Abstract
Ractopamine, a synthetic β2-adrenoceptor agonist, is widely used as a feed additive in the United States to promote a reduction in body fat and enhance muscle growth in cattle, pigs, and turkeys. It has the potential for illegal use in show and racing animals because it may affect performance via its β-adrenergic agonist properties or anabolic activities. Nine greyhounds were orally administered 1 mg/kg of ractopamine to investigate the ability to detect the drug in urine. Postdosing, 7 of 9 dogs developed cardiac arrhythmias and had elevated troponin levels indicating myocardial damage. One dog necropsied 4 days postdosing had massive myocardial necrosis, mild to focally moderate skeletal muscle necrosis, and widespread segmental arterial mediolysis. A second dog necropsied 17 days postdosing had mild myocardial necrosis and fibrosis. Scattered arteries exhibited segmental medial and perimedial fibromuscular dysplasia. This is the first reported case of arterial, cardiac, and skeletal muscle damage associated with ractopamine.
Synthetic β2-adrenoceptor agonists cause smooth muscle relaxation resulting in bronchial and vasodilation and were developed for the treatment of bronchial disease. 1 Later it was discovered that these compounds were efficient partitioning agents capable of promoting a reduction in body fat and enhancing muscle growth in cattle, sheep, and swine. 10 Ractopamine is a synthetic β2-adrenoceptor agonist that is widely used in US swine, cattle, and turkeys as a feed additive to increase feed efficiency and carcass leanness by inhibiting fat production, stimulating lipolysis, and increasing protein synthesis. 10
The cardiotoxicity of endogenous adrenoceptor agonists (eg, norepinephrine) and a synthetic analogue (isoprenaline) is widely recognized. 3–5,8 Cardiotoxic effects have also been reported for synthetic β-adrenoceptor agonists (eg, fenoterol) and selective β2-agonists, including salbutamol and clenbuterol. 2,5,7,8 Studies have demonstrated that cardiotoxicity is mediated by β1- rather than β2- adrenoceptors. 4 When clenbuterol is given in vivo, its stimulation of β2-adrenoceptors of the peripheral vasculature and sympathetic nervous system potentiates norepinephrine release from the sympathetic varicosities, which is sufficient to stimulate cardiomyocyte β1-adrenoceptors and induce apoptosis. 3,4 This mechanism was conclusively demonstrated when prior administration of either a selective β1-adrenoceptor antagonist (bisoprolol) or reserpine, a compound that depletes norepinephrine from sympathetic varicosities, significantly protected the myocardium from apoptosis induced by clenbuterol. 2,3 The cardiotoxicity of selective β2-adrenoreceptor agonists such as clenbuterol and salbutamol is less than that of agonists that also stimulate cardiomyocyte β1-adrenoceptors and directly induce cardiomyocyte death, such as fenoterol and isoprenaline. 3,4
Ractopamine is an Association of Racing Commissioners International class 3 drug and is not recognized as a therapeutic medication in racing animals. 6 It has considerable potential for illegal use in humans and show and performance animals because it may have the ability to significantly affect performance via its β-adrenergic agonist properties and its anabolic activities. 2,3,6
Nine adult greyhounds were maintained by a pari-mutuel testing laboratory for the purpose of validating assays to detect illicit drugs in canine urine. Annually, all 9 dogs were vaccinated and tested for heartworm and intestinal parasites; all received a complete physical examination by a licensed veterinarian; and blood was collected for serum chemistries and a complete blood count. Annual hematology and serum chemistry results on all dogs were within published reference intervals for greyhounds, and no abnormalities were identified on physical examination before ractopamine administration. All 9 dogs were administered a single oral dose (1 mg/kg) of ractopamine to investigate the ability of the pari-mutuel testing laboratory to detect this compound in urine. Two hours postdosing, all dogs were lethargic; several had vomited; and abnormal heart rhythms were ausculted. Electrocardiograms (ECGs) recorded 12 hours postdosing revealed elevated heart rates in all 9 dogs and abnormal rhythms in 7 of 9 animals (Table 1).
Nine Greyhounds Orally Administered Ractopamine, 1 mg/kg
VPC, ventricular premature complex; V-tach, ventricular tachycardia.
aEight to 12 hours postdosing.
bReference range, 0.05–0.16 ng/ml; 17 and 60 days postdosing.
cNot available, animal euthanized 17 days postdosing.
dTest result 1 day postdosing.
eNot available, animal euthanized 4 days postdosing.
