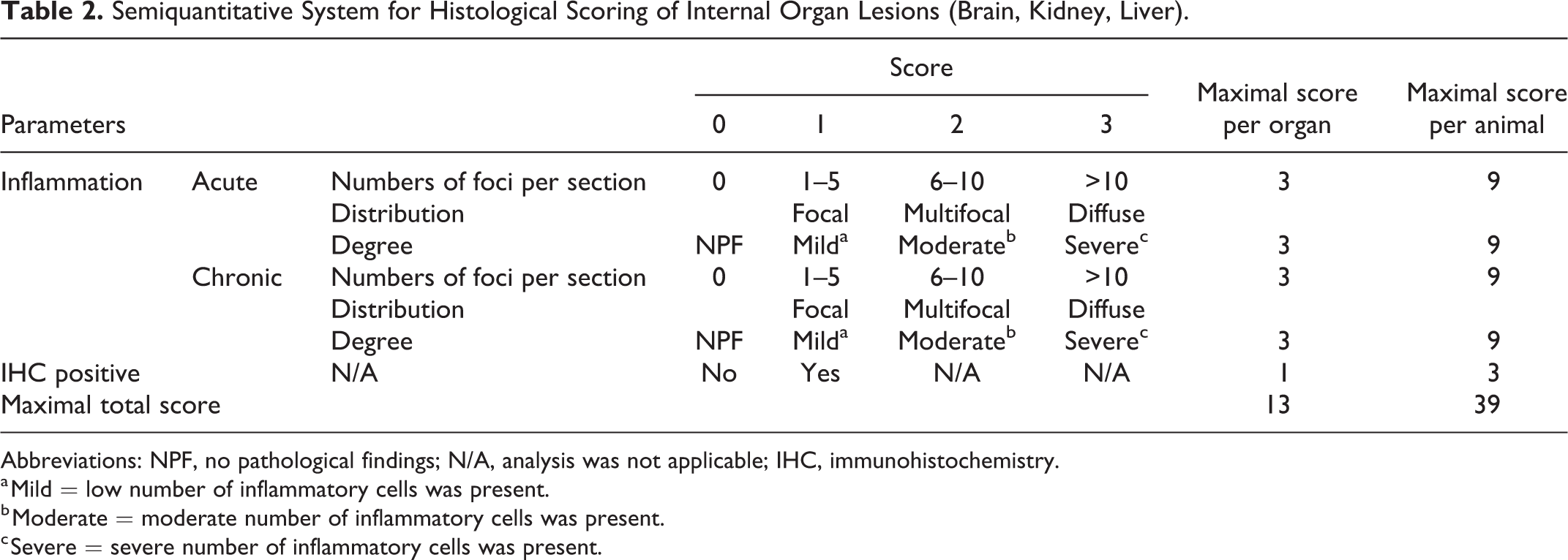
Abbreviations: NPF, no pathological findings; N/A, analysis was not applicable; IHC, immunohistochemistry.
a Mild = low number of inflammatory cells was present.
b Moderate = moderate number of inflammatory cells was present.
c Severe = severe number of inflammatory cells was present.
For lung tissues, high-resolution digital scans were acquired using the Hamamatsu Photonics’s NanoZoomer HT2.0 (Hamamatsu). All bronchi and bronchioles in each lung section were measured. For those with a minimal diameter of 200 μm, peribronchial lymph follicles (representing BALT hyperplasia) and peribronchiolar lymphocyte accumulations were assessed.
Lymph nodes and spleen were analyzed for the presence of hyperplasia as previously described. 3 The area of the whole tissue on each section was measured, the number of follicles were counted at a magnification of 100×, and assessed semiquantitatively using a newly developed scoring system (0 = 10, 1 = >20, 2 = >50 lymph follicles per section; and 0 = 1–4, 1 = 5–10, 2 = >10 secondary follicles with germinal centers). For lymph nodes and spleen, a minimum of 5 randomly chosen slides for each group was examined in a blinded manner. All tissue assessments were performed according to the targeted masked review, as previously described. 31
Immunohistochemistry
Immunohistochemistry (IHC) to detect chlamydial antigen in tissues of joints and internal organs of experimentally infected, naturally infected and control animals was performed as previously described. 21 A total of 312 FFPE tissues were examined using a light microscope at a magnification of 100× and 400×. A positive immunohistochemistry signal was included in the scoring system (Supplemental Tables S2, S3, and S4, Tables 1 and 2). IHC was considered positive if an intracytoplasmic granular labeling in at least one cell (organ-specific) was present.
Histologic sections of synovial tissue (n = 45) were reevaluated for the presence and percentage of inflammatory cells (macrophages, B and T cells) by immunohistochemical labeling to detect Iba1 (macrophages), CD20 (B cells), and CD3 (T cells) antigens. Briefly, the sections were mounted on positively charged slides and dried overnight at 37 °C. After deparaffinization, different antigen demasking protocols for each antibody were applied (Table 3). For the semiquantitative evaluation, areas with inflammatory infiltrations were first selected in HE stained slides at 100× magnification. Then, the percentage of IHC-positive labelled cells (ranging from 0% to 100%, in 5% steps) was counted in 5 randomly selected regions in corresponding IHC slides for Iba1, CD20, and CD3 at 400× magnification.
Antibodies Used for Immunophenotyping of Macrophages, B and T Lymphocytes in Joints.

a Basic buffer heat.
b Envision mouse antibody.
c Envision rabbit antibody.
d Citrate buffer heat
Statistical Analysis
Statistical analyses were performed on data from experimentally infected animals and the corresponding control group using the software R 39 version R3.6.3. Generalized mixed effect models with a Poisson distribution were performed using the lme4 package to evaluate the results of histological assessment of synovial and internal organ lesions using the newly developed scoring system. 2 Because liver lesions were only mild and also present in control and UV-irradiated groups, we can assume that they were not related to the C. pecorum infection and were therefore excluded from the statistical evaluation. As fixed effects, group and joints or organs, and as random effects, animals were considered. To adjust for multiple comparisons, Dunnett’s or Tukey’s approach containing the package multcomp 19 were chosen.
In lung tissues, bronchioles with a minimal diameter of 200 micrometer were assessed for peribronchiolar lymphocytic accumulation and lymph follicles. Lymph nodes and spleen were analyzed for the presence of hyperplasia by the assessment of lymph follicles as well as secondary follicles using a newly developed scoring system in consideration of the area-size of each tissue section. Linear models were used for the evaluation of lung tissues and tissues of spleen and lymph nodes. The measurements of the immunohistochemical markers (CD3, CD20, Iba1) were dichotomized into absence and presence and the proportion of present biomarkers was described with Wilson’s binomial 95% confidence intervals using the package, Desctools. 45 Generalized estimation equations were used to assess if these markers differed between the experimental groups and locations by using the package, geepack. 18 The qPCR measurements in the joints and organs were analyzed with mixed Poisson and zero-inflated Poisson models using the packages lme42 and glmmTMB. 8
Results
Histologic Findings in Synovial Tissues
Synovial tissues of animals from control and IPA UV-irradiated groups did not show any signs of inflammation. In joints of experimentally infected animals, the arthritis/synovitis were most often chronic (lymphoplasmacytic and histiocytic including granulation tissue; Figs. 3, 4), followed by chronic-active (lymphoplasmacytic and histiocytic as well as purulent including fibrin accumulation; Figs. 5, 6), and less frequently acute arthritis/synovitis (purulent inflammation including fibrin accumulation, without lymphoplasmacytic or histiocytic inflammation). According to the semiquantitative parameters in the scoring system, the most severe histological joint lesions were present in the IPA iv (intravenous) group, followed by the E58 ia (intraarticular) group. Moderate inflammatory changes were present in the IPA ia group, followed by mild lesions in the joints of the E58 iv group (Table 4).
