Abstract
Dogs with and without lymphoma have undergone hematopoietic cell transplantation in a research setting for decades. North Carolina State University is currently treating dogs with B- and T-cell lymphoma in a clinical setting with autologous peripheral blood progenitor cell transplants, using peripheral blood CD34+ progenitor cells harvested using an apheresis machine. Complete blood counts were performed daily for 15 to 19 days posttransplantation to monitor peripheral blood cell nadirs and subsequent CD34+ cell engraftment. This study documents the hematologic toxicities of total body irradiation in 10 dogs and the subsequent recovery of the affected cell lines after peripheral blood progenitor cell transplant, indicating successful CD34+ engraftment. All peripheral blood cell lines, excluding red blood cells, experienced grade 4 toxicities. All dogs had ≥ 500 neutrophils/μl by day 12, while thrombocytopenia persisted for many weeks. All dogs were clinically normal at discharge.
For close to 5 decades, dogs have been utilized as a large animal preclinical model for hematopoietic cell transplantation. 5 Given this research data and recent reports documenting the successful harvesting of peripheral blood CD34+ progenitor cells or whole bone marrow and reinfusion after myeloablative therapy in a clinical setting, 3,4,6 North Carolina State University began treating dogs diagnosed with B- and T-cell lymphoma with autologous peripheral blood progenitor cell transplant following lethal total body irradiation (TBI). In this report, we document the bone marrow toxicities associated with the pretransplant TBI and the subsequent hematopoietic reconstitution in the ensuing weeks.
Procedures
Ten dogs diagnosed cytologically with lymphoma (8 B-cell, 1 T-cell, 1 unknown phenotype) were admitted to North Carolina State University Bone Marrow Transplant Unit. All dogs were in clinical remission upon arrival. Seven to 10 days prior to admission, the dogs received 500 mg/m 2 of cyclophosphamide (Cytoxan, cyclophosphamide for injection, USP) intravenously. Six days before apheresis, 5 μg/kg of rhG-CSF (Neupogen, Amgen; subcutaneous, 2 times daily) was initiated. After a double dose of rhG-CSF on day 6, peripheral blood mononuclear cell apheresis was performed using a Baxter-Fenwal CS3000 Plus cell separator. The percentage of CD34+ cells in the apheresis product ranged from 0.98% to 4.8% (mean, 2.6%). The following day, the dogs received two 5-Gy doses of TBI (Clinac 1800, Varian Medical Systems, Palo Alto, CA) with a 3-hour interfraction interval. 1,2 Immediately following TBI, the cell harvest was infused intravenously over 1 hour (day 0). All dogs received > 1.8 × 10 6 CD34+ cells/kg (mean, 5.7 × 10 6 /kg; range, 1.8 × 10 6 /kg–1.3 × 10 7 /kg).
Bone marrow toxicities and subsequent hematopoietic reconstitution after TBI were monitored using daily complete blood counts and 100-cell manual differential counts. All patients exhibited marked leukocytosis on the day of apheresis (day –1) consistent with rhuG-CSF administration (mean white blood cell count, 56 260/ml; range, 42 130–90 770/ml; reference interval, 4390–11 610 cells/μl; mean neutrophil count, 46 450/ml; range, 34 550–70 800/ml; reference interval, 2841–9112/μl; mean monocyte count, 2883/μl; range, 1582–5417/μl; reference interval, 75–850/μl). Six out of 10 dogs were lymphopenic at this time (mean, 1160/μl; range, 0–2279/μl; reference interval, 594–3505/μl). Nine of 10 dogs were also thrombocytopenic (mean, 146 000/μl; range, 88 000–199 000/μl; reference interval, 190 000–468 000/μl). The mean hematocrit (Hct) at this time was 37.8% (range, 31.4%–45.3%; reference interval, 39.2%–55.9%).
Findings
Eight out of 10 dogs were anemic on day –1; 10 of 10 dogs were anemic on day +1 (mean, 31.1%; range, 26.1%–39%). The lowest group mean Hct of 23.2% occurred on day +7 (Fig. 1a). Progressive Hct recovery to baseline was observed by day +20 in all dogs, and Hcts were within the reference interval in all dogs by day +50.