Serum chemistry and hematologic abnormalities in these 9 dogs 1 day postractopamine administration were variable. When compared to annual testing data and published reference intervals, elevated serum alanine aminotransferase was the only abnormality detected in the majority of animals (7 of 9) (mean, 256.4 IU/liter, range: 134–522 IU/liter, reference interval: 24–90 IU/liter). Elevated alanine aminotransferase in canines is typically associated with hepatocellular injury, but increases may also follow muscle necrosis. Creatine kinase and aspartate aminotransferase were not requested.
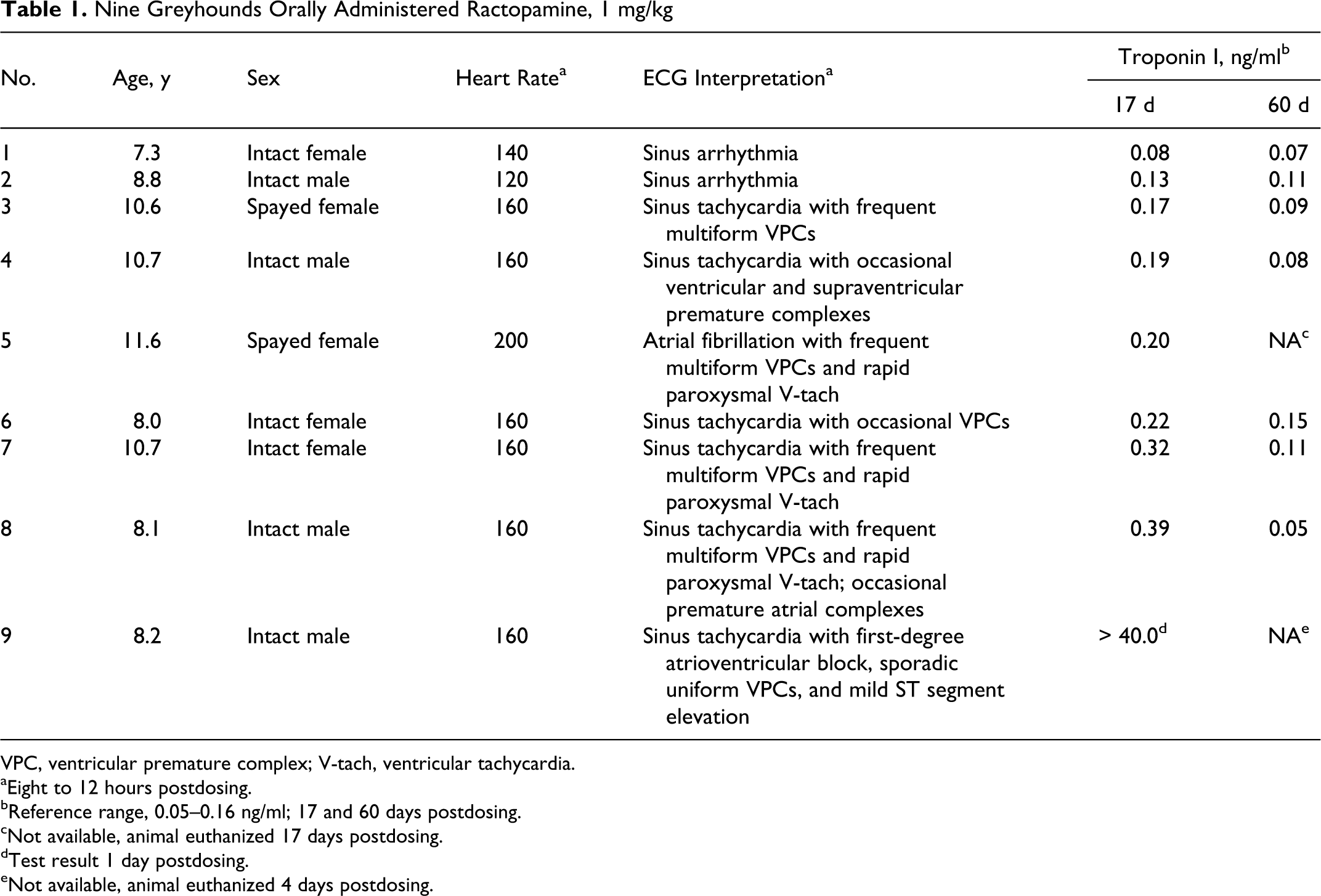
Twelve hours postdosing, dog No. 9, a 7-year-old intact male greyhound, was severely depressed and anorectic. An ECG revealed sinus tachycardia with first-degree atrioventricular block, sporadic uniform ventricular premature complexes, and mild ST segment elevation. Circulating cardiac troponin I concentrations were markedly elevated (> 40 ng/ml). An echocardiogram showed mild pericardial effusion, mildly decreased left ventricular systolic function, and mild left atrial enlargement. Four days postdosing, conduction abnormalities persisted and were characterized by atrial fibrillation, frequent multiform ventricular premature complexes, paroxysmal ventricular tachycardia, and profound ST segment elevation. Echocardiography demonstrated progressive left ventricular systolic dysfunction with chamber enlargement, and the dog was euthanized because of its deteriorating clinical condition. At necropsy, the heart was a mottled pale tan to dark red. The apex and subendocardium were most severely affected (Figure 1). An estimated 75% of the heart, including both atria, were pale tan.