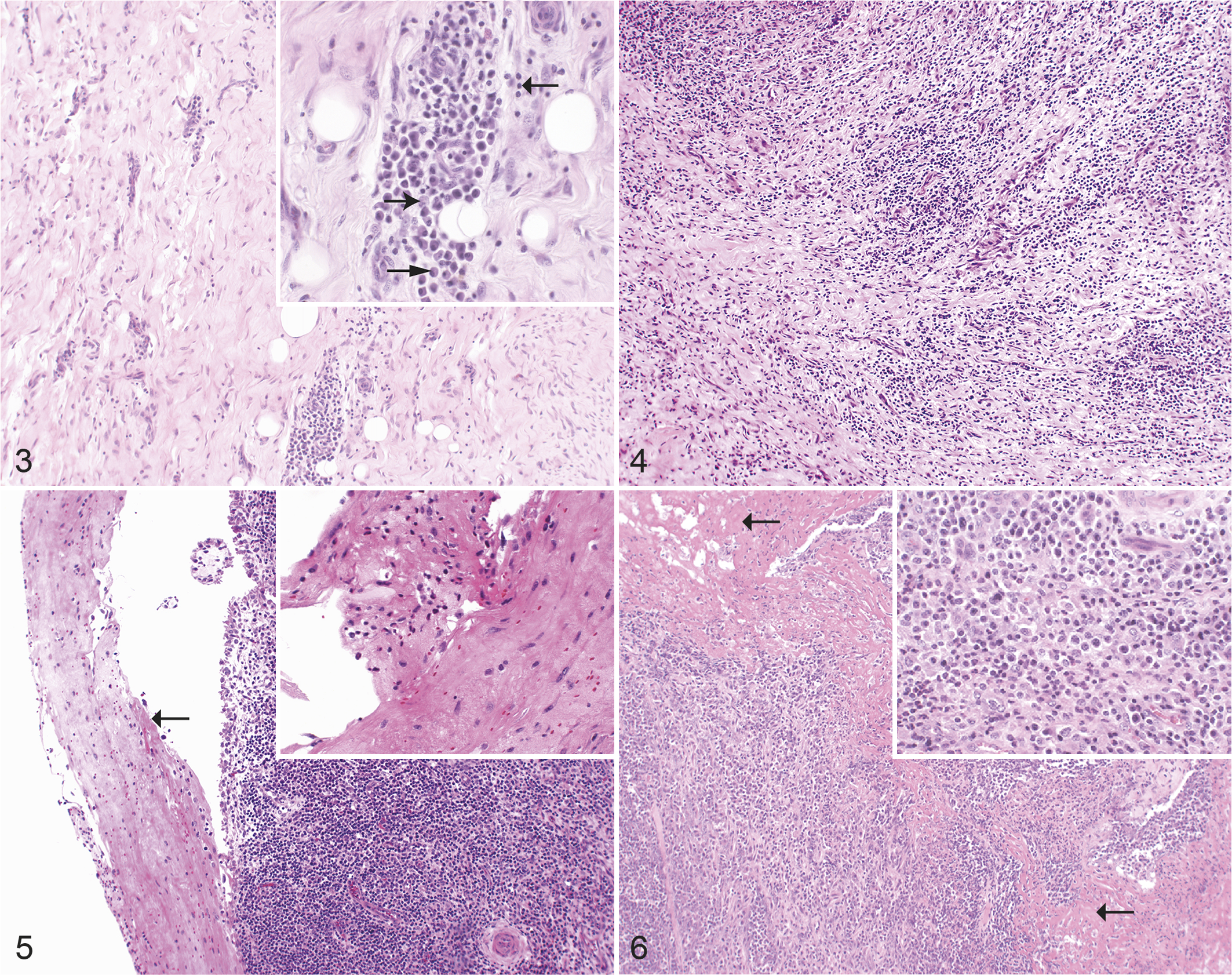
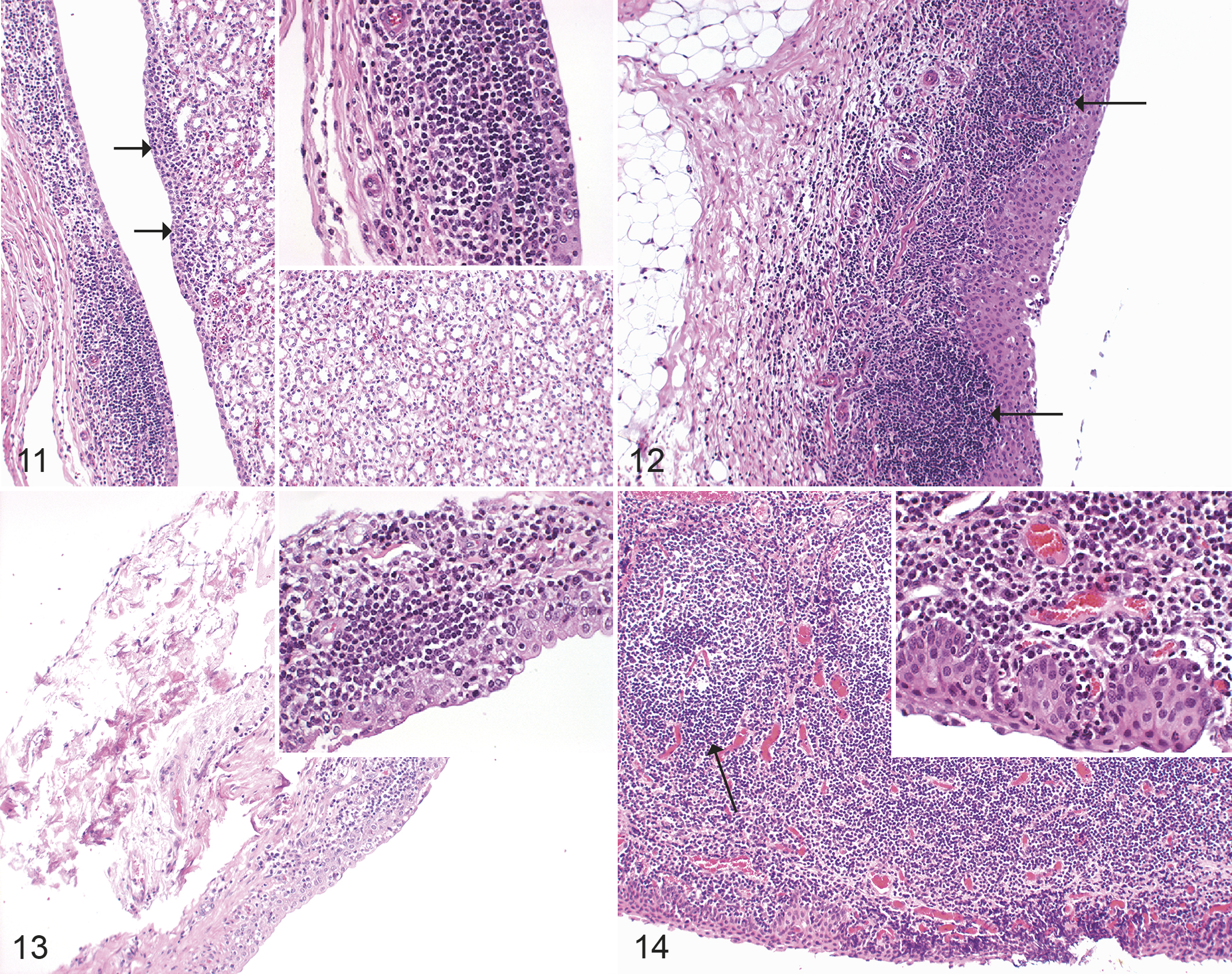
Chlamydia pecorum arthritis, carpal joint, sheep, experimental infections. Hematoxylin and eosin.
Frequency and Scoring of Histologic Lesions in Joints of Sheep Experimentally Infected With Chlamydia pecorum, Grouped According to Inoculum and Route of Administration.