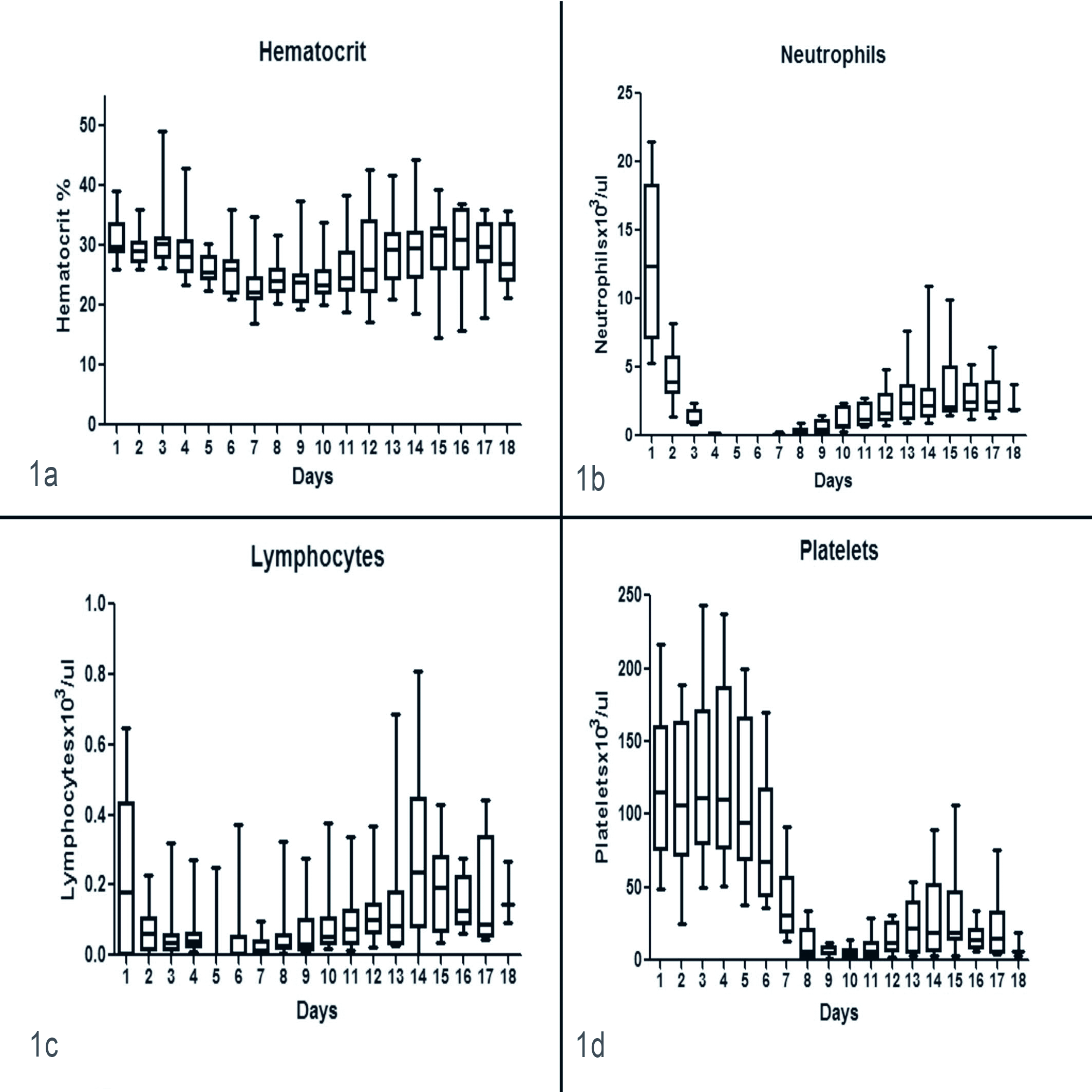
Box and whisker plots of red blood cell (a), neutrophil (b), lymphocyte (c), and platelet (d) dyscrasias for 18 days after lethal total body irradiation, followed by peripheral blood CD34+ progenitor cell transplantation.
In all dogs, the neutrophil count nadir occurred on day +5, where 9 out of 10 (90%) had 0/μl and 1 dog had 10 cells/μl (grade IV toxicity 7 ). Within 24 to 36 hours, a progressive and rapid recovery of neutrophils was observed (Fig. 1b). Neutrophil counts were > 1000/μl in all dogs by day +15 (mean, 2930/μl) and recovered to normal values while either in the hospital or soon after discharge.
Although 6 dogs were lymphopenic at day –1, 9 were lymphopenic on day +1 (mean, 230/μl). The remaining dog, at 645 cells/μl, was at the low end of the reference interval. The lowest lymphocyte counts (mean, 20/μl) were seen on days +5 and +7 (Fig. 1c), although progressive recovery was observed after this time. All dogs remained lymphopenic at discharge and remained lymphopenic until day +30.
Platelet counts progressively decreased after TBI, with the nadir in all dogs occurring on days +9 or +10 (mean, 4450/μl; range, 0–15 000/μl). Similar to the neutrophils, within 24 to 36 hours, platelet recovery was observed (Fig. 1d). This recovery was slow, with ∽60% of patients having platelet counts below 20 000/μl at day +20. In 6 patients, platelet counts day +75 or more were available. Four had platelet counts within the reference interval; 1 was markedly thrombocytopenic on day +89 (48 000/μl); and 1 was mildly thrombocytopenic on day +103 (162 000/μl).
Comments
In humans, engraftment after lethal myeloablative therapy and hematopoietic cell transplantation is documented by absolute neutrophil counts consistently greater than 500/μl. 4 In this study, this occurred in 10 of 10 dogs between days +9 and +11. Therefore, it is concluded that engraftment did occur in these patients following lethal TBI. These encouraging results lend credence to the notion that client-owned canine lymphoma patients can undergo successful autologous peripheral blood progenitor cell transplant in a clinical setting. This lays the groundwork for future studies using a larger cohort of patients to determine the effectiveness of this procedure when treating dogs with B- and T-cell lymphoma.
Footnotes
Acknowledgements
We wish to thank Tillie Laws and Joanna Carroll from the Clinical Pathology Laboratory at the Veterinary Teaching Hospital, North Carolina State University, for their support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