Heart; dog No. 9. The heart is irregularly mottled tan to dark red in color. Tan areas represent extensive regions of necrosis involving the left and right ventricular free walls.
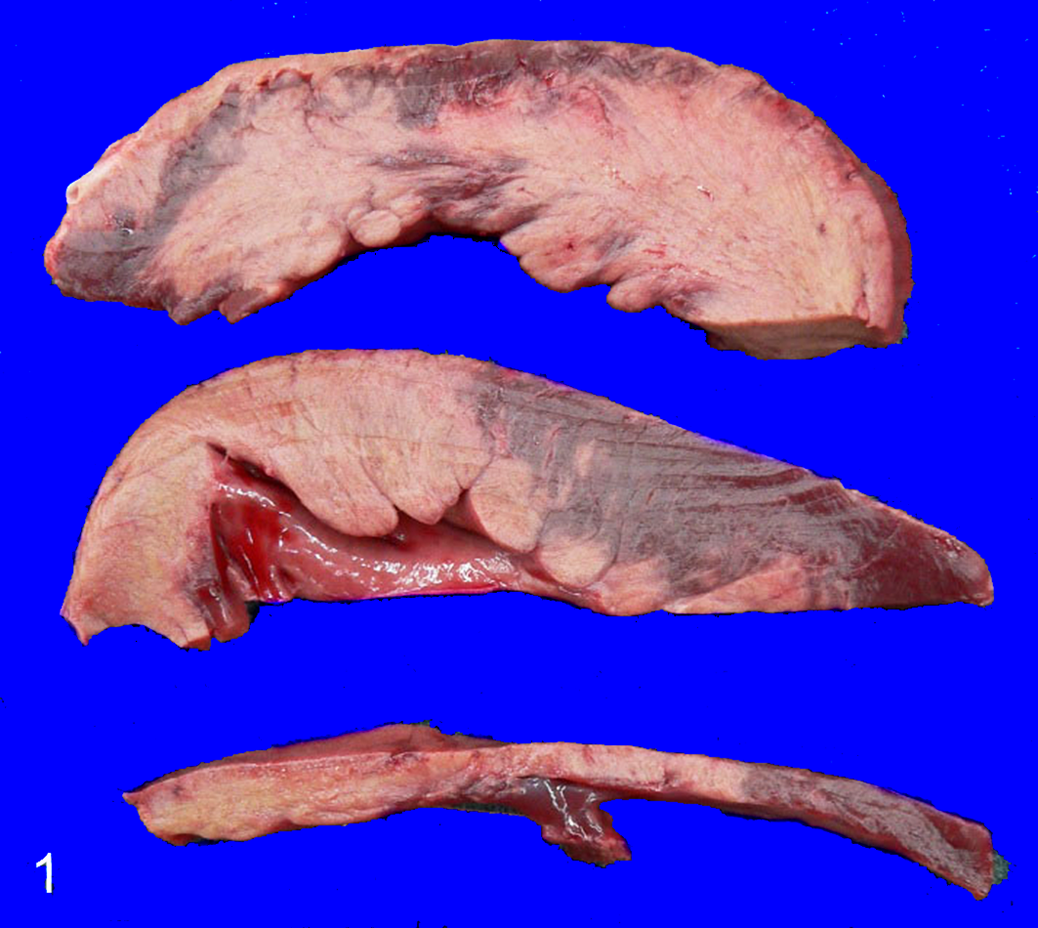
Tissues were fixed in 10% neutral buffered formalin, processed routinely, sectioned at approximately 5 μm, and stained with hematoxylin and eosin. On histologic examination, cardiac myofibers were shrunken, pale, and extensively fragmented and had lost cross striations (Figure 2). Myofiber voids were filled by clusters of plump spindle cells with enlarged nuclei (fibroblasts) (Figure 2). These changes involved scattered small groups of muscle fibers and focally extensive to submassive regions of the heart.