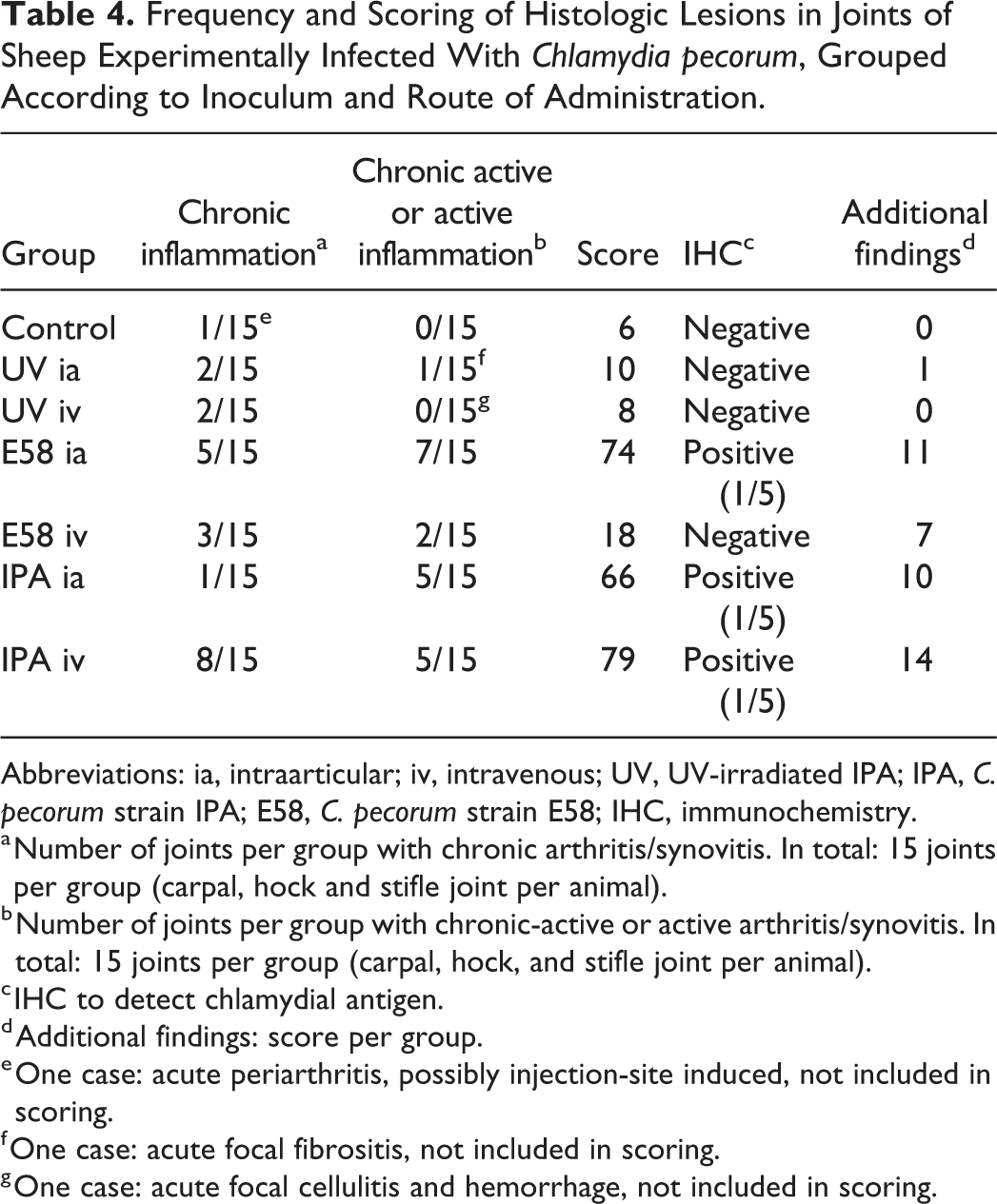
Abbreviations: ia, intraarticular; iv, intravenous; UV, UV-irradiated IPA; IPA, C. pecorum strain IPA; E58, C. pecorum strain E58; IHC, immunochemistry.
a Number of joints per group with chronic arthritis/synovitis. In total: 15 joints per group (carpal, hock and stifle joint per animal).
b Number of joints per group with chronic-active or active arthritis/synovitis. In total: 15 joints per group (carpal, hock, and stifle joint per animal).
c IHC to detect chlamydial antigen.
d Additional findings: score per group.
e One case: acute periarthritis, possibly injection-site induced, not included in scoring.
f One case: acute focal fibrositis, not included in scoring.
g One case: acute focal cellulitis and hemorrhage, not included in scoring.
Additional findings (synoviocyte proliferation, neovascularization, and periarterial fibrosis; Table 5) were evaluated (Figs. 7, 8) and the IPA iv group had the highest number of animals (14), which showed one or more of these lesions. Stifle joints had the highest score followed by carpal and hock joints (Supplemental Table S5). An investigation of synovial pathology score differences between the experimental groups and the control group were analyzed using generalized mixed effect models with a Poisson distribution. The groups E58 ia, IPA iv, and IPA ia differed significantly from the control group (P < .001, Supplemental Table S6).
Frequency of Additional Histologic Parameters in Joints of Sheep Experimentally Infected With Chlamydia pecoruma.
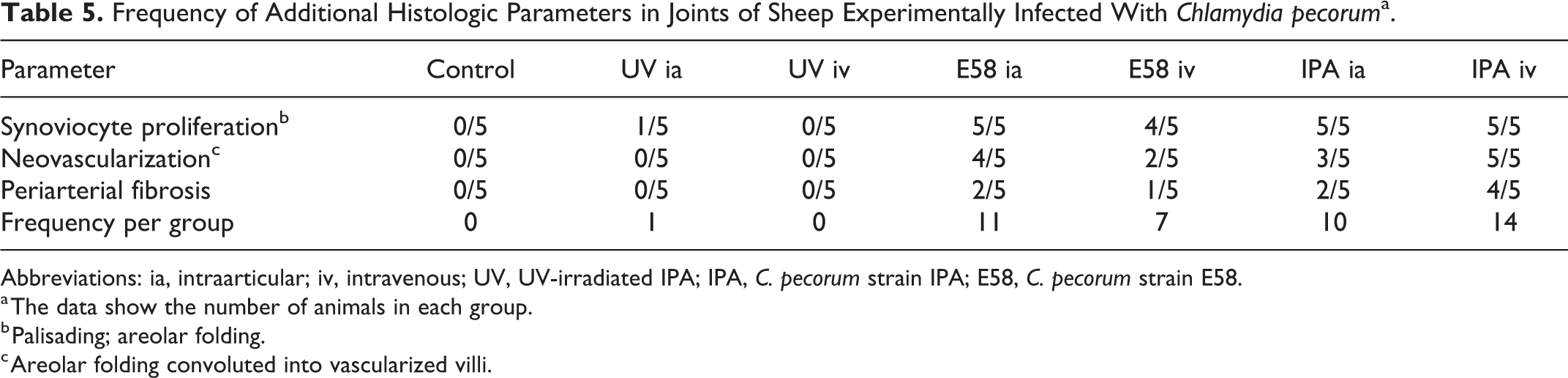
Abbreviations: ia, intraarticular; iv, intravenous; UV, UV-irradiated IPA; IPA, C. pecorum strain IPA; E58, C. pecorum strain E58.
a The data show the number of animals in each group.
b Palisading; areolar folding.
c Areolar folding convoluted into vascularized villi.
The presence of C. pecorum DNA in joints (synovia) for all 5 naturally infected sheep (5/5) was confirmed by PCR. Histologically, joints of 5/5 animals had inflammatory lesions. The majority of inflammatory lesions were chronic-active processes (lymphoplasmacytic and histiocytic as well as purulent arthritis/synovitis; Figs. 9, 10). The right carpus of animal 3 did not show any inflammatory signs.
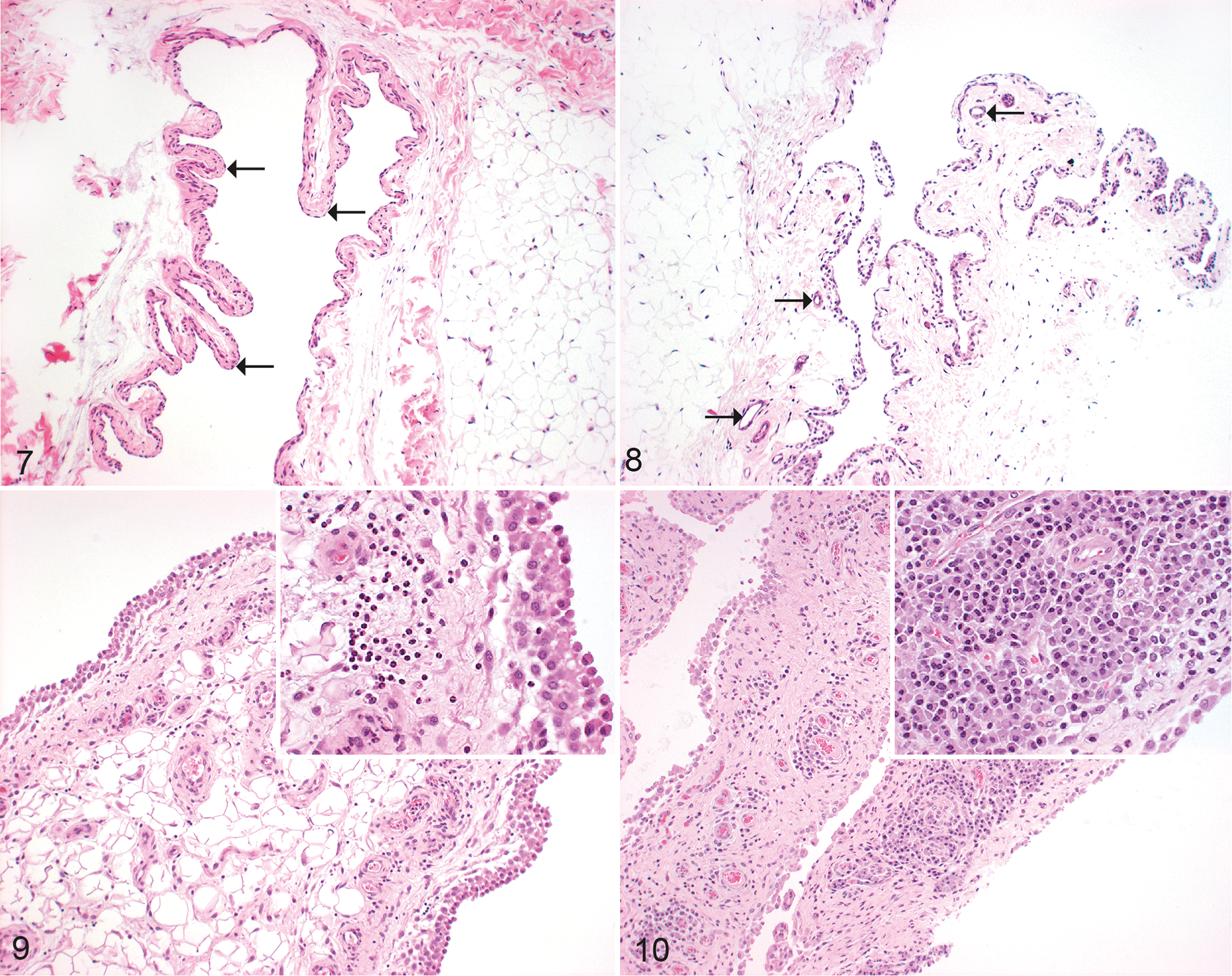
Chlamydia pecorum arthritis, joint, sheep. Hematoxylin and eosin.
Histologic Findings in Internal Organs
The control and IPA UV-irradiated groups did not exhibit any lesions in the kidneys (Table 6). In other groups of infected animals, the most obvious kidney lesion was a chronic or chronic-active pyelitis, which partially ascended into the interstitium (Figs. 11, 12) and was most severe in the IPA iv group. Statistical analyses of differences between the experimental groups compared to the control group revealed significant differences in group IPA iv (P value: <.001, Supplemental Table S6).
Frequency and Scoring of Histologic Lesions in Internal Organs of Sheep Experimentally Infected With Chlamydia pecorum, Grouped According to Inoculum and Route of Administration.

Abbreviations: ia, intraarticular; iv, intravenous; UV, UV-irradiated IPA; IPA, C. pecorum strain IPA; E58, C. pecorum strain E58.
a Number of inner organs per group with chronic inflammation.
b Number of inner organs per group with chronic-active or active inflammation.
c Immunohistochemistry to detect chlamydial antigen.
d1/5 = mild and focal chronic encephalitis, not included in scoring.
e1/5 = mild and focal chronic periportal hepatitis, not included in scoring.
f Leukocytostasis.
g Focal glia nodule, negative tested for toxoplasma antigen.
h Also interstitial nephritis.
i1/5 = mild chronic and focal lymphocytic pyelitis, not included in scoring.
Chlamydia pecorum infection, sheep. Hematoxylin and eosin.
All naturally infected animals had a chronic (animals 1 and 3) or chronic-active (animals 2, 4, and 5) pyelitis (Fig. 13), which partially ascended into the interstitium. A severe chronic (lymphoplasmahistiocytic) multifocal conjunctivitis was seen in animal 1 (Fig. 14).
The brains of animals from the control and IPA UV-irradiated groups did not show any signs of inflammation. In brains of the experimentally infected animals, the most obvious inflammatory lesion was a mild chronic lymphohistiocytic encephalitis or meningo-encephalitis (5/20 animals; Table 6, Fig. 15). Glial nodules were present in 3/35 brains (1 each from the UVA-IPA ia, E58 iv, and IPA iv groups; Fig. 15, inset). In these cases, Toxoplasma gondii infection was ruled out by examination of brain sections using immunohistochemistry (rabbit polyclonal antibody RB-282-A0, Thermo Fisher Scientific; dilution 1:50).
Lung tissues from experimental animals (n = 35) did not have any signs of inflammation, and thus no significant differences were found between the groups. BALT hyperplasia and lymphocytic peribronchitis/peribrochiolitis were present in 23/35 lungs in animals from all groups (Fig. 16). Secondary follicles were found in 28/35 lymph nodes in animals from all groups (Supplemental Fig. S1).
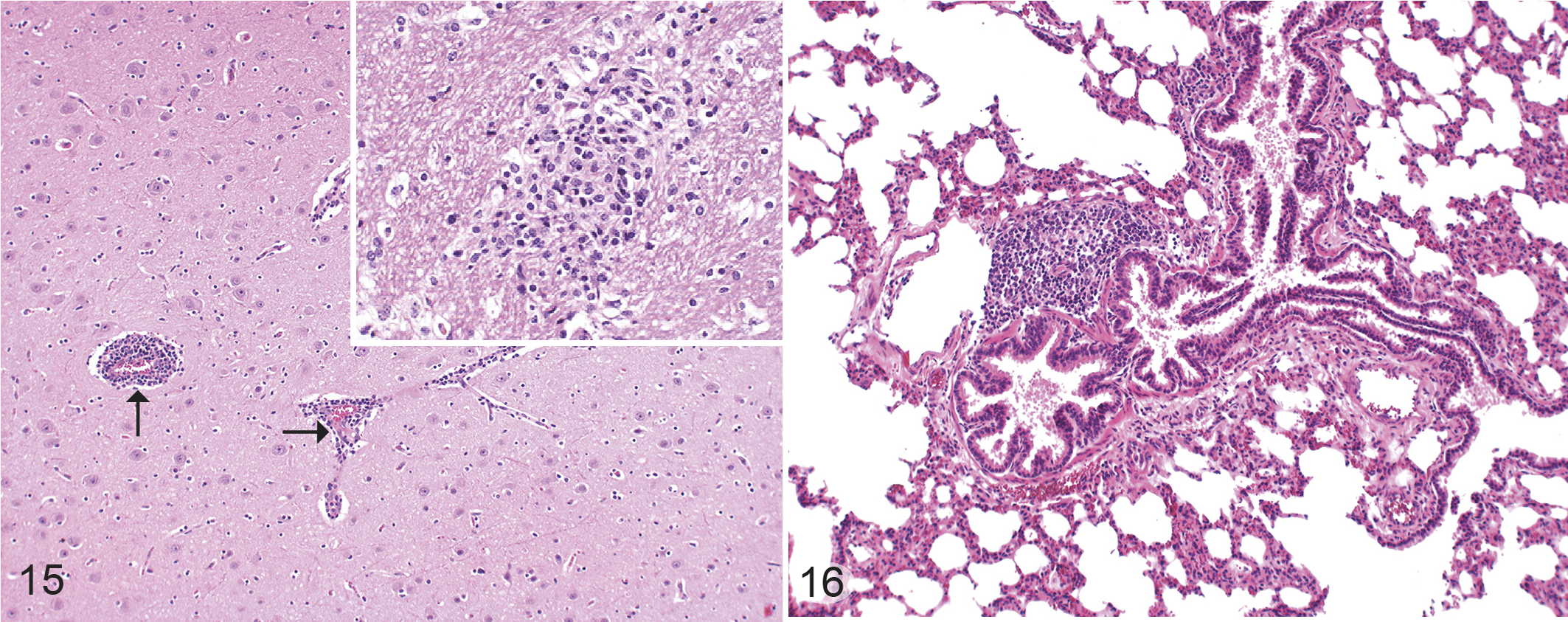
Chlamydia pecorum infection, sheep. Hematoxylin and eosin.
A mild chronic multifocal periportal hepatitis was present in 5/15 livers of animals from the control and the IPA UV-irradiated groups (Supplemental Fig. S2). Similar lesions were also observed in 8/20 livers of animals in the other experimentally infected groups. Occasional leukocytostasis in portal veins was present in all groups (Table 6).
Tissues of spleen and lymph nodes from all experimentally infected animals (n = 35) did not reveal any signs of inflammation and no significant differences were found (Supplemental Table S5). For the investigation of differences between the experimental groups compared to the control group in lymph nodes, linear model measurement was performed and revealed a significant difference (P < .01) between numbers of follicles in the E58 iv group compared to the control group but no significant differences regarding the numbers of secondary follicles (Supplemental Table S6).
In naturally infected animals, lungs, lymph nodes, and spleen did not show any inflammatory lesions. A mild chronic multifocal periportal hepatitis was present in livers of all animals (n = 5).