Heart; dog No. 9. There are extensive regions of cardiac myofiber fragmentation with early reparative fibrosis. HE.
Samples of skeletal muscle from the hind limb (biceps femoris, semitendinosus), fore limb (triceps), and epaxial musculature (longissimus) were evaluated. Mild histologic changes were observed in all sections of skeletal muscle. Cross sections revealed scattered individual myofibers and small groups of myofibers that were slightly shrunken and hypereosinophilic, with frayed margins and pyknotic nuclei. These myofibers were separated by clear spaces (edema). Rare myofibers were interrupted by small accumulations of plump spindle cells.
Dramatic changes were identified in small- and medium-sized arteries in several tissues, including the heart, kidney, liver, and vessels surrounding abdominal lymphoid tissue. There was multifocal absence of smooth muscle cells in the outer tunica media, which was extensively expanded by eosinophilic fibrillar material (fibrin) and free red blood cells (Figure 3). The nuclei of smooth muscle cells in the inner tunica media were enlarged and had multiple large nucleoli. Lining endothelial cells were mild to moderately hypertrophied. Affected vessels were surrounded by several haphazardly arranged layers of large, plump spindle cells with markedly enlarged nuclei and occasional mitotic figures (fibroblasts) (Figure 3). In longitudinal sections, the arterial lesions were segmental and did not involve the entire length of the vessel.
Cardiac troponin I concentrations were measured on day 1 postdosing in dog No. 9 (euthanized day 4), day 17 in dog No. 5 (euthanized day 17), and days 17 and 60 in the remaining 7 animals (Table 1). All 7 dogs with postdosing arrhythmias had cardiac troponin I values above the published reference interval of 0.05–0.16 ng/ml. Troponin I values in the 7 surviving dogs were within the published reference interval 60 days postdosing (Table 1).
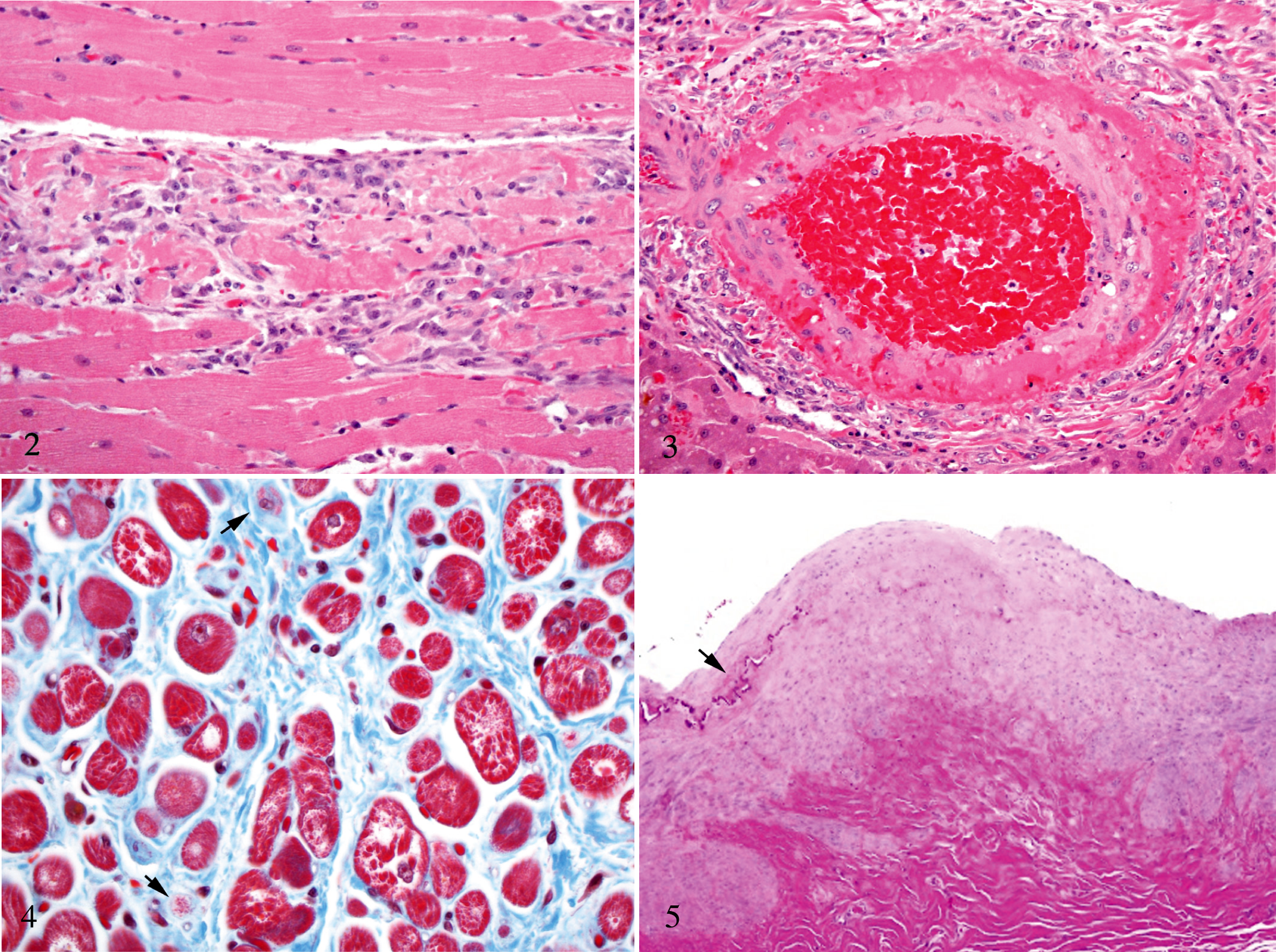
Greyhound No. 5, an 11.5-year-old spayed female, was euthanized 17 days postractopamine administration. An initial ECG 12 hours postdosing demonstrated atrial fibrillation and severe multiform ventricular tachyarrhythmias. The atrial fibrillation persisted, and this animal was euthanized. On gross inspection, the right auricle appeared slightly thickened and fibrotic. A small fibrous scar (4 × 1 × 1 mm) was noted in the right ventricular free wall. Histologic changes were most dramatic in the right atrium and consisted of multifocal variation in myofiber diameter, scattered degenerate fibers, apparent loss of cardiac myofibers, and replacement by loose fibrous connective tissue accompanied by occasional hemosiderin-laden macrophages (Figure 4).
This animal also had lesions in scattered arteries, including arteries of the renal crest, renal, coronary, hepatic, and celiac arteries. These changes consisted of focal extension of fibrous connective tissue from the adventitia deep into the media, replacing smooth muscle cells (Figure 5). The media was focally expanded by disorganized smooth muscle cells, mildly separated by collagenous stroma. Focal mineralization and disruption of the internal elastic lamina was also observed (Figure 5). The celiac artery had a cleftlike space partially filled with free red blood cells between the tunica media and tunica adventitia.
Ractopamine is abused by body builders and has the potential for illicit use in show and performance animals. 2,3,6 This is the first reported case of cardiac and skeletal muscle damage associated with the administration of ractopamine. Changes are comparable to those described for clenbuterol, another selective β2-adrenoceptor agonist. 2–4 Although all dogs were given the same dose on a body weight basis, there was considerable variability in heart damage. The identification of ventricular tachyarrhythmias in 7 of 9 dogs indicate that myocardial damage occurred in the majority of animals shortly after receiving a single oral dose (1.0 mg/kg). Two dogs with persistent cardiac arrhythmias were made available for necropsy. Dog No. 9, necropsied 4 days postdosing, had massive myocardial necrosis, whereas dog No. 5, necropsied 17 days postdosing, had mild myocardial necrosis and fibrosis. Five dogs had arrhythmias, which resolved by 14 days postractopamine administration. Clinical or biochemical evidence of myocardial damage was not identified in the 2 remaining animals.
Cardiac troponin I is a sensitive and specific marker for myocardial injury. Cardiac troponin I concentrations were measured on day 1 postdosing in dog No. 9 and at 17 days postdosing in all remaining animals. Although the delayed sampling time in the majority of dogs was not ideal, all 7 animals with arrhythmias had elevations in serum troponin I (Table 1), indicating myocardial damage.