Immunohistochemistry
Immunohistochemical labeling of chlamydial antigen in experimentally infected animals revealed positive results in the carpal joint of one animal from the IPA ia group (Fig. 17), the stifle joint of one animal from the IPA iv group, the carpal, hock, and stifle joints of one animal from the E58 ia group, and the kidney of one animal from the IPA iv group (Fig. 18). In naturally infected animals, immunohistochemical labeling of chlamydial antigen revealed positive results in the left and right carpal joints of animal 1 (Fig. 19), the right carpal joint and kidney of animal 2 (Fig. 20), the left carpal joint of animal 3, the right carpal joint of animal 4, and the kidney of animal 5.
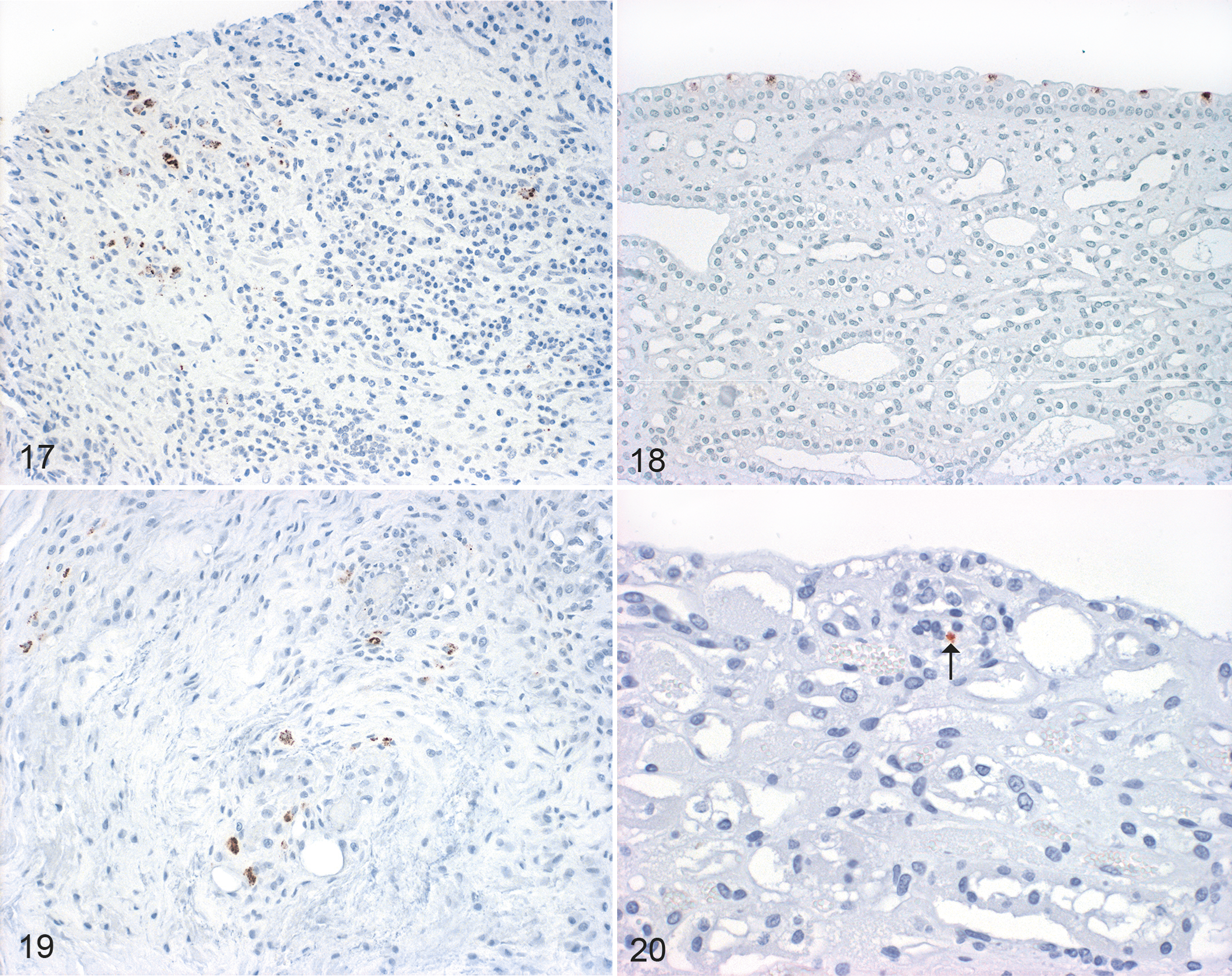
Chlamydia pecorum infection, sheep. Immunohistochemistry for Chlamydia sp.
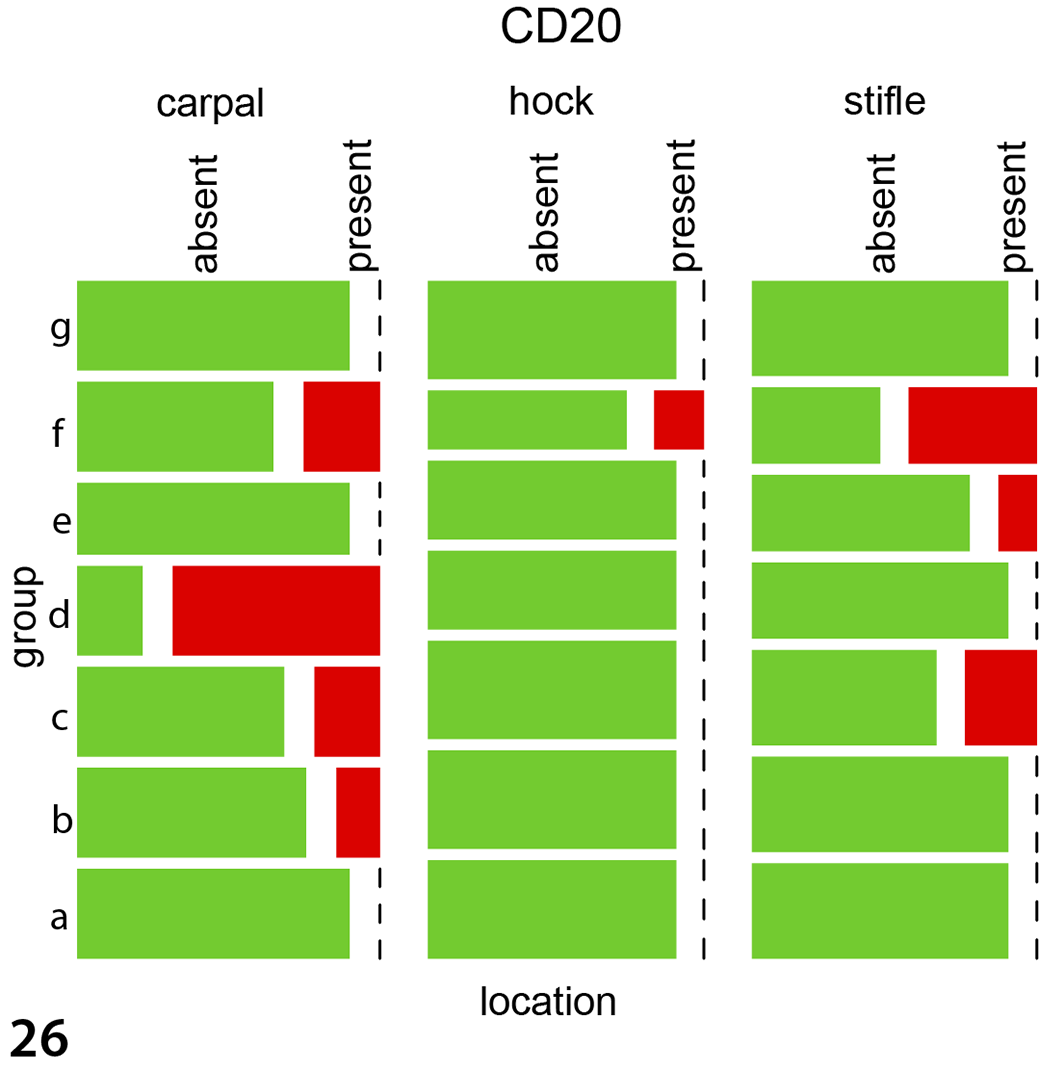
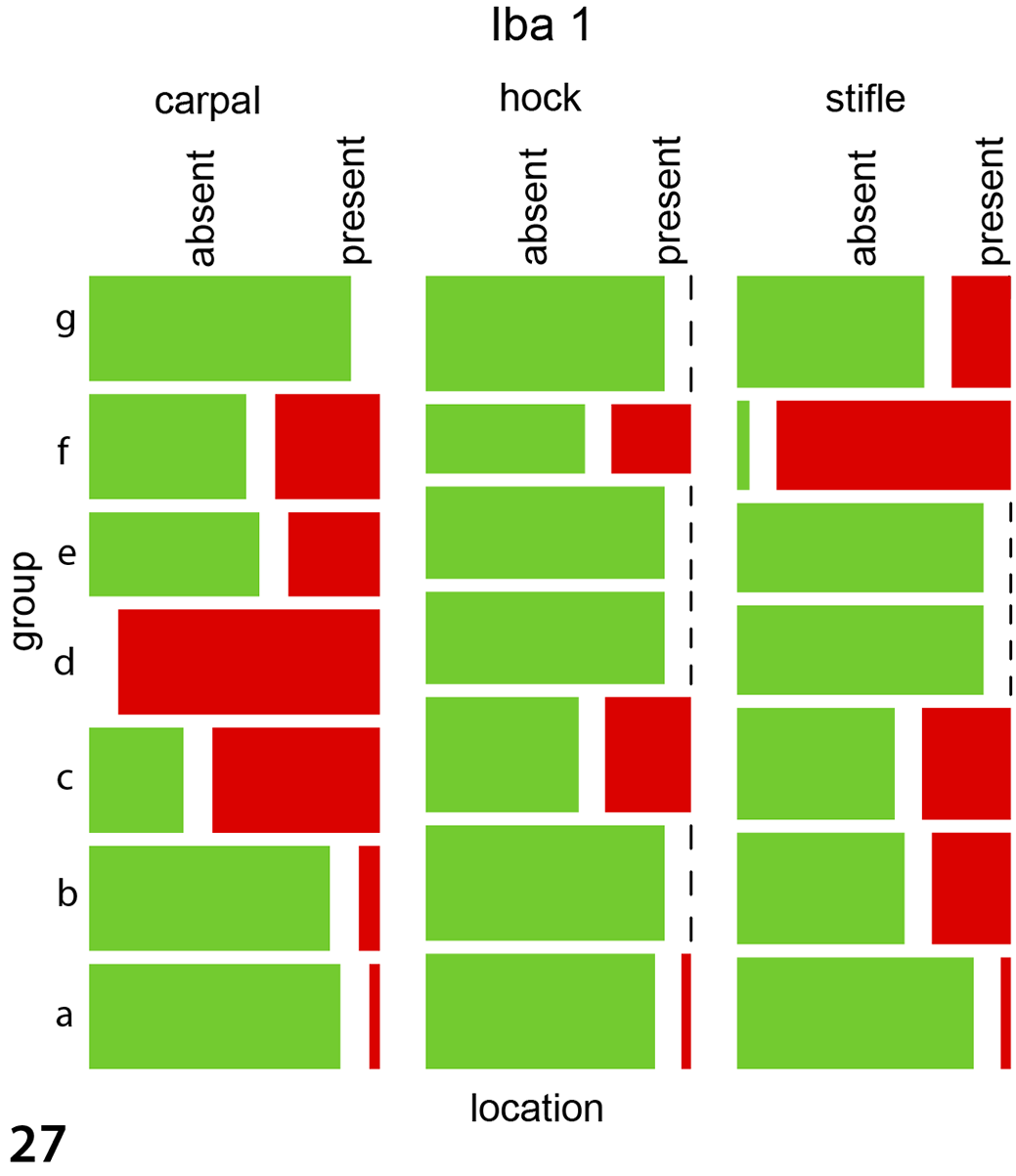
Immunohistochemical labeling in experimentally infected animals to detect Iba1 (macrophages, Fig. 21), CD20 (B cells, Fig. 22), and CD3 (T cells, Fig. 23) antigens was histologically most obvious for detecting macrophages (Iba1). The same pattern was observed in the 5 naturally infected animals using antibodies against Iba1 (Fig. 24), CD20 (Supplemental Fig. S3), and CD3 (Supplemental Fig. S4). For the immunohistological labeling (Iba1, CD20, CD3) in experimentally infected animals, a statistical evaluation was performed. Wilson’s binomial 95% confidence intervals revealed the highest proportions of cells with Iba1 labeling (24.5% of cells were labelled), followed by CD20 (12.2%) and CD3 (11.50%) with the highest amount of positive labelled cells in carpal joints, followed by stifle and hock joints. For CD3 labeling, the groups IPA ia, IPA iv, and E58 ia differed significantly from the control group with the highest presence in carpal joints followed by stifle and hock joints (P < .004 for all joints, Supplemental Table S6, Fig. 25). CD20 labeling revealed a significant difference in the E58 ia, IPA ia, and IPA iv group when compared to the control group with the highest presence in carpal joints followed by stifle and hock joints (P < .01, Supplemental Table S6, Fig. 26). For Iba1 labeling, the groups E58 ia, IPA iv, and IPA ia group differed significantly from the control group with the highest presence in carpal joints followed by stifle and hock joints (P < .001, Supplemental Table S6, Fig. 27).
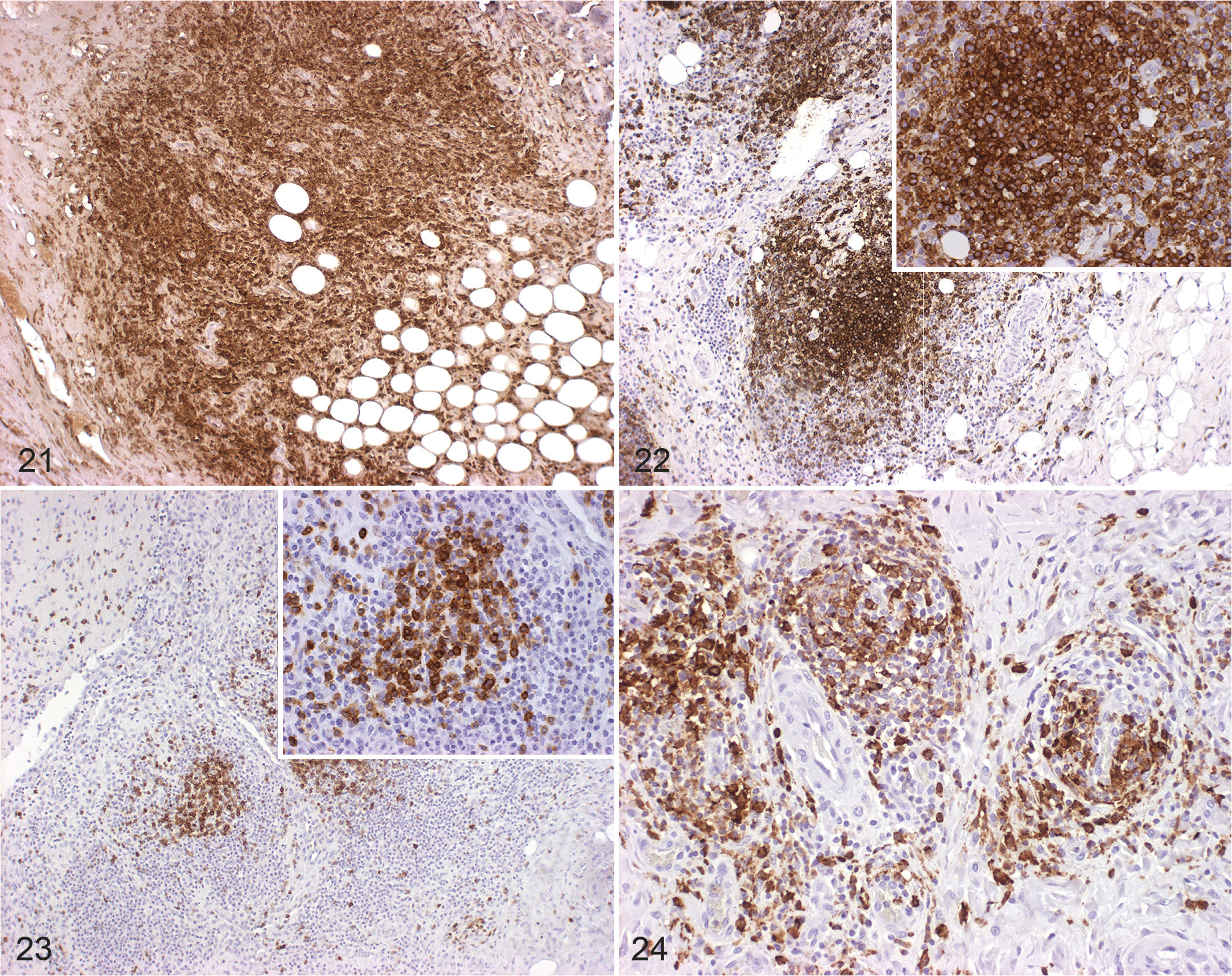
Chlamydia pecorum infection, joints, sheep. Immunohistochemistry for detecting Iba1 (macrophages), CD20 (B cells), and CD3 (T cells).