Two potential mechanisms were considered for the development of the severe myocardial lesions in dog No. 9. Heart changes may have resulted from an indirect, adrenoceptor-mediated effect similar to that described for clenbuterol, 2–4 or the necrosis may have been associated with the striking vascular lesions. The pattern of myocardial damage (focally involving only a few myofibers), the lack of identifiable vascular thrombi, and the absence of necrotizing lesions in other tissues (eg, kidney, which had similar arterial changes) indicate that the massive myocardial necrosis was not likely due to infarction associated with the arterial lesions.
Dog No. 5 was necropsied 17 days postdosing and had atrial fibrillation associated with chronic atrial lesions. Because prior ECG data were not available on any of the dogs, it was unclear whether the atrial fibrillation represented a preexisting condition or was the result of ractopamine administration. Regular monitoring of the heart during annual physical examinations and dental cleaning had failed to detect a rapid or rapid/irregular heartbeat that may have indicated preexisting atrial fibrillation. The elevated cardiac troponin I and the development of ventricular arrhythmias, which resolved, both indicate postdosing myocardial damage in this animal.
Greyhound No. 9 had skeletal muscle damage that was less severe than the myocardial changes. Skeletal myofiber death has been induced with clenbuterol and was demonstrated to be mediated via the β2-adrenoceptor. 3,4 Differences in the relative degree of myotoxicity between heart and skeletal muscle have been reported for other β2-adrenoceptor agonists, including fenoterol and clenbuterol. 3,4
Toxicity following ractopamine administration was not anticipated, because the drug was administered at a level that was less than one-fifth of a dose reported to be well tolerated by Beagles during a long-term study (http://www.inchem.org/documents/jecfa/jecmono/v31je09.htm). Compared to mongrel dogs, trained racing greyhounds are reported to have cardiomegaly, higher cardiac output, higher mean arterial pressure, lower systemic arterial resistances, a lower vascular collagen:elastin ratio, and lower plasma renin activity. Greyhounds have distinctive hemodynamic characteristics, and the physiologic manifestations of breeding and training for coursing and racing may have increased their cardiovascular susceptibility to ractopamine.
Dog No. 9 had striking arterial lesions, which consisted of segmental loss of smooth muscle cells with accumulation of fibrin and hemorrhage in the outer tunica media, hypertrophy of smooth muscle cells in the inner tunica media, and extensive early reparative fibrosis in the surrounding tissue. Lesions are consistent with changes described for a rare human condition termed segmental arterial mediolysis (SAM). The hallmark of SAM is the segmental destruction of arterial media. 9 SAM does not appear to be an infectious, immunologic, or degenerative disorder. 9 It has been proposed that SAM is a vasospastic disease, potentially associated with norepinephrine release, which when applied directly to medial smooth muscle leads to cytoplasmic vacuolization. 9 Norepinephrine is synthesized and released from nerve endings in the outer media of muscular arteries, which is the precise site of injury in SAM. 9
Dog No. 5 was necropsied 17 days postractopamine administration. It had chronic arterial lesions consistent with medial and perimedial fibromuscular dysplasia. The cause of these dysplastic vascular changes in humans is unclear, but it has been speculated that they may represent a chronic manifestation of SAM. 9 A small dissecting hemorrhage was identified between the media and adventitia in the celiac artery. In humans, arterial dissections are an important sequela to SAM and may lead to clinically significant hemorrhage. 9
This report is the first to describe cardiac and skeletal muscle lesions associated with the widely used β2-adrenoceptor agonist ractopamine. It is also the first report of SAM and medial and perimedial fibromuscular dysplasia in the veterinary literature. Other selective β2-adrenoceptor agonists, such as clenbuterol, are thought to cause cardiac myocyte apoptosis because of the potentiated release of norepinephrine. SAM is a proposed vasospastic disease, also likely associated with norepinephrine release, and perimedial fibromuscular dysplasia is speculated to be a chronic manifestation of SAM. The striking myocardial and arterial lesions identified in the 2 most severely affected animals potentially share a common pathogenesis. Specifically, the vascular and myocardial lesions are likely the result of the potentiated release of norepinephrine following β2-adrenoceptor agonist administration.
Footnotes
Acknowledgement
We thank James Fosse (Animal and Plant Health Inspection Service, US Department of Agriculture, Ames, Iowa) for his invaluable technical assistance.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