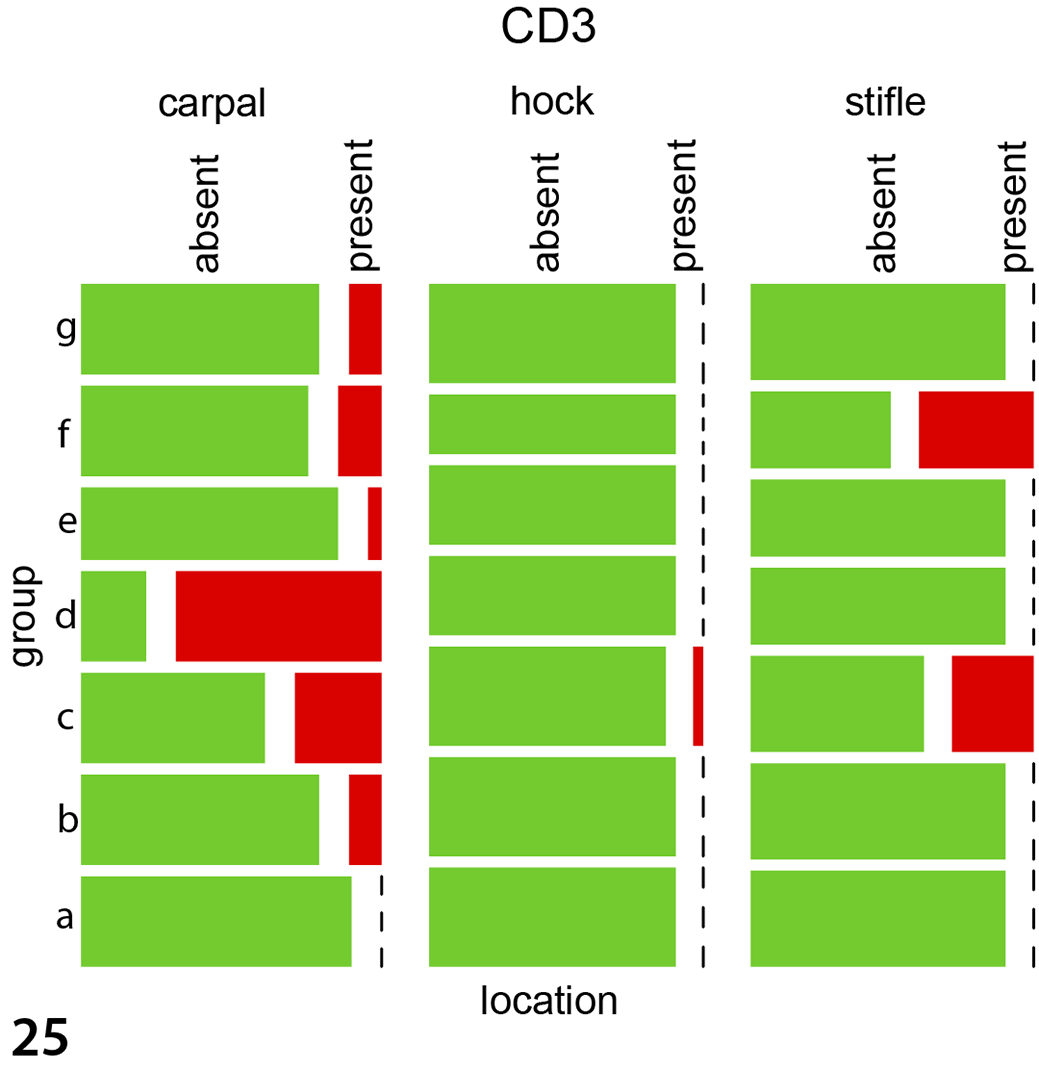
Mosaicplots, immunohistochemistry data for the joint tissues of experimentally infected animals. The groups IPA ia, IPA iv, and E58 ia differed significantly from the control group for CD3 (Fig. 25), CD20 (Fig. 26), and Iba1 (Fig. 27). Group a: UVA iv, b: UVA ia, c: IPA iv, d: IPA ia, e: E58 iv, f: E58 ia, g: control. For all antigens, the greatest immunolabeling was in carpal joints followed by stifle and hock joints. Data were evaluated using mixed models.
qPCR Results
By qPCR, 8/15 joint tissues from the IPA ia group were positive for chlamydial DNA (5/5 carpal joints, 1/5 hock joint, and 2/5 stifle joints) with a range of 71 to 142 313 copies/μl. In the IPA iv group, 11/15 joint tissues tested positive for chlamydial DNA (qPCR, 2/5 carpal joints, 5/5 hock joint, and 4/5 stifle joints) with a range of 130 to 2506 copies/μl. In the E58 ia group, 5/15 tissues of carpal joints were positive for chlamydial DNA via qPCR with a range of 9641 to 167 349 copies/μl. In the E58 iv group, 8/15 joint tissues were positive by qPCR (2/5 carpal joints, 4/5 hock joint, and 2/5 stifle joints) with a range of 92 to 1884 copies/μl. Wilson’s binomial 95% confidence intervals revealed the highest proportions in carpal joints followed by stifle and hock joints (Supplemental Table S5). This was also confirmed by mixed Poisson and zero-inflated Poisson models. The groups which received intraarticular inoculation had significantly more copies per µl than the intravenously inoculated groups (Supplemental Table S6). In carpal, hock, and stifle joints, the highest qPCR loads (copies/μl) were evaluated in the IPA ia group (Supplemental Table S5, loads per joints and groups).
Regarding the internal organs, Wilson’s binomial 95% confidence intervals revealed the highest proportions of qPCR loads in lymph nodes, followed by liver, brain, kidney, spleen, and lung (Supplemental Table S5). Mixed Poisson and zero-inflated Poisson models revealed no significant differences between the qPCR loads of the organs (lung, lymph nodes, liver, spleen, and kidney). There was no significant difference between the groups for all organs assessed (Supplemental Table S6). In brains, kidneys, livers, and lungs, maximal qPCR loads (copies/μl) were evaluated in the E58 iv and in the IPA iv groups in lymph nodes (Supplemental Table S5, loads per organs and groups).
In naturally infected animals, 4/5 joint tissues (left hock of animal 3619, left carpus of animal 7629, right carpus of animal 7579, and right carpus of animal 3619) were positive for chlamydial DNA with a range of 6.76 to 163 copies/μl.
Discussion
Chlamydia pecorum–associated arthritis in sheep is an economically important infectious disease in Australia causing losses to the sheep industry. 50 In this study, we investigated histomorphological lesions of joints and internal organs from experimentally and naturally infected sheep to elucidate the pathogenesis of C. pecorum–associated arthritis. Tissue samples were retrieved from an experimental challenge model in sheep. 21 This challenge model evaluated 2 different inoculation routes (intraarticular, intravenous) and the disease progression of 2 virulent C. pecorum isolates (E58, IPA). The challenge study focused on clinical outcomes and gross lesions with the aim of establishing an infection model that can be utilized for potential testing for vaccines. C. pecorum E58 was isolated from the brain of a calf with sporadic bovine encephalomyelitis 33 whereas C. pecorum IPA originated from a joint of a polyarthritic sheep. 35 Both strains were able to establish systemic infections as shown in previous studies 48,49 and confirmed in this study. Potential limitations of this study were the variability of tissue harvest dates due to different time points of euthanasia and the small animal sizes per group.
The most severe histologic synovial lesions were observed in the IPA iv group with a predominantly chronic inflammatory type (lymphoplasmacytic and histiocytic infiltrates and granulation tissue). Chlamydiae are able to induce chronic disease by persisting in their host and triggering ongoing inflammatory responses. 7 The intravenously inoculated E58 strain resulted in mild histological synovial lesions while the same strain inoculated intraarticularly resulted in severe lesions. For the IPA strain, both inoculation routes resulted in moderate to severe lesions. Altogether, the results suggest that the IPA isolate is more virulent than E58 in this challenge model. Previous potential explanations for this difference 21 include that (1) the IPA strain was originally isolated from a sheep with polyarthritis and is better adapted to this host; (2) the conversion of reticulate to elementary bodies in vitro is 6 hours earlier in IPA than E58, possibly resulting in more replication competence 22 ; and (3) previous studies demonstrated pathogenetic diversity caused by minor genomic variations among different Chlamydia strains. 27,40 Moreover, previous in vivo and in vitro investigations 22,23 indicated that the C. pecorum IPA isolate is a plasmid-bearing strain while E58 is not and a direct relationship between the presence of a plasmid and the strain virulence is strongly suspected. Chlamydial release via host cell lysis appears to be plasmid regulated 53 and results in more infectious EBs available to infect the neighboring cells.
In naturally infected sheep (n = 5), the majority of joint lesions were chronic-active inflammatory lesions (lymphoplasmacytic, histiocytic, and purulent arthritis/synovitis; 4/5 animals), while the majority of joints of experimentally infected animals showed chronic inflammatory lesions without active components (lymphoplasmacytic and histiocytic including granulation tissue). Although experimentally and naturally infected animals were in the same age range, the duration of the C. pecorum infection in naturally infected animals is unknown and thus these studies cannot be directly compared.
Immunohistochemistry detected chlamydial antigen in the carpal joint of animal 628 from the IPA ia group, the stifle joint of animal 658 from the IPA iv group, and in carpal, hock, and stifle joints of animal 710 from E58 ia group. Chlamydial inclusions were found in macrophages and occasional in synovial fibroblasts and synoviocytes. Similar to these investigations, other studies described epithelial or epithelial-like cells as primary chlamydial host cells. 12 Also, synoviocytes have been found to be suitable host cells for C. trachomatis, 20 and C. pecorum isolates can infect circulating monocytes/macrophages. 14,22 This, and the detection of C. pecorum DNA in blood and organs, the detection of IgG and IgM antibodies in sera of infected animals, and the isolation of live organisms from joints of iv-infected animals 21 leads to the inference that there is systemic, hematogenous spreading of C. pecorum, possibly with the help of macrophages.
Chlamydial antigen was rarely detected by IHC in joint tissues and tissues of internal organs of experimentally infected animals, whereas the qPCR test was positive in joints (53%) and internal organs (41%). In naturally infected animals, chlamydial antigen was detected in 5/5 animals by IHC in joint tissues and tissues of internal organs, whereas the corresponding qPCRs were positive in 4/5 joint tissues from 3 animals. A mild or subclinial infection might have been present resulting in only single IHC-positive cells as reported in koala tissues. 9 Furthermore, IHC only detects inclusions while PCR also detects individual elementary and reticulate bodies and is therefore the more sensitive method. Moreover, PCR and IHC data cannot be compared in sections taken from various levels of a FFPE block. 5
In this study, chlamydial IHC results have been included in our newly developed semiquantitative scoring system for the assessment of histomorphological joint lesions. The first step toward the development of our scoring system was the identification of previously established scoring systems. 16,28,34 After the designation of inflammatory features, it was important to avoid extreme ranges of severity levels 42 to create an adaptive, reproducible, and easily applicable scoring system that started with an unblinded initial evaluation, followed by a targeted masked review. 31 The inflammatory features were determined (1) on the basis of standard definitions for chronic and active inflammation under the supervision of a dipl. ECVP pathologist and (2) based on the comparison of synovial pathologic features to normal synovial tissue as previously assessed for human rheumatology research. 10,15 Such semiquantitative scoring systems might aid for characterization or standardization of pathogenesis. They could also have a practical value if clinical or gross findings correlate with histologic findings, although this was not assessed during this study. Interestingly, calculation of the mean scores of all groups revealed the highest score in stifle joints, followed by carpal and hock joints, although the administration route in the ia group was the carpal joint. The reasons for the stifle joint as a predilection site remains unclear.
To our knowledge, a thorough description of C. pecorum–induced histological lesions in internal organs of sheep has not been performed until now. Histologically, severe lesions in experimentally infected (most severe in the IPA iv group) and in all naturally infected animals were observed in the kidneys. The kidneys showed a chronic or chronic-active pyelitis, which partially ascended into the interstitium. In koalas, a thorough investigation of organ distribution and histopathological findings due to chlamydial infection was performed and 6/23 koalas showed pan-glomerular sclerosis, mild glomerulonephritis, and also chronic interstitial nephritis with severe chronic-active pyelitis. 9 Another study revealed that genetically distinct C. pecorum strains have distinct tissue tropisms in the koala and that plasmid-bearing C. pecorum strains in the upper genital tract correlates with urogenital disease, suggesting that plasmids expand the genetic diversity of the infecting Chlamydia and can increase virulence. 36 Thus, there are similarities between kidney lesions in koalas and sheep due to infection with C. pecorum.
Interestingly, 4/20 infected sheep (IPA ia [n = 1], IPA iv [n = 2], E58 iv [n = 2]) had mild chronic lymphohistiocytic encephalitis or meningoencephalitis, which to our knowledge have been previously reported only in calves due to C. pecorum and in dogs due to the chlamydial agent of ovine polyarthritis, strains LW 679 and LW 38. 46,47,54
In experimentally infected animals, immunohistochemical labeling was most obvious for detecting macrophages (Iba-1), followed by B cells (CD20) and T cells (CD3), with the highest presence in carpal joints followed by stifle and hock joints. In naturally infected animals, immunohistochemical labeling was similarly most obvious for detecting macrophages. Macrophages, monocytes, and mucosal epithelial cells of respiratory, gastrointestinal, urogenital tracts, and conjunctival and placental trophoblastic epithelium are well known as chlamydial target cells. 32,38,44 In humans, monocytic cells constitute the primary host cell type for C. trachomatis–induced reactive arthritis in synovial tissue. 11 Earlier research groups detected C. trachomatis inclusions within blood monocytes via transmission electron microscopy and chlamydial major outer membrane protein via immunoelectron microscopy. 29
Conclusions
The objective of the present study was to investigate histologic changes due to C. pecorum in tissues of carpal, hock, and stifle joints as well as spleen, liver, kidney, lymph nodes, and brain of 35 sheep. The most obvious inflammatory lesions were observed in joints and renal pelvis from experimentally infected animals (especially those infected with IPA) and in naturally infected animals. The immunohistochemical assessment of the presence and distribution of macrophages, T and B cells in joints revealed the highest presence of macrophages. Former observations that C. pecorum infected circulating monocytes along with our findings of histological lesions in joints and internal organs with detection of C. pecorum by qPCR, underlines the assumption of a hematogenous spread of this agent. In humans, macrophages constitute the primary host cell type for C. trachomatis-induced reactive arthritis in synovial tissue. The immunohistochemical assessment, reported here, with the highest presence of macrophages might lead to the hypothesis that monocytic cells constitute a primary host cell type for C. pecorum, among others. This is a valuable finding but further confirmative investigations are necessary. Ultimately, a better understanding of C. pecorum pathogenesis and the development of a vaccine will help minimize the consequences of chlamydial polyarthritis and the economic losses due to C. pecorum infection.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Ostfeld_et_al - Chlamydia pecorum–Induced Arthritis in Experimentally and Naturally Infected Sheep
Supplemental Material, Combined_supplemental_materials-Ostfeld_et_al for Chlamydia pecorum–Induced Arthritis in Experimentally and Naturally Infected Sheep by Nina Ostfeld, Mominul M. Islam, Martina Jelocnik, Monika Hilbe, Titus Sydler, Sonja Hartnack, Caroline Jacobson, Tom Clune, Ian Marsh, Narelle Sales, Adam Polkinghorne and Nicole Borel in Veterinary Pathology
Footnotes
Acknowledgements
The authors wish to thank all collaborators from the University of the Sunshine Coast, Australia. We thank the laboratory of the Institute of Veterinary Pathology, Vetuisse Faculty, University of Zurich, Switzerland, for preparing the FFPE blocks and histological and immunohistochemical sections (Theresa Pesch, Sabina Wunderlin, Barbara Prähauser). We acknowledge the NSW DPI, Elizabeth Macarthur Agricultural Institute, where the experimental infection trial was conducted. Many thanks to Mark Hazelton, Sam Gilchrist, Ronald Coilparampil, Leah Stroud, Shayne Fell, Greg Glasgow, and Kim Koeford. We thank the owners and staff from the farm in Western Australia for their assistance in providing access to their sheep and facilities for this study, and ongoing support for research into sheep health, welfare, and production. We would like to thank Jeanne Peter from Vetcom, Vetsuisse Faculty, University of Zurich, Switzerland, for her help in designing the figure plates.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Meat and Livestock Australia and the Australian Wool Innovation supported Science and Innovation Award for Young People in Agriculture, Fisheries and Forestry provided funding to projects for which naturally infected sheep were identified and samples were provided for this study.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
